Application of Corn Straw, an Agro-Waste, to Remove Dyes in an Aqueous Medium, Producing Blue or Red Fibers
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Treatment CS Fibers
2.3. Characterization of CS Fibers
2.4. Study of Dye Removal by CS Fibers
2.5. Characterization of Dye-Pigmented CS Fibers (After Study of Dyes Removal)
3. Results and Discussion
3.1. Characterization of CS Fibers
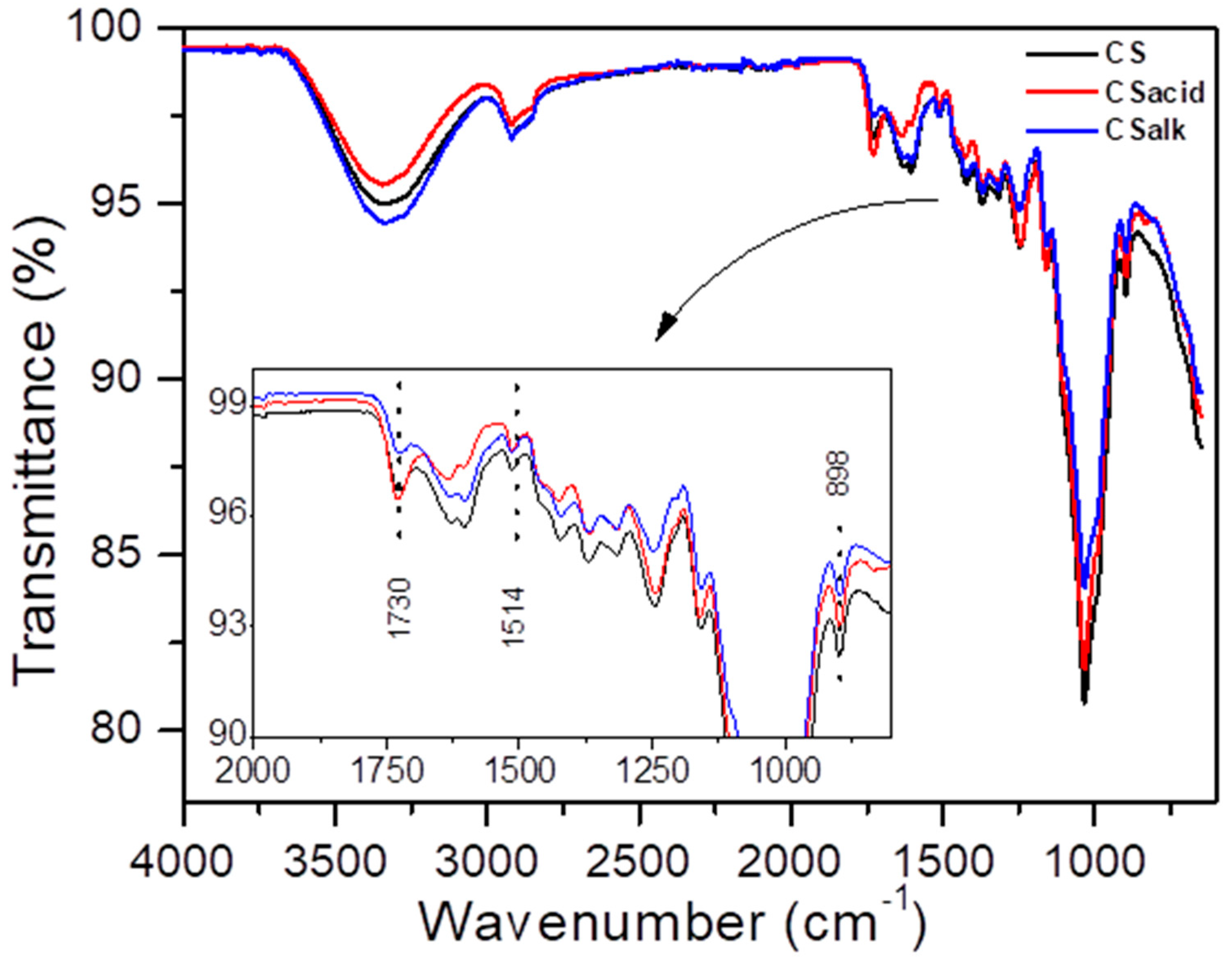
3.1.1. ATR-FTIR
3.1.2. SEM-EDS
3.1.3. Zeta Potential
3.2. Dyes Removal Test
3.3. Characterization of Dyed Corn Straw Fibers
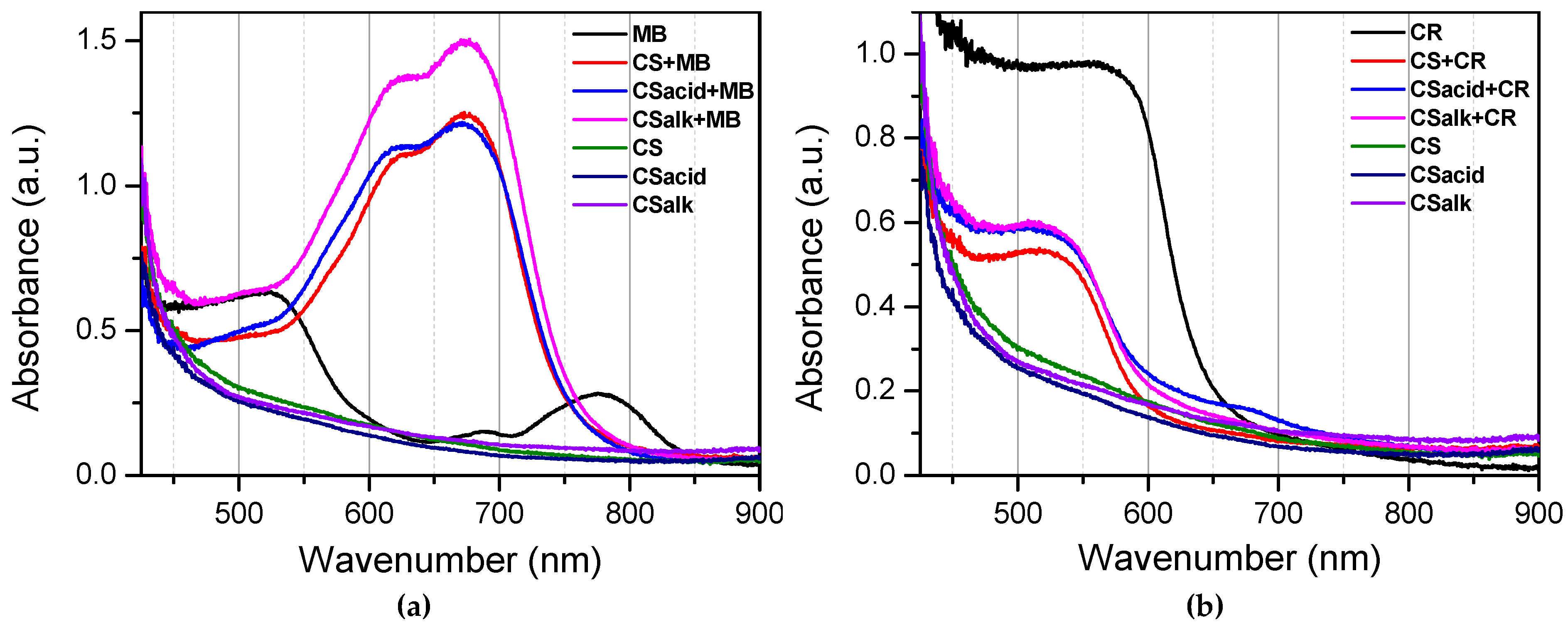
3.3.1. Diffuse Spectra
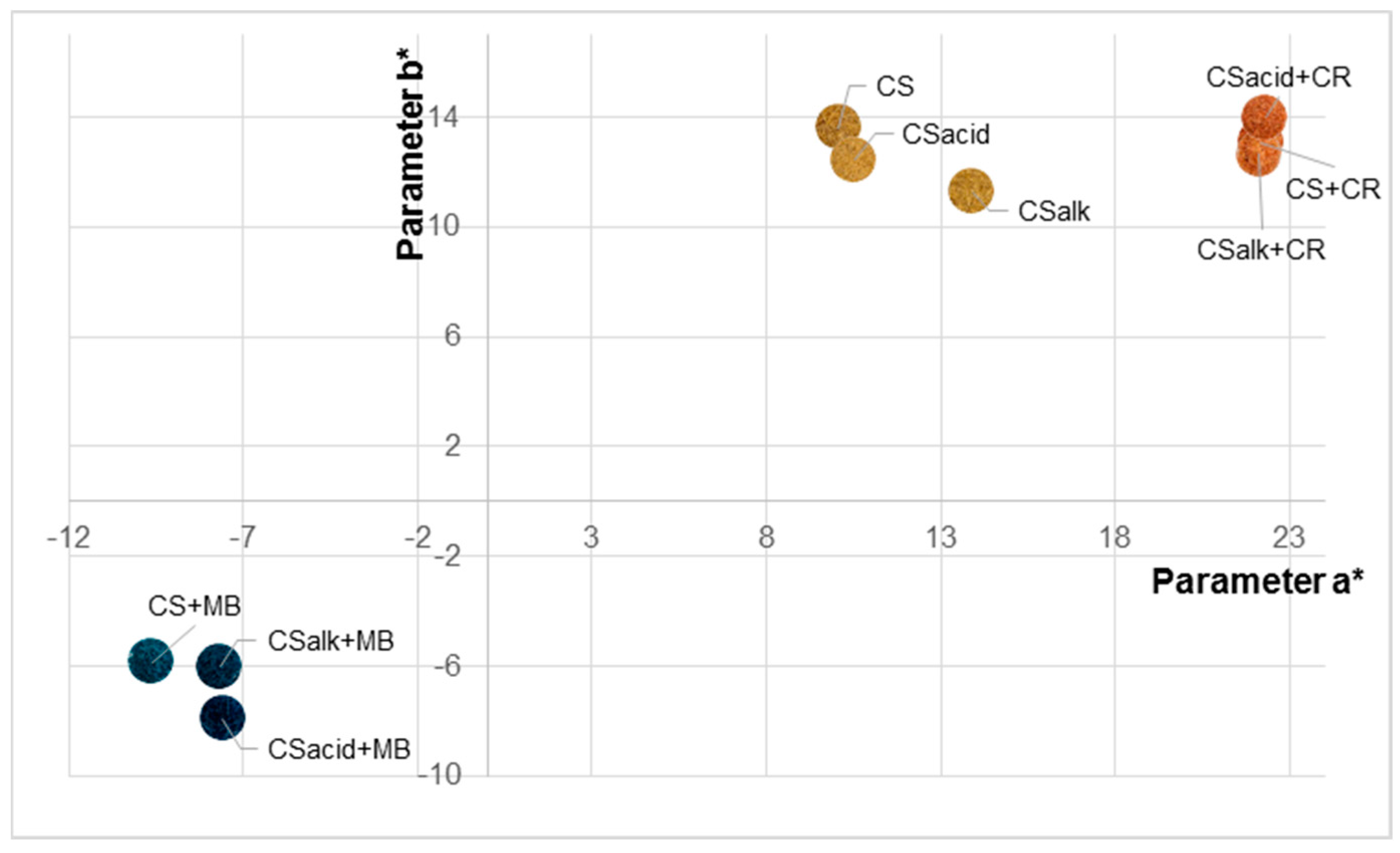
3.3.2. Colorimetry
3.3.3. Chemical Color Stability
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Corn Industry Worldwide—Statistics & Facts. Available online: https://www.statista.com/topics/7169/corn-industry-worldwide/ (accessed on 3 January 2024).
- Shibukawa, V.P.; Reis, C.E.R.; dos Santos, J.C.; Da Rós, P.C.M. Utilization of Co-Products from Corn Ethanol Industry in a Biorefinery Context: A Review on the Biotechnological Potential of Thin Stillage. Braz. J. Chem. Eng. 2023, 17. [Google Scholar] [CrossRef]
- Plantio Direto a Palhada Do Bem. Available online: https://www.embrapa.br/contando-ciencia/agricultura/-/asset_publisher/FcDEMJIbvFle/content/plantio-direto-a-plahada-do-bem/1355746?inheritRedirect=false#:~:text=O sistema de plantio direto,com isso%2C o aquecimento global (accessed on 24 January 2024).
- de Barcellos Ferreira, A.C.; Borin, A.L.D.C.; Lamas, F.M.; Sofiatti, V. Cover Plants in Second Crop: Nutrients in Straw and Cotton Yield in Succession. Pesqui. Agropecuária Trop. 2023, 53, e75032. [Google Scholar] [CrossRef]
- Lange, A.; Cabezas, W.A.R.L.; Trivelin, P.C.O. Produtividade de Palha e de Milho No Sistema Semeadura Direta, Em Função Da Época Da Aplicação Do Nitrogênio No Milho. Rev. Bras. Milho Sorgo 2009, 8, 57–68. [Google Scholar] [CrossRef]
- Crespo, A.M.; Souza, M.N.; Favarato, L.F.; Guarçoni, R.C.; Araújo, J.B.S.; Rangel, O.J.P.; de Souza, J.L.; da Costa Gonçalves, D. The Green Corn Development and Yield on Different Summer Soil Covering Plants in the Organic No-Tillage System. Int. J. Adv. Eng. Res. Sci. 2022, 9, 217–225. [Google Scholar] [CrossRef]
- Ruedell, J. Manejo Do Solo e Da Água Nos Sistemas de Produção Da Atualidade e Do Futuro. Rev. Plantio Direto Tecnol. Agrícola 2017, 155, 36–48. [Google Scholar]
- No Plantio Direto o Milho é o Melhor. Available online: https://revistacultivar.com.br/artigos/no-plantio-direto-o-milho-e-o-melhor (accessed on 23 January 2024).
- Resíduo Da Colheita de Cana, Palha Protege o Solo e Tem Alto Potencial Gerador de Bioenergia. Available online: https://cnpem.br/residuo-da-colheita-de-cana-palha-protege-o-solo-e-tem-alto-potencial-gerador-de-bioenergia-2/ (accessed on 2 February 2024).
- Değermenci, G.D.; Değermenci, N.; Ayvaoğlu, V.; Durmaz, E.; Çakır, D.; Akan, E. Adsorption of Reactive Dyes on Lignocellulosic Waste; Characterization, Equilibrium, Kinetic and Thermodynamic Studies. J. Clean. Prod. 2019, 225, 1220–1229. [Google Scholar] [CrossRef]
- Ma, P.; Yao, S.; Wang, Z.; Qi, F.; Liu, X. Preparation of Nitrogen-Doped Hierarchical Porous Carbon Aerogels from Agricultural Wastes for Efficient Pollution Adsorption. Sep. Purif. Technol. 2023, 311, 123250. [Google Scholar] [CrossRef]
- Liu, Z.; Li, L.; Liu, C.; Xu, A. Pretreatment of Corn Straw Using the Alkaline Solution of Ionic Liquids. Bioresour. Technol. 2018, 260, 417–420. [Google Scholar] [CrossRef]
- Monlau, F.; Barakat, A.; Trably, E.; Dumas, C.; Steyer, J.; Carrère, H. Lignocellulosic Materials Into Biohydrogen and Biomethane: Impact of Structural Features and Pretreatment. Crit. Rev. Environ. Sci. Technol. 2013, 43, 260–322. [Google Scholar] [CrossRef]
- Hendriks, A.T.W.M.; Zeeman, G. Pretreatments to Enhance the Digestibility of Lignocellulosic Biomass. Bioresour. Technol. 2009, 100, 10–18. [Google Scholar] [CrossRef]
- Mosbah, A.; Chouchane, H.; Abdelwahed, S.; Redissi, A.; Hamdi, M.; Kouidhi, S.; Neifar, M.; Slaheddine Masmoudi, A.; Cherif, A.; Mnif, W. Peptides Fixing Industrial Textile Dyes: A New Biochemical Method in Wastewater Treatment. J. Chem. 2019, 2019, 5081807. [Google Scholar] [CrossRef]
- dos Santos, A.; Viante, M.F.; dos Anjos, P.P.; Naidek, N.; Moises, M.P.; de Castro, E.G.; Downs, A.J.; Almeida, C.A.P. Removal of Astrazon Blue Dye from Aqueous Media by a Low-Cost Adsorbent from Coal Mining. Desalin. Water Treat. 2016, 57, 27213–27225. [Google Scholar] [CrossRef]
- Ranjbari, S.; Ayati, A.; Niknam Shahrak, M.; Tanhaei, B.; Hamidi Tabrizi, S. Design of [BmIm]3PW12O40 Ionic Liquid Encapsulated-ZIF-8 Nanocomposite for Cationic Dye Adsorptive Removal: Modeling by Response Surface Methodology. Ind. Eng. Chem. Res. 2023, 62, 4636–4645. [Google Scholar] [CrossRef]
- Trentini, J.D.; Jaerger, S.; Balaba, N.; Alves, H.J.; Wypych, F.; Anaissi, F.J. Adsorptive Removal of Congo Red by Macroporous ZnO Obtained from Citrus Pectin Gelation and Reuse as a Hybrid Pigment. Part. Sci. Technol. 2023, 41, 120–130. [Google Scholar] [CrossRef]
- Favaro, S.L.; Ganzerli, T.A.; de Carvalho Neto, A.G.V.; da Silva, O.R.R.F.; Radovanovic, E. Chemical, Morphological and Mechanical Analysis of Sisal Fiber-Reinforced Recycled High-Density Polyethylene Composites. Express Polym. Lett. 2010, 4, 465–473. [Google Scholar] [CrossRef]
- dos Santos, F.D.D.; Batistela, V.R.; dos Santos, A.; Halison de Oliveira, J.; Radovanovic, E.; Granzotto, D.C.T.; Fávaro, S.L. Hybrid Polyurethane/Natural Fibers Composites Optimized by Simplex-Centroid Mixture Design. J. Compos. Mater. 2022, 56, 1039–1052. [Google Scholar] [CrossRef]
- Laverde, V.; Marin, A.; Benjumea, J.M.; Rincón Ortiz, M. Use of Vegetable Fibers as Reinforcements in Cement-Matrix Composite Materials: A Review. Constr. Build. Mater. 2022, 340, 127729. [Google Scholar] [CrossRef]
- Li, V.C. From Micromechanics to Structural Engineering—The Design of Cementitious Composites for Civil Engineering Applications—Review. Jpn. Socienty Civ. Eng. 1993, 10, 37s–48s. [Google Scholar]
- The 17 Goals—Sustainable Development. Available online: https://sdgs.un.org/goals (accessed on 20 February 2024).
- Gunam, I.B.W.; Setiyo, Y.; Antara, N.S.; Wijaya, I.M.M.; Arnata, I.W.; Putra, I.W.W.P. Enhanced Delignification of Corn Straw with Alkaline Pretreatment at Mild Temperature. Rasayan J. Chem. 2020, 13, 1022–1029. [Google Scholar] [CrossRef]
- Chen, D.; Gao, D.; Huang, S.; Capareda, S.C.; Liu, X.; Wang, Y.; Zhang, T.; Liu, Y.; Niu, W. Influence of Acid-Washed Pretreatment on the Pyrolysis of Corn Straw: A Study on Characteristics, Kinetics and Bio-Oil Composition. J. Anal. Appl. Pyrolysis 2021, 155, 105027. [Google Scholar] [CrossRef]
- YANG, C.; LU, X.; LIN, W.; YANG, X.; YAO, J. TG-FTIR Study on Corn Straw Pyrolysis-Influence of Minerals. Chem. Res. Chinese Univ. 2006, 22, 524–532. [Google Scholar] [CrossRef]
- Song, Z.; Yang, G.; Liu, X.; Yan, Z.; Yuan, Y.; Liao, Y. Comparison of Seven Chemical Pretreatments of Corn Straw for Improving Methane Yield by Anaerobic Digestion. PLoS ONE 2014, 9, e101617. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.; Sun, X.; Sun, R. Chemical, Structural, and Thermal Characterizations of Alkali-Soluble Lignins and Hemicelluloses, and Cellulose from Maize Stems, Rye Straw, and Rice Straw. Polym. Degrad. Stab. 2001, 74, 307–319. [Google Scholar] [CrossRef]
- Li, C.; Knierim, B.; Manisseri, C.; Arora, R.; Scheller, H.V.; Auer, M.; Vogel, K.P.; Simmons, B.A.; Singh, S. Comparison of Dilute Acid and Ionic Liquid Pretreatment of Switchgrass: Biomass Recalcitrance, Delignification and Enzymatic Saccharification. Bioresour. Technol. 2010, 101, 4900–4906. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Wang, C.; Liu, S.; Huang, Z. Synthesis of Citric Acid Functionalized Magnetic Graphene Oxide Coated Corn Straw for Methylene Blue Adsorption. Bioresour. Technol. 2016, 221, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Guha, N.; Krishnan, S.; Singh, A.K.; Mathur, P.; Rai, D.K. Selective and Recyclable Congo Red Dye Adsorption by Spherical Fe3O4 Nanoparticles Functionalized with 1,2,4,5-Benzenetetracarboxylic Acid. Sci. Rep. 2020, 10, 111. [Google Scholar] [CrossRef] [PubMed]
- Meenakshisundaram, S.; Calcagno, V.; Ceballos, C.; Fayeulle, A.; Léonard, E.; Herledan, V.; Krafft, J.-M.; Millot, Y.; Liu, X.; Jolivalt, C.; et al. Chemically and Physically Pretreated Straw in Moderate Conditions: Poor Correlation between Biogas Production and Commonly Used Biomass Characterization. Energies 2023, 16, 1146. [Google Scholar] [CrossRef]
- Nunes, C.S.; de Souza Castillo, A.; Ramos, J.P.; Gomes, L.G.; Cardoso, F.J.B.; do Socorro de Souza Vilhena, K. Potencial de Resíduo Não Processado Proveniente Da Obtenção Do Silício Metálico Como Adsorvente Do Corante Vermelho de Metila. Matéria (Rio Janeiro) 2023, 28. [Google Scholar] [CrossRef]
- Almeida, C.A.P.; Machado, C.; Debacher, N.A. Adsorption of Methylene Blue as a Model for the Use of Barro Branco as an Alternative Adsorbent for Color Removal. Surf. Colloid Sci. 2004, 278–282. [Google Scholar] [CrossRef]
- Hu, Z.; Chen, H.; Ji, F.; Yuan, S. Removal of Congo Red from Aqueous Solution by Cattail Root. J. Hazard. Mater. 2010, 173, 292–297. [Google Scholar] [CrossRef]
- Wang, G.; Chen, H. Fractionation of Alkali-Extracted Lignin from Steam-Exploded Stalk by Gradient Acid Precipitation. Sep. Purif. Technol. 2013, 105, 98–105. [Google Scholar] [CrossRef]
- Wen, J.; Xue, Z.; Yin, X.; Wang, X. Insights into Aqueous Reduction of Cr(VI) by Biochar and Its Iron-Modified Counterpart in the Presence of Organic Acids. Chemosphere 2022, 286, 131918. [Google Scholar] [CrossRef]
- Balbina, F.T.C.d.S. Fenômeno de Modulação Termo-Óptica Do Corante Azul de Metileno Utilizado Em Terapia Fotodinâmica. Ph.D. Thesis, Universidade Anhembi Morumbi, São Paulo, Brazil, 2023. [Google Scholar]
- Toma, H.E.; da Silva Bonifácio, L.; Anaissi, F.J. Da Cor à Cor Inexistente: Uma Reflexão Sobre Espectros Eletrônicos e Efeitos Cromáticos. Quim. Nova 2005, 28, 897–900. [Google Scholar] [CrossRef][Green Version]
- Fernández-Pérez, A.; Marbán, G. Visible Light Spectroscopic Analysis of Methylene Blue in Water; What Comes after Dimer? ACS Omega 2020, 5, 29801–29815. [Google Scholar] [CrossRef]
- The Dimensions of Colours. Available online: http://www.huevaluechroma.com/045.php (accessed on 24 January 2024).
- Hirschler, R. Electronic Colour Communication in the Textile and Apparel Industry. Redige Rev. Des. Innov. Strateg. Manag. 2010, 1, 43–61. [Google Scholar]
- Horsth, D.F.L.; Primo, J.O.; Dalpasquale, M.; Bittencourt, C.; Anaissi, F.J. Colored Aluminates Pigments Obtained from Metallic Aluminum Waste, an Opportunity in the Circular Economy. Clean. Eng. Technol. 2021, 5, 100313. [Google Scholar] [CrossRef]
- Debrassi, A.; Largura, M.C.T.; Rodrigues, C.A. Adsorção Do Corante Vermelho Congo Por Derivados Da O-Carboximetilquitosana Hidrofobicamente Modificados. Quim. Nova 2011, 34, 764–770. [Google Scholar] [CrossRef]
- de Oliveira, B.; Moriyama, L.; Bagnato, V. Colorimetric Analysis of Cotton Textile Bleaching through H2O2 Activated by UV Light. J. Braz. Chem. Soc. 2017, 29, 1360–1365. [Google Scholar] [CrossRef]
- Beg, M.D.H.; Pickering, K.L.; Gauss, C. The Effects of Alkaline Digestion, Bleaching and Ultrasonication Treatment of Fibre on 3D Printed Harakeke Fibre Reinforced Polylactic Acid Composites. Compos. Part A Appl. Sci. Manuf. 2023, 166, 107384. [Google Scholar] [CrossRef]




| Elements | Sample | ||
|---|---|---|---|
| CS (%) | CSacid (%) | CSalk (%) | |
| O | 55.59 (±0.28) | 49.99 (±0.14) | 54.47 (±0.31) |
| C | 41.43 (±0.29) | 49.87 (±0.14) | 43.88 (±0.32) |
| Mg | 1.11 (±0.03) | -- | 0.29 (±0.02) |
| K | 0.64 (±0.01) | -- | 0.24 (±0.01) |
| Ca | 1.23 (±0.02) | -- | 0.25 (±0.01) |
| Si | -- | 0.14 (±0.01) | 0.30 (±0.02) |
| Na | -- | -- | 0.57 (±0.04) |
| Sample | Methylene Blue | Congo Red | Amount Removed Ration MB/CR (qe/qe) | ||
|---|---|---|---|---|---|
| Removal (%) | qe (mg g−1) | Removal (%) | qe (mg g−1) | ||
| CS | 93.5 (±0.8) | 10.8 (±0.2) | 44.2 (±1.2) | 1.37 (±0.2) | 7.88 |
| CSacid | 85.2 (±3.0) | 9.80 (±0.7) | 77.2 (±0.5) | 2.13 (±0.1) | 4.60 |
| CSalk | 99.2 (±0.2) | 16.7 (±0.1) | 33.9 (±3.2) | 1.05 (±0.2) | 15.9 |
| Sample | Colorimetric Parameters * | Photo | |||||
|---|---|---|---|---|---|---|---|
| L* | a* | b* | c | h | ΔE | ||
| CSalk+MB | 13.32 | −7.65 | −6.02 | 9.74 | 248.20 | - |  |
| CSalk+MB/natural water | 14.93 | −8.78 | −6.28 | 10.31 | 217.52 | 3.61 |  |
| CSalk+MB/acetic acid | 16.01 | −9.66 | −5.53 | 11.13 | 209.73 | 3.39 |  |
| CSalk+MB/sodium bicarbonate | 25.01 | −11.88 | 10.03 | 15.55 | 139.83 | 5.90 |  |
| CSacid+CR | 70.31 | 22.32 | 13.98 | 26.34 | 32.05 | - |  |
| CSacid+CR/natural water | 79.02 | 10.48 | 22.42 | 24.75 | 64.96 | 16.95 |  |
| CSacid+CR/acetic acid | 78.42 | 14.28 | 24.48 | 28.34 | 59.75 | 15.51 |  |
| CSacid+CR/sodium bicarbonate | 81.67 | 19.31 | 25.75 | 27.38 | 70.13 | 16.63 |  |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
dos Santos, A.; Sotiles, A.R.; Anaissi, F.J. Application of Corn Straw, an Agro-Waste, to Remove Dyes in an Aqueous Medium, Producing Blue or Red Fibers. Processes 2024, 12, 694. https://doi.org/10.3390/pr12040694
dos Santos A, Sotiles AR, Anaissi FJ. Application of Corn Straw, an Agro-Waste, to Remove Dyes in an Aqueous Medium, Producing Blue or Red Fibers. Processes. 2024; 12(4):694. https://doi.org/10.3390/pr12040694
Chicago/Turabian Styledos Santos, Andressa, Anne R. Sotiles, and Fauze J. Anaissi. 2024. "Application of Corn Straw, an Agro-Waste, to Remove Dyes in an Aqueous Medium, Producing Blue or Red Fibers" Processes 12, no. 4: 694. https://doi.org/10.3390/pr12040694
APA Styledos Santos, A., Sotiles, A. R., & Anaissi, F. J. (2024). Application of Corn Straw, an Agro-Waste, to Remove Dyes in an Aqueous Medium, Producing Blue or Red Fibers. Processes, 12(4), 694. https://doi.org/10.3390/pr12040694








