Regenerable Kiwi Peels as an Adsorbent to Remove and Reuse the Emerging Pollutant Propranolol from Water
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of the Adsorbent
2.3. UV-Visible Analyses
2.4. ATR-FTIR Spectroscopy Analyses
2.5. In-Batch Adsorption Experiments
2.6. Kinetics of Adsorption
2.7. Thermodynamic Analysis
2.8. Isotherms of Adsorption
2.9. In-Batch Mode Experiments of Desorption
2.10. Zero-Point Charge Determination of Kiwi Peels’ Surfaces
3. Results and Discussion
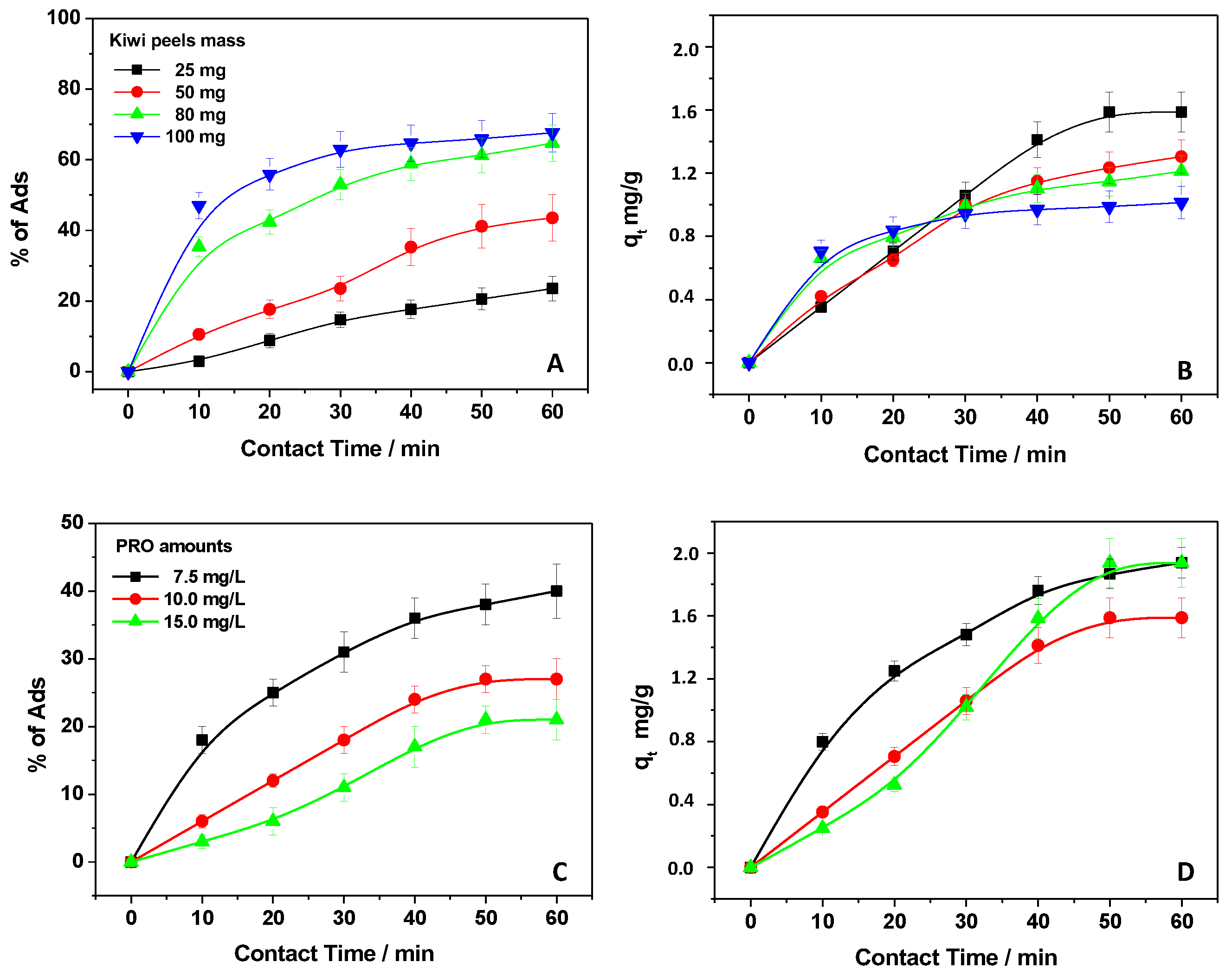
3.1. Roles of PRO and Kiwi Peel Amounts in the Adsorption Process
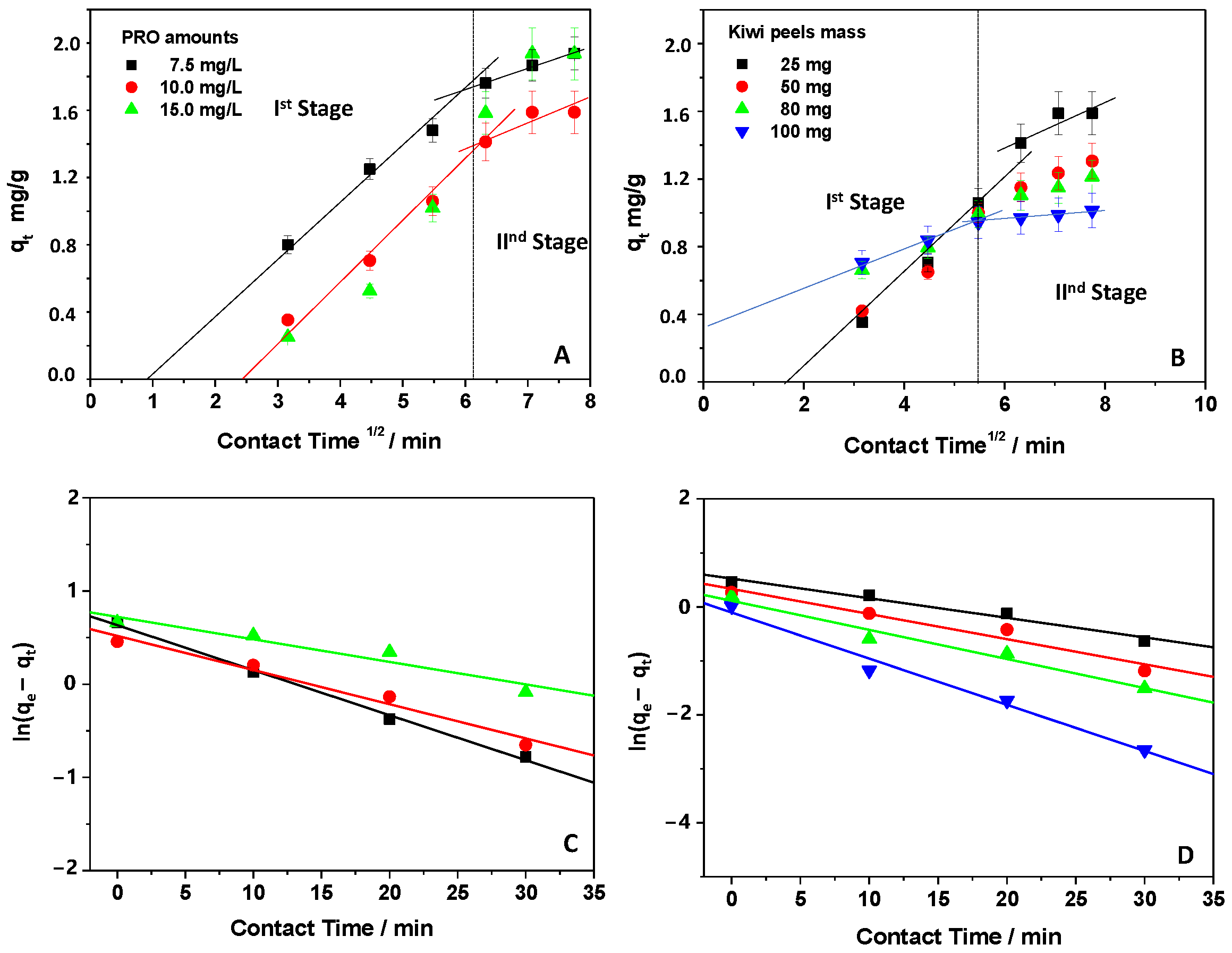
3.2. Kinetic Analysis
3.3. Thermodynamic Analysis
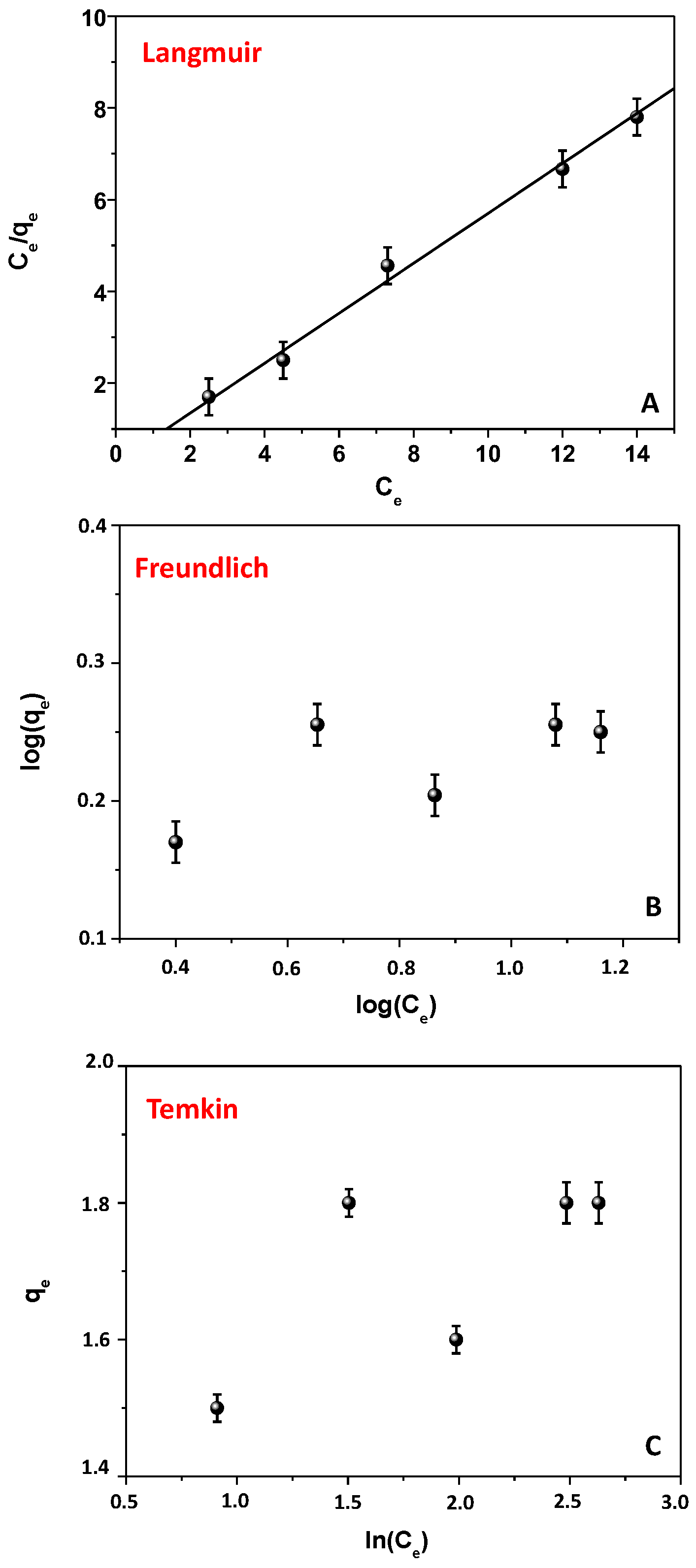
3.4. Isotherms of Adsorption
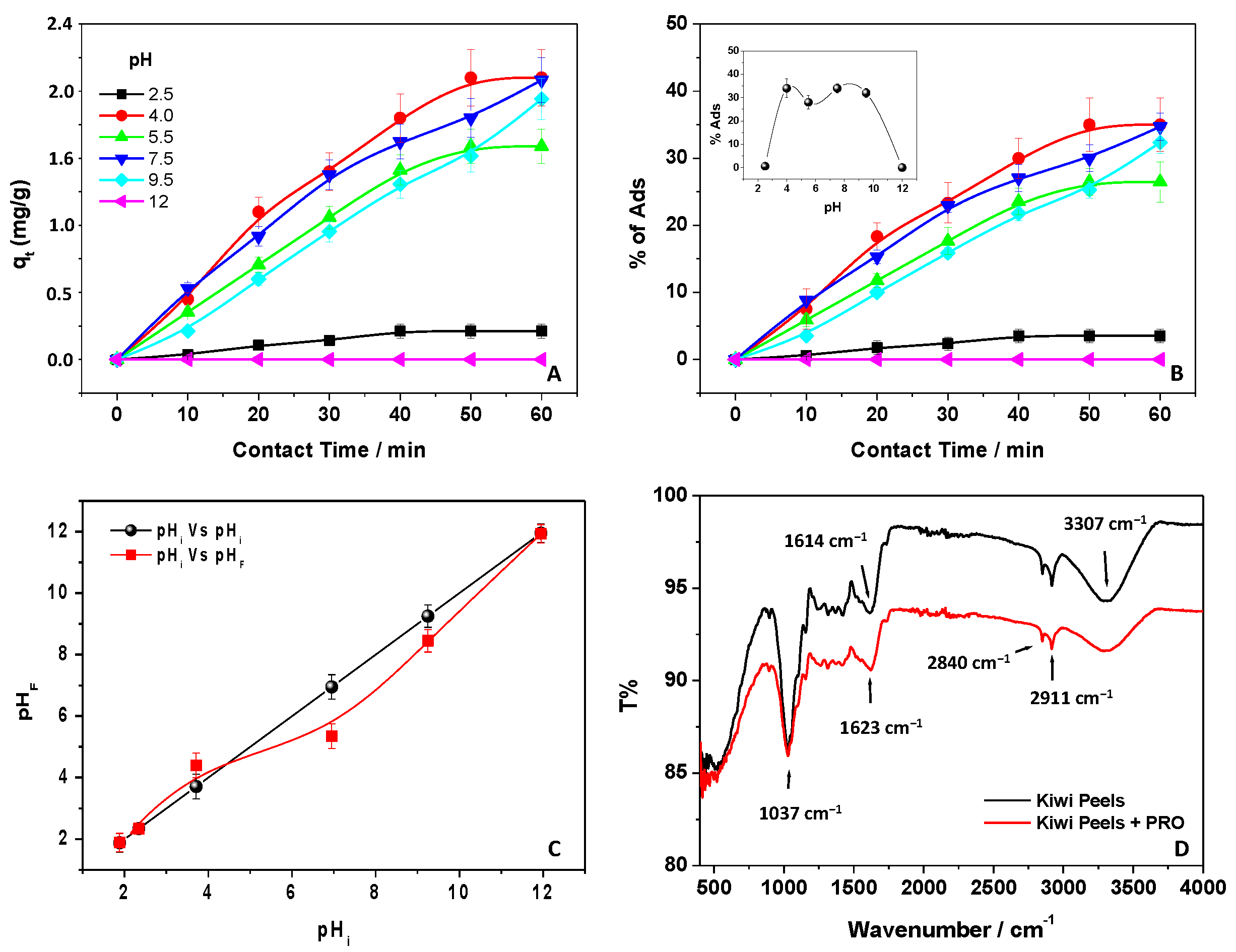
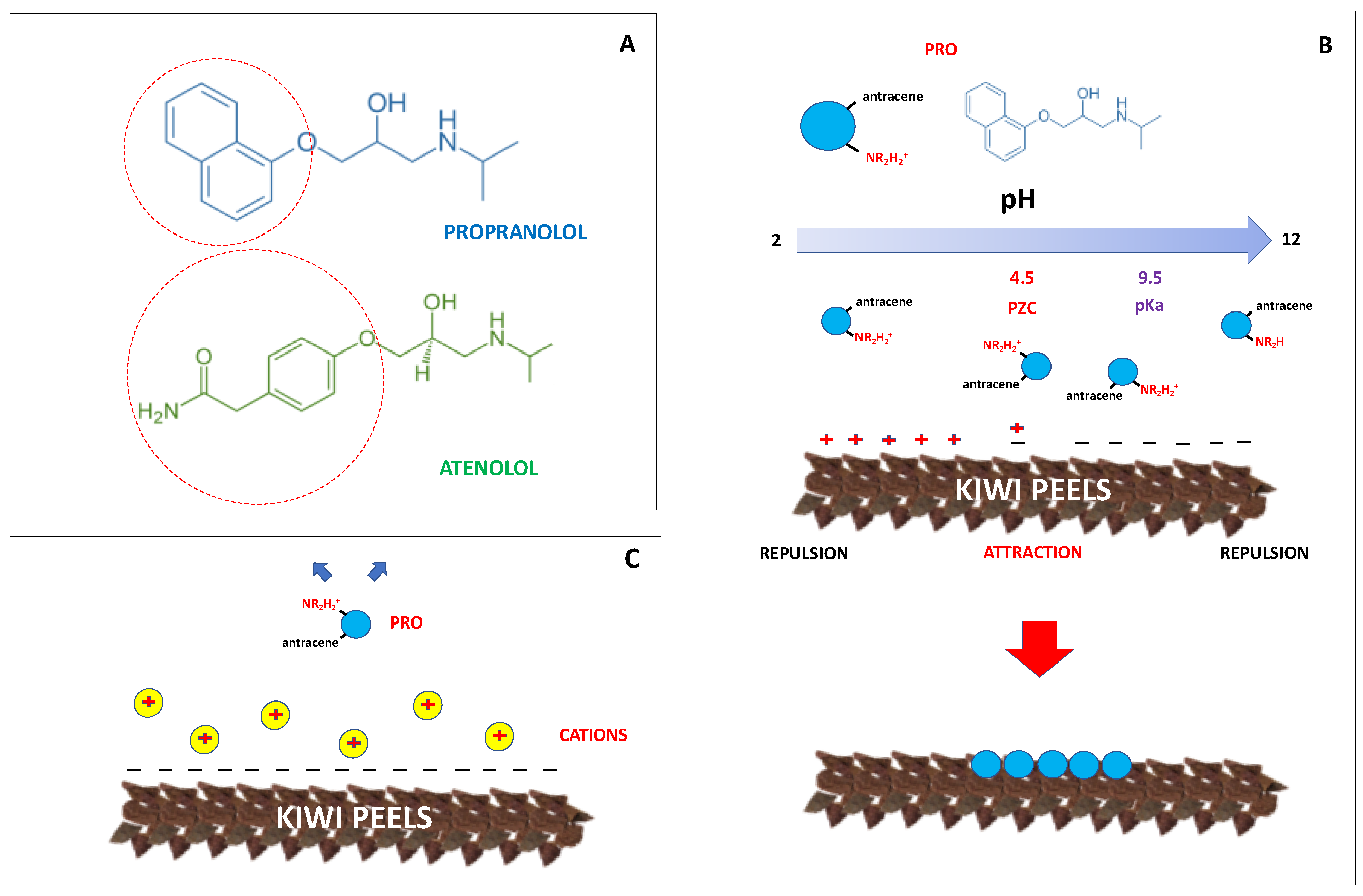
3.5. Effect of pH
3.6. FTIR-ATR Measurements
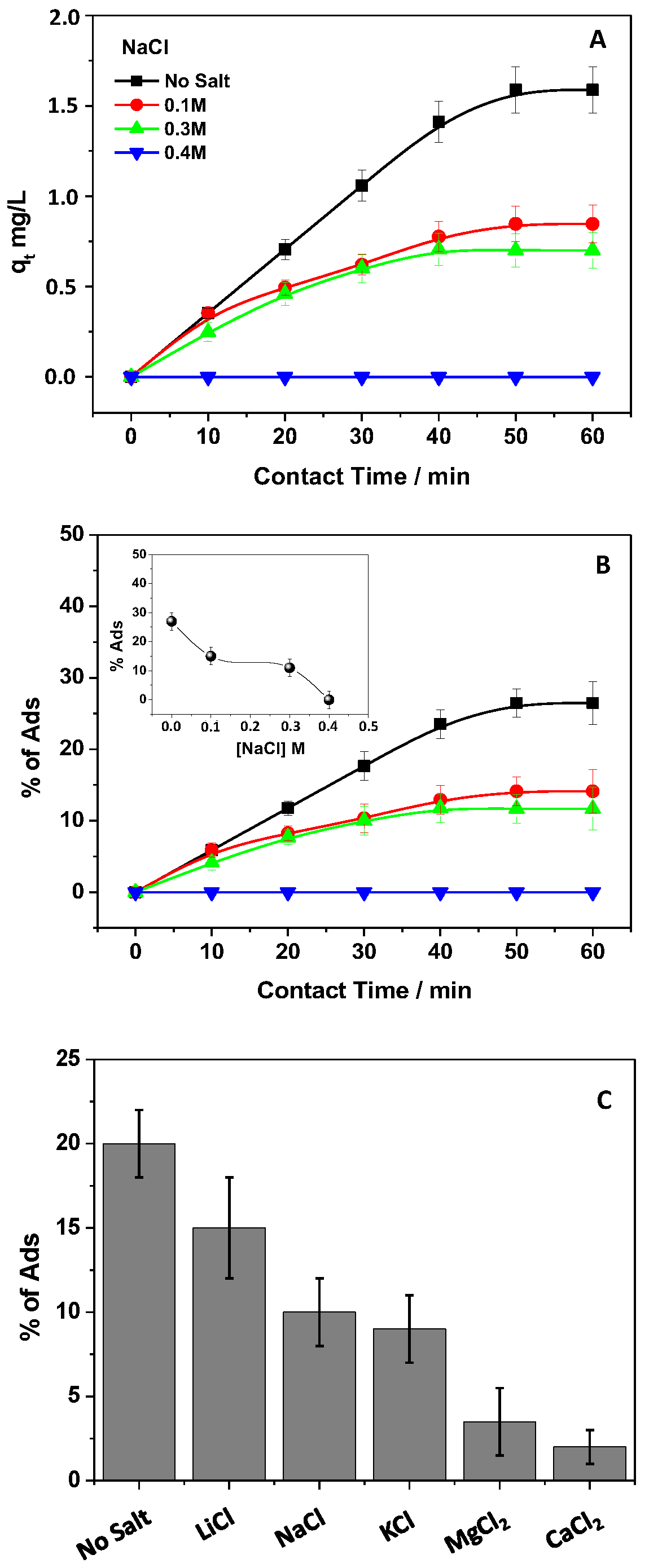
3.7. Effect of Salts in PRO Solutions
3.8. Desorption of PRO and Adsorbent Recycling
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liakos, E.V.; Rekos, K.; Giannakoudakis, D.A.; Mitropoulos, A.C.; Fu, J.; Kyzas, G.Z. Activated Porous Carbon Derived from Tea and Plane Tree Leaves Biomass for the Removal of Pharmaceutical Compounds from Wastewaters. Antibiotics 2021, 10, 65. [Google Scholar] [CrossRef] [PubMed]
- Petrella, A.; Spasiano, D.; Cosma, P.; Rizzi, V.; Race, M.; Mascolo, M.C.; Ranieri, E. Methyl orange photo-degradation by TiO2 in a pilot unit under different chemical, physical, and hydraulic conditions. Processes 2021, 9, 205. [Google Scholar] [CrossRef]
- Rizzi, V.; Gubitosa, J.; Fini, P.; Nuzzo, S.; Cosma, P. Amino-grafted mesoporous MCM-41 and SBA-15 recyclable adsorbents: Desert-rose-petals-like SBA-15 type as the most efficient to remove azo textile dyes and their mixture from water. Sustain. Mater. Technol. 2020, 26, e00231. [Google Scholar] [CrossRef]
- Petrella, A.; Spasiano, D.; Rizzi, V.; Cosma, P.; Race, M.; De Vietro, N. Thermodynamic and kinetic investigation of heavy metals sorption in packed bed columns by recycled lignocellulosic materials from olive oil production. Chem. Eng. Commun. 2019, 206, 1715–1730. [Google Scholar] [CrossRef]
- Rizzi, V.; Prasetyanto, E.A.; Chen, P.; Gubitosa, J.; Fini, P.; Agostiano, A.; De Cola, L.; Cosma, P. Amino grafted MCM-41 as highly efficient and reversible ecofriendly adsorbent material for the Direct Blue removal from wastewater. J. Mol. Liq. 2019, 273, 435–446. [Google Scholar] [CrossRef]
- Rizzi, V.; Fiorini, F.; Lamanna, G.; Gubitosa, J.; Prasetyanto, E.A.; Fini, P.; Fanelli, F.; Nacci, A.; De Cola, L.; Cosma, P. Polyamidoamine-Based Hydrogel for Removal of Blue and Red Dyes from Wastewater. Adv. Sustain. Syst. 2018, 2, 1700146. [Google Scholar] [CrossRef]
- Rizzi, V.; Longo, A.; Placido, T.; Fini, P.; Gubitosa, J.; Sibillano, T.; Giannini, C.; Semeraro, P.; Franco, E.; Ferrandiz, M.; et al. A comprehensive investigation of dye–chitosan blended films for green chemistry applications. J. Appl. Polym. Sci. 2018, 135, 45945. [Google Scholar] [CrossRef]
- Rizzi, V.; D’Agostino, F.; Gubitosa, J.; Fini, P.; Petrella, A.; Agostiano, A.; Semeraro, P.; Cosma, P. An Alternative Use of Olive Pomace as a Wide-Ranging Bioremediation Strategy to Adsorb and Recover Disperse Orange and Disperse Red Industrial Dyes from Wastewater. Separations 2017, 4, 29. [Google Scholar] [CrossRef] [Green Version]
- Rizzi, V.; D’Agostino, F.; Fini, P.; Semeraro, P.; Cosma, P. An interesting environmental friendly cleanup: The excellent potential of olive pomace for disperse blue adsorption/desorption from wastewater. Dyes Pigment. 2017, 140, 480–490. [Google Scholar] [CrossRef]
- Rizzi, V.; Gubitosa, J.; Signorile, R.; Fini, P.; Cecone, C.; Matencio, A.; Trotta, F.; Cosma, P. Cyclodextrin nanosponges as adsorbent material to remove hazardous pollutants from water: The case of ciprofloxacin. Chem. Eng. J. 2021, 411, 128514. [Google Scholar] [CrossRef]
- Rizzi, V.; Gubitosa, J.; Fini, P.; Romita, R.; Nuzzo, S.; Gabaldón, J.A.; Gorbe, M.I.F.; Gómez-Morte, T.; Cosma, P. Chitosan film as recyclable adsorbent membrane to remove/recover hazardous pharmaceutical pollutants from water: The case of the emerging pollutant Furosemide. J. Environ. Sci. Health Part A 2021, 56, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Rizzi, V.; Gubitosa, J.; Fini, P.; Romita, R.; Agostiano, A.; Nuzzo, S.; Cosma, P. Commercial bentonite clay as low-cost and recyclable “natural” adsorbent for the Carbendazim removal/recover from water: Overview on the adsorption process and preliminary photodegradation considerations. Colloids Surf. A Physicochem. Eng. Asp. 2020, 602, 125060. [Google Scholar] [CrossRef]
- Rizzi, V.; Gubitosa, J.; Fini, P.; Petrella, A.; Romita, R.; Agostiano, A.; Cosma, P. A “classic” material for capture and detoxification of emergent contaminants for water purification: The case of tetracycline. Environ. Technol. Innov. 2020, 19, 100812. [Google Scholar] [CrossRef]
- Rizzi, V.; Gubitosa, J.; Fini, P.; Romita, R.; Nuzzo, S.; Cosma, P. Chitosan Biopolymer from Crab Shell as Recyclable Film to Remove/Recover in Batch Ketoprofen from Water: Understanding the Factors Affecting the Adsorption Process. Materials 2019, 12, 3810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rizzi, V.; Romanazzi, F.; Gubitosa, J.; Fini, P.; Romita, R.; Agostiano, A.; Petrella, A.; Cosma, P. Chitosan Film as Eco-Friendly and Recyclable Bio-Adsorbent to Remove/Recover Diclofenac, Ketoprofen, and their Mixture from Wastewater. Biomolecules 2019, 9, 571. [Google Scholar] [CrossRef] [Green Version]
- Romita, R.; Rizzi, V.; Semeraro, P.; Gubitosa, J.; Gabaldón, J.A.; Gorbe, M.I.F.; López, V.M.G.; Cosma, P.; Fini, P. Operational parameters affecting the atrazine removal from water by using cyclodextrin based polymers as efficient adsorbents for cleaner technologies. Environ. Technol. Innov. 2019, 16, 100454. [Google Scholar] [CrossRef]
- Rizzi, V.; Lacalamita, D.; Gubitosa, J.; Fini, P.; Petrella, A.; Romita, R.; Agostiano, A.; Gabaldón, J.A.J.A.; Fortea Gorbe, M.I.M.I.; Gómez-Morte, T.; et al. Removal of tetracycline from polluted water by chitosan-olive pomace adsorbing films. Sci. Total Environ. 2019, 693, 133620. [Google Scholar] [CrossRef]
- Qureshi, U.A.; Hameed, B.H.; Ahmed, M.J. Adsorption of endocrine disrupting compounds and other emerging contaminants using lignocellulosic biomass-derived porous carbons: A review. J. Water Process Eng. 2020, 38, 101380. [Google Scholar] [CrossRef]
- Delgado-Moreno, L.; Bazhari, S.; Gasco, G.; Méndez, A.; El Azzouzi, M.; Romero, E. New insights into the efficient removal of emerging contaminants by biochars and hydrochars derived from olive oil wastes. Sci. Total Environ. 2021, 752, 141838. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E. Advantages and disadvantages of techniques used for wastewater treatment. Environ. Chem. Lett. 2019, 17, 145–155. [Google Scholar] [CrossRef]
- Moosavi, S.; Lai, C.W.; Gan, S.; Zamiri, G.; Akbarzadeh Pivehzhani, O.; Johan, M.R. Application of Efficient Magnetic Particles and Activated Carbon for Dye Removal from Wastewater. ACS Omega 2020, 5, 20684–20697. [Google Scholar] [CrossRef] [PubMed]
- Afolabi, I.C.; Popoola, S.I.; Bello, O.S. Modeling pseudo-second-order kinetics of orange peel-paracetamol adsorption process using artificial neural network. Chemom. Intell. Lab. Syst. 2020, 203, 104053. [Google Scholar] [CrossRef]
- Khan, T.; Mustafa, M.R.U.; Isa, M.H.; Manan, T.S.B.A.; Ho, Y.-C.; Lim, J.-W.; Yusof, N.Z. Artificial Neural Network (ANN) for Modelling Adsorption of Lead (Pb (II)) from Aqueous Solution. Water Air Soil Pollut. 2017, 228, 426. [Google Scholar] [CrossRef]
- Suzaimi, N.D.; Goh, P.S.; Malek, N.A.N.N.; Lim, J.W.; Ismail, A.F. Performance of branched polyethyleneimine grafted porous rice husk silica in treating nitrate-rich wastewater via adsorption. J. Environ. Chem. Eng. 2019, 7, 103235. [Google Scholar] [CrossRef]
- Suzaimi, N.D.; Goh, P.S.; Malek, N.A.N.N.; Lim, J.W.; Ismail, A.F. Enhancing the performance of porous rice husk silica through branched polyethyleneimine grafting for phosphate adsorption. Arab. J. Chem. 2020, 13, 6682–6695. [Google Scholar] [CrossRef]
- Isiyaka, H.A.; Jumbri, K.; Sambudi, N.S.; Lim, J.W.; Saad, B.; Ramli, A.; Zango, Z.U. Experimental and Modeling of Dicamba Adsorption in Aqueous Medium Using MIL-101(Cr) Metal-Organic Framework. Processes 2021, 9, 419. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Sillanpää, M.; Witek-Krowiak, A. Agricultural waste peels as versatile biomass for water purification—A review. Chem. Eng. J. 2015, 270, 244–271. [Google Scholar] [CrossRef]
- Xu, G.F.; Jing, H.M.; Guo, R.X. The Adsorption Isotherm Studies of Orange Peel on Pesticide Furadan. Appl. Mech. Mater. 2014, 477–478, 1331–1335. [Google Scholar] [CrossRef]
- Shuaibing, Z.M.Z.S.L.; Qihua, Z. Adsorption of dye wastewater by banana peel powder immobilized by sodium alginate. Chin. J. Environ. Eng. 2013, 7, 2208–2212. [Google Scholar]
- Aman, T.; Kazi, A.A.; Sabri, M.U.; Bano, Q. Potato peels as solid waste for the removal of heavy metal copper(II) from waste water/industrial effluent. Colloids Surf. B Biointerfaces 2008, 63, 116–121. [Google Scholar] [CrossRef]
- Gubitosa, J.; Rizzi, V.; Cignolo, D.; Fini, P.; Fanelli, F.; Cosma, P. From agricultural wastes to a resource: Kiwi Peels, as long-lasting, recyclable adsorbent, to remove emerging pollutants from water. The case of Ciprofloxacin removal. Sustain. Chem. Pharm. 2022, 29, 100749. [Google Scholar] [CrossRef]
- Sanz, V.; López-Hortas, L.; Torres, M.D.; Domínguez, H. Trends in kiwifruit and byproducts valorization. Trends Food Sci. Technol. 2021, 107, 401–414. [Google Scholar] [CrossRef]
- Al-Qahtani, K.M. Water purification using different waste fruit cortexes for the removal of heavy metals. J. Taibah Univ. Sci. 2016, 10, 700–708. [Google Scholar] [CrossRef] [Green Version]
- Mokif, L.A.; Abdulhusain, N.A.; AL-Mamoori, S.O.H. The Possibility of Using the kiwi Peels as an Adsorbent for Removing Nitrate from Water. J. Univ. Babylon Eng. Sci. 2018, 26, 192–197. [Google Scholar]
- Saleh Jafer, A.; Hassan, A.A. Removal of oil content in oilfield produced water using chemically modified kiwi peels as efficient low-cost adsorbent. J. Phys. Conf. Ser. 2019, 1294, 72013. [Google Scholar] [CrossRef] [Green Version]
- Rahimnejad, M.; Pirzadeh, K.; Mahdavi, I.; Peyghambarzadeh, S.M. Pb(II) removal from aqueous solution by adsorption on activated carbon from kiwi peel. Environ. Eng. Manag. J. 2018, 17, 1293–1300. [Google Scholar] [CrossRef]
- Ali, I.; Alothman, Z.A.; Alwarthan, A. Uptake of propranolol on ionic liquid iron nanocomposite adsorbent: Kinetic, thermodynamics and mechanism of adsorption. J. Mol. Liq. 2017, 236, 205–213. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Koltsakidou, A.; Nanaki, S.G.; Bikiaris, D.N.; Lambropoulou, D.A. Removal of beta-blockers from aqueous media by adsorption onto graphene oxide. Sci. Total Environ. 2015, 537, 411–420. [Google Scholar] [CrossRef]
- Barbieri, A.; Robinson, N.; Palma, G.; Maurea, N.; Desiderio, V.; Botti, G. Can Beta-2-Adrenergic Pathway Be a New Target to Combat SARS-CoV-2 Hyperinflammatory Syndrome?—Lessons Learned From Cancer. Front. Immunol. 2020, 11, 2615. [Google Scholar] [CrossRef]
- Colpani, G.L.; Santos, V.F.; Frezza Zeferino, R.C.; Zanetti, M.; Muneron de Mello, J.M.; Silva, L.L.; Padoin, N.; de Fátima Peralta Muniz Moreira, R.; Fiori, M.A.; Soares, C. Propranolol hydrochloride degradation using La@TiO2 functionalized with CMCD. J. Rare Earths 2021, 40, 579–585. [Google Scholar] [CrossRef]
- Deng, Y.; Wu, F.; Liu, B.; Hu, X.; Sun, C. Sorptive removal of β-blocker propranolol from aqueous solution by modified attapulgite: Effect factors and sorption mechanisms. Chem. Eng. J. 2011, 174, 571–578. [Google Scholar] [CrossRef]
- Deng, Y.; Li, Y. Surface-Bound Humic Acid Increased Propranolol Sorption on Fe3O4/Attapulgite Magnetic Nanoparticles. Nanomaterials 2020, 10, 205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deng, Y.; Li, Y.; Nie, W.; Gao, X.; Zhang, L.; Yang, P.; Tan, X. Fast Removal of Propranolol from Water by Attapulgite/Graphene Oxide Magnetic Ternary Composites. Materials 2019, 12, 924. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- del Mar Orta, M.; Martín, J.; Medina-Carrasco, S.; Santos, J.L.; Aparicio, I.; Alonso, E. Adsorption of propranolol onto montmorillonite: Kinetic, isotherm and pH studies. Appl. Clay Sci. 2019, 173, 107–114. [Google Scholar] [CrossRef]
- Nie, W.; Li, Y.; Chen, L.; Zhao, Z.; Zuo, X.; Wang, D.; Zhao, L.; Feng, X. Interaction between multi-walled carbon nanotubes and propranolol. Sci. Rep. 2020, 10, 10259. [Google Scholar] [CrossRef] [PubMed]
- Salvador, R.; Puglieri, F.N.; Halog, A.; de Andrade, F.G.; Piekarski, C.M.; De Francisco, A.C. Key aspects for designing business models for a circular bioeconomy. J. Clean. Prod. 2021, 278, 124341. [Google Scholar] [CrossRef]
- Stegmann, P.; Londo, M.; Junginger, M. The circular bioeconomy: Its elements and role in European bioeconomy clusters. Resour. Conserv. Recycl. X 2020, 6, 100029. [Google Scholar] [CrossRef]
- Yu, F.; Chen, D.; Ma, J. Adsorptive removal of ciprofloxacin by ethylene diaminetetraacetic acid/β-cyclodextrin composite from aqueous solution. New J. Chem. 2018, 42, 2216–2223. [Google Scholar] [CrossRef]
- Wang, Y.X.; Ngo, H.H.; Guo, W.S. Preparation of a specific bamboo based activated carbon and its application for ciprofloxacin removal. Sci. Total Environ. 2015, 533, 32–39. [Google Scholar] [CrossRef]
- Lu, D.; Xu, S.; Qiu, W.; Sun, Y.; Liu, X.; Yang, J.; Ma, J. Adsorption and desorption behaviors of antibiotic ciprofloxacin on functionalized spherical MCM-41 for water treatment. J. Clean. Prod. 2020, 264, 121644. [Google Scholar] [CrossRef]
- Yu, F.; Sun, S.; Han, S.; Zheng, J.; Ma, J. Adsorption removal of ciprofloxacin by multi-walled carbon nanotubes with different oxygen contents from aqueous solutions. Chem. Eng. J. 2016, 285, 588–595. [Google Scholar] [CrossRef]
- Hubbe, M.A.; Azizian, S.; Douven, S. Implications of apparent pseudo-second-order adsorption kinetics onto cellulosic materials: A review. BioResources 2019, 14, 7582–7626. [Google Scholar] [CrossRef]
- N’diaye, A.D.; Kankou, M.S. Modeling of adsorption isotherms of pharmaceutical products onto various adsorbents: A Short Review. J. Mater. Environ. Sci. 2020, 11, 1264–1276. [Google Scholar]



| Adsorbent | Maximum Adsorption Capacity qmax mg/g | Reference |
|---|---|---|
| Multi-walled carbon nanotubes | 54.17 | Nie et al. [45] 2020 |
| Fe3O4/attapulgite magnetic nanoparticles | 12.87–16.87 | Deng et al. [42] 2020 |
| Attapulgite/graphene oxide magnetic ternary composites | 46.8 | Deng et al. [43] 2019 |
| Montmorillonite | 25.9 | Del Mar Orta et al. [44] 2019 |
| Ionic liquid iron nanocomposite adsorbent | 0.1 | Ali et al. [37] 2017 |
| Graphene oxide | 67–77 | Kyzas et al. [38] 2015 |
| Modified attapulgite | 24.56–48.55 | Deng et al. [41] 2011 |
| Kiwi peels | 2 | This work |
| Concentration (PRO) mg/L | Pseudo-First-Order | Pseudo-Second-Order | ||||||
|---|---|---|---|---|---|---|---|---|
| qeexp mg/g | qecalc mg/g | k1 1/min | R2 | qeexp mg/g | qecalc mg/g | k2 g/(mg·min) | R2 | |
| 15 | 1.88 | 2.05 | 0.025 | 0.9250 | 1.88 | 2.70 | 0.014 | 0.5532 |
| 10 | 1.63 | 1.68 | 0.040 | 0.9740 | 1.63 | 6.25 | 0.025 | 0.6310 |
| 7.5 | 1.88 | 1.88 | 0.050 | 0.9964 | 1.88 | 4.16 | 0.06 | 0.9960 |
| Kiwi Peels (mg) | Pseudo-First-Order | Pseudo-Second-Order | ||||||
|---|---|---|---|---|---|---|---|---|
| qeexp mg/g | qecalc mg/g | k1 1/min | R2 | qeexp mg/g | qecalc mg/g | k2 g/(mg·min) | R2 | |
| 100 | 1.00 | 0.96 | 0.085 | 0.9818 | 1.00 | 1.12 | 0.14 | 0.9994 |
| 80 | 1.23 | 1.18 | 0.055 | 0.9704 | 1.23 | 1.51 | 0.045 | 0.9916 |
| 50 | 1.25 | 1.35 | 0.046 | 0.9557 | 1.25 | 2.43 | 0.010 | 0.9478 |
| 25 | 1.63 | 1.68 | 0.040 | 0.9740 | 1.63 | 6.25 | 0.025 | 0.6310 |
| ΔH°298K (KJ/mol) | ΔS°298K (J/mol·K) | ΔG°289K (KJ/mol) | ΔG°298K (KJ/mol) | ΔG°305K (KJ/mol) |
|---|---|---|---|---|
| +70 ± 5 | +300 ± 10 | −10 ± 1 | −12 ± 1 | −15 ± 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gubitosa, J.; Rizzi, V.; Fini, P.; Nuzzo, S.; Cosma, P. Regenerable Kiwi Peels as an Adsorbent to Remove and Reuse the Emerging Pollutant Propranolol from Water. Processes 2022, 10, 1417. https://doi.org/10.3390/pr10071417
Gubitosa J, Rizzi V, Fini P, Nuzzo S, Cosma P. Regenerable Kiwi Peels as an Adsorbent to Remove and Reuse the Emerging Pollutant Propranolol from Water. Processes. 2022; 10(7):1417. https://doi.org/10.3390/pr10071417
Chicago/Turabian StyleGubitosa, Jennifer, Vito Rizzi, Paola Fini, Sergio Nuzzo, and Pinalysa Cosma. 2022. "Regenerable Kiwi Peels as an Adsorbent to Remove and Reuse the Emerging Pollutant Propranolol from Water" Processes 10, no. 7: 1417. https://doi.org/10.3390/pr10071417
APA StyleGubitosa, J., Rizzi, V., Fini, P., Nuzzo, S., & Cosma, P. (2022). Regenerable Kiwi Peels as an Adsorbent to Remove and Reuse the Emerging Pollutant Propranolol from Water. Processes, 10(7), 1417. https://doi.org/10.3390/pr10071417









