Long-Term Impact of COVID-19: A Systematic Review of the Literature and Meta-Analysis
Abstract
:1. Background
2. Methods
2.1. Protocol and Registration
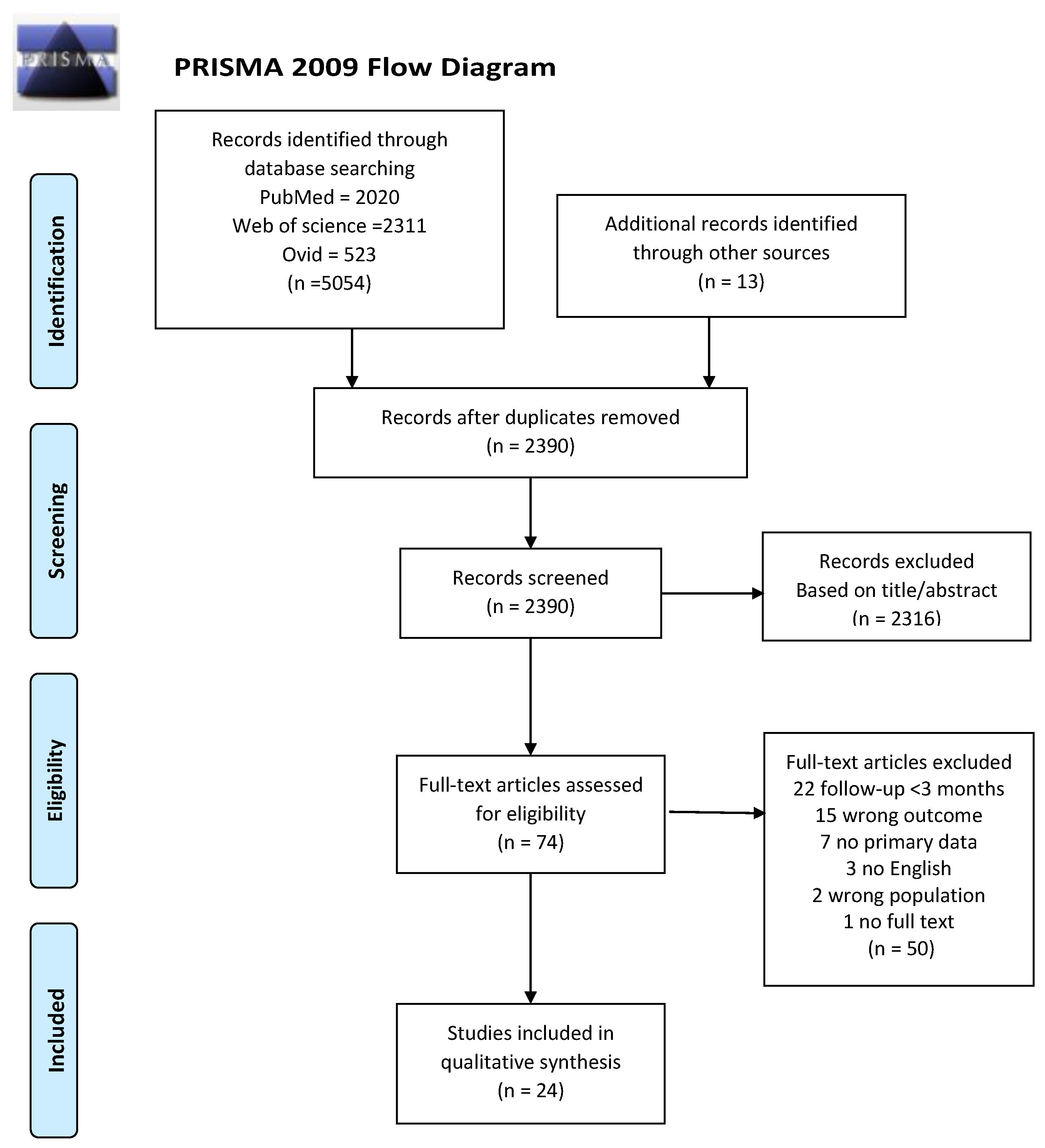
2.2. Literature Search and Study Selection
2.3. Data Extraction and Synthesis
3. Results
3.1. Lung Imaging
3.2. Lung Function
3.3. Fatigue and Respiratory Symptoms
3.4. Functional Capacity, HRQoL, and Return to Work/No Work Impairment
4. Discussion
4.1. Implications and Considerations
4.2. Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- John Hopkins University of Medicine Coronavirus Resource Center. Available online: https://coronavirus.jhu.edu/ (accessed on 21 July 2021).
- Shi, H.; Han, X.; Jiang, N.; Cao, Y.; Alwalid, O.; Gu, J.; Fan, Y.; Zheng, C. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: A descriptive study. Lancet Infect. Dis. 2020, 20, 425–434. [Google Scholar] [CrossRef]
- Wu, Z.; McGoogan, J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef]
- Nöbauer-Huhmann, I.-M.; Eibenberger, K.; Schaefer-Prokop, C.; Steltzer, H.; Schlick, W.; Strasser, K.; Fridrich, P.; Herold, C.J. Changes in lung parenchyma after acute respiratory distress syndrome (ARDS): Assessment with high-resolution computed tomography. Eur. Radiol. 2001, 11, 2436–2443. [Google Scholar] [CrossRef]
- Xie, L.; Liu, Y.; Xiao, Y.; Tian, Q.; Fan, B.; Zhao, H.; Chen, W. Follow-up Study on Pulmonary Function and Lung Radiographic Changes in Rehabilitating Severe Acute Respiratory Syndrome Patients After Discharge. Chest 2005, 127, 2119–2124. [Google Scholar] [CrossRef] [Green Version]
- Hui, D.S.; Joynt, G.; Wong, K.T.; Gomersall, C.; Li, T.S.; Antonio, G.; Ko, F.W.S.; Chan, M.C.; Chan, D.P.; Tong, M.W.; et al. Impact of severe acute respiratory syndrome (SARS) on pulmonary function, functional capacity and quality of life in a cohort of survivors. Thorax 2005, 60, 401–409. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ngai, J.C.; Ko, F.W.S.; Ng, S.; To, K.-W.; Tong, M.; Hui, D. The long-term impact of severe acute respiratory syndrome on pulmonary function, exercise capacity and health status. Respirology 2010, 15, 543–550. [Google Scholar] [CrossRef] [Green Version]
- Ong, K.-C.; Ng, A.W.-K.; Lee, L.; Kaw, G.; Kwek, S.-K.; Leow, M.K.-S.; Earnest, A. 1-Year Pulmonary Function and Health Status in Survivors of Severe Acute Respiratory Syndrome. Chest 2005, 128, 1393–1400. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Peng, L.; Liu, H.; Hua, S. Pulmonary Function and Clinical Manifestations of Patients Infected with Mild Influenza A Virus Subtype H1N1: A One-Year Follow-Up. PLoS ONE 2015, 10, e0133698. [Google Scholar] [CrossRef]
- Luyt, C.-E.; Combes, A.; Becquemin, M.-H.; Beigelman-Aubry, C.; Hatem, S.; Brun, A.-L.; Zraik, N.; Carrat, F.; Grenier, P.A.; Richard, J.-C.M.; et al. Long-term Outcomes of Pandemic 2009 Influenza A(H1N1)-Associated Severe ARDS. Chest 2012, 142, 583–592. [Google Scholar] [CrossRef] [PubMed]
- Bein, T.; Weber-Carstens, S.; Apfelbacher, C. Long-term outcome after the acute respiratory distress syndrome: Different from general critical illness? Curr. Opin. Crit. Care 2018, 24, 35–40. [Google Scholar] [CrossRef]
- Schelling, G.; Stoll, C.; Vogelmeier, C.; Hummel, T.; Behr, J.; Kapfhammer, H.-P.; Rothenhäusler, H.-B.; Haller, M.; Durst, K.; Krauseneck, T.; et al. Pulmonary function and health-related quality of life in a sample of long-term survivors of the acute respiratory distress syndrome. Intensive Care Med. 2000, 26, 1304–1311. [Google Scholar] [CrossRef] [PubMed]
- Wilcox, M.E.; Herridge, M.S. Lung function and quality of life in survivors of the acute respiratory distress syndrome (ARDS). La Presse Médicale 2011, 40, e595–e603. [Google Scholar] [CrossRef] [PubMed]
- Mahase, E. COVID-19: What do we know about long covid? BMJ 2020, 370, m2815. [Google Scholar] [CrossRef]
- Huang, Y.; Tan, C.; Wu, J.; Chen, M.; Wang, Z.; Luo, L.; Zhou, X.; Liu, X.; Huang, X.; Yuan, S.; et al. Impact of coronavirus disease 2019 on pulmonary function in early convalescence phase. Respir. Res. 2020, 21, 1–10. [Google Scholar] [CrossRef]
- Cellai, M.; O’Keefe, J.B. Characterization of Prolonged COVID-19 Symptoms in an Outpatient Telemedicine Clinic. Open Forum Infect. Dis. 2020, 7, ofaa420. [Google Scholar] [CrossRef]
- Daher, A.; Balfanz, P.; Cornelissen, C.; Müller, A.; Bergs, I.; Marx, N.; Müller-Wieland, D.; Hartmann, B.; Dreher, M.; Müller, T. Follow up of patients with severe coronavirus disease 2019 (COVID-19): Pulmonary and extrapulmonary disease sequelae. Respir. Med. 2020, 174, 106197. [Google Scholar] [CrossRef]
- Jacobs, L.G.; Paleoudis, E.G.; Bari, D.L.-D.; Nyirenda, T.; Friedman, T.; Gupta, A.; Rasouli, L.; Zetkulic, M.; Balani, B.; Ogedegbe, C.; et al. Persistence of symptoms and quality of life at 35 days after hospitalization for COVID-19 infection. PLoS ONE 2020, 15, e0243882. [Google Scholar] [CrossRef]
- Halpin, S.J.; McIvor, C.; Whyatt, G.; Adams, A.; Harvey, O.; McLean, L.; Walshaw, C.; Kemp, S.; Corrado, J.; Singh, R.; et al. Postdischarge symptoms and rehabilitation needs in survivors of COVID-19 infection: A cross-sectional evaluation. J. Med. Virol. 2021, 93, 1013–1022. [Google Scholar] [CrossRef]
- Pizarro-Pennarolli, C.; Sánchez-Rojas, C.; Torres-Castro, R.; Vera-Uribe, R.; Sanchez-Ramirez, D.C.; Vasconcello-Castillo, L.; Solís-Navarro, L.; Rivera-Lillo, G. Assessment of activities of daily living in patients post COVID-19: A systematic review. PeerJ 2021, 9, e11026. [Google Scholar] [CrossRef]
- Carfì, A.; Bernabei, R.; Landi, F. Persistent Symptoms in Patients After Acute COVID-19. JAMA 2020, 324, 603–605. [Google Scholar] [CrossRef]
- Townsend, L.; Dyer, A.H.; Jones, K.; Dunne, J.; Mooney, A.; Gaffney, F.; O’Connor, L.; Leavy, D.; O’Brien, K.; Dowds, J.; et al. Persistent fatigue following SARS-CoV-2 infection is common and independent of severity of initial infection. PLoS ONE 2020, 15, e0240784. [Google Scholar] [CrossRef] [PubMed]
- D’Cruz, R.F.; Waller, M.D.; Perrin, F.; Periselneris, J.; Norton, S.; Smith, L.-J.; Patrick, T.; Walder, D.; Heitmann, A.; Lee, K.; et al. Chest radiography is a poor predictor of respiratory symptoms and functional impairment in survivors of severe COVID-19 pneumonia. ERJ Open Res. 2021, 7. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [Green Version]
- National Heart, Lung and Blood Institute. Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 4 June 2021).
- Higgins, J.; Thomas, J. Cochrane Handbook for Systematic Reviews of Interventions. Available online: https://training.cochrane.org/handbook/current (accessed on 4 June 2021).
- Bellan, M.; Soddu, D.; Balbo, P.E.; Baricich, A.; Zeppegno, P.; Avanzi, G.C.; Baldon, G.; Bartolomei, G.; Battaglia, M.; Battistini, S.; et al. Respiratory and Psychophysical Sequelae Among Patients With COVID-19 Four Months After Hospital Discharge. JAMA Netw. Open 2021, 4, e2036142. [Google Scholar] [CrossRef]
- Han, X.; Fan, Y.; Alwalid, O.; Li, N.; Jia, X.; Yuan, M.; Li, Y.; Cao, Y.; Gu, J.; Wu, H.; et al. Six-month Follow-up Chest CT Findings after Severe COVID-19 Pneumonia. Radiology 2021, 299, E177–E186. [Google Scholar] [CrossRef]
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef]
- Lerum, T.V.; Aaløkken, T.M.; Brønstad, E.; Aarli, B.; Ikdahl, E.; Lund, K.M.A.; Durheim, M.T.; Rodriguez, J.R.; Meltzer, C.; Tonby, K.; et al. Dyspnoea, lung function and CT findings three months after hospital admission for COVID-19. Eur. Respir. J. 2020, 57, 2003448. [Google Scholar] [CrossRef]
- Liang, L.; Yang, B.; Jiang, N.; Fu, W.; He, X.; Zhou, Y.; Ma, W.-L.; Wang, X. Three-Month Follow-Up Study of Survivors of Coronavirus Disease 2019 after Discharge. J. Korean Med. Sci. 2020, 35, e418. [Google Scholar] [CrossRef]
- Sonnweber, T.; Sahanic, S.; Pizzini, A.; Luger, A.; Schwabl, C.; Sonnweber, B.; Kurz, K.; Koppelstätter, S.; Haschka, D.; Petzer, V.; et al. Cardiopulmonary recovery after COVID-19—An observational prospective multi-center trial. Eur. Respir J. 2020, 57, 2003481. [Google Scholar]
- Tabatabaei, S.M.H.; Rajebi, H.; Moghaddas, F.; Ghasemiadl, M.; Talari, H. Chest CT in COVID-19 pneumonia: What are the findings in mid-term follow-up? Emerg. Radiol. 2020, 27, 711–719. [Google Scholar] [CrossRef]
- Guler, S.A.; Ebner, L.; Aubry-Beigelman, C.; Bridevaux, P.-O.; Brutsche, M.; Clarenbach, C.; Garzoni, C.; Geiser, T.K.; Lenoir, A.; Mancinetti, M.; et al. Pulmonary function and radiological features 4 months after COVID-19: First results from the national prospective observational Swiss COVID-19 lung study. Eur. Respir. J. 2021, 57, 2003690. [Google Scholar] [CrossRef]
- Wong, A.W.; Shah, A.S.; Johnston, J.C.; Carlsten, C.; Ryerson, C.J. Patient-reported outcome measures after COVID-19: A prospective cohort study. Eur. Respir. J. 2020, 56, 2003276. [Google Scholar] [CrossRef] [PubMed]
- The Writing Committee for the COMEBAC Study Group; Morin, L.; Savale, L.; Pham, T.; Colle, R.; Figueiredo, S.; Harrois, A.; Gasnier, M.; Lecoq, A.-L.; Meyrignac, O.; et al. Four-Month Clinical Status of a Cohort of Patients After Hospitalization for COVID-19. JAMA 2021, 325, 1525. [Google Scholar] [CrossRef]
- Shah, A.S.; Wong, A.W.; Hague, C.J.; Murphy, D.T.; Johnston, J.C.; Ryerson, C.J.; Carlsten, C. A prospective study of 12-week respiratory outcomes in COVID-19-related hospitalisations. Thorax 2021, 76, 402–404. [Google Scholar] [CrossRef]
- Walle-Hansen, M.M.; Ranhoff, A.H.; Mellingsæter, M.; Wang-Hansen, M.S.; Myrstad, M. Health-related quality of life, functional decline, and long-term mortality in older patients following hospitalisation due to COVID-19. BMC Geriatr. 2021, 21, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Logue, J.K.; Franko, N.M.; McCulloch, D.J.; McDonald, D.; Magedson, A.; Wolf, C.R.; Chu, H.Y. Sequelae in Adults at 6 Months After COVID-19 Infection. JAMA Netw. Open 2021, 4, e210830. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.-M.; Shang, Y.-M.; Song, W.-B.; Li, Q.-Q.; Xie, H.; Xu, Q.-F.; Jia, J.-L.; Li, L.-M.; Mao, H.-L.; Zhou, X.-M.; et al. Follow-up study of the pulmonary function and related physiological characteristics of COVID-19 survivors three months after recovery. EClinicalMedicine 2020, 25, 100463. [Google Scholar] [CrossRef]
- Wu, Q.; Zhong, L.; Li, H.; Guo, J.; Li, Y.; Hou, X.; Yang, F.; Xie, Y.; Li, L.; Xing, Z. A Follow-Up Study of Lung Function and Chest Computed Tomography at 6 Months after Discharge in Patients with Coronavirus Disease. Can. Respir. J. 2021, 2021, 1–7. [Google Scholar] [CrossRef]
- Xiong, Q.; Xu, M.; Li, J.; Liu, Y.; Zhang, J.; Xu, Y.; Dong, W. Clinical sequelae of COVID-19 survivors in Wuhan, China: A single-centre longitudinal study. Clin. Microbiol. Infect. 2021, 27, 89–95. [Google Scholar] [CrossRef]
- Cao, J.; Zheng, X.; Wei, W.; Chu, X.; Chen, X.; Wang, Y.; Liu, Q.; Luo, S.; Weng, J.; Hu, X. Three-month outcomes of recovered COVID-19 patients: Prospective observational study. Ther. Adv. Respir. Dis. 2021, 15. [Google Scholar] [CrossRef]
- Garrigues, E.; Janvier, P.; Kherabi, Y.; Le Bot, A.; Hamon, A.; Gouze, H.; Doucet, L.; Berkani, S.; Oliosi, E.; Mallart, E.; et al. Post-discharge persistent symptoms and health-related quality of life after hospitalization for COVID-19. J. Infect. 2020, 81, e4–e6. [Google Scholar] [CrossRef]
- van den Borst, B.; Peters, J.B.; Brink, M.; Schoon, Y.; Bleeker-Rovers, C.P.; Schers, H.; van Hees, H.W.H.; van Helvoort, H.; van den Boogard, M.; van der Hoeven, H.; et al. Comprehensive health assessment three months after recovery from acute COVID-19. Clin. Infect. Dis. 2020, ciaa1750. [Google Scholar] [CrossRef]
- Abdallah, S.J.; Voduc, N.; Corrales-Medina, V.F.; McGuinty, M.; Pratt, A.; Chopra, A.; Law, A.; Garuba, H.; Thavorn, K.; Reid, R.E.; et al. Symptoms, Pulmonary Function and Functional Capacity Four Months after COVID-19. Ann. Am. Thorac. Soc. 2021. [Google Scholar] [CrossRef]
- Anastasio, F.; Barbuto, S.; Scarnecchia, E.; Cosma, P.; Fugagnoli, A.; Rossi, G.; Parravicini, M.; Parravicini, P. Medium-term impact of COVID-19 on pulmonary function, functional capacity and quality of life. Eur. Respir. J. 2021, 2004015. [Google Scholar] [CrossRef] [PubMed]
- Baricich, A.; Borg, M.B.; Cuneo, D.; Cadario, E.; Azzolina, D.; Balbo, P.; Bellan, M.; Zeppegno, P.; Pirisi, M.; Cisari, C.; et al. Midterm functional sequelae and implications in rehabilitation after COVIDA cross-sectional study. Eur. J. Phys. Rehabil. Med. 2021, 57, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, K.B.; Rao, M.; Bonilla, H.; Subramanian, A.; Hack, I.; Madrigal, M.; Singh, U.; Jagannathan, P.; Grant, P. Patients With Uncomplicated Coronavirus Disease 2019 (COVID-19) Have Long-Term Persistent Symptoms and Functional Impairment Similar to Patients with Severe COVID-19: A Cautionary Tale During a Global Pandemic. Clin. Infect. Dis. 2021. [Google Scholar] [CrossRef] [PubMed]
- Sykes, D.L.; Holdsworth, L.; Jawad, N.; Gunasekera, P.; Morice, A.H.; Crooks, M.G. Post-COVID-19 Symptom Burden: What is Long-COVID and How Should We Manage It? Lung 2021, 199, 113–119. [Google Scholar] [CrossRef]
- Hui, D.S.; Wong, K.T.; Ko, F.W.S.; Tam, L.-S.; Chan, D.P.; Woo, J.; Sung, J.J.Y. The 1-Year Impact of Severe Acute Respiratory Syndrome on Pulmonary Function, Exercise Capacity, and Quality of Life in a Cohort of Survivors. Chest 2005, 128, 2247–2261. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Jiang, H.; Xie, Y.; Zhang, T.; Liu, S.; Wu, S.; Sun, Q.; Song, S.; Wang, W.; Deng, X.; et al. Long-term clinical prognosis of human infections with avian influenza A(H7N9) viruses in China after hospitalization. EClinicalMedicine 2020, 20, 100282. [Google Scholar] [CrossRef] [Green Version]
- Park, C.M.; Goo, J.M.; Lee, H.J.; Lee, C.H.; Chun, E.J.; Im, J.-G. Nodular Ground-Glass Opacity at Thin-Section CT: Histologic Correlation and Evaluation of Change at Follow-up. Radiographics 2007, 27, 391–408. [Google Scholar] [CrossRef] [Green Version]
- Ranu, H.; Wilde, M.; Madden, B. Pulmonary Function Tests. Ulst. Med. J. 2011, 80, 84–90. [Google Scholar]
- Ozeki, N.; Kawaguchi, K.; Fukui, T.; Fukumoto, K.; Nakamura, S.; Hakiri, S.; Kato, T.; Hirakawa, A.; Okasaka, T.; Yokoi, K. The diffusing capacity of the lung for carbon monoxide is associated with the histopathological aggressiveness of lung adenocarcinoma. Eur. J. Cardio-Thoracic Surg. 2017, 52, 969–974. [Google Scholar] [CrossRef] [PubMed]
- Lundbäck, B.; Stjernberg, N.; Nyström, L.; Forsberg, B.; Lindström, M.; Lundbäck, K.; Jonsson, E.; Rosenhall, L. Epidemiology of respiratory symptoms, lung function and important determinants. Report from the Obstructive Lung Disease in Northern Sweden Project. Tuber. Lung Dis. 1994, 75, 116–126. [Google Scholar] [CrossRef]
- Dani, M.; Dirksen, A.; Taraborrelli, P.; Torocastro, M.; Panagopoulos, D.; Sutton, R.; Lim, P.B. Autonomic dysfunction in ‘long COVID’: Rationale, physiology and management strategies. Clin. Med. 2021, 21, e63–e67. [Google Scholar] [CrossRef]
- Lam, M.H.; Wing, Y.K.; Yu, M.W.; Leung, C.M.; Ma, R.C.; Kong, A.P.; So, W.Y.; Fong, S.Y.-Y.; Lam, S.-P. Mental morbidities and chronic fatigue in severe acute respiratory syndrome survivors: Long-term follow-up. Arch. Intern. Med. 2009, 169, 2142–2147. [Google Scholar] [CrossRef] [Green Version]
- Wilson, C. Concern coronavirus may trigger post-viral fatigue syndromes. New Sci. 2020, 246, 10–11. [Google Scholar] [CrossRef]
| Author Year | Country | Study Design | Follow-Up Time | Post COVID-19 Participants | Disease Severity at Baseline ¥ | ||
|---|---|---|---|---|---|---|---|
| N | Age (Years) | Male (%) | Severe Cases n (%) | ||||
| Abdallah, S. 2021 [46] | Canada | Cross-sectional | 119.9 (SD 16.2) days after positive COVID-19 test for hospitalized patients and 129.8 (SD 16.5) for non-hospitalized patients | 63 | 59.1 (SD 13.5) for hospitalized 42.4 (SD 12.9) days non-hospitalized | 57% | 25 (40%) hospitalized |
| Anastasio, F. 2021 [47] | Italy | Cross-sectional | 135 (IQR 102–175) days after symptoms onset | 379 | 56.0 (IQR 49–63) | 46% | 222 (58.6%) pneumonia |
| Baricich, A. 2021 [48] | Italy | Cross-sectional | 124 (SD 17.5) days after hospital discharge | 204 | 57.9 (SD 12.8) | 60% | 27 (13%) ICU |
| Bellan, M. 2021 [27] | Italy | Prospective | 4 months after hospital discharge | 238 | 61.0 (IQR 50–71) | 60% | 28 (12%) ICU |
| Cao, J. 2021 [43] | China | Prospective | 3 months after hospital discharge | 61 | 43.5 (SD 15.9) | 54% | 57 (94%) pneumonia severe pneumonia critical illness |
| Garrigues, E. 2020 [44] | France | Cross-sectional | 110.9 (SD 11.1) days following admission | 120 | 63.2 (SD 15.7) | 63% | 24 (20%) ICU |
| Guler, S. 2021 [34] | Switzerland | Prospective | 128 (IQR 108–144) days from initial symptoms | 113 | 60.3 (SD 12) Severe/critical COVID 52.9 (SD 11) Mild/moderate COVID-19 | 59% | 66 (58.4%) severe/ critical disease |
| Han, X. 2021 [28] | China | Prospective | 175 (SD 20) days after symptoms onset | 114 | 54.0 (SD 12) | 70% | 114 (100%) severe disease |
| Huang, Ch. 2020 [29] | China | Ambi-directional | 186 (175–199) days after symptom onset | 1733 | 57.0 (IQR 47–65) | 52% | 1294 (75%) required supplemental oxygen (n = 1172) or HFNC, NIV, or IMV (n = 122) |
| Jacobson, K. 2021 [49] | USA | Cross-sectional | 119.3 (SD 33) days after COVID-19 diagnosis | 118 | 43.3 (SD 14.4) | 53% | 22 (18.6%) hospitalized |
| Lerum, T. 2020 [30] | Norway | Prospective | 83 (73–90) days after hospital admission | 103 | 59.0 (IQR 49–72) | 52% | 15 (14.6%) ICU |
| Liang, L. 2020 [31] | China | Prospective | 3 months after hospital discharge | 76 | 41.3 (SD 13.8) | 28% | 7 (9.2%) ICU |
| Logue, J. 2021 [39] | USA | Prospective | 169 (SD 39.5) days after illness onset | 177 | 48.0 (SD 15.2) | 43% | 16 (10%) hospitalized |
| Morin, L. 2021 [36] | France | Prospective | 4 months after hospital or ICU discharge | 478 | 60.9 (SD 16.1) | 58% | 142 (30%) ICU |
| * Shah, A. 2020 [37] | Canada | Prospective | 11.7 weeks after symptoms onset | 60 | 67.0 (IQR 54–74) | 68% | 46 (76.7%) required supplemental oxygen |
| Sonnweber, T. 2020 [32] | Austria | Prospective | 103 (SD 21) days after diagnosis for second visit (100 days after onset) | 145 | 57.0 (14) | 55% | 109 (75%) hospitalized |
| Sykes, D. 2021 [50] | UK | Cross-sectional | 113 days (46–167) days post discharge | 134 | 59.6 (SD 14.0) | 66% | 27 (20.1%) ICU |
| Tabatabaei, S. 2020 [33] | Iran | Retrospective | 91 (SD 15.5) days after initial CT | 52 | 50.2 (SD 13.1) | 62% | NR |
| van den Borst, B. 2020 [45] | the Netherlands | Prospective | 13.0 (SD 2.2) weeks after symptoms onset | 124 | 59.0 (SD 14) | 60% | 46 (37%) severe/ critical disease |
| Walle-Hansen, M. 2021 [38] | Norway | Prospective | 186 days after discharge | 106 | 74.3 (range 60–96) | 57% | 27 (25.4%) ICU or intermediary ward |
| * Wong, A. 2020 [35] | Canada | Prospective | 13 (IQR 11–14) weeks after symptoms onset | 78 | 62.0 (SD 16) | 64% | NR |
| Wu, Q. 2021 [41] | China | Prospective | 6 months after discharge | 54 | 47.0 (IQR 36–57) | 59% | 23 (42.5%) severe disease |
| Xiong, Q. 2021 [42] | China | Prospective | 97 (95–102) days after hospital discharge | 538 | 52.0 (SD 41–62) | 46% | 207 (38.5%) severe/ critical disease |
| Zhao, Y. 2020 [40] | China | Retrospective | 3 months after symptom onset (64–93 days after discharged from hospitals) | 55 | 47.7 (SD 15.49) | 58% | 4 (7.3%) severe disease |
| Outcome | No. of Studies | No. of Patients Analyzed | Mean (SD) Follow-Up Time (Months) | Pooled Prevalence (95% CI) | I2 % | p-Value |
|---|---|---|---|---|---|---|
| Chest CT [28,29,30,31,32,33,34,36,37,40,41,43,45] | ||||||
| Chest CT abnormalities | 10 | 987 | 3.9 (1.4) | 59% (44–73%) | 96 | <0.001 |
| GGO | 13 | 1313 | 3.9 (1.3) | 39% (26–52%) | 97 | <0.001 |
| Interstitial thickening or interlobular septal thickening | 7 | 885 | 3.8 (1.2) | 33% (13–52%) | 98 | <0.001 |
| Parenchymal band or fibrous stripe | 6 | 815 | 3.8 (1.2) | 31% (17–44%) | 95 | <0.001 |
| Bronchovascular bundle distortion or bronchiectasis | 5 | 437 | 4.5 (1.3) | 26% (9–43%) | 97 | <0.001 |
| Thickening or adjacent pleura | 4 | 573 | 5.5 (0.8) | 11% (2–20%) | 94 | <0.001 |
| Consolidation | 4 | 652 | 4.9 (1.3) | 6% (2–11%) | 89 | <0.001 |
| Crazy paving | 1 | 55 | 3.0 | 5% (1–11%) | NA | NA |
| Pulmonary function [27,28,29,30,31,32,36,37,40,41,43,45,46] | ||||||
| Pulmonary function abnormalities | 6 | 439 | 3.5 (1.2) | 39% (24–55%) | 94 | <0.001 |
| Diffusion pattern | 12 | 1490 | 4.0 (1.3) | 31% (24–38%) | 89 | <0.001 |
| Restrictive Pattern | 8 | 921 | 3.8 (1.5) | 12% (8–17%) | 82 | <0.001 |
| Obstructive pattern | 7 | 858 | 3.6 (1.3) | 8% (6–9%) | 7 | <0.001 |
| Fatigue [29,31,32,35,36,39,40,41,43,44,45,46,47,49,50] and respiratory symptoms [27,28,29,30,31,32,33,35,36,40,41,42,43,44,46,47,49,50] | ||||||
| Fatigue | 15 | 4118 | 4.0 (1.3) | 38% (27–49%) | 98 | <0.001 |
| Dyspnea | 16 | 3526 | 4.0 (1.1) | 32% (24–40%) | 98 | <0.001 |
| Chest pain/Tightness | 10 | 3728 | 3.9 (0.9) | 16% (12–21%) | 94 | <0.001 |
| Cough | 14 | 2539 | 3.8 (0.9) | 13% (9–17%) | 94 | <0.001 |
| Sputum | 4 | 783 | 3.8 (1.4) | 12% (3–21%) | 94 | <0.001 |
| Sore Throat | 6 | 2554 | 4.4 (1.3) | 4% (2–7%) | 66 | 0.02 |
| Functional capacity [27,29,45,48], Health-related quality of live (HRQoL) [35,38,39,45], and return to work/work impairment [31,40,44,49] | ||||||
| * Decreased functional capacity | 5 | 2364 | 4.2 (1.3) | 36% (22–49%) | 97 | <0.001 |
| Decreased HRQoL | 4 | 474 | 4.4 (1.6) | 52% (33–71%) | 94 | <0.001 |
| ¥ Return to work/No work impairment | 4 | 259 | 3.4 (0.4) | 80% | NA | NA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanchez-Ramirez, D.C.; Normand, K.; Zhaoyun, Y.; Torres-Castro, R. Long-Term Impact of COVID-19: A Systematic Review of the Literature and Meta-Analysis. Biomedicines 2021, 9, 900. https://doi.org/10.3390/biomedicines9080900
Sanchez-Ramirez DC, Normand K, Zhaoyun Y, Torres-Castro R. Long-Term Impact of COVID-19: A Systematic Review of the Literature and Meta-Analysis. Biomedicines. 2021; 9(8):900. https://doi.org/10.3390/biomedicines9080900
Chicago/Turabian StyleSanchez-Ramirez, Diana C., Kaylene Normand, Yang Zhaoyun, and Rodrigo Torres-Castro. 2021. "Long-Term Impact of COVID-19: A Systematic Review of the Literature and Meta-Analysis" Biomedicines 9, no. 8: 900. https://doi.org/10.3390/biomedicines9080900
APA StyleSanchez-Ramirez, D. C., Normand, K., Zhaoyun, Y., & Torres-Castro, R. (2021). Long-Term Impact of COVID-19: A Systematic Review of the Literature and Meta-Analysis. Biomedicines, 9(8), 900. https://doi.org/10.3390/biomedicines9080900








