Vivo-Morpholino-Based Antiviral for SARS-CoV-2: Implications for Novel Therapies in the Treatment of Acute COVID-19 Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Development of MRCV-1
2.2. Assay Methods for Testing MRCV-19
2.2.1. Screening Strategy
2.2.2. Compound Preparation
2.3. Method for Measuring Antiviral Effect of Compounds
2.4. Method for Measuring Cytotoxic Effect of Compounds
3. Data Analysis
4. Results
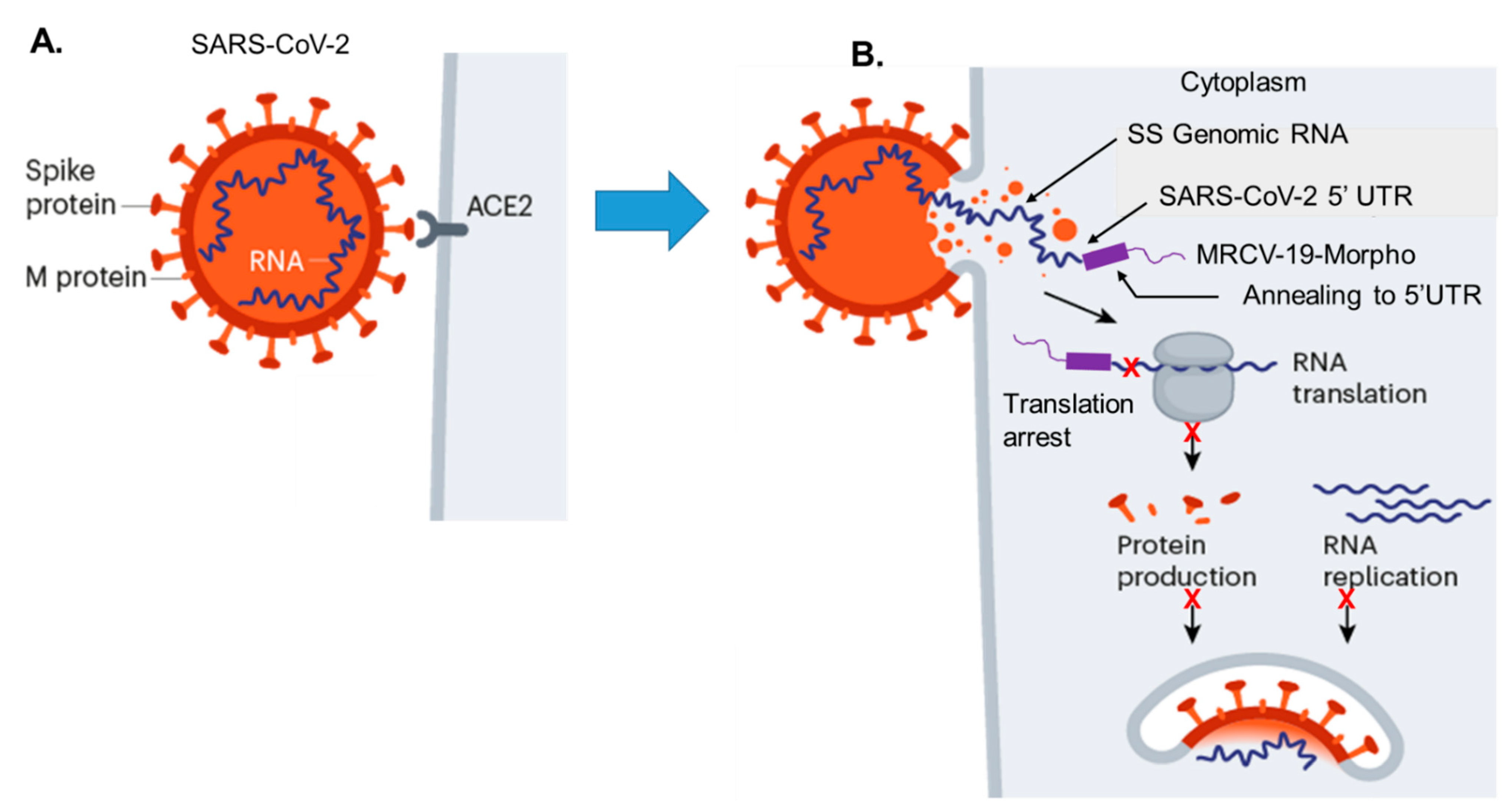
4.1. Strategy for MRCV-19 Inhibition of SARS-CoV-2 Replication
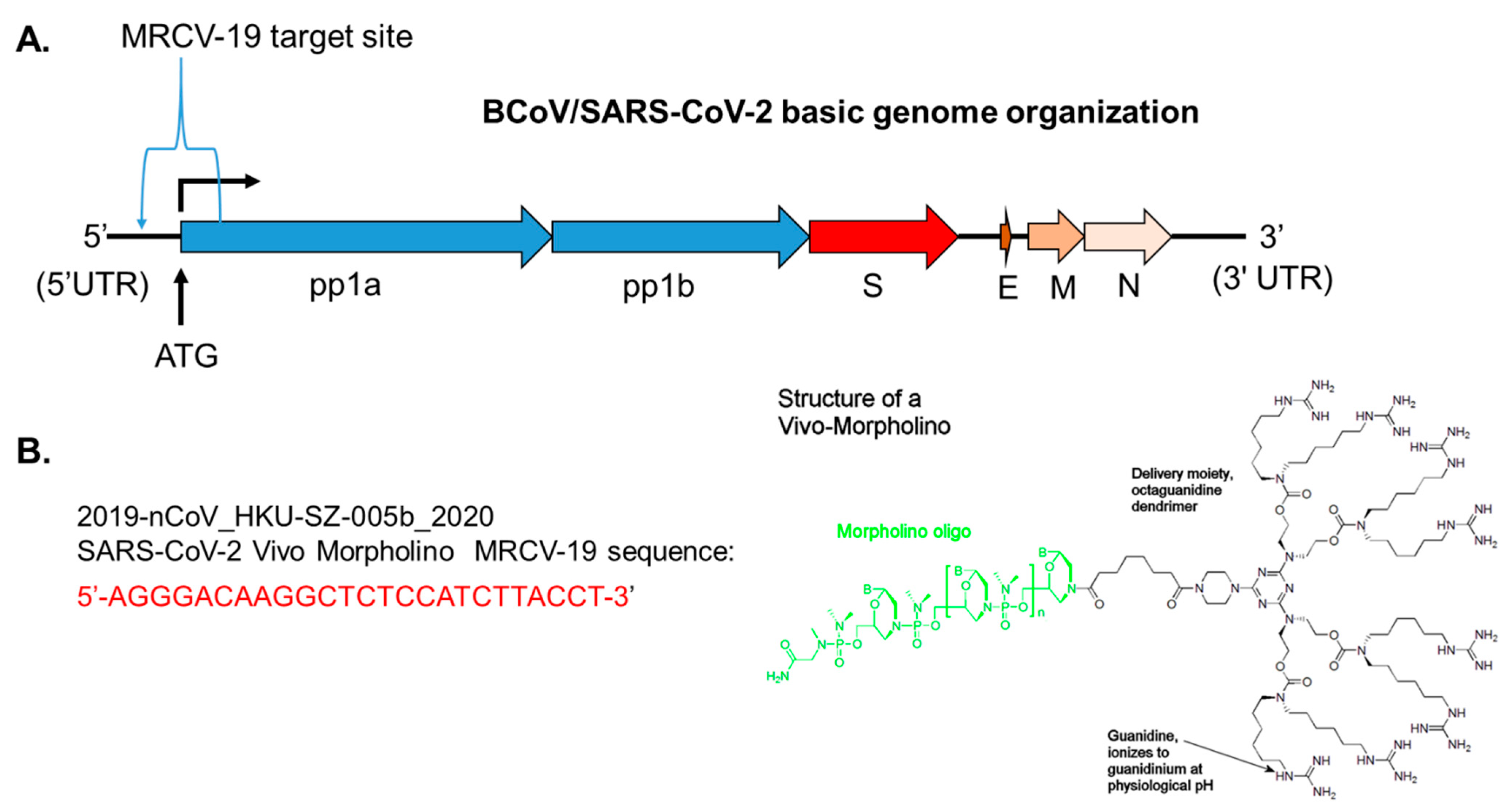
4.2. Sequence Alignments and Morpholino Targeting in the 5′UTR
4.3. SARS-CoV-2 Antiviral (MRCV-19)
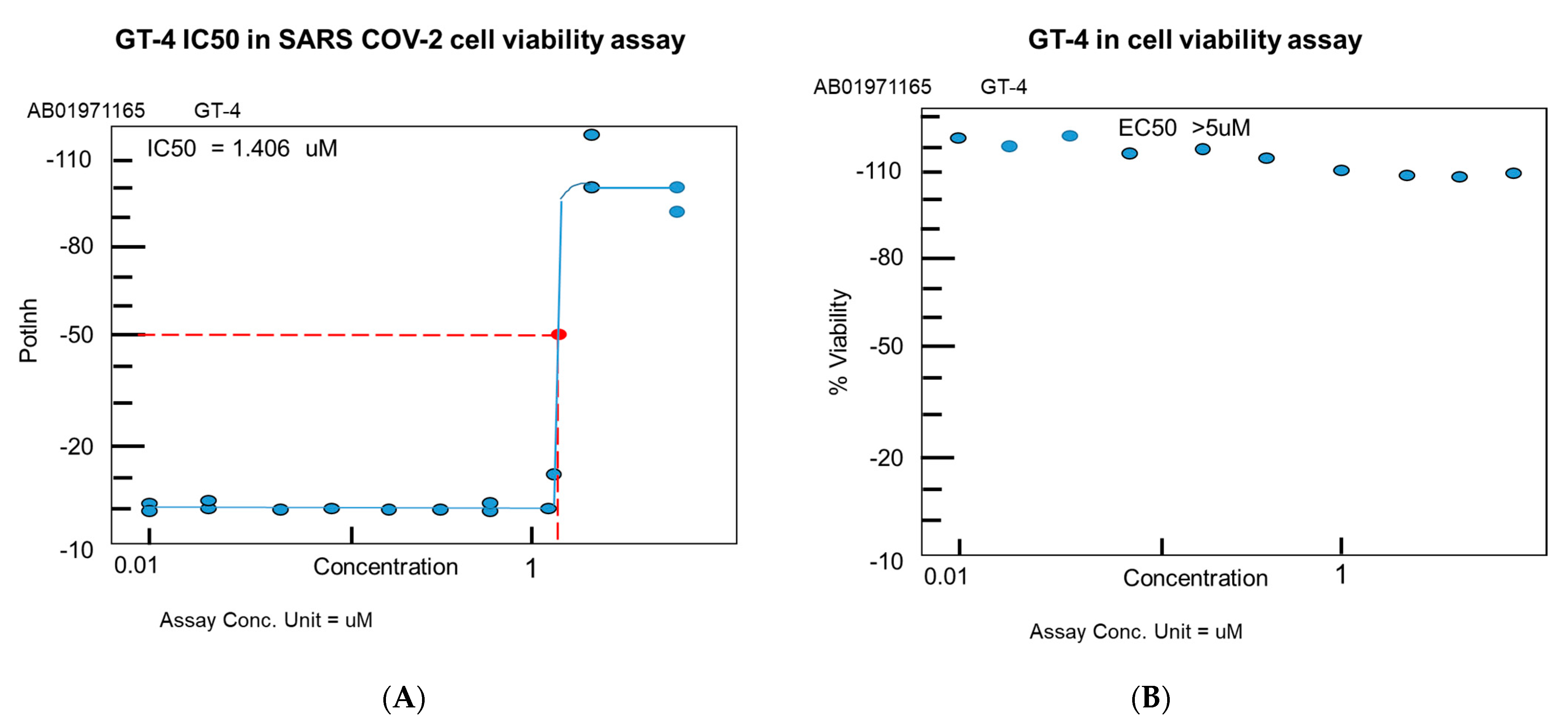
4.4. MRCV-19 Inhibition of SARS-CoV-2 Infection
4.5. MRCV Exhibits Low Toxicity In Vitro
4.6. MRVC-19, More Effective Than Hydroxychloroquine and Remdesivir to Inhibit SARS-CoV-2 Infection In Vitro
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Disclaimer
References
- WHO. Pneumonia of Unknown Cause: China. Available online: www.who.int/csr/don/05-january-2020-pneumonia-of-unkown-cause-china/en/ (accessed on 13 February 2020).
- WHO. Coronavirus Disease (COVID-19) Technical Guidance: Early Investigations. Available online: www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance/early-investigations (accessed on 15 February 2020).
- Johns Hopkins Whiting School of Engineering. Coronavirus COVID-19 Global Cases. Available online: gisanddata.maps.arcgis.com/apps/opsdashboard/index.html#/bda7594740fd40299423467b48e9ecf6 (accessed on 20 February 2020).
- Bassetti, M.; Vena, A.; Giacobbe, D.R. The novel Chinese coronavirus (2019nCoV) infections: Challenges for fighting the storm. Eur. J. Clin. Investig. 2020, 50, e13209. [Google Scholar] [CrossRef] [PubMed]
- Woo, P.C.Y.; Lau, S.K.P.; Lam, C.S.F.; Lau, C.C.Y.; Tsang, A.K.L.; Lau, J.H.L.; Bai, R.; Teng, J.L.L.; Tsang, C.C.C.; Wang, M.; et al. Discovery of seven novel mammalian and avian coronaviruses in the genus deltacoronavirus supports bat coronaviruses as the gene source of alphacoronavirus and betacoronavirus and avian coronaviruses as the gene source of gammacoronavirus and deltacoronavirus. J. Virol. 2012, 86, 3995–4008. [Google Scholar] [PubMed]
- International Committee on Taxonomy of Viruses. Virus Taxonomy: 2017 Release. Available online: https://talk.ictvonline.org/taxonomy/ (accessed on 13 February 2020).
- Du Toit, A. Outbreak of a novel coronavirus. Nat. Rev. Microbiol. 2020, 18, 123. [Google Scholar] [CrossRef] [PubMed]
- Rothan, H.A.; Byrareddy, S.N. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J. Autoimmun. 2020, 109, 102433. [Google Scholar] [CrossRef] [PubMed]
- Hui, D.S.; Azhar, E.I.; Madani, T.A.; Ntoumi, F.; Kock, R.; Dar, O.; Ippolito, G.; Mchugh, T.D.; Memish, Z.A.; Drosten, C.; et al. The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health—The latest 2019 novel coronavirus outbreak in Wuhan, China. Int. J. Infect. Dis. 2020, 91, 264–266. [Google Scholar] [CrossRef] [PubMed]
- Woo, P.C.; Lau, S.K.; Chy, C.-M.; Chan, K.-H.; Tsoi, H.-W.; Huang, Y.; Wong, B.H.L.; Poon, R.S.W.; Cai, J.J.; Luk, W.-K.; et al. Characterization and complete genome sequence of a novel coronavirus, coronavirus HKU1, from patients with pneumonia. J Virol. 2005, 2, 884–895. [Google Scholar] [CrossRef] [PubMed]
- Fehr, A.R.; Perlman, S. Coronaviruses: An overview of their replication and pathogenesis. Methods Mol. Biol. 2015, 1282, 1–23. [Google Scholar] [PubMed]
- Chan, J.F.; To, K.; Tse, H.; Jin, D.-Y.; Yuen, K.-Y. Interspecies transmission and emergence of novel viruses: Lessons from bats and birds. Trends Microbiol. 2013, 10, 544–555. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Wong, G.; Shi, W.; Liu, J.; Lai, A.C.K.; Zhou, J.; Liu, W.; Bi, Y.; Gao, G.F. Epidemiology, genetic recombination, and pathogenesis of coronaviruses. Trends Microbiol. 2016, 24, 490–502. [Google Scholar] [CrossRef] [PubMed]
- Cheng, V.C.; Lau, S.K.P.; Woo, P.C.Y.; Yuen, K.Y. Severe acute respiratory syndrome coronavirus as an agent of emerging and reemerging infection. Clin. Microbiol. Rev. 2007, 4, 660–694. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.F.; Lau, S.K.; To, K.K.W.; Cheng, V.C.C.; Woo, P.C.Y.; Yuen, K.-Y. Middle East respiratory syndrome coronavirus: Another zoonotic betacoronavirus causing SARS-like disease. Clin. Microbiol. Rev. 2015, 2, 465–522. [Google Scholar] [CrossRef] [PubMed]
- Arabi, Y.M.; Arifi, A.A.; Balkhy, H.H.; Najm, H.; Aldawood, A.S.; Ghabashi, H.H.; Alothman, A.; Khaldi, A.; Al Raiy, B. Clinical course and outcomes of critically ill patients with Middle East respiratory syndrome coronavirus infection. Ann. Intern. Med. 2014, 6, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Chafekar, A.; Fielding, B.C. MERS-CoV: Understanding the latest human coronavirus threat. Viruses 2018, 2, 93. [Google Scholar] [CrossRef]
- Summerton, J.; Weller, D. Morpholino antisense oligomers: Design, preparation, and properties. Antisense Nucleic Acid Drug Dev. 1997, 7, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.F.; Morcos, P.A. Design and synthesis of dendritic molecular transporter that achieves efficient in vivo delivery of morpholino antisense oligo. Bioconjug. Chem. 2008, 7, 1464–1470. [Google Scholar] [CrossRef]
- Maddox, C.B.; Rasmussen, L.; White, E.L. Adapting cell based assays to the high throughput screening platform: Problems encountered and lessons learned. JALA 2008, 13, 168–173. [Google Scholar] [CrossRef][Green Version]
- Severson, W.E.; Shindo, N.; Sosa, M.; Fletcher, T., III; White, E.L.; Ananthan, S.; Jonsson, C.B. Development and validation of a high-throughput screen for inhibitors of SARS CoV and its application in screening of a 100,000-compound library. J. Biomol. Screen. 2007, 1, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Moore, M.J.; Vasilieva, N.; Sui, J.; Wong, S.K.; Berne, M.A.; Somasundaran, M.; Sullivan, J.L.; Luzuriaga, K.; Greenough, T.C.; et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 2003, 6965, 450–454. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.F.; Yuan, F.; Kok, K.-H.; To, K.K.-W.; Chu, H.; Yang, J.; Xing, F.; Liu, J.; Yip, C.; Poon, R.; et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: A study of a family cluster. Lancet 2020, 10223, 514–523. [Google Scholar] [CrossRef]
- Hu, D.; Zhu, C.; Ai, L.; He, T.; Wang, Y.; Ye, F.; Yang, L.; Ding, C.; Zhu, X.; Lv, R.; et al. Genomic characterization and infectivity of a novel SARS-like coronavirus in Chinese bats. Emerg. Microbes Infect. 2018, 1, 154. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hildreth, J.E.K.; Moulton, J.D.; Alcendor, D.J. Vivo-Morpholino-Based Antiviral for SARS-CoV-2: Implications for Novel Therapies in the Treatment of Acute COVID-19 Disease. Biomedicines 2021, 9, 1018. https://doi.org/10.3390/biomedicines9081018
Hildreth JEK, Moulton JD, Alcendor DJ. Vivo-Morpholino-Based Antiviral for SARS-CoV-2: Implications for Novel Therapies in the Treatment of Acute COVID-19 Disease. Biomedicines. 2021; 9(8):1018. https://doi.org/10.3390/biomedicines9081018
Chicago/Turabian StyleHildreth, James E. K., Jon D. Moulton, and Donald J. Alcendor. 2021. "Vivo-Morpholino-Based Antiviral for SARS-CoV-2: Implications for Novel Therapies in the Treatment of Acute COVID-19 Disease" Biomedicines 9, no. 8: 1018. https://doi.org/10.3390/biomedicines9081018
APA StyleHildreth, J. E. K., Moulton, J. D., & Alcendor, D. J. (2021). Vivo-Morpholino-Based Antiviral for SARS-CoV-2: Implications for Novel Therapies in the Treatment of Acute COVID-19 Disease. Biomedicines, 9(8), 1018. https://doi.org/10.3390/biomedicines9081018






