Generation and Evaluation of Novel Biomaterials Based on Decellularized Sturgeon Cartilage for Use in Tissue Engineering
Abstract
:1. Introduction
2. Materials and Methods
2.1. Human and Sturgeon Collagen Sequence Alignment
2.2. Biochemical Characterization of Human and Sturgeon Cartilage with Ultra-Performance Liquid Chromatography
2.3. Obtaining Novel Biomaterials by Sturgeon Cartilage Decellularization
2.4. Cell Cultures and Recellularization
2.5. In Vivo Evaluation
2.6. Histology and Histochemistry
2.7. Immunohistochemistry
2.8. Quantification and Statistical Analysis
3. Results
3.1. Comparative Analysis of Human and Sturgeon Cartilage Proteins
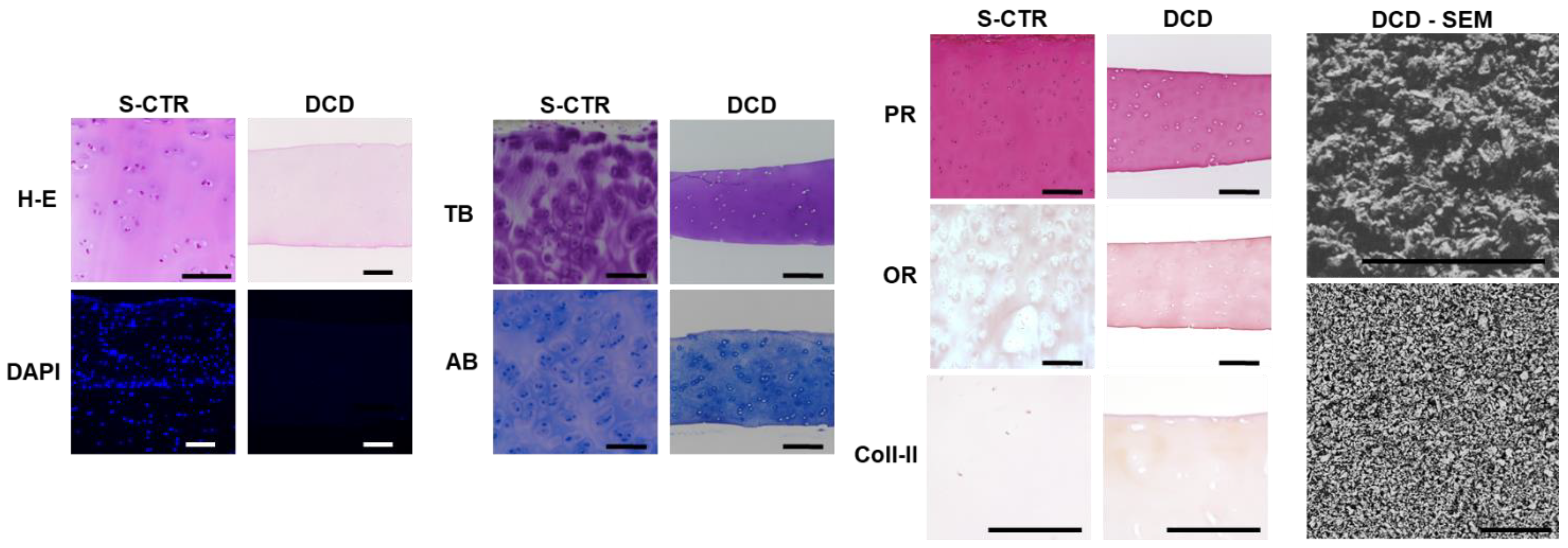
3.2. Characterization of Native Human and Sturgeon Control Cartilage
3.3. Obtaining Novel Biomaterials by Sturgeon Cartilage Decellularization
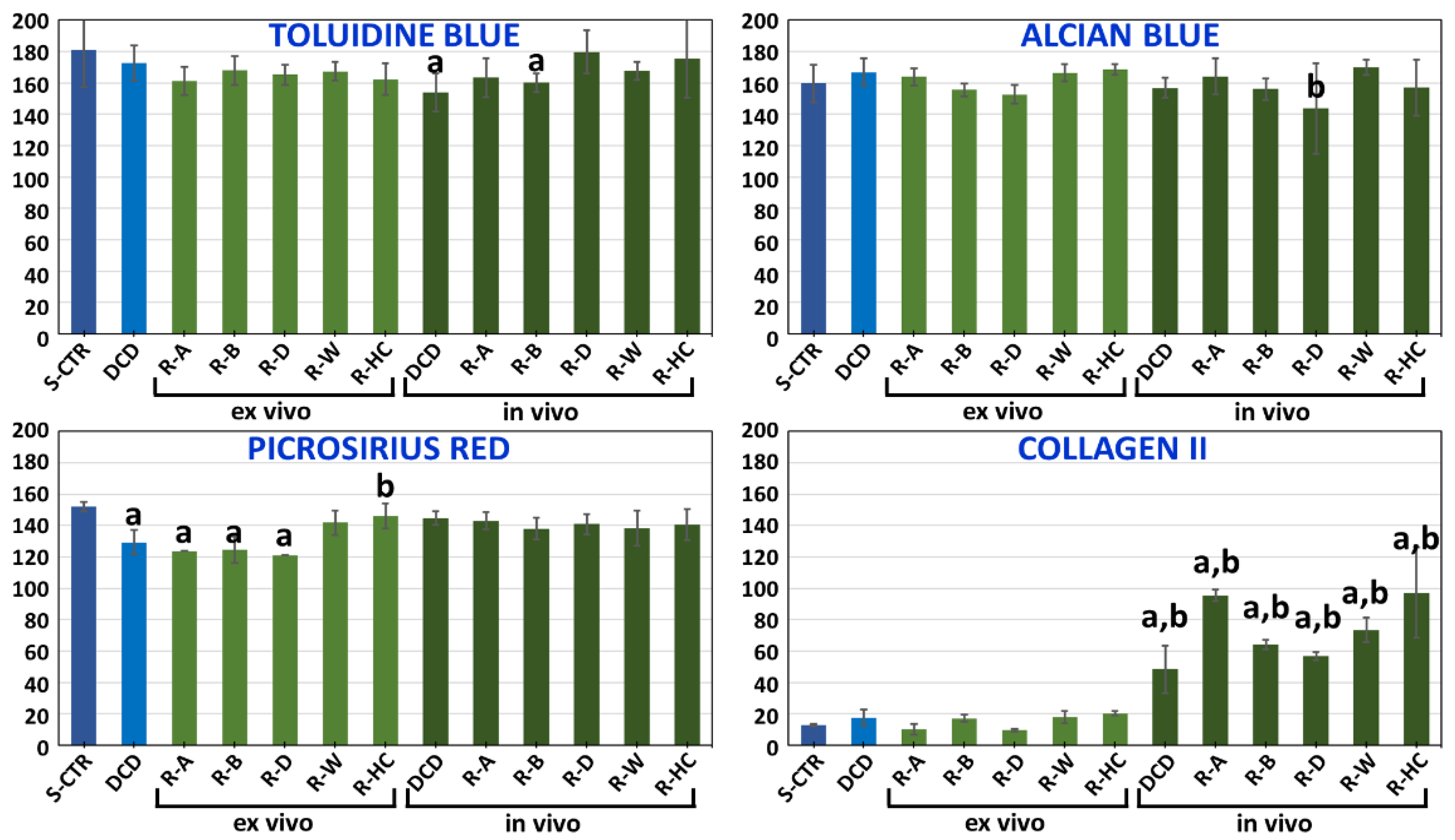
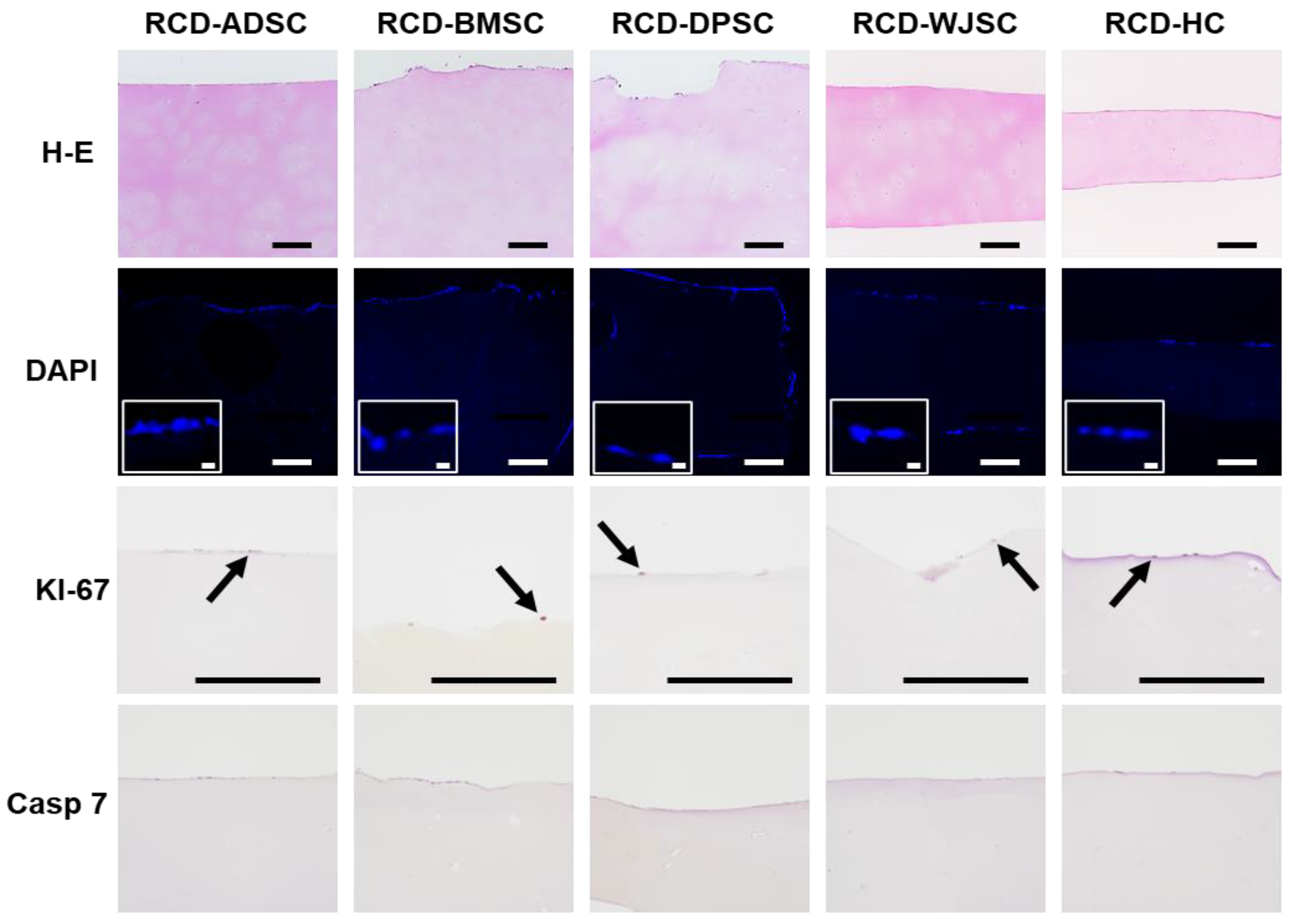
3.4. Ex Vivo Characterization of Recellularized Cartilage Disks
3.5. In Vivo Characterization of Decellularized Cartilage Disks and Recellularized Cartilage Disks
4. Discussion
5. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Langer, R.; Vacanti, J.P. Tissue Engineering. Science 1993, 260, 920–926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blanco-Elices, C.; España-Guerrero, E.; Mateu-Sanz, M.; Sánchez-Porras, D.; García-García, Ó.D.; Sánchez-Quevedo, M.D.C.; Fernández-Valadés, R.; Alaminos, M.; Martín-Piedra, M.Á.; Garzón, I. In Vitro Generation of Novel Functionalized Biomaterials for Use in Oral and Dental Regenerative Medicine Applications. Running Title: Fibrin-Agarose Functionalized Scaffolds. Materials 2020, 13, 1692. [Google Scholar] [CrossRef] [Green Version]
- Fu, L.; Li, P.; Li, H.; Gao, C.; Yang, Z.; Zhao, T.; Chen, W.; Liao, Z.; Peng, Y.; Cao, F.; et al. The Application of Bioreactors for Cartilage Tissue Engineering: Advances, Limitations, and Future Perspectives. Stem Cells Int. 2021, 2021, 6621806. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Florit, M.; Pardo, A.; Domingues, R.M.A.; Graça, A.L.; Babo, P.S.; Reis, R.L.; Gomes, M.E. Natural-Based Hydrogels for Tissue Engineering Applications. Molecules 2020, 25, 5858. [Google Scholar] [CrossRef] [PubMed]
- Kabir, W.; Di Bella, C.; Jo, I.; Gould, D.; Choong, P.F.M. Human Stem Cell Based Tissue Engineering for In Vivo Cartilage Repair: A Systematic Review. Tissue Eng. Part B Rev. 2021, 27, 74–93. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Kennedy, P.; Bonazza, N.; Yu, Y.; Dhawan, A.; Ozbolat, I. Three-Dimensional Bioprinting of Articular Cartilage: A Systematic Review. Cartilage 2021, 12, 76–92. [Google Scholar] [CrossRef]
- Jacob, G.; Shimomura, K.; Nakamura, N. Osteochondral Injury, Management and Tissue Engineering Approaches. Front. Cell Dev. Biol. 2020, 8, 580868. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, H.; Luo, W.; Cai, T.; Li, Z.; Liu, Y.; Gao, W.; Wan, Q.; Wang, X.; Wang, J.; et al. Regeneration of Skeletal System with Genipin Crosslinked Biomaterials. J. Tissue Eng. 2020, 11, 2041731420974861. [Google Scholar] [CrossRef]
- Entekhabi, E.; Haghbin Nazarpak, M.; Sedighi, M.; Kazemzadeh, A. Predicting Degradation Rate of Genipin Cross-Linked Gelatin Scaffolds with Machine Learning. Mater. Sci. Eng. C 2020, 107, 110362. [Google Scholar] [CrossRef]
- Taylor, D.A.; Sampaio, L.C.; Ferdous, Z.; Gobin, A.S.; Taite, L.J. Decellularized Matrices in Regenerative Medicine. Acta Biomater. 2018, 74, 74–89. [Google Scholar] [CrossRef]
- Pina, S.; Ribeiro, V.P.; Marques, C.F.; Maia, F.R.; Silva, T.H.; Reis, R.L.; Oliveira, J.M. Scaffolding Strategies for Tissue Engineering and Regenerative Medicine Applications. Materials 2019, 12, 1824. [Google Scholar] [CrossRef] [Green Version]
- Lammi, M.J.; Piltti, J.; Prittinen, J.; Qu, C. Challenges in Fabrication of Tissue-Engineered Cartilage with Correct Cellular Colonization and Extracellular Matrix Assembly. Int. J. Mol. Sci. 2018, 19, 2700. [Google Scholar] [CrossRef] [Green Version]
- Ibsirlioglu, T.; Elçin, A.E.; Elçin, Y.M. Decellularized Biological Scaffold and Stem Cells from Autologous Human Adipose Tissue for Cartilage Tissue Engineering. Methods 2019, 171, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Safari, F.; Fani, N.; Eglin, D.; Alini, M.; Stoddart, M.J.; Baghaban Eslaminejad, M. Human Umbilical Cord-Derived Scaffolds for Cartilage Tissue Engineering. J. Biomed. Mater. Res. A 2019, 107, 1793–1802. [Google Scholar] [CrossRef] [PubMed]
- Vas, W.J.; Shah, M.; Blacker, T.S.; Duchen, M.R.; Sibbons, P.; Roberts, S.J. Decellularized Cartilage Directs Chondrogenic Differentiation: Creation of a Fracture Callus Mimetic. Tissue Eng. Part A 2018, 24, 1364–1376. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, S.; Ren, S.; Zhang, X.; Yang, F.; Chen, Y.; Wang, B. Structural Characterization and Proliferation Activity of Chondroitin Sulfate from the Sturgeon, Acipenser Schrenckii. Int. J. Biol. Macromol. 2020, 164, 3005–3011. [Google Scholar] [CrossRef]
- Zhang, X.; Ookawa, M.; Tan, Y.; Ura, K.; Adachi, S.; Takagi, Y. Biochemical Characterisation and Assessment of Fibril-Forming Ability of Collagens Extracted from Bester Sturgeon Huso Huso × Acipenser Ruthenus. Food Chem. 2014, 160, 305–312. [Google Scholar] [CrossRef]
- Zhang, X.; Azuma, N.; Hagihara, S.; Adachi, S.; Ura, K.; Takagi, Y. Characterization of Type I and II Procollagen A1 Chain in Amur Sturgeon (Acipenser Schrenckii) and Comparison of Their Gene Expression. Gene 2016, 579, 8–16. [Google Scholar] [CrossRef]
- Lai, C.-S.; Tu, C.-W.; Kuo, H.-C.; Sun, P.-P.; Tsai, M.-L. Type II Collagen from Cartilage of Acipenser Baerii Promotes Wound Healing in Human Dermal Fibroblasts and in Mouse Skin. Mar. Drugs 2020, 18, 511. [Google Scholar] [CrossRef]
- Im, A.-R.; Park, Y.; Kim, Y.S. Isolation and Characterization of Chondroitin Sulfates from Sturgeon (Acipenser Sinensis) and Their Effects on Growth of Fibroblasts. Biol. Pharm. Bull. 2010, 33, 1268–1273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Chen, W.; Dai, Y.; Huang, Y.; Chen, Z.; Xi, T.; Zhou, Z.; Liu, H. Decellularized Sturgeon Cartilage Extracellular Matrix Scaffold Inhibits Chondrocyte Hypertrophy in vitro and in vivo. J. Tissue Eng. Regen. Med. 2021. [Google Scholar] [CrossRef]
- Khajavi, M.; Hajimoradloo, A.; Zandi, M.; Pezeshki-Modaress, M.; Bonakdar, S.; Zamani, A. Fish Cartilage: A Promising Source of Biomaterial for Biological Scaffold Fabrication in Cartilage Tissue Engineering. J. Biomed. Mater. Res. A 2021. [Google Scholar] [CrossRef]
- Youngstrom, D.W.; Cakstina, I.; Jakobsons, E. Cartilage-Derived Extracellular Matrix Extract Promotes Chondrocytic Phenotype in Three-Dimensional Tissue Culture. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1040–1047. [Google Scholar] [CrossRef]
- Garzon, I.; Chato-Astrain, J.; Campos, F.; Fernandez-Valades, R.; Sanchez-Montesinos, I.; Campos, A.; Alaminos, M.; D’Souza, R.N.; Martin-Piedra, M.A. Expanded Differentiation Capability of Human Wharton’s Jelly Stem Cells Toward Pluripotency: A Systematic Review. Tissue Eng. Part B Rev. 2020, 26, 301–312. [Google Scholar] [CrossRef]
- Lu, Z.; Yan, L.; Pei, M. Commentary on “Surface Markers Associated with Chondrogenic Potential of Human Mesenchymal Stromal/Stem Cells”. F1000Research 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Corpet, F. Multiple Sequence Alignment with Hierarchical Clustering. Nucleic Acids Res. 1988, 16, 10881–10890. [Google Scholar] [CrossRef]
- Pu, L.; Wu, J.; Pan, X.; Hou, Z.; Zhang, J.; Chen, W.; Na, Z.; Meng, M.; Ni, H.; Wang, L.; et al. Determining the Optimal Protocol for Preparing an Acellular Scaffold of Tissue Engineered Small-Diameter Blood Vessels. J. Biomed. Mater. Res. B Appl. Biomater. 2018, 106, 619–631. [Google Scholar] [CrossRef]
- Oliveira, A.C.; Garzón, I.; Ionescu, A.M.; Carriel, V.; Cardona, J.d.l.C.; González-Andrades, M.; Pérez, M.d.M.; Alaminos, M.; Campos, A. Evaluation of Small Intestine Grafts Decellularization Methods for Corneal Tissue Engineering. PLoS ONE 2013, 8, e66538. [Google Scholar] [CrossRef] [Green Version]
- Philips, C.; Campos, F.; Roosens, A.; Sánchez-Quevedo, M.D.C.; Declercq, H.; Carriel, V. Qualitative and Quantitative Evaluation of a Novel Detergent-Based Method for Decellularization of Peripheral Nerves. Ann. Biomed. Eng. 2018, 46, 1921–1937. [Google Scholar] [CrossRef] [PubMed]
- Martin-Piedra, M.A.; Alfonso-Rodriguez, C.A.; Zapater, A.; Durand-Herrera, D.; Chato-Astrain, J.; Campos, F.; Sanchez-Quevedo, M.C.; Alaminos, M.; Garzon, I. Effective Use of Mesenchymal Stem Cells in Human Skin Substitutes Generated by Tissue Engineering. Eur. Cell Mater. 2019, 37, 233–249. [Google Scholar] [CrossRef]
- Alaminos, M.; Pérez-Köhler, B.; Garzón, I.; García-Honduvilla, N.; Romero, B.; Campos, A.; Buján, J. Transdifferentiation Potentiality of Human Wharton’s Jelly Stem Cells towards Vascular Endothelial Cells. J. Cell Physiol. 2010, 223, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Porras, D.; Durand-Herrera, D.; Paes, A.B.; Chato-Astrain, J.; Verplancke, R.; Vanfleteren, J.; Sánchez-López, J.D.; García-García, Ó.D.; Campos, F.; Carriel, V. Ex Vivo Generation and Characterization of Human Hyaline and Elastic Cartilaginous Microtissues for Tissue Engineering Applications. Biomedicines 2021, 9, 292. [Google Scholar] [CrossRef] [PubMed]
- Alfonso-Rodríguez, C.-A.; Garzón, I.; Garrido-Gómez, J.; Oliveira, A.-C.-X.; Martín-Piedra, M.-Á.; Scionti, G.; Carriel, V.; Hernández-Cortés, P.; Campos, A.; Alaminos, M. Identification of Histological Patterns in Clinically Affected and Unaffected Palm Regions in Dupuytren’s Disease. PLoS ONE 2014, 9, e112457. [Google Scholar] [CrossRef] [Green Version]
- Vela-Romera, A.; Carriel, V.; Martín-Piedra, M.A.; Aneiros-Fernández, J.; Campos, F.; Chato-Astrain, J.; Prados-Olleta, N.; Campos, A.; Alaminos, M.; Garzón, I. Characterization of the Human Ridged and Non-Ridged Skin: A Comprehensive Histological, Histochemical and Immunohistochemical Analysis. Histochem. Cell Biol. 2019, 151, 57–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodriguez-Pozo, J.A.; Ramos-Lopez, J.F.; Gonzalez-Gallardo, M.C.; Campos, F.; Sanchez-Porras, D.; Oyonarte, S.; Oruezabal, R.I.; Campos, A.; Martin-Piedra, M.A.; Alaminos, M. Evaluation of Myopic Cornea Lenticules. A Histochemical and Clinical Correlation. Exp. Eye Res. 2020, 196, 108066. [Google Scholar] [CrossRef] [PubMed]
- Carriel, V.; Garzón, I.; Campos, A.; Cornelissen, M.; Alaminos, M. Differential Expression of GAP-43 and Neurofilament during Peripheral Nerve Regeneration through Bio-Artificial Conduits. J. Tissue Eng. Regen. Med. 2017, 11, 553–563. [Google Scholar] [CrossRef]
- Crapo, P.M.; Gilbert, T.W.; Badylak, S.F. An Overview of Tissue and Whole Organ Decellularization Processes. Biomaterials 2011, 32, 3233–3243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Samsudin, E.Z.; Kamarul, T. The Comparison between the Different Generations of Autologous Chondrocyte Implantation with Other Treatment Modalities: A Systematic Review of Clinical Trials. Knee Surg. Sports Traumatol. Arthrosc. 2016, 24, 3912–3926. [Google Scholar] [CrossRef]
- Lim, T.; Tang, Q.; Zhu, Z.-Z.; Feng, Y.; Zhan, S.; Wei, X.-J.; Zhang, C.-Q. A Decellularized Scaffold Derived from Squid Cranial Cartilage for Use in Cartilage Tissue Engineering. J. Mater. Chem. B 2020, 8, 4516–4526. [Google Scholar] [CrossRef]
- Atef, M.; Ojagh, S.M.; Latifi, A.M.; Esmaeili, M.; Udenigwe, C.C. Biochemical and Structural Characterization of Sturgeon Fish Skin Collagen (Huso Huso). J. Food Biochem. 2020, 44, e13256. [Google Scholar] [CrossRef]
- Mredha, M.T.I.; Kitamura, N.; Nonoyama, T.; Wada, S.; Goto, K.; Zhang, X.; Nakajima, T.; Kurokawa, T.; Takagi, Y.; Yasuda, K.; et al. Anisotropic Tough Double Network Hydrogel from Fish Collagen and Its Spontaneous in vivo Bonding to Bone. Biomaterials 2017, 132, 85–95. [Google Scholar] [CrossRef]
- Mredha, M.T.I.; Zhang, X.; Nonoyama, T.; Nakajima, T.; Kurokawa, T.; Takagi, Y.; Gong, J.P. Swim Bladder Collagen Forms Hydrogel with Macroscopic Superstructure by Diffusion Induced Fast Gelation. J. Mater. Chem. B 2015, 3, 7658–7666. [Google Scholar] [CrossRef]
- Hung, S.S.O. Recent Advances in Sturgeon Nutrition. Anim. Nutr. 2017, 3, 191–204. [Google Scholar] [CrossRef] [PubMed]
- Viegas, C.S.B.; Simes, D.C.; Laizé, V.; Williamson, M.K.; Price, P.A.; Cancela, M.L. Gla-Rich Protein (GRP), a New Vitamin K-Dependent Protein Identified from Sturgeon Cartilage and Highly Conserved in Vertebrates. J. Biol. Chem. 2008, 283, 36655–36664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.B.; Gadsbøll, V.; Whittaker, J.; Barton, B.A.; Conlon, J.M. Gastroenteropancreatic Hormones (Insulin, Glucagon, Somatostatin, and Multiple Forms of PYY) from the Pallid Sturgeon, Scaphirhynchus Albus (Acipenseriformes). Gen. Comp. Endocrinol. 2000, 120, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Howe, K.; Clark, M.D.; Torroja, C.F.; Torrance, J.; Berthelot, C.; Muffato, M.; Collins, J.E.; Humphray, S.; McLaren, K.; Matthews, L.; et al. The Zebrafish Reference Genome Sequence and Its Relationship to the Human Genome. Nature 2013, 496, 498–503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuhlmann, C.; Schenck, T.L.; Tluczynski, K.; Aszodi, A.; Metzger, P.; Giunta, R.; Wiggenhauser, P.S. Experimental Approach to Nasal Septal Cartilage Regeneration with Adipose Tissue-Derived Stem Cells and Decellularized Porcine Septal Cartilage. Xenotransplantation 2020, 20, e12660. [Google Scholar] [CrossRef]
- Xia, C.; Mei, S.; Gu, C.; Zheng, L.; Fang, C.; Shi, Y.; Wu, K.; Lu, T.; Jin, Y.; Lin, X.; et al. Decellularized Cartilage as a Prospective Scaffold for Cartilage Repair. Mater. Sci. Eng. C 2019, 101, 588–595. [Google Scholar] [CrossRef] [PubMed]
- Badylak, S.F.; Freytes, D.O.; Gilbert, T.W. Extracellular Matrix as a Biological Scaffold Material: Structure and Function. Acta Biomater. 2009, 5, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Terry, D.E.; Chopra, R.K.; Ovenden, J.; Anastassiades, T.P. Differential Use of Alcian Blue and Toluidine Blue Dyes for the Quantification and Isolation of Anionic Glycoconjugates from Cell Cultures: Application to Proteoglycans and a High-Molecular-Weight Glycoprotein Synthesized by Articular Chondrocytes. Anal. Biochem. 2000, 285, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Corrao, S.; La Rocca, G.; Lo Iacono, M.; Corsello, T.; Farina, F.; Anzalone, R. Umbilical Cord Revisited: From Wharton’s Jelly Myofibroblasts to Mesenchymal Stem Cells. Histol. Histopathol. 2013, 28, 1235–1244. [Google Scholar] [CrossRef] [PubMed]
- Main, B.J.; Valk, J.A.; Maffulli, N.; Rodriguez, H.C.; Gupta, M.; Stone, I.W.; El-Amin, S.F.; Gupta, A. Umbilical Cord-Derived Wharton’s Jelly for Regenerative Medicine Applications in Orthopedic Surgery: A Systematic Review Protocol. J. Orthop. Surg. 2020, 15, s13018–s13020. [Google Scholar] [CrossRef] [PubMed]
- Xiao, T.; Guo, W.; Chen, M.; Hao, C.; Gao, S.; Huang, J.; Yuan, Z.; Zhang, Y.; Wang, M.; Li, P.; et al. Fabrication and In Vitro Study of Tissue-Engineered Cartilage Scaffold Derived from Wharton’s Jelly Extracellular Matrix. BioMed Res. Int. 2017, 2017, 5839071. [Google Scholar] [CrossRef]
- Paduszyński, P.; Aleksander-Konert, E.; Zajdel, A.; Wilczok, A.; Jelonek, K.; Witek, A.; Dzierżewicz, Z. Changes in Expression of Cartilaginous Genes during Chondrogenesis of Wharton’s Jelly Mesenchymal Stem Cells on Three-Dimensional Biodegradable Poly(L-Lactide-Co-Glycolide) Scaffolds. Cell. Mol. Biol. Lett. 2016, 21, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gallo, V.; Cirillo, E.; Giardino, G.; Pignata, C. FOXN1 Deficiency: From the Discovery to Novel Therapeutic Approaches. J. Clin. Immunol. 2017, 37, 751–758. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharp, A.K.; Colston, M.J. Elevated Macrophage Activity in Nude Mice. Pathobiology 1984, 52, 44–47. [Google Scholar] [CrossRef]
- Nash, T.E. Antigenic Variation in Giardia Lamblia and the Host’s Immune Response. Philos. Trans. R Soc. Lond. B. Biol. Sci. 1997, 352, 1369–1375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mariani, E.; Lisignoli, G.; Borzì, R.M.; Pulsatelli, L. Biomaterials: Foreign Bodies or Tuners for the Immune Response? Int. J. Mol. Sci. 2019, 20, 636. [Google Scholar] [CrossRef] [Green Version]
- Klopfleisch, R. Macrophage Reaction against Biomaterials in the Mouse Model—Phenotypes, Functions and Markers. Acta Biomater. 2016, 43, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Humberto Valencia, C. Hydrolytic Degradation and in Vivo Resorption of Poly-l-Lactic Acid-Chitosan Biomedical Devices in the Parietal Bones of Wistar Rats. J. Int. Med. Res. 2019, 47, 1705–1716. [Google Scholar] [CrossRef] [Green Version]
- Nenna, R.; Turchetti, A.; Mastrogiorgio, G.; Midulla, F. COL2A1 Gene Mutations: Mechanisms of Spondyloepiphyseal Dysplasia Congenita. Appl. Clin. Genet. 2019, 12, 235–238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kabir, W.; Di Bella, C.; Choong, P.F.M.; O’Connell, C.D. Assessment of Native Human Articular Cartilage: A Biomechanical Protocol. Cartilage 2020, 1947603520973240. [Google Scholar] [CrossRef]
- Schuurman, W.; Klein, T.J.; Dhert, W.J.A.; van Weeren, P.R.; Hutmacher, D.W.; Malda, J. Cartilage Regeneration Using Zonal Chondrocyte Subpopulations: A Promising Approach or an Overcomplicated Strategy? J. Tissue Eng. Regen. Med. 2015, 9, 669–678. [Google Scholar] [CrossRef] [PubMed] [Green Version]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortiz-Arrabal, O.; Carmona, R.; García-García, Ó.-D.; Chato-Astrain, J.; Sánchez-Porras, D.; Domezain, A.; Oruezabal, R.-I.; Carriel, V.; Campos, A.; Alaminos, M. Generation and Evaluation of Novel Biomaterials Based on Decellularized Sturgeon Cartilage for Use in Tissue Engineering. Biomedicines 2021, 9, 775. https://doi.org/10.3390/biomedicines9070775
Ortiz-Arrabal O, Carmona R, García-García Ó-D, Chato-Astrain J, Sánchez-Porras D, Domezain A, Oruezabal R-I, Carriel V, Campos A, Alaminos M. Generation and Evaluation of Novel Biomaterials Based on Decellularized Sturgeon Cartilage for Use in Tissue Engineering. Biomedicines. 2021; 9(7):775. https://doi.org/10.3390/biomedicines9070775
Chicago/Turabian StyleOrtiz-Arrabal, Olimpia, Ramón Carmona, Óscar-Darío García-García, Jesús Chato-Astrain, David Sánchez-Porras, Alberto Domezain, Roke-Iñaki Oruezabal, Víctor Carriel, Antonio Campos, and Miguel Alaminos. 2021. "Generation and Evaluation of Novel Biomaterials Based on Decellularized Sturgeon Cartilage for Use in Tissue Engineering" Biomedicines 9, no. 7: 775. https://doi.org/10.3390/biomedicines9070775
APA StyleOrtiz-Arrabal, O., Carmona, R., García-García, Ó.-D., Chato-Astrain, J., Sánchez-Porras, D., Domezain, A., Oruezabal, R.-I., Carriel, V., Campos, A., & Alaminos, M. (2021). Generation and Evaluation of Novel Biomaterials Based on Decellularized Sturgeon Cartilage for Use in Tissue Engineering. Biomedicines, 9(7), 775. https://doi.org/10.3390/biomedicines9070775







