Interactions between Autophagy, Proinflammatory Cytokines, and Apoptosis in Neuropathic Pain: Granulocyte Colony Stimulating Factor as a Multipotent Therapy in Rats with Chronic Constriction Injury
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Surgical Procedure and G-CSF Treatment
2.3. Behavioral Tests for Mechanical Allodynia
2.4. Western Blotting
2.5. Immunohistochemistry
2.6. Statistical Analyses
3. Results
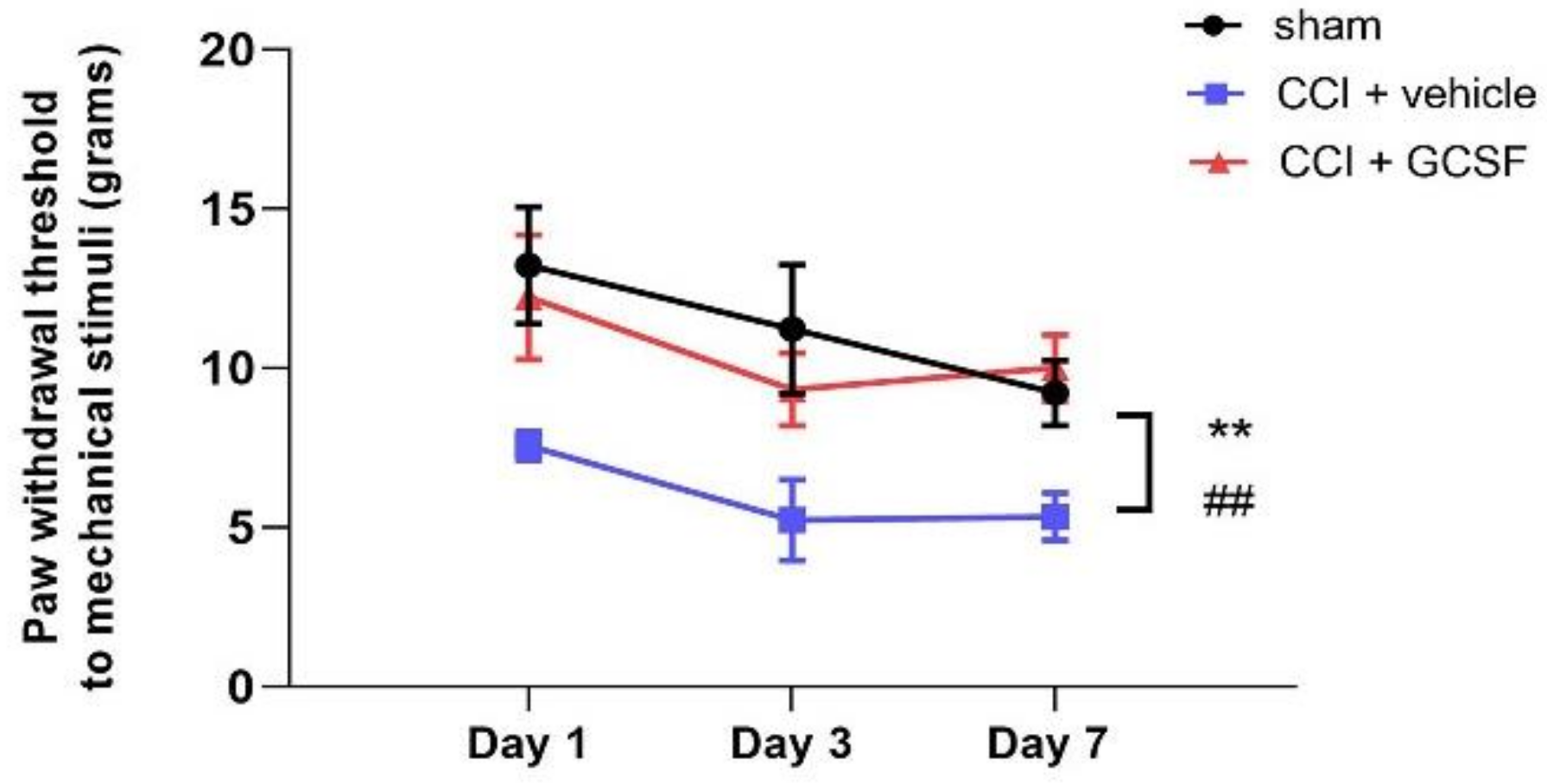
3.1. Early Systemic G-CSF Treatment Alleviated Mechanical Allodynia in CCI Rats from the 1st to the 7th Day after Nerve Injury
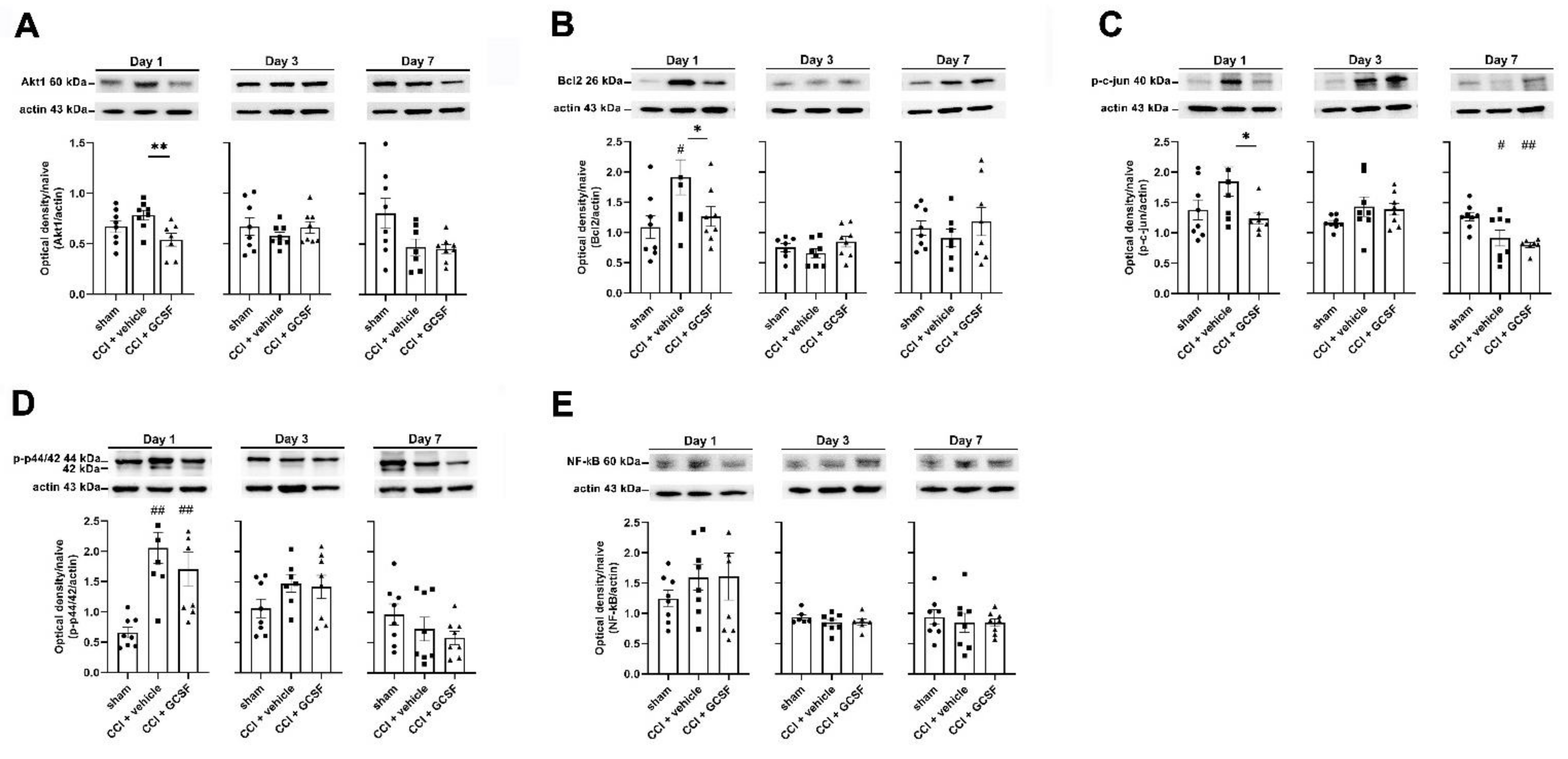
3.2. G-CSF Downregulated Akt1, Bcl-2, and Phospho-c-Jun Protein Expression but Did Not Alter Phospho-p44/42 or Nuclear Factor Kappa B (NF-κB) Protein Expression in the Injured Sciatic Nerve on the 1st Day after Nerve Injury
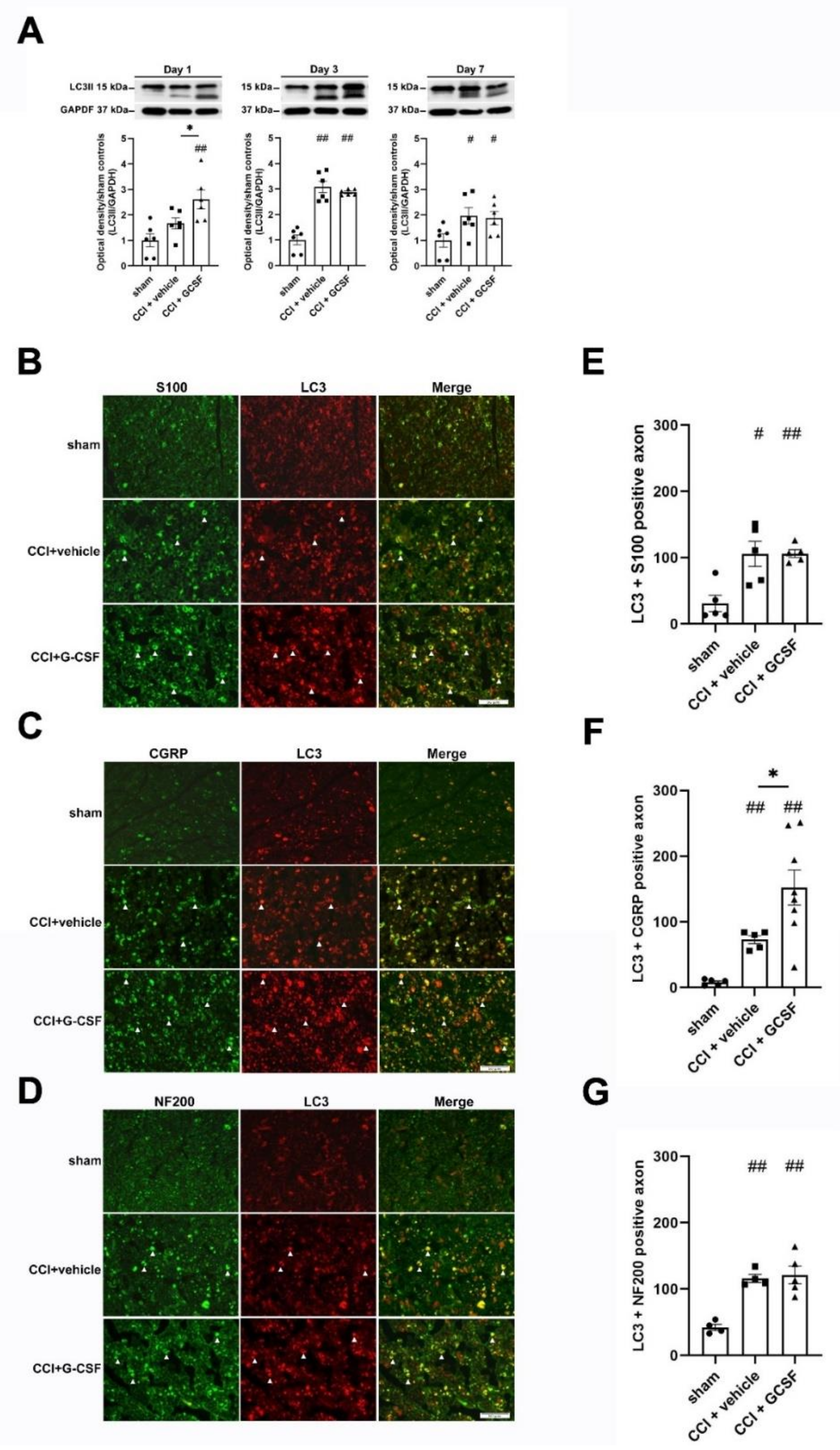
3.3. G-CSF Treatment Upregulated Autophagic Activity in the Injured Sciatic Nerve and DRG Neurons at the Early Phase (1st Day) after Nerve Injury
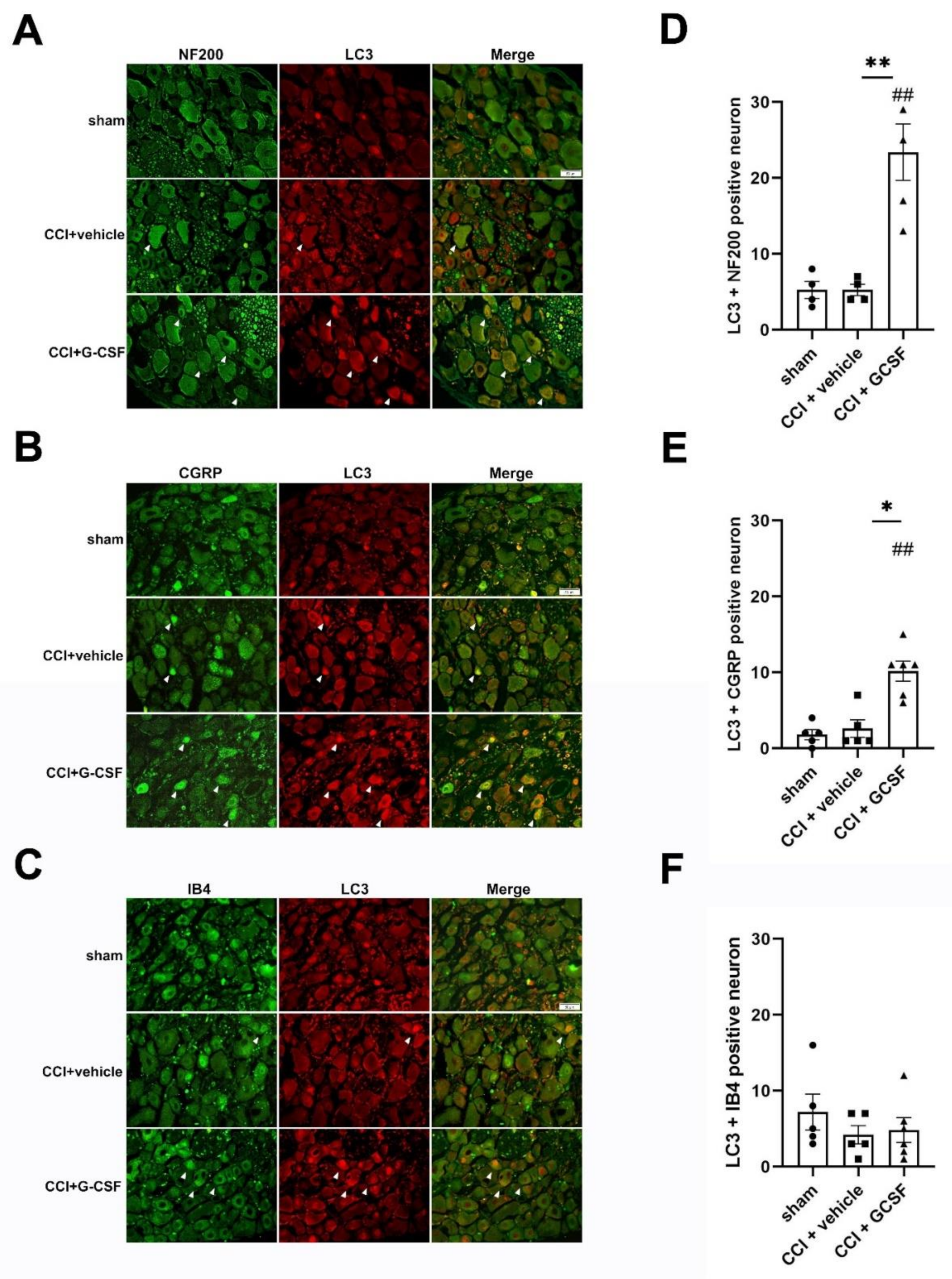
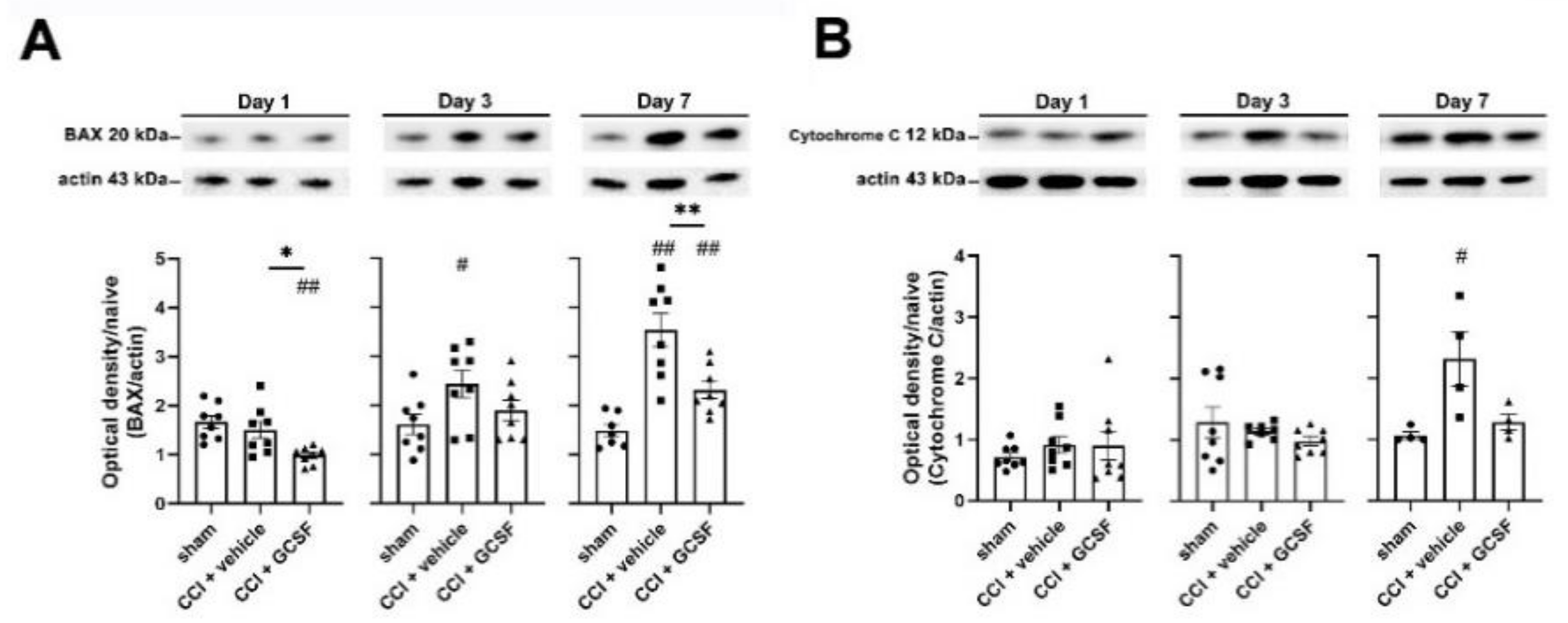
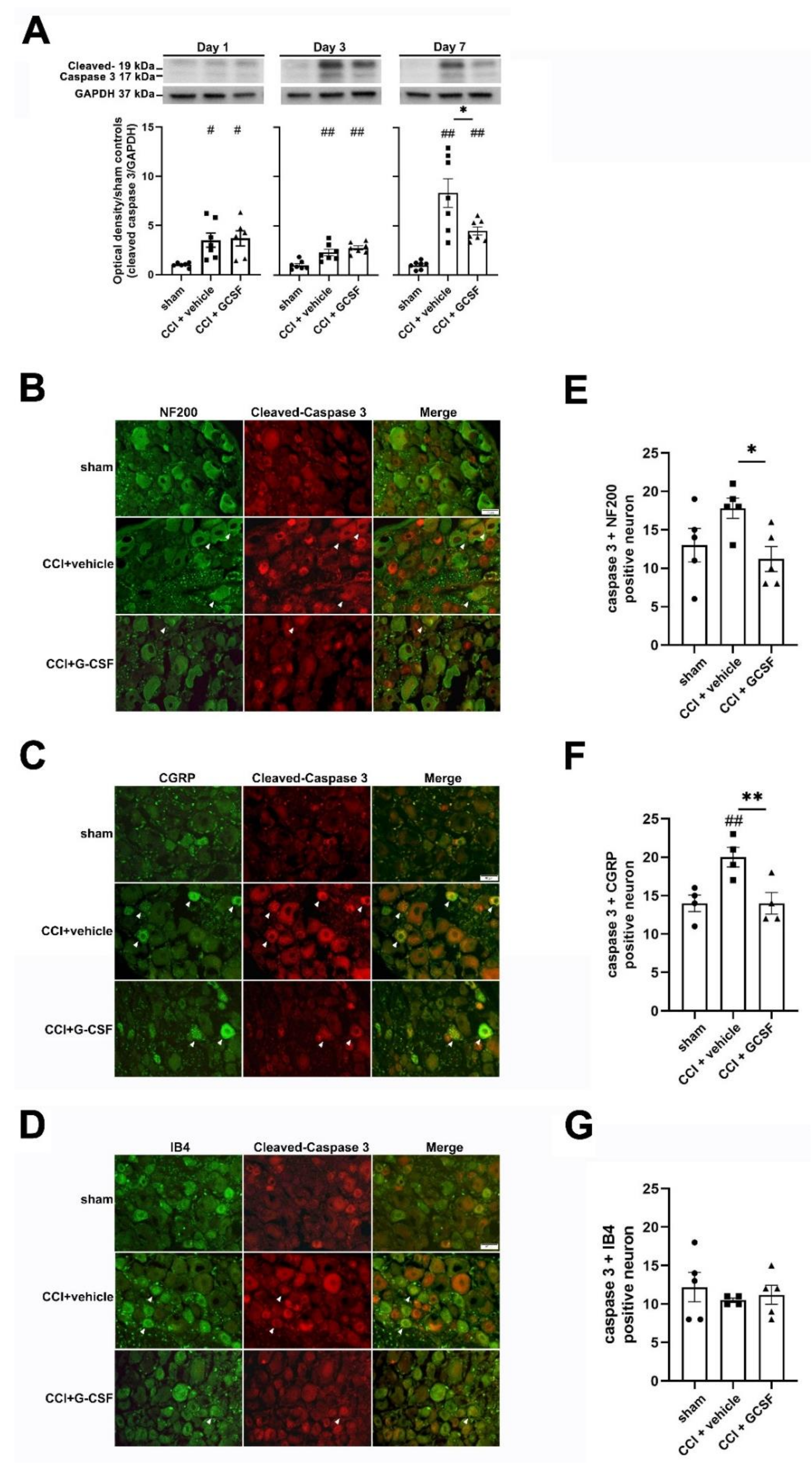
3.4. G-CSF Treatment Suppressed Apoptotic Activity in the Injured Sciatic Nerve and DRG Neurons in the Late Phase (7 Days) after Nerve Injury
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chao, P.K.; Lu, K.T.; Lee, Y.L.; Chen, J.C.; Wang, H.L.; Yang, Y.L.; Cheng, M.Y.; Liao, M.F.; Ro, L.S. Early systemic granulocyte-colony stimulating factor treatment attenuates neuropathic pain after peripheral nerve injury. PLoS ONE 2012, 7, e43680. [Google Scholar] [CrossRef] [PubMed]
- Liao, M.F.; Yeh, S.R.; Lo, A.L.; Chao, P.K.; Lee, Y.L.; Hung, Y.H.; Lu, K.T.; Ro, L.S. An early granulocyte colony-stimulating factor treatment attenuates neuropathic pain through activation of mu opioid receptors on the injured nerve. Sci. Rep. 2016, 6, 25490. [Google Scholar] [CrossRef] [PubMed]
- Koda, M.; Furuya, T.; Kato, K.; Mannoji, C.; Hashimoto, M.; Inada, T.; Kamiya, K.; Ota, M.; Maki, S.; Okawa, A.; et al. Delayed granulocyte colony-stimulating factor treatment in rats attenuates mechanical allodynia induced by chronic constriction injury of the sciatic nerve. Spine 2014, 39, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Kato, K.; Koda, M.; Takahashi, H.; Sakuma, T.; Inada, T.; Kamiya, K.; Ota, M.; Maki, S.; Okawa, A.; Takahashi, K.; et al. Granulocyte colony-stimulating factor attenuates spinal cord injury-induced mechanical allodynia in adult rats. J. Neurol. Sci. 2015, 355, 79–83. [Google Scholar] [CrossRef]
- Liao, M.F.; Hsu, J.L.; Lu, K.T.; Chao, P.K.; Cheng, M.Y.; Hsu, H.C.; Lo, A.L.; Lee, Y.L.; Hung, Y.H.; Lyu, R.K.; et al. Granulocyte Colony Stimulating Factor (GCSF) Can Attenuate Neuropathic Pain by Suppressing Monocyte Chemoattractant Protein-1 (MCP-1) Expression, through Upregulating the Early MicroRNA-122 Expression in the Dorsal Root Ganglia. Cells 2020, 9, 1669. [Google Scholar] [CrossRef]
- Yamazaki, M.; Sakuma, T.; Kato, K.; Furuya, T.; Koda, M. Granulocyte colony-stimulating factor reduced neuropathic pain associated with thoracic compression myelopathy: Report of two cases. J. Spinal Cord Med. 2013, 36, 40–43. [Google Scholar] [CrossRef][Green Version]
- Kato, K.; Yamazaki, M.; Okawa, A.; Furuya, T.; Sakuma, T.; Takahashi, H.; Kamiya, K.; Inada, T.; Takahashi, K.; Koda, M. Intravenous administration of granulocyte colony-stimulating factor for treating neuropathic pain associated with compression myelopathy: A phase I and IIa clinical trial. Eur. Spine J. 2013, 22, 197–204. [Google Scholar] [CrossRef]
- Anderlini, P.; Przepiorka, D.; Champlin, R.; Korbling, M. Biologic and clinical effects of granulocyte colony-stimulating factor in normal individuals. Blood 1996, 88, 2819–2825. [Google Scholar] [CrossRef]
- Guo, Y.; Liu, S.; Zhang, X.; Wang, L.; Gao, J.; Han, A.; Hao, A. G-CSF promotes autophagy and reduces neural tissue damage after spinal cord injury in mice. Lab. Investig. 2015, 95, 1439–1449. [Google Scholar] [CrossRef]
- Solaroglu, I.; Cahill, J.; Jadhav, V.; Zhang, J.H. A novel neuroprotectant granulocyte-colony stimulating factor. Stroke 2006, 37, 1123–1128. [Google Scholar] [CrossRef]
- Schneider, A.; Kruger, C.; Steigleder, T.; Weber, D.; Pitzer, C.; Laage, R.; Aronowski, J.; Maurer, M.H.; Gassler, N.; Mier, W.; et al. The hematopoietic factor G-CSF is a neuronal ligand that counteracts programmed cell death and drives neurogenesis. J. Clin. Investig. 2005, 115, 2083–2098. [Google Scholar] [CrossRef]
- Komine-Kobayashi, M.; Zhang, N.; Liu, M.; Tanaka, R.; Hara, H.; Osaka, A.; Mochizuki, H.; Mizuno, Y.; Urabe, T. Neuroprotective effect of recombinant human granulocyte colony-stimulating factor in transient focal ischemia of mice. J. Cereb. Blood Flow Metab. 2006, 26, 402–413. [Google Scholar] [CrossRef]
- Modi, J.; Menzie-Suderam, J.; Xu, H.; Trujillo, P.; Medley, K.; Marshall, M.L.; Tao, R.; Prentice, H.; Wu, J.Y. Mode of action of granulocyte-colony stimulating factor (G-CSF) as a novel therapy for stroke in a mouse model. J. Biomed. Sci. 2020, 27, 19. [Google Scholar] [CrossRef]
- Menzie-Suderam, J.M.; Modi, J.; Xu, H.; Bent, A.; Trujillo, P.; Medley, K.; Jimenez, E.; Shen, J.; Marshall, M.; Tao, R.; et al. Granulocyte-colony stimulating factor gene therapy as a novel therapeutics for stroke in a mouse model. J. Biomed. Sci. 2020, 27, 99. [Google Scholar] [CrossRef]
- Leveque-El Mouttie, L.; Vu, T.; Lineburg, K.E.; Kuns, R.D.; Bagger, F.O.; Teal, B.E.; Lor, M.; Boyle, G.M.; Bruedigam, C.; Mintern, J.D.; et al. Autophagy is required for stem cell mobilization by G-CSF. Blood 2015, 125, 2933–2936. [Google Scholar] [CrossRef]
- Van Raam, B.J.; Drewniak, A.; Groenewold, V.; van den Berg, T.K.; Kuijpers, T.W. Granulocyte colony-stimulating factor delays neutrophil apoptosis by inhibition of calpains upstream of caspase-3. Blood 2008, 112, 2046–2054. [Google Scholar] [CrossRef]
- Glick, D.; Barth, S.; Macleod, K.F. Autophagy: Cellular and molecular mechanisms. J. Pathol. 2010, 221, 3–12. [Google Scholar] [CrossRef]
- Khandia, R.; Dadar, M.; Munjal, A.; Dhama, K.; Karthik, K.; Tiwari, R.; Yatoo, M.I.; Iqbal, H.M.N.; Singh, K.P.; Joshi, S.K.; et al. A Comprehensive Review of Autophagy and Its Various Roles in Infectious, Non-Infectious, and Lifestyle Diseases: Current Knowledge and Prospects for Disease Prevention, Novel Drug Design, and Therapy. Cells 2019, 8, 674. [Google Scholar] [CrossRef]
- Mizushima, N.; Levine, B. Autophagy in Human Diseases. N. Engl. J. Med. 2020, 383, 1564–1576. [Google Scholar] [CrossRef]
- Liu, X.; Zhu, M.; Ju, Y.; Li, A.; Sun, X. Autophagy dysfunction in neuropathic pain. Neuropeptides 2019, 75, 41–48. [Google Scholar] [CrossRef]
- Berliocchi, L.; Russo, R.; Maiaru, M.; Levato, A.; Bagetta, G.; Corasaniti, M.T. Autophagy impairment in a mouse model of neuropathic pain. Mol. Pain 2011, 7, 83. [Google Scholar] [CrossRef]
- Kosacka, J.; Nowicki, M.; Bluher, M.; Baum, P.; Stockinger, M.; Toyka, K.V.; Kloting, I.; Stumvoll, M.; Serke, H.; Bechmann, I.; et al. Increased autophagy in peripheral nerves may protect Wistar Ottawa Karlsburg W rats against neuropathy. Exp. Neurol. 2013, 250, 125–135. [Google Scholar] [CrossRef]
- Shi, G.; Shi, J.; Liu, K.; Liu, N.; Wang, Y.; Fu, Z.; Ding, J.; Jia, L.; Yuan, W. Increased miR-195 aggravates neuropathic pain by inhibiting autophagy following peripheral nerve injury. Glia 2013, 61, 504–512. [Google Scholar] [CrossRef]
- Marinelli, S.; Nazio, F.; Tinari, A.; Ciarlo, L.; D’Amelio, M.; Pieroni, L.; Vacca, V.; Urbani, A.; Cecconi, F.; Malorni, W.; et al. Schwann cell autophagy counteracts the onset and chronification of neuropathic pain. Pain 2014, 155, 93–107. [Google Scholar] [CrossRef]
- Guo, J.S.; Jing, P.B.; Wang, J.A.; Zhang, R.; Jiang, B.C.; Gao, Y.J.; Zhang, Z.J. Increased autophagic activity in dorsal root ganglion attenuates neuropathic pain following peripheral nerve injury. Neurosci. Lett. 2015, 599, 158–163. [Google Scholar] [CrossRef]
- Xie, X.; Ma, L.; Xi, K.; Zhang, W.; Fan, D. MicroRNA-183 Suppresses Neuropathic Pain and Expression of AMPA Receptors by Targeting mTOR/VEGF Signaling Pathway. Cell. Physiol. Biochem. 2017, 41, 181–192. [Google Scholar] [CrossRef]
- Piao, Y.; Gwon, D.H.; Kang, D.W.; Hwang, T.W.; Shin, N.; Kwon, H.H.; Shin, H.J.; Yin, Y.; Kim, J.J.; Hong, J.; et al. TLR4-mediated autophagic impairment contributes to neuropathic pain in chronic constriction injury mice. Mol. Brain 2018, 11, 11. [Google Scholar] [CrossRef]
- Weng, W.; Yao, C.; Poonit, K.; Zhou, X.; Sun, C.; Zhang, F.; Yan, H. Metformin relieves neuropathic pain after spinal nerve ligation via autophagy flux stimulation. J. Cell. Mol. Med. 2019, 23, 1313–1324. [Google Scholar] [CrossRef]
- Liu, Y.D.; Wang, Z.B.; Han, G.; Jin, L.; Zhao, P. Hyperbaric oxygen relieves neuropathic pain through AKT/TSC2/mTOR pathway activity to induce autophagy. J. Pain Res. 2019, 12, 443–451. [Google Scholar] [CrossRef]
- Cai, W.; Zhang, Y.; Su, Z. ciRS-7 targeting miR-135a-5p promotes neuropathic pain in CCI rats via inflammation and autophagy. Gene 2020, 736, 144386. [Google Scholar] [CrossRef]
- Sekiguchi, M.; Sekiguchi, Y.; Konno, S.; Kobayashi, H.; Homma, Y.; Kikuchi, S. Comparison of neuropathic pain and neuronal apoptosis following nerve root or spinal nerve compression. Eur. Spine J. 2009, 18, 1978–1985. [Google Scholar] [CrossRef] [PubMed]
- Wiberg, R.; Novikova, L.N.; Kingham, P.J. Evaluation of apoptotic pathways in dorsal root ganglion neurons following peripheral nerve injury. Neuroreport 2018, 29, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Schaeffer, V.; Meyer, L.; Patte-Mensah, C.; Eckert, A.; Mensah-Nyagan, A.G. Sciatic nerve injury induces apoptosis of dorsal root ganglion satellite glial cells and selectively modifies neurosteroidogenesis in sensory neurons. Glia 2010, 58, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, S.; Stevens, M.; Wiley, J.W. Diabetic peripheral neuropathy: Evidence for apoptosis and associated mitochondrial dysfunction. Diabetes 2000, 49, 1932–1938. [Google Scholar] [CrossRef]
- Campana, W.M.; Myers, R.R. Exogenous erythropoietin protects against dorsal root ganglion apoptosis and pain following peripheral nerve injury. Eur. J. Neurosci. 2003, 18, 1497–1506. [Google Scholar] [CrossRef]
- Amin, B.; Abnous, K.; Motamedshariaty, V.; Hosseinzadeh, H. Attenuation of oxidative stress, inflammation and apoptosis by ethanolic and aqueous extracts of Crocus sativus L. stigma after chronic constriction injury of rats. An. Acad. Bras. Ciências 2014, 86, 1821–1832. [Google Scholar] [CrossRef]
- Fu, H.; Li, F.; Thomas, S.; Yang, Z. Hyperbaric oxygenation alleviates chronic constriction injury (CCI)-induced neuropathic pain and inhibits GABAergic neuron apoptosis in the spinal cord. Scand. J. Pain 2017, 17, 330–338. [Google Scholar] [CrossRef]
- Hu, Q.; Fang, L.; Li, F.; Thomas, S.; Yang, Z. Hyperbaric oxygenation treatment alleviates CCI-induced neuropathic pain and decreases spinal apoptosis. Eur. J. Pain 2015, 19, 920–928. [Google Scholar] [CrossRef]
- Bennett, G.J.; Xie, Y.K. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 1988, 33, 87–107. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Guo, N.; Peng, G.; Wang, X.; Han, M.; Raincrow, J.; Chiu, C.H.; Coolen, L.M.; Wenthold, R.J.; Zhao, Z.Q.; et al. Role of SIP30 in the development and maintenance of peripheral nerve injury-induced neuropathic pain. Pain 2009, 146, 130–140. [Google Scholar] [CrossRef]
- Ciaccio, M.F.; Wagner, J.P.; Chuu, C.P.; Lauffenburger, D.A.; Jones, R.B. Systems analysis of EGF receptor signaling dynamics with microwestern arrays. Nat. Methods 2010, 7, 148–155. [Google Scholar] [CrossRef]
- Gomez-Sanchez, J.A.; Carty, L.; Iruarrizaga-Lejarreta, M.; Palomo-Irigoyen, M.; Varela-Rey, M.; Griffith, M.; Hantke, J.; Macias-Camara, N.; Azkargorta, M.; Aurrekoetxea, I.; et al. Schwann cell autophagy, myelinophagy, initiates myelin clearance from injured nerves. J. Cell Biol. 2015, 210, 153–168. [Google Scholar] [CrossRef]
- Ma, Z.; Han, Q.; Wang, X.; Ai, Z.; Zheng, Y. Galectin-3 Inhibition Is Associated with Neuropathic Pain Attenuation after Peripheral Nerve Injury. PLoS ONE 2016, 11, e0148792. [Google Scholar] [CrossRef]
- Scheubel, R.J.; Bartling, B.; Simm, A.; Silber, R.E.; Drogaris, K.; Darmer, D.; Holtz, J. Apoptotic pathway activation from mitochondria and death receptors without caspase-3 cleavage in failing human myocardium: Fragile balance of myocyte survival? J. Am. Coll. Cardiol. 2002, 39, 481–488. [Google Scholar] [CrossRef]
- White, K.; Dempsie, Y.; Caruso, P.; Wallace, E.; McDonald, R.A.; Stevens, H.; Hatley, M.E.; Van Rooij, E.; Morrell, N.W.; MacLean, M.R.; et al. Endothelial apoptosis in pulmonary hypertension is controlled by a microRNA/programmed cell death 4/caspase-3 axis. Hypertension 2014, 64, 185–194. [Google Scholar] [CrossRef]
- Ge, Y.; Huang, M.; Yao, Y.M. Autophagy and proinflammatory cytokines: Interactions and clinical implications. Cytokine Growth Factor Rev. 2018, 43, 38–46. [Google Scholar] [CrossRef]
- Bussi, C.; Peralta Ramos, J.M.; Arroyo, D.S.; Gaviglio, E.A.; Gallea, J.I.; Wang, J.M.; Celej, M.S.; Iribarren, P. Autophagy down regulates pro-inflammatory mediators in BV2 microglial cells and rescues both LPS and alpha-synuclein induced neuronal cell death. Sci. Rep. 2017, 7, 43153. [Google Scholar] [CrossRef]
- Harris, J. Autophagy and IL-1 Family Cytokines. Front. Immunol. 2013, 4, 83. [Google Scholar] [CrossRef]
- Grace, P.M.; Hutchinson, M.R.; Maier, S.F.; Watkins, L.R. Pathological pain and the neuroimmune interface. Nat. Rev. Immunol. 2014, 14, 217–231. [Google Scholar] [CrossRef]
- Qin, L.; Wang, Z.; Tao, L.; Wang, Y. ER stress negatively regulates AKT/TSC/mTOR pathway to enhance autophagy. Autophagy 2010, 6, 239–247. [Google Scholar] [CrossRef]
- Zhao, L.; Zhu, Y.; Wang, D.; Chen, M.; Gao, P.; Xiao, W.; Rao, G.; Wang, X.; Jin, H.; Xu, L.; et al. Morphine induces Beclin 1- and ATG5-dependent autophagy in human neuroblastoma SH-SY5Y cells and in the rat hippocampus. Autophagy 2010, 6, 386–394. [Google Scholar] [CrossRef]
- Wan, J.; Ma, J.; Anand, V.; Ramakrishnan, S.; Roy, S. Morphine potentiates LPS-induced autophagy initiation but inhibits autophagosomal maturation through distinct TLR4-dependent and independent pathways. Acta Physiol. 2015, 214, 189–199. [Google Scholar] [CrossRef]
- Scholz, J.; Broom, D.C.; Youn, D.H.; Mills, C.D.; Kohno, T.; Suter, M.R.; Moore, K.A.; Decosterd, I.; Coggeshall, R.E.; Woolf, C.J. Blocking caspase activity prevents transsynaptic neuronal apoptosis and the loss of inhibition in lamina II of the dorsal horn after peripheral nerve injury. J. Neurosci. 2005, 25, 7317–7323. [Google Scholar] [CrossRef]
- Joseph, E.K.; Levine, J.D. Caspase signalling in neuropathic and inflammatory pain in the rat. Eur. J. Neurosci. 2004, 20, 2896–2902. [Google Scholar] [CrossRef]
- Jacobs, J.M.; Ro, L.S. A morphological study of experimental mononeuropathy in the rat: Early ischemic changes. J. Neurol. Sci. 1994, 127, 143–152. [Google Scholar] [CrossRef]
- Maiuri, M.C.; Zalckvar, E.; Kimchi, A.; Kroemer, G. Self-eating and self-killing: Crosstalk between autophagy and apoptosis. Nat. Rev. Mol. Cell Biol. 2007, 8, 741–752. [Google Scholar] [CrossRef]
- Gump, J.M.; Thorburn, A. Autophagy and apoptosis: What is the connection? Trends Cell Biol. 2011, 21, 387–392. [Google Scholar] [CrossRef]
- Ye, L.; Huang, Y.; Zhao, L.; Li, Y.; Sun, L.; Zhou, Y.; Qian, G.; Zheng, J.C. IL-1beta and TNF-alpha induce neurotoxicity through glutamate production: A potential role for neuronal glutaminase. J. Neurochem. 2013, 125, 897–908. [Google Scholar] [CrossRef] [PubMed]
- Robertson, J.; Beaulieu, J.M.; Doroudchi, M.M.; Durham, H.D.; Julien, J.P.; Mushynski, W.E. Apoptotic death of neurons exhibiting peripherin aggregates is mediated by the proinflammatory cytokine tumor necrosis factor-alpha. J. Cell. Biol. 2001, 155, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Baldo, M.P.; Davel, A.P.; Damas-Souza, D.M.; Nicoletti-Carvalho, J.E.; Bordin, S.; Carvalho, H.F.; Rodrigues, S.L.; Rossoni, L.V.; Mill, J.G. The antiapoptotic effect of granulocyte colony-stimulating factor reduces infarct size and prevents heart failure development in rats. Cell. Physiol. Biochem. 2011, 28, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.H.; Lim, Y.H.; Song, Y.S.; So, B.I.; Park, J.Y.; Fang, C.H.; Lee, Y.; Kim, H.; Kim, K.S. Granulocyte-colony stimulating factor reduces cardiomyocyte apoptosis and ameliorates diastolic dysfunction in Otsuka Long-Evans Tokushima Fatty rats. Cardiovasc. Drugs Ther. 2014, 28, 211–220. [Google Scholar] [CrossRef]
- Baez, A.; Martin-Antonio, B.; Piruat, J.I.; Prats, C.; Alvarez-Laderas, I.; Barbado, M.V.; Carmona, M.; Urbano-Ispizua, A.; Perez-Simon, J.A. Granulocyte colony-stimulating factor produces long-term changes in gene and microRNA expression profiles in CD34+ cells from healthy donors. Haematologica 2014, 99, 243–251. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, Z.; Zhang, W.; Zhang, L. MicroRNAs play an essential role in autophagy regulation in various disease phenotypes. Biofactors 2019, 45, 844–856. [Google Scholar] [CrossRef]
- Xu, J.; Wang, Y.; Tan, X.; Jing, H. MicroRNAs in autophagy and their emerging roles in crosstalk with apoptosis. Autophagy 2012, 8, 873–882. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, J.; Liu, W.; Wang, H.; Zhao, L.; Liu, S.; Li, P.; Zhang, S.; Sun, C.; Wu, Y.; et al. microRNA-378 promotes autophagy and inhibits apoptosis in skeletal muscle. Proc. Natl. Acad. Sci. USA 2018, 115, E10849–E10858. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, H.; Guo, M.; Fei, D.; Zhang, L.; Xing, M. Targeting the miR-122/PKM2 autophagy axis relieves arsenic stress. J. Hazard. Mater. 2020, 383, 121217. [Google Scholar] [CrossRef]
 , Mu opioid receptor; ↑: increase; ↓: decrease; →: enhance; ―|: suppress).
, Mu opioid receptor; ↑: increase; ↓: decrease; →: enhance; ―|: suppress).
 , Mu opioid receptor; ↑: increase; ↓: decrease; →: enhance; ―|: suppress).
, Mu opioid receptor; ↑: increase; ↓: decrease; →: enhance; ―|: suppress).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, M.-F.; Yeh, S.-R.; Lu, K.-T.; Hsu, J.-L.; Chao, P.-K.; Hsu, H.-C.; Peng, C.-H.; Lee, Y.-L.; Hung, Y.-H.; Ro, L.-S. Interactions between Autophagy, Proinflammatory Cytokines, and Apoptosis in Neuropathic Pain: Granulocyte Colony Stimulating Factor as a Multipotent Therapy in Rats with Chronic Constriction Injury. Biomedicines 2021, 9, 542. https://doi.org/10.3390/biomedicines9050542
Liao M-F, Yeh S-R, Lu K-T, Hsu J-L, Chao P-K, Hsu H-C, Peng C-H, Lee Y-L, Hung Y-H, Ro L-S. Interactions between Autophagy, Proinflammatory Cytokines, and Apoptosis in Neuropathic Pain: Granulocyte Colony Stimulating Factor as a Multipotent Therapy in Rats with Chronic Constriction Injury. Biomedicines. 2021; 9(5):542. https://doi.org/10.3390/biomedicines9050542
Chicago/Turabian StyleLiao, Ming-Feng, Shin-Rung Yeh, Kwok-Tung Lu, Jung-Lung Hsu, Po-Kuan Chao, Hui-Ching Hsu, Chi-Hao Peng, Yun-Lin Lee, Yu-Hui Hung, and Long-Sun Ro. 2021. "Interactions between Autophagy, Proinflammatory Cytokines, and Apoptosis in Neuropathic Pain: Granulocyte Colony Stimulating Factor as a Multipotent Therapy in Rats with Chronic Constriction Injury" Biomedicines 9, no. 5: 542. https://doi.org/10.3390/biomedicines9050542
APA StyleLiao, M.-F., Yeh, S.-R., Lu, K.-T., Hsu, J.-L., Chao, P.-K., Hsu, H.-C., Peng, C.-H., Lee, Y.-L., Hung, Y.-H., & Ro, L.-S. (2021). Interactions between Autophagy, Proinflammatory Cytokines, and Apoptosis in Neuropathic Pain: Granulocyte Colony Stimulating Factor as a Multipotent Therapy in Rats with Chronic Constriction Injury. Biomedicines, 9(5), 542. https://doi.org/10.3390/biomedicines9050542






