Updates on Anticoagulation and Laboratory Tools for Therapy Monitoring of Heparin, Vitamin K Antagonists and Direct Oral Anticoagulants
Abstract
1. Introduction
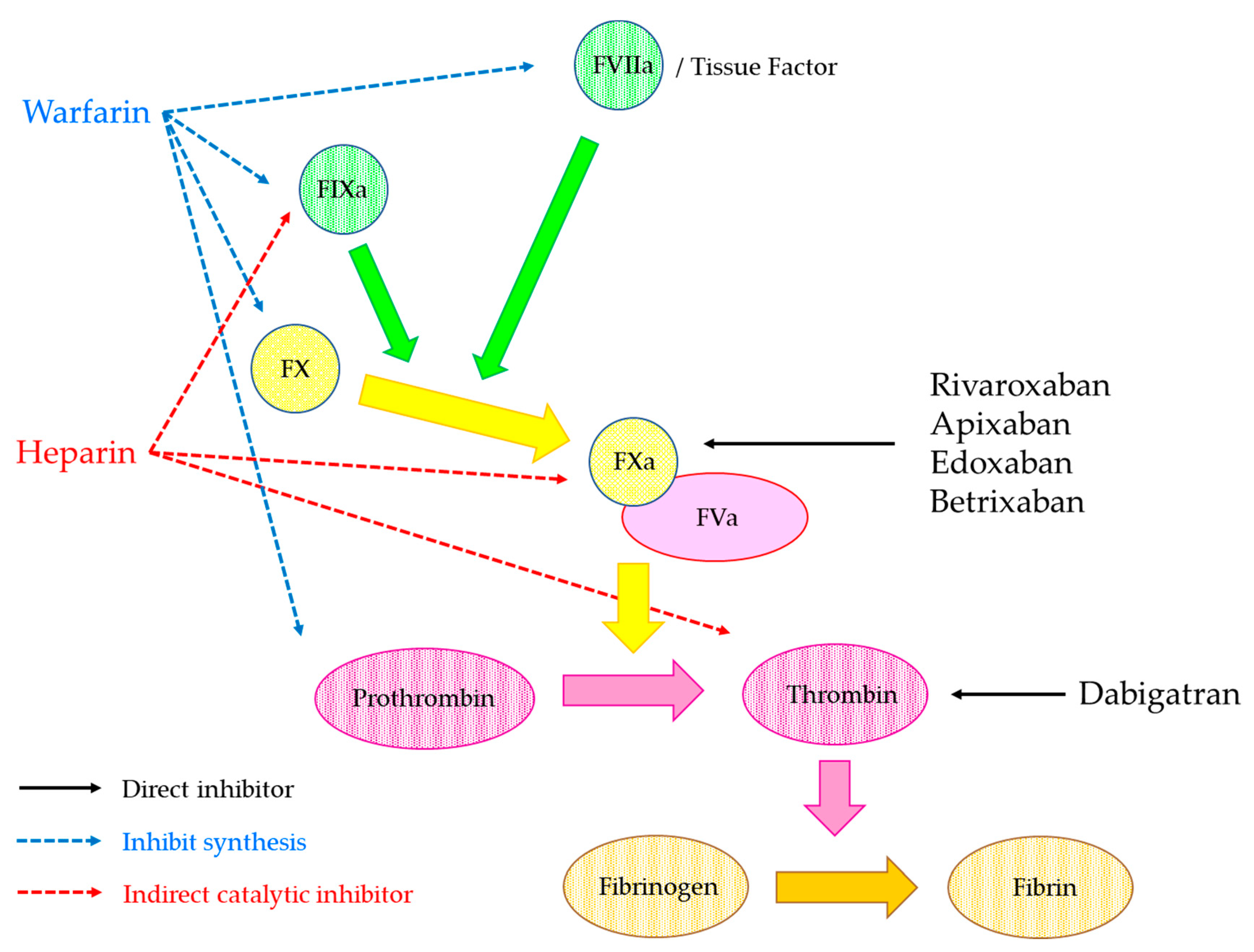
2. Anticoagulants
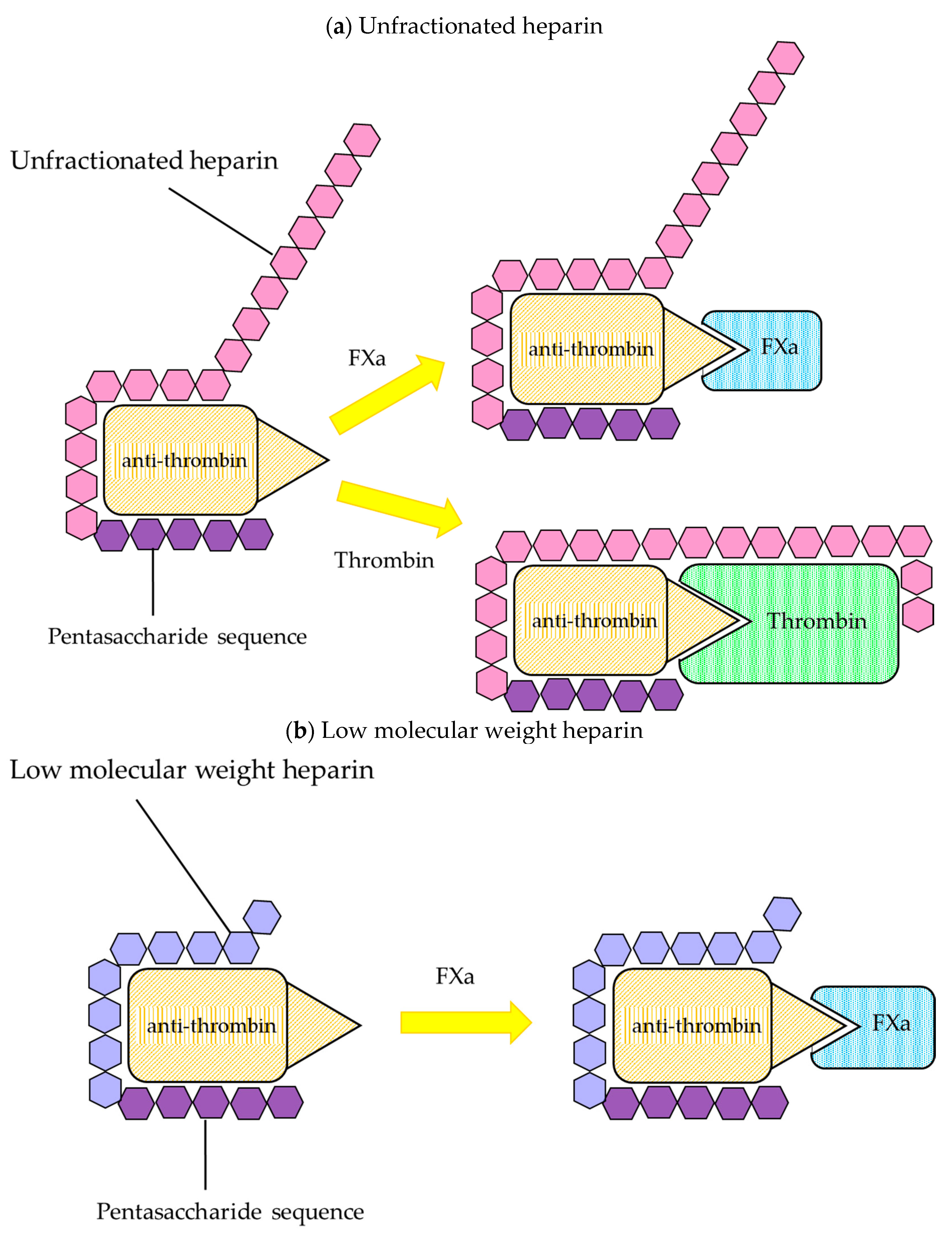
2.1. Heparin
2.1.1. The Properties of Heparin
2.1.2. Biological Properties
Unfractionated Heparin
Low Molecular Weight Heparin
2.1.3. Monitoring of Heparin
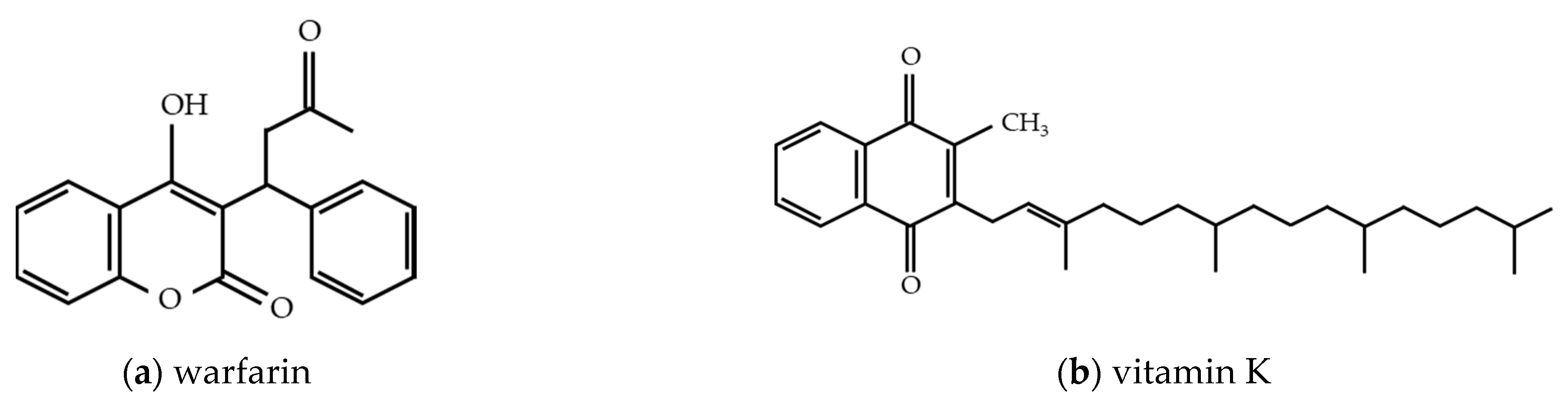
2.2. Warfarin
2.2.1. Properties of Warfarin
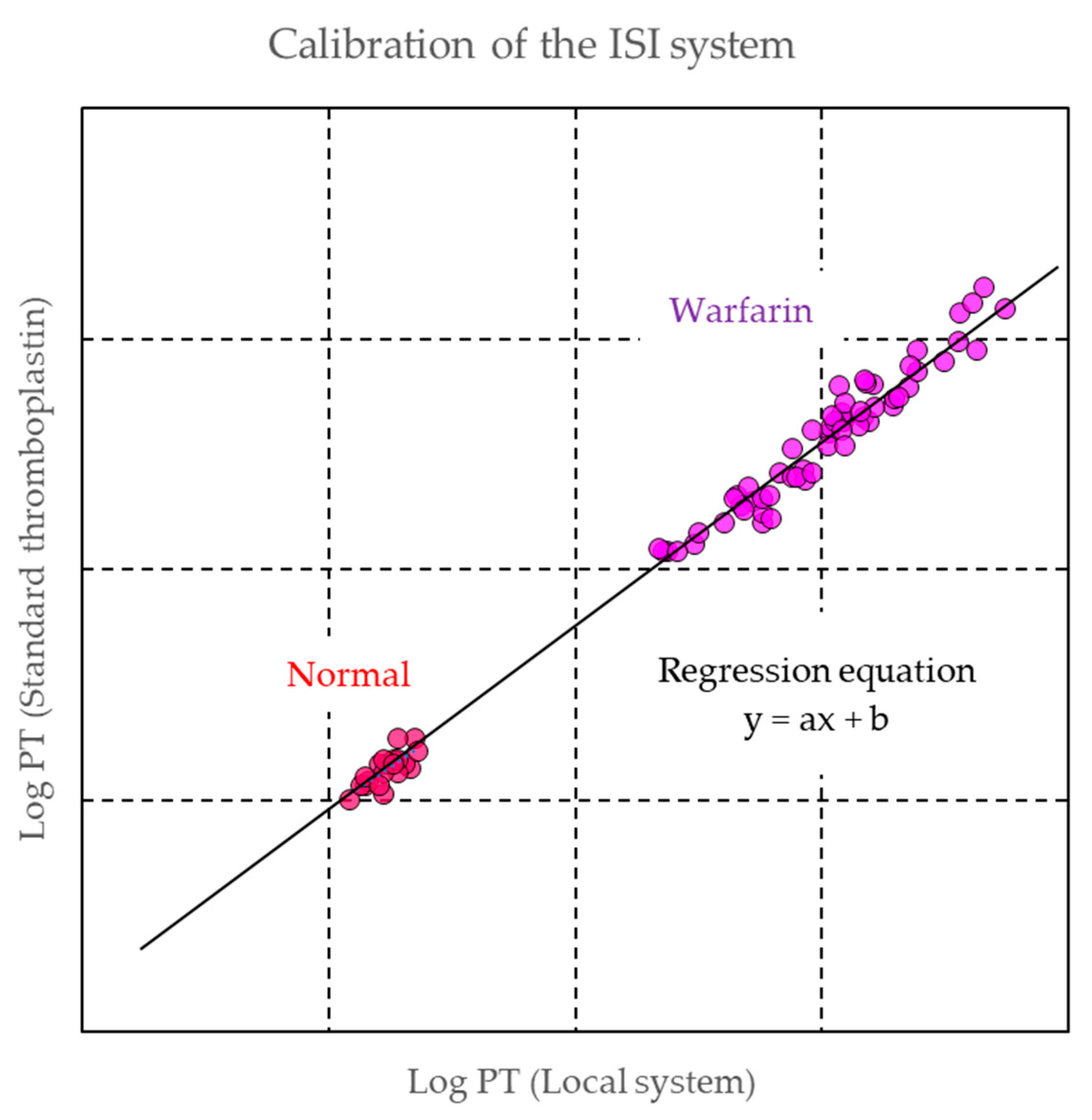
2.2.2. Monitoring of Warfarin
2.3. Direct Oral Anticoagulant (DOAC)
2.3.1. Properties of DOAC
2.3.2. Measurement of DOACs
- (i)
- Persistent bleeding or thrombosis
- (ii)
- Decreased drug clearance resulting from impaired kidney function or liver disease
- (iii)
- Identification of subtherapeutic or supratherapeutic levels in patients taking other drugs that are known to significantly affect pharmacokinetics
- (iv)
- Extremely body weight: <40 kg or >120 kg
- (v)
- Perioperative management
- (vi)
- Reversal of anticoagulant
- (vii)
- Suspicion of overdosage
- (viii)
- Adherence to treatment protocol
Qualitative Assays for DOACs
Quantitative Assay for DOAC Measurement
3. Coronavirus Disease 2019 and Thrombosis
3.1. The Thrombosis Mechanisms
3.2. COVID-19 Therapy and Anticoagulants
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Hirsh, J.; Raschke, R. Heparin and low-molecular-weight heparin. Chest 2004, 126, 188–203. [Google Scholar] [CrossRef]
- Dorgalaleh, A.; Favaloro, E.J.; Bahraini, M.; Rad, F. Standardization of Prothrombin time/international normalized ratio (PT/INR). Int. J. Lab. Hematol. 2020, in press. [Google Scholar] [CrossRef]
- Schulman, S.; Kearon, C.; Kakkar, A.K.; Mismetti, P.; Schellong, S.; Eriksson, H.; Baanstra, D.; Schnee, J.; Goldhaber, S.Z. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N. Engl. J. Med. 2009, 361, 2342–2352. [Google Scholar] [CrossRef]
- Bauersachs, R.; Berkowitz, S.D.; Brenner, B.; Buller, H.R.; Decousus, H.; Gallus, A.S.; Lensing, A.W.; Misselwitz, F.; Prins, M.H.; Raskob, G.E.; et al. Oral rivaroxaban for symptomatic venous thromboembolism. N. Engl. J. Med. 2010, 363, 2499–2510. [Google Scholar] [CrossRef]
- Büller, H.R.; Prins, M.H.; Lensin, A.W.A.; Decousus, H.; Jacobson, B.F.; Minar, E.; Chlumsky, J.; Verhamme, P.; Wells, P.; Agnelli, G.; et al. Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N. Engl. J. Med. 2012, 366, 1287–1297. [Google Scholar] [PubMed]
- Agnelli, G.; Buller, H.R.; Cohen, A.; Curto, M.; Gallus, A.S.; Johnson, M.; Masiukiewicz, U.; Pak, R.; Thompson, J.; Raskob, G.E.; et al. Oral apixaban for the treatment of acute venous thromboembolism. N. Engl. J. Med. 2013, 369, 799–808. [Google Scholar] [CrossRef] [PubMed]
- Büller, H.R.; Décousus, H.; Grosso, M.A.; Mercuri, M.; Middeldorp, S.; Prins, M.H.; Raskob, G.E.; Schellong, S.M.; Schwocho, L.; Segers, A.; et al. Edoxaban versus warfarin for the treatment of symptomatic venous thromboembolism. N. Engl. J. Med. 2013, 369, 1406–1415. [Google Scholar]
- Schulman, S.; Kakkar, A.K.; Goldhaber, S.Z.; Schellong, S.; Eriksson, H.; Mismetti, P.; Christiansen, A.V.; Friedman, J.; Le Maulf, F.; Peter, N.; et al. Treatment of acute venous thromboembolism with dabigatran or warfarin and pooled analysis. Circulation 2014, 129, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Schulman, S.; Kearon, C.; Kakkar, A.K.; Schellong, S.; Eriksson, H.; Baanstra, D.; Kvamme, A.M.; Friedman, J.; Mismetti, P.; Goldhaber, S.Z. Extended use of dabigatran, warfarin, or placebo in venous thromboembolism. N. Engl. J. Med. 2013, 368, 709–718. [Google Scholar] [CrossRef]
- Agnelli, G.; Buller, H.R.; Cohen, A.; Curto, M.; Gallus, A.S.; Johnson, M.; Porcari, A.; Raskob, G.E.; Weitz, J.I. Apixaban for extended treatment of venous thromboembolism. N. Engl. J. Med. 2013, 368, 699–708. [Google Scholar] [CrossRef]
- Lancaster, T.R.; Singer, D.E.; Sheehan, M.A.; Oertel, L.B.; Maraventano, S.W.; Hughes, R.A.; Kistler, J.P. The impact of long-term warfarin therapy on quality of life. Arch. Intern. Med. 1991, 151, 1944–1949. [Google Scholar] [CrossRef] [PubMed]
- Ghate, S.R.; Biskupiak, J.; Ye, X.; Kwong, W.J.; Brixner, D.I. All-cause and bleeding-related health care costs in warfarin-treated patients with atrial fibrillation. J. Manag. Care Pharm. 2011, 17, 672–684. [Google Scholar] [CrossRef] [PubMed]
- Carrier, M.; Le Gal, G.; Wells, P.S.; Rodger, M.A. Systematic review: Case-fatality rates of recurrent venous thromboembolism and major bleeding events among patients treated for venous thromboembolism. Ann. Intern. Med. 2010, 152, 578–589. [Google Scholar] [CrossRef] [PubMed]
- Klok, F.A.; Kooiman, J.; Huisman, M.V.; Konstantinides, S.; Lankeit, M. Predicting anticoagulant-related bleeding in patients with venous thromboembolism: A clinically oriented review. Eur. Respir. J. 2014, 45, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Tang, N.; Li, D.; Wang, X.; Sun, Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 844–847. [Google Scholar] [CrossRef]
- McLean, J. The thromboplastic action of cephalin. Am. J. Physiol. Content 1916, 41, 250–257. [Google Scholar] [CrossRef]
- Rosenberg, R.D.; Lam, L. Correlation between structure and function of heparin. Proc. Natl. Acad. Sci. USA 1979, 76, 1218–1222. [Google Scholar] [CrossRef]
- Turpie, A.G.; Gallus, A.S.; Hoek, J.A. A synthetic pentasaccharide for the prevention of deep-vein thrombosis after total hip re-placement. N. Engl. J. Med. 2001, 344, 619–625. [Google Scholar] [CrossRef]
- Herbert, J.M.; Herault, J.P.; Bernat, A.; van Amsterdam, G.M.; Lormeau, J.C.; Petitou, M.; van Boeckel, C.; Hoffmann, P.; Meuleman, D.G. Biochemical and pharmacological properties of SANORG 34006, a potent and long-acting synthetic pentasaccharide. Blood 1998, 91, 4197–4205. [Google Scholar] [CrossRef]
- Eriksson, B.I.; Bauer, K.A.; Lassen, M.R.; Turpie, A.G. Fondaparinux compared with enoxaparin for the prevention of venous thromboembolism after hip-fracture surgery. N. Engl. J. Med. 2001, 345, 1298–1304. [Google Scholar] [CrossRef]
- Lam, L.; Silbert, J.; Rosenberg, R. The separation of active and inactive forms of heparin. Biochem. Biophys. Res. Commun. 1976, 69, 570–577. [Google Scholar] [CrossRef]
- Andersson, L.-O.; Barrowcliffe, T.; Holmer, E.; Johnson, E.; Sims, G. Anticoagulant properties of heparin fractionated by affinity chromatography on matrix-bound antithrombin III and by gel filtration. Thromb. Res. 1976, 9, 575–583. [Google Scholar] [CrossRef]
- Bârzu, T.; Molho, P.; Tobelem, G.; Petitou, M.; Caen, J. Binding and endocytosis of heparin by human endothelial cells in culture. Biochim. Biophys. Acta Bioenergy 1985, 845, 196–203. [Google Scholar] [CrossRef]
- Cook, B.W. Anticoagulation management. Semin. Interv. Radiol. 2010, 27, 360–367. [Google Scholar] [CrossRef]
- de Swart, C.A.; Nijmeyer, B.; Roelofs, J.M.; Sixma, J.J. Kinetics of intravenously administered heparin in normal humans. Blood 1982, 60, 1251–1258. [Google Scholar] [CrossRef]
- Bjornsson, T.D.; Wolfram, K.M.; Kitchell, B.B. Heparin kinetics determined by three assay methods. Clin. Pharmacol. Ther. 1982, 31, 104–113. [Google Scholar] [CrossRef]
- Dawes, J.; Papper, D.S.; Pepper, D.S. Catabolism of low-dose heparin in man. Thromb. Res. 1979, 14, 845–860. [Google Scholar] [CrossRef]
- Leentjens, J.; Peters, M.; Esselink, A.C.; Smulders, Y.; Kramers, C. Initial anticoagulation in patients with pulmonary embolism: Thrombolysis, unfractionated heparin, LMWH, fondaparinux, or DOACs? Br. J. Clin. Pharmacol. 2017, 83, 2356–2366. [Google Scholar] [CrossRef] [PubMed]
- Hirsh, J.; Levine, M.N. Low molecular weight heparin. Blood 1992, 79, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Casu, B.; Oreste, P.; Torri, G.; Zoppetti, G.; Choay, J.; Lormeau, J.C.; Petitou, M.; Sinaɕ, P. The structure of heparin oligosaccharide fragments with high anti-(factor Xa) activity containing the minimal antithrombin III-binding sequence Chemical and13C nuclear-magnetic-resonance studies. Biochem. J. 1981, 197, 599–609. [Google Scholar] [CrossRef] [PubMed]
- Lindahl, U.; Thunberg, L.; Backstrom, G.; Riesenfeld, J.; Nordling, K.; Bjork, I. Extension and structural variability of the an-tithrombin-binding sequence in heparin. J. Biol. Chem. 1984, 259, 12368–12376. [Google Scholar] [CrossRef]
- Gray, E.; Mulloy, B.; Barrowcliffe, T.W. Heparin and low-molecular-weight heparin. Thromb. Haemost. 2008, 99, 807–818. [Google Scholar] [PubMed]
- Basu, D.; Gallus, A.; Hirsh, J.; Cade, J. A Prospective study of the value of monitoring heparin treatment with the activated partial thromboplastin time. N. Engl. J. Med. 1972, 287, 324–327. [Google Scholar] [CrossRef] [PubMed]
- Kitchen, S.; Cartwright, I.; Woods, T.A.; Jennings, I.; Preston, F.E. Lipid composition of seven APTT reagents in relation to heparin sensitivity. Br. J. Haematol. 1999, 106, 801–808. [Google Scholar] [CrossRef] [PubMed]
- Smythe, M.A.; Priziola, J.; Dobesh, P.P.; Wirth, D.; Cuker, A.; Wittkowsky, A.K. Guidance for the practical management of the heparin anticoagulants in the treatment of venous thromboembolism. J. Thromb. Thrombolysis 2016, 41, 165–186. [Google Scholar] [CrossRef] [PubMed]
- Garcia, D.A.; Baglin, T.P.; Weitz, J.I.; Samama, M.M. Parenteral anticoagulants: Antithrombotic therapy and prevention of thrombosis: 9th ed.: American college of chest physicians evidence-based clinical practice guidelines. Chest 2012, 141, 24–43. [Google Scholar] [CrossRef] [PubMed]
- Vandiver, J.W.; Vondracek, T.G. Antifactor Xa levels versus activated partial thromboplastin time for monitoring unfractionated heparin. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2012, 32, 546–558. [Google Scholar] [CrossRef]
- Arachchillage, D.; Kamani, F.; Deplano, S.; Banya, W.; Laffan, M. Should we abandon the APTT for monitoring unfractionated heparin? Thromb. Res. 2017, 157, 157–161. [Google Scholar] [CrossRef]
- Samama, M.M. Contemporary laboratory monitoring of low molecular weight heparins. Clin. Lab. Med. 1995, 15, 119–123. [Google Scholar] [CrossRef]
- Abbate, R.; Gori, A.M.; Farsi, A.; Attanasio, M.; Pepe, G. Monitoring of low-molecular-weight heparins in cardiovascular disease. Am. J. Cardiol. 1998, 82, 33L–36L. [Google Scholar] [CrossRef]
- Kessler, C.M. Low molecular weight heparins: Practical considerations. Semin. Hematol. 1997, 34, 35–42. [Google Scholar] [PubMed]
- Linkins, L.-A.; Julian, J.A.; Rischke, J.; Hirsh, J.; Weitz, J.I. In vitro comparison of the effect of heparin, enoxaparin and fondaparinux on tests of coagulation. Thromb. Res. 2002, 107, 241–244. [Google Scholar] [CrossRef]
- Thomas, O.; Lybeck, E.; Strandberg, K.; Tynngård, N.; Schött, U. Monitoring low molecular weight heparins at therapeutic levels: Dose-responses of, and correlations and differences between aPTT, anti-factor Xa and Thrombin generation assays. PLoS ONE 2015, 10, e0116835. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef]
- Lip, G.Y.H. Recommendations for thromboprophylaxis in the 2012 focused update of the ESC guidelines on atrial fibrillation: A commentary. J. Thromb. Haemost. 2013, 11, 615–626. [Google Scholar] [CrossRef] [PubMed]
- Leite, P.M.; Martins, M.A.P.; Castilho, R.O. Review on mechanisms and interactions in concomitant use of herbs and warfarin therapy. Biomed. Pharmacother. 2016, 83, 14–21. [Google Scholar] [CrossRef]
- Garcia, D.; Libby, E.; Crowther, M.A. The new oral anticoagulants. Blood 2010, 115, 15–20. [Google Scholar] [CrossRef]
- Poller, L.; Ibrahim, S.; Keown, M.; Pattison, A.; Jespersen, J. The prothrombin time/international normalized ratio (PT/INR) Line: Derivation of local INR with commercial thromboplastinsand coagulometers—Two independent studies. J. Thromb. Haemost. 2011, 9, 140–148. [Google Scholar] [CrossRef]
- Poller, L. International Normalized Ratios (INR): The first 20 years. J. Thromb. Haemost. 2004, 2, 849–860. [Google Scholar] [CrossRef]
- Meijer, P.; Kynde, K.; van den Besselaar, A.M.; Van Blerk, M.; Woods, T.A. International normalized ratio (INR) testing in Europe: Between-laboratory comparability of test results obtained by Quick and Owren reagents. Clin. Chem. Lab. Med. 2018, 56, 1698–1703. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization Expert Committee on Biological Standardization. Sixty-Second Report; No.979; WHO: Geneva, Switzerland, 2013. [Google Scholar]
- Favaloro, E.J. How to generate a more accurate laboratory-based international normalized ratio: Solutions to obtaining or ver-ifying the mean normal prothrombin time and international sensitivity index. Semin. Thromb. Hemost. 2019, 45, 10–21. [Google Scholar] [CrossRef]
- Van den Besselaar, A.M.H.P.; Barrowcliffe, T.W.; Houbouyan-Reveillard, L.L.; Jespersen, J.; Johnston, M.; Poller, L.; Tripodi, A. Subcommittee on Control of Anticoagulation of the Scientific and Standardization Committee of the ISTH. Guidelines on preparation, certification, and use of certified plasmas for ISI calibration and INR determination. J. Thromb. Haemost. 2004, 2, 1946–1953. [Google Scholar] [CrossRef]
- Clinical Laboratory Standards Institute. H57—A Protocol for the Evaluation, Validation, and Implementation of Coagulometers; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008; Volume 28, pp. 1–33. [Google Scholar]
- Kearon, C.; Ginsberg, J.S.; Kovacs, M.J.; Anderson, D.R.; Wells, P.; Julian, J.A.; MacKinnon, B.; Weitz, J.I.; Crowther, M.A.; Dolan, S.; et al. Comparison of low-intensity warfarin therapy with conventional-intensity warfarin therapy for long-term prevention of re-current venous thromboembolism. N. Engl. J. Med. 2003, 349, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Hylek, E.M.; Skates, S.J.; Sheehan, M.A.; Singer, D.E. An analysis of the lowest effective intensity of prophylactic anticoagulation for patients with nonrheumatic atrial fibrillation. N. Engl. J. Med. 1996, 335, 540–546. [Google Scholar] [CrossRef]
- Hering, D.; Piper, C.; Bergemann, R.; Hillenbach, C.; Dhm, M.; Huth, C.; Horstkotte, D. Thromboembolic and bleeding compli-cations following St. Jude Medical valve replacement: Results of the German Experience with low-intensity anticoagulation study. Chest 2005, 127, 53–59. [Google Scholar] [CrossRef]
- Makris, M.; van Veen, J.J.; Maclean, R. Warfarin anticoagulation reversal: Management of the asymptomatic and bleeding pa-tient. J. Thromb. Thromblysis 2010, 29, 171–181. [Google Scholar] [CrossRef]
- Ageno, W.; Gallus, A.S.; Wittkowsky, A.; Crowther, M.; Hylek, E.M.; Palareti, G. Oral anticoagulant therapy: Antithrombotic therapy and prevention of thrombosis, 9th ed.: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012, 141, 44S–88S. [Google Scholar] [CrossRef]
- Holbrook, A.; Schulman, S.; Witt, D.M.; Vandvik, P.O.; Fish, J.; Kovacs, M.J.; Svensson, P.J.; Veenstra, D.L.; Crowther, M.; Guyatt, G.H. Evidence-based management of anticoagulant therapy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed.: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012, 141, 152S–184S. [Google Scholar] [CrossRef] [PubMed]
- Baglin, T.; Hillarp, A.; Tripodi, A.; Elalamy, I.; Buller, H.; Ageno, W. Measuring oral direct inhibitors (ODIs) of thrombin and factor Xa: A recommendation from the Subcommittee on Control of Anticoagulation of the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. J. Thromb. Haemost. 2013, 11, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Barnes, G.D.; Ageno, W.; Ansell, J.; Kaatz, S. Recommendation on the nomenclature for oral anticoagulants: Communication from the SSC of the ISTH. J. Thromb. Haemost. 2015, 13, 1154–1156. [Google Scholar] [CrossRef]
- Dale, B.J.; Chan, N.C.; Eikelboom, J.W. Laboratory measurement of the direct oral anticoagulants. Br. J. Haematol. 2016, 172, 315–336. [Google Scholar] [CrossRef]
- Stangier, J. Clinical pharmacokinetics and pharmacodynamics of the oral direct thrombin inhibitor dabigatran etexilate. Clin. Pharmacokinet. 2008, 47, 285–295. [Google Scholar] [CrossRef]
- Perzborn, E.; Strassburger, J.; Wilmen, A.; Pohlmann, J.; Roehrig, S.; Schlemmer, K.-H.; Straub, A. In vitro and in vivo studies of the novel antithrombotic agent BAY 59-7939-an oral, direct Factor Xa inhibitor. J. Thromb. Haemost. 2005, 3, 514–521. [Google Scholar] [CrossRef] [PubMed]
- Ogata, K.; Mendell-Harary, J.; Tachibana, M.; Masumoto, H.; Oguma, T.; Kojima, M.; Kunitada, S. Clinical safety, tolerability, pharmacokinetics, and pharmacodynamics of the novel factor Xa inhibitor edoxaban in healthy volunteers. J. Clin. Pharmacol. 2010, 50, 743–753. [Google Scholar] [CrossRef]
- Weitz, J.I.; Harenberg, J. New developments in anticoagulants: Past, present and future. Thromb. Haemost. 2017, 117, 1283–1288. [Google Scholar] [CrossRef]
- Huisman, M.V.; Klok, F.A. Pharmacological properties of betrixaban. Eur. Heart J. Suppl. 2018, 20, E12–E15. [Google Scholar] [CrossRef] [PubMed]
- Mueck, W.; Schwers, S.; Stampfuss, J. Rivaroxaban and other novel oral anticoagulants: Pharmacokinetics in healthy subjects, specific patient populations and relevance of coagulation monitoring. Thromb. J. 2013, 11, 10. [Google Scholar] [CrossRef] [PubMed]
- Cuker, A.; Siegal, D. Monitoring and reversal of direct oral anticoagulants. Hematol. Am. Soc. Hematol. Educ. Program. 2015, 2015, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Gosselin, R.C.; Adcock, D.M. The laboratory’s 2015 perspective on direct oral anticoagulant testing. J. Thromb. Haemost. 2016, 14, 886–893. [Google Scholar] [CrossRef]
- Douxfils, J.; Mullier, F.; Loosen, C.; Chatelain, C.; Chatelain, B.; Dogne, J.M. Assessment of the impact of rivaroxaban on coagu-lation assays: Laboratory recommendations for the monitoring of rivaroxaban and review of the literature. Thromb. Res. 2012, 130, 956–966. [Google Scholar] [CrossRef]
- Robert, S.; Chatelain, C.; Douxfils, J.; Mullier, F.; Chatelain, B.; Dogné, J.-M. Impact of dabigatran on a large panel of routine or specific coagulation assays. Thromb. Haemost. 2012, 107, 985–997. [Google Scholar] [CrossRef]
- Thom, I.; Cameron, G.; Robertson, D.; Watson, H.G. Measurement of rivaroxaban concentrations demonstrates lack of clinical utility of a PT, dPT and APTT test in estimating levels. Int. J. Lab. Hematol. 2018, 40, 493–499. [Google Scholar] [CrossRef]
- Gosselin, R.; Grant, R.P.; Adcock, D.M. Comparison of the effect of the anti-Xa direct oral anticoagulants apixaban, edoxaban, and rivaroxaban on coagulation assays. Int. J. Lab. Hematol. 2016, 38, 505–513. [Google Scholar] [CrossRef]
- Flaujac, C.; Delavenne, X.; Quenet, S.; Horellou, M.-H.; Laporte, S.; Siguret, V.; Lecompte, T.; Gouin-Thibault, I. Assessment of apixaban plasma levels by laboratory tests: Suitability of three anti-Xa assays. Thromb. Haemost. 2014, 111, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Chatelain, C.; Chatelain, B.; Douxfils, J.; Dogné, J.-M.; Mullier, F. Impact of apixaban on routine and specific coagulation assays: A practical laboratory guide. Thromb. Haemost. 2013, 110, 283–294. [Google Scholar] [CrossRef]
- Zafar, M.U.; Vorchheimer, D.A.; Gaztanaga, J.; Velez, M.; Yadegar, D.; Moreno, P.R.; Kunitada, S.; Pagan, J.; Fuster, V.; Badimon, J.J. Antithrombotic effects of factor Xa inhibition with DU-176b: Phase-I study of an oral, direct factor Xa inhibitor using an ex-vivo flow chamber. Thromb. Haemost. 2007, 98, 883–888. [Google Scholar] [CrossRef]
- Morishima, Y.; Kamisato, C. Laboratory measurements of the oral direct factor Xa inhibitor edoxaban: Comparison of pro-thrombin time, activated partial thromboplastin time, and thrombin generation assay. Am. J. Clin. Pathol. 2015, 143, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Helin, T.A.; Pakkanen, A.; Lassila, R.; Joutsi-Korhonen, L. Laboratory assessment of novel oral anticoagulants: Method suitability and variability between coagulation laboratories. Clin. Chem. 2013, 59, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Halbmayer, W.-M.; Weigel, G.; Quehenberger, P.; Tomasits, J.; Haushofer, A.C.; Aspoeck, G.; Loacker, L.; Schnapka-Koepf, M.; Goebel, G.; Griesmacher, A. Interference of the new oral anticoagulant dabigatran with frequently used coagulation tests. Clin. Chem. Lab. Med. 2012, 50, 1601–1605. [Google Scholar] [CrossRef] [PubMed]
- Hillarp, A.; Baghaei, F.; Blixter, I.F.; Gustafsson, K.M.; Stigendal, L.; Sten-Linder, M.; Strandberg, K.; Lindahl, T.L. Effects of the oral, direct factor Xa inhibitor rivaroxaban on commonly used coagulation assays. J. Thromb. Haemost. 2011, 9, 133–139. [Google Scholar] [CrossRef]
- Liesenfeld, K.-H.; Schäfer, H.G.; Trocóniz, I.F.; Tillmann, C.; Eriksson, B.I.; Stangier, J. Effects of the direct thrombin inhibitor dabigatran on ex vivo coagulation time in orthopaedic surgery patients: A population model analysis. Br. J. Clin. Pharmacol. 2006, 62, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Kher, A.; Dieri, R.A.; Hemker, H.C.; Beguin, S. Laboratory assessment of antithrombotic therapy: What tests and if so why? Pathophysiol. Haemost. Thromb. 1997, 27, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Conrad, K.A. Clinical pharmacology and drug safety: Lessons from hirudin. Clin. Pharmacol. Ther. 1995, 58, 123–126. [Google Scholar] [CrossRef]
- Fox, I.; Dawson, A.; Loynds, P.; Eisner, J.; Findlen, K.; Levin, E.; Hanson, D.; Mant, T.; Wagner, J.; Maraganore, J. Anticoagulant activity of hirulog™, a direct thrombin inhibitor, in humans. Thromb. Haemost. 1993, 69, 157–163. [Google Scholar] [CrossRef]
- Lessire, S.; Douxfils, J.; Baudar, J.; Bailly, N.; Dincq, A.-S.; Gourdin, M.; Dogné, J.-M.; Chatelain, B.; Mullier, F. Is thrombin time useful for the assessment of dabigatran concentrations? An in vitro and ex vivo study. Thromb. Res. 2015, 136, 693–696. [Google Scholar] [CrossRef] [PubMed]
- Dager, W.E.; Gosselin, R.C.; Kitchen, S.; Dwyre, D. Dabigatran Effects on the international normalized ratio, activated partial thromboplastin time, thrombin time, and fibrinogen: A multicenter, in vitro study. Ann. Pharmacother. 2012, 46, 1627–1636. [Google Scholar] [CrossRef]
- Martinoli, J.-L.; Leflem, L.; Guinet, C.; Plu-Bureau, G.; Depasse, F.; Perzborn, E.; Samama, M.M. Assessment of laboratory assays to measure rivaroxaban—An oral, direct factor Xa inhibitor. Thromb. Haemost. 2010, 103, 815–825. [Google Scholar] [CrossRef]
- Ieko, M.; Ohmura, K.; Naito, S.; Yoshida, M.; Sakuma, I.; Ikeda, K.; Ono, S.; Suzuki, T.; Takahashi, N. Novel assay based on diluted prothrombin time reflects anticoagulant effects of direct oral factor Xa inhibitors: Results of multicenter study in Japan. Thromb. Res. 2020, 195, 158–164. [Google Scholar] [CrossRef]
- Kumano, O.; Suzuki, S.; Yamazaki, M.; An, Y.; Yasaka, M.; Ieko, M. for the Japanese study group for the effect confirmation of direct oral anticoagulants. The basic evaluation of the newly developing modified diluted prothrombin time reagent for direct oral anticoagulants measurements. In Proceedings of the XXVIII Virtual Congress on International Society on Thrombosis and Haemostasis, Online. 12–14 July 2020. [Google Scholar]
- Kumano, O.; Suzuki, S.; Yamazaki, M.; An, Y.; Yasaka, M.; Ieko, M. for the Japanese study group for the effect confirmation of direct oral anticoagulants. New formula of “ratio of inhibited thrombin generation” based on modified diluted prothrombin time reagent predicts bleeding risk of patients with low coagulation activity in direct oral anticoagulant therapy. In Proceedings of the XXVIII Virtual Congress on International Society on Thrombosis and Haemostasis, Online. 12–14 July 2020. [Google Scholar]
- Letertre, L.R.; Gudmundsdottir, B.R.; Francis, C.W.; Gosselin, R.C.; Skeppholm, M.; Malmstrom, R.E.; Moll, S.; Hawes, E.; Francart, S.; Onundarson, P.T. A single test to assay warfarin, dabigatran, rivaroxaban, apixaban, unfractionated heparin, and enoxaparin in plasma. J. Thromb. Haemost. 2016, 14, 1043–1053. [Google Scholar] [CrossRef][Green Version]
- Rohde, G. Determination of rivaroxaban—A novel, oral, direct Factor Xa inhibitor—In human plasma by high-performance liquid chromatography–tandem mass spectrometry. J. Chromatogr. B 2008, 872, 43–50. [Google Scholar] [CrossRef]
- Wang, J.; Song, Y.; Pursley, J.; Wastall, P.; Wright, R.; Lacreta, F.; Frost, C.; Barrett, Y.C. A randomised assessment of the pharmacokinetic, pharmacodynamic and safety interaction between apixaban and enoxaparin in healthy subjects. Thromb. Haemost. 2012, 107, 916–924. [Google Scholar] [CrossRef] [PubMed]
- Bathala, M.S.; Masumoto, H.; Oguma, T.; He, L.; Lowrie, C.; Mendell, J. Pharmacokinetics, biotransformation, and mass balance of edoxaban, a selective, direct factor Xa Inhibitor, in humans. Drug Metab. Dispos. 2012, 40, 2250–2255. [Google Scholar] [CrossRef]
- Schmitz, E.E.; Boonen, K.K.; van den Heuvel, D.J.A.; Van Dongen, J.J.; Schellings, M.W.M.; Emmen, J.J.; Van Der Graaf, F.; Brunsveld, L.L.; Van De Kerkhof, D.D. Determination of dabigatran, rivaroxaban and apixaban by ultra-performance liquid chromatography—Tandem mass spectrometry (UPLC-MS/MS) and coagulation assays for therapy monitoring of novel direct oral anticoagulants. J. Thromb. Haemost. 2014, 12, 1636–1646. [Google Scholar] [CrossRef]
- Dogné, J.-M.; Mullier, F.; Chatelain, B.; Rönquist-Nii, Y.; Malmström, R.E.; Hjemdahl, P.; Douxfils, J. Comparison of calibrated dilute thrombin time and aPTT tests with LC-MS/MS for the therapeutic monitoring of patients treated with dabigatran etexilate. Thromb. Haemost. 2013, 110, 543–549. [Google Scholar] [CrossRef]
- Douxfils, J.; Tamigniau, A.; Chatelain, B.; Chatelain, C.; Wallemacq, P.; Dogne, J.M.; Mullier, F. Comparison of calibrated chro-mogenic anti-Xa assay and PT tests with LC-MS/MS for the therapeutic monitoring of patients treated with rivaroxaban. Thromb. Haemost. 2013, 110, 723–731. [Google Scholar]
- Antovic, J.P.; Skeppholm, M.; Eintrei, J.; Bojia, E.E.; Soderblom, L.; Norberg, E.M.; Onelov, L.; Ronquist-Nii, Y.; Pohanka, A.; Beck, O.; et al. Evaluation of coagulation assays versus LC-MS/MS for determinations of dabigatran concentrations in plasma. Eur. J. Clin. Pharmacol. 2013, 69, 1875–1881. [Google Scholar] [CrossRef]
- Skeppholm, M.; Hjemdahl, P.; Antovic, J.P.; Muhrbeck, J.; Eintrei, J.; Rönquist-Nii, Y.; Pohanka, A.; Beck, O.; Malmström, R.E. On the monitoring of dabigatran treatment in “real life” patients with atrial fibrillation. Thromb. Res. 2014, 134, 783–789. [Google Scholar] [CrossRef] [PubMed]
- Skeppholm, M.; Al-Aieshy, F.; Berndtsson, M.; Al-Khalili, F.; Rönquist-Nii, Y.; Söderblom, L.; Östlund, A.Y.; Pohanka, A.; Antovic, J.; Malmström, R.E. Clinical evaluation of laboratory methods to monitor apixaban treatment in patients with atrial fibrillation. Thromb. Res. 2015, 136, 148–153. [Google Scholar] [CrossRef]
- Stangier, J.; Feuring, M. Using the HEMOCLOT direct thrombin inhibitor assay to determine plasma concentrations of dabigatran. Blood Coagul. Fibrinolysis 2012, 23, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Butler, J.; Malan, E.; Chunilal, S.; Tran, H.; Hapgood, G. The effect of dabigatran on the activated partial thromboplastin time and thrombin time as determined by the Hemoclot thrombin inhibitor assay in patient plasma samples. Thromb. Haemost. 2013, 110, 308–315. [Google Scholar] [CrossRef]
- Samoš, M.; Stančiaková, L.; Ivanková, J.; Staško, J.; Kovář, F.; Dobrotová, M.; Galajda, P.; Kubisz, P.; Mokáň, M. Monitoring of dabigatran therapy using hemoclot thrombin inhibitor assay in patients with atrial fibrillation. J. Thromb. Thrombolysis 2015, 39, 95–100. [Google Scholar] [CrossRef]
- Studt, J.-D.; Alberio, L.; Angelillo-Scherrer, A.; Asmis, L.M.; Fontana, P.; Korte, W.; Mendez, A.; Schmid, P.; Stricker, H.; Tsakiris, D.A.; et al. Accuracy and consistency of anti-Xa activity measurement for determination of rivaroxaban plasma levels. J. Thromb. Haemost. 2017, 15, 1576–1583. [Google Scholar] [CrossRef] [PubMed]
- Lessire, S.; Dincq, A.S.; Siriez, R.; Pochet, L.; Sennesael, A.L.; Vornicu, O.; Hardy, M.; Deceuninck, O.; Douxifils, J.; Mullier, F. Assessment of low plasma concentrations of apixaban in the periprocedural setting. Int. J. Lab. Hematol. 2020, 42, 394–402. [Google Scholar] [CrossRef]
- Hirsh, J. Low-molecular-weight heparin. A review of the results of recent studies of the treatment of venous thromboembolism and unstable angina. Circulation 1998, 98, 1575–1582. [Google Scholar] [CrossRef]
- Li, Q.; Guan, X.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Ren, R.; Leung, S.M.K.; Lau, E.H.Y.; Wong, J.Y.; et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N. Engl. J Med. 2020, 382, 1199–1207. [Google Scholar] [CrossRef]
- Hou, Y.J.; Okuda, K.; Edwards, C.E.; Martinez, D.R.; Asakura, T.; Dinnon, K.H., 3rd; Kato, T.; Lee, R.E.; Yount, B.L.; Mascenik, T.M.; et al. SARS-CoV-2 reverse genetics reveals a variable infection gradient in the respiratory tract. Cell 2020, 182, 429–446. [Google Scholar] [CrossRef]
- McFadyen, J.D.; Stevens, H.; Peter, K. The emerging threat of (Micro)thrombosis in COVID-19 and Its therapeutic implications. Circ. Res. 2020, 127, 571–587. [Google Scholar] [CrossRef] [PubMed]
- Vidali, S.; Morosetti, D.; Cossu, E.; Luisi, M.L.E.; Pancani, S.; Semeraro, V.; Consales, G. D-dimer as an indicator of prognosis in SARS-CoV-2 infection: A systematic review. ERJ Open Res. 2020, 6, 00260–02020. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Li, L.; Xu, M.; Wu, J.; Luo, D.; Zhu, Y.; Li, B.; Song, X.; Zhou, X. Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19. J. Clin. Virol. 2020, 127, 104370. [Google Scholar] [CrossRef]
- Levi, M.; van der Poll, T. Coagulation and sepsis. Thromb. Res. 2017, 149, 38–44. [Google Scholar] [CrossRef]
- Barnes, B.J.; Adrover, J.M.; Baxter-Stoltzfus, A.; Borczuk, A.; Cools-Lartigue, J.; Crawford, J.M.; Daßler-Plenker, J.; Guerci, P.; Huynh, C.; Knight, J.S.; et al. Targeting potential drivers of COVID-19: Neutrophil extracellular traps. J. Exp. Med. 2020, 217. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Y.; Yalavarthi, S.; Shi, H.; Gockman, K.; Zuo, M.; Madison, J.A.; Blair, C.; Weber, A.; Barnes, B.J.; Egeblad, M.; et al. Neutrophil extracellular traps in COVID-19. JCI Insight 2020, 5, e138999. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, S.; Opneja, A.; Nayak, L. The role of neutrophils in thrombosis. Thromb. Res. 2018, 170, 87–96. [Google Scholar] [CrossRef]
- Twaddell, S.H.; Baines, K.J.; Grainge, C.; Gibson, P.G. The Emerging role of neutrophil extracellular traps in respiratory disease. Chest 2019, 156, 774–782. [Google Scholar] [CrossRef]
- Thachil, J.; Tang, N.; Gando, S.; Falanga, A.; Cattaneo, M.; Levi, M.; Clark, C.; Iba, T. ISTH interim guidance on recognition and management of coagulopathy in COVID-19. J. Thromb. Haemost. 2020, 18, 1023–1026. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Clinical Management of COVID-19 Interim Guidance 27 May 2020; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Song, J.C.; Wang, G.; Zhang, W.; Zhang, Y.; Li, W.Q.; Zhou, Z. People’s liberation army professional committee of critical care medicine, chinese society on thrombosis and haemostasis. Chinese expert consensus on diagnosis and treatment of coagulation dysfunction in COVID-19. Mil. Med. Res. 2020, 7, 19. [Google Scholar]
- Kumano, O.; Ieko, M.; Komiyama, Y.; Naito, S.; Yoshida, M.; Takahashi, N.; Ohmura, K.; Hayakawa, M. Characterization of fibrin/fibrinogen degradation products reagents and their utility in critical care patients with enhanced fibrinolysis. Int. J. Lab. Hematol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Klok, F.A.; Kruip, M.J.H.A.; van der Meer, N.J.M.; Arbous, M.S.; Gommers, D.A.M.P.J.; Kant, K.M.; Kaptein, F.H.J.; van Paassen, J.; Stals, M.A.M.; Huisman, M.V.; et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 2020, 191, 145–147. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.; Meade, M.; Lauzier, F.; Zarychanski, R.; Mehta, S.; Lamontagne, F.; Dodek, P.; McIntyre, L.; Hall, R.; Heels-Ansdell, D.; et al. Failure of anticoagulant thromboprophylaxis: Risk factors in medical-surgical critically ill patients. Crit. Care Med. 2015, 43, 401–410. [Google Scholar] [CrossRef]
- White, D.; Macdonald, S.; Bull, T.; Hayman, M.; De Monteverde-Robb, R.; Sapsford, D.; Lavinio, A.; Varley, J.; Johnston, A.; Besser, M.; et al. Heparin resistance in COVID-19 patients in the intensive care unit. J. Thromb. Thrombolysis 2020, 50, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Finley, A.; Greenberg, C. Heparin sensitivity and resistance: Management during cardiopulmonary bypass. Anesth. Analg. 2013, 116, 1210–1222. [Google Scholar] [CrossRef] [PubMed]

| Dabigatran | Rivaroxaban | Apixaban | Edoxaban | Betrixaban | |
|---|---|---|---|---|---|
| Target | Thrombin | Factor Xa | Factor Xa | Factor Xa | Factor Xa |
| Primary clearance | Renal | Renal | Fecal | Renal | Fecal |
| Tmax | 1.5–3 h | 2–3 h | 3–4 h | 1–2 h | 3–4 h |
| Half-life | 12–14 h | 5–13 h | 12 h | 10–14 h | 19–27 h |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumano, O.; Akatsuchi, K.; Amiral, J. Updates on Anticoagulation and Laboratory Tools for Therapy Monitoring of Heparin, Vitamin K Antagonists and Direct Oral Anticoagulants. Biomedicines 2021, 9, 264. https://doi.org/10.3390/biomedicines9030264
Kumano O, Akatsuchi K, Amiral J. Updates on Anticoagulation and Laboratory Tools for Therapy Monitoring of Heparin, Vitamin K Antagonists and Direct Oral Anticoagulants. Biomedicines. 2021; 9(3):264. https://doi.org/10.3390/biomedicines9030264
Chicago/Turabian StyleKumano, Osamu, Kohei Akatsuchi, and Jean Amiral. 2021. "Updates on Anticoagulation and Laboratory Tools for Therapy Monitoring of Heparin, Vitamin K Antagonists and Direct Oral Anticoagulants" Biomedicines 9, no. 3: 264. https://doi.org/10.3390/biomedicines9030264
APA StyleKumano, O., Akatsuchi, K., & Amiral, J. (2021). Updates on Anticoagulation and Laboratory Tools for Therapy Monitoring of Heparin, Vitamin K Antagonists and Direct Oral Anticoagulants. Biomedicines, 9(3), 264. https://doi.org/10.3390/biomedicines9030264







