Oral Squamous Cell Carcinoma Is Associated with a Low Thrombosis Risk Due to Storage Pool Deficiency in Platelets
Abstract
1. Introduction
2. Materials and Methods
2.1. Antibodies and Reagent
2.2. Cell Culture
2.3. Mice
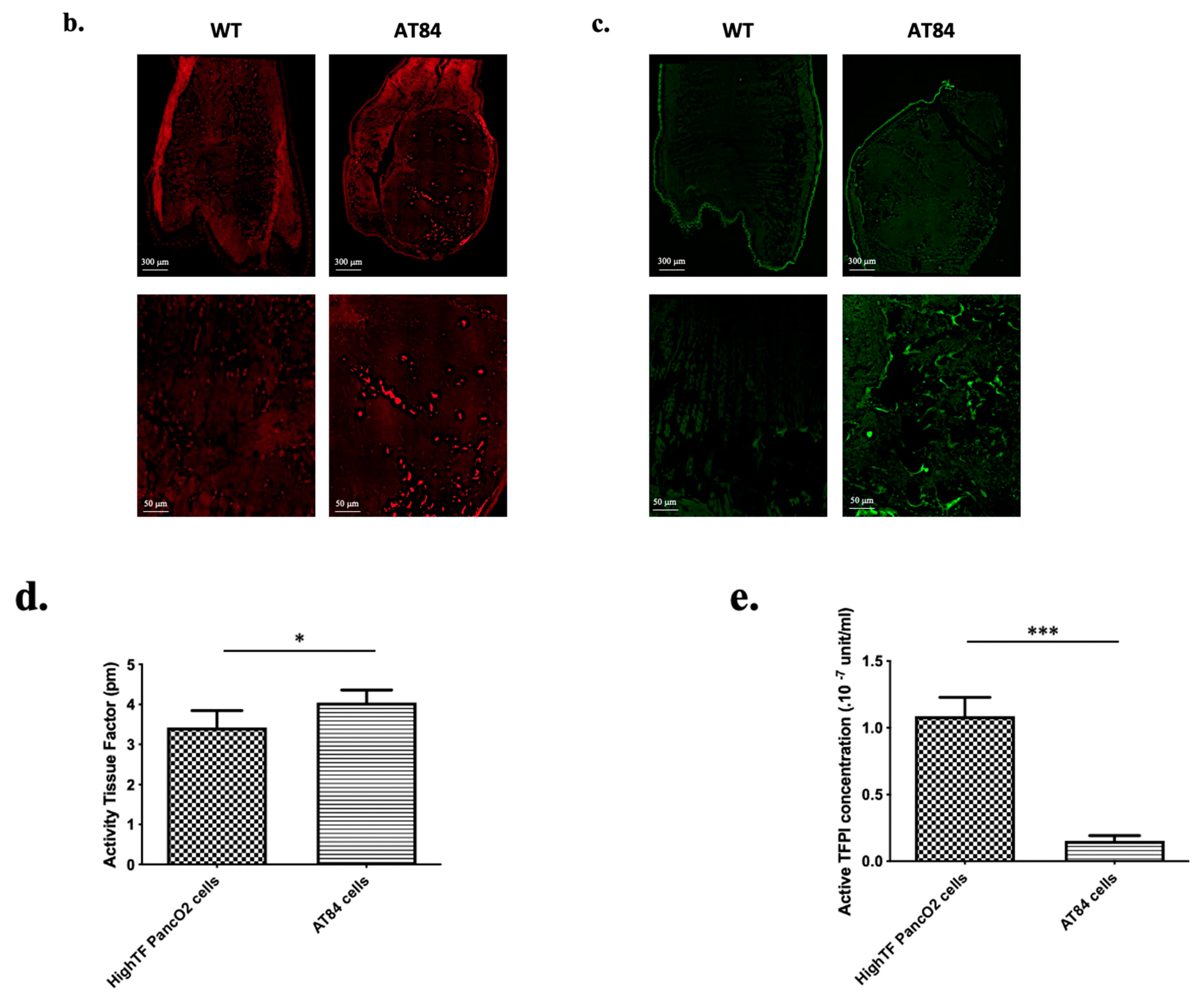
2.4. TF and TFPI Activity Assay
2.5. Mice Platelet Rich Plasma Preparation (PRP)
2.6. Induction of Platelet Aggregation by OSCC Cell
2.7. Platelet Aggregometry
2.8. Mouse Model of Tongue Squamous Cell Carcinoma
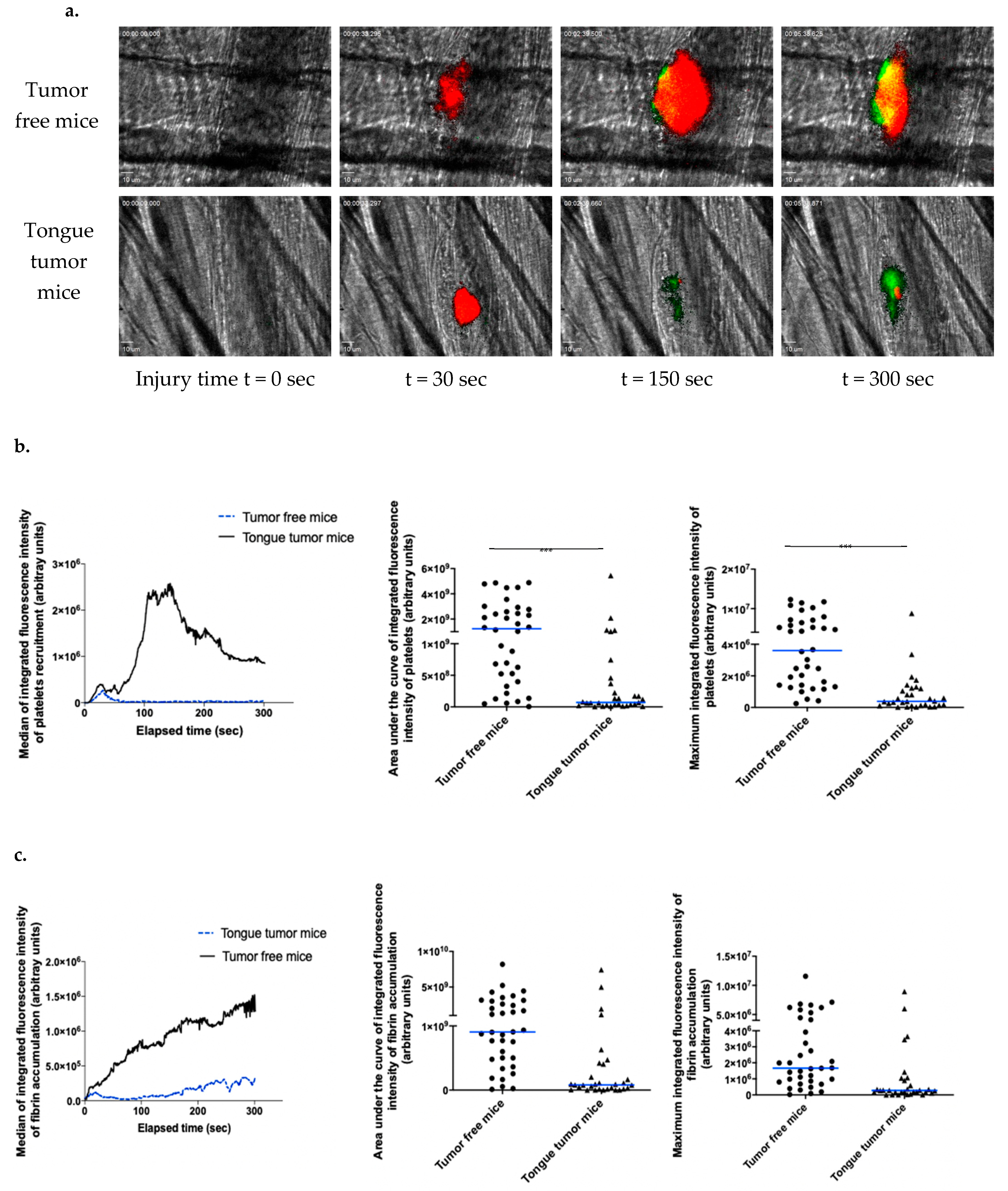
2.9. Intravital Microscopy and Laser-Induced Injury
2.10. Optical Microscopy
2.11. Fluorescent Microscopy
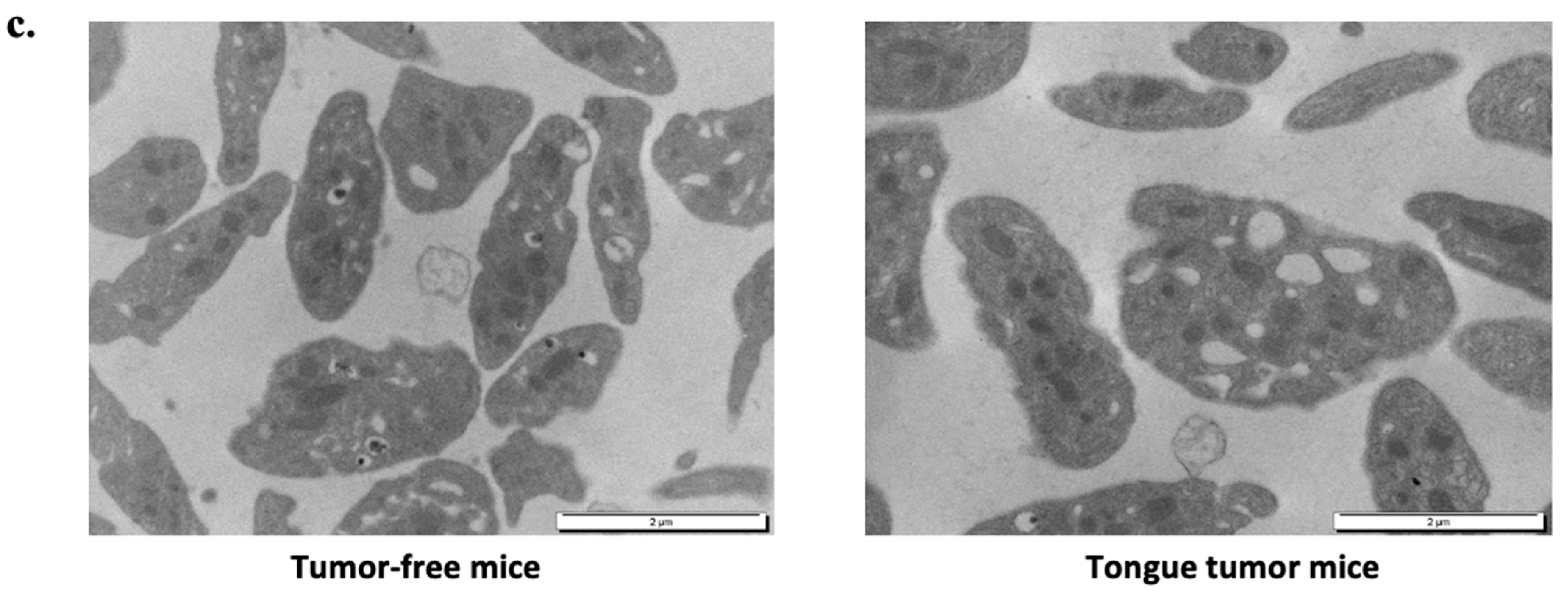
2.12. Transmission Electron Microscopy (TEM)
2.13. Statistics
3. Results
3.1. OSCC Cells Induce Platelet Aggregation In Vitro
3.2. Platelet Aggregates and Fibrin Deposits Are Locally Detected at the Site of the Primary Squamous Cell Carcinoma Tumors
3.3. Tongue SCC Is Associated with a Reduction in Systemic Thrombosis Risk in Living Mice
3.4. Tongue Tumors Are Associated with a Decrease in Platelet Reactivity
3.5. Tongue Tumors Are Associated with Granule Pool Deficiency
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Varki, A. Trousseau’s syndrome: Multiple definitions and multiple mechanisms. Blood 2007, 110, 1723–1729. [Google Scholar] [CrossRef]
- Palacios-Acedo, A.L.; Mège, D.; Crescence, L.; Dignat-George, F.; Dubois, C.; Panicot-Dubois, L. Platelets, Thrombo-Inflammation, and Cancer: Collaborating With the Enemy. Front. Immunol. 2019, 10, 1805. [Google Scholar] [CrossRef] [PubMed]
- Key, N.S.; Khorana, A.A.; Kuderer, N.M.; Bohlke, K.; Lee, A.Y.Y.; Arcelus, J.I.; Wong, S.L.; Balaban, E.P.; Flowers, C.R.; Francis, C.W.; et al. Venous Thromboembolism Prophylaxis and Treatment in Patients With Cancer: ASCO Clinical Practice Guideline Update. J. Clin. Oncol. 2020, 38, 496–520. [Google Scholar] [CrossRef] [PubMed]
- Chew, H.K.; Wun, T.; Harvey, D.; Zhou, H.; White, R.H. Incidence of venous thromboembolism and its effect on survival among patients with common cancers. Arch. Intern. Med. 2006, 166, 458–464. [Google Scholar] [CrossRef]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef]
- Pires, F.R.; Ramos, A.B.; de Oliveira, J.B.C.; Tavares, A.S.; da Luz, P.S.R.; dos Santos, T.C.R.B. Oral squamous cell carcinoma: Clinicopathological features from 346 cases from a single oral pathology service during an 8-year period. J. Appl. Oral Sci. 2013, 21, 460–467. [Google Scholar] [CrossRef] [PubMed]
- Ng, J.H.; Iyer, N.G.; Tan, M.-H.; Edgren, G. Changing epidemiology of oral squamous cell carcinoma of the tongue: A global study. Head Neck 2017, 39, 297–304. [Google Scholar] [CrossRef]
- Sørensen, H.T.; Mellemkjaer, L.; Steffensen, F.H.; Olsen, J.H.; Nielsen, G.L. The risk of a diagnosis of cancer after primary deep venous thrombosis or pulmonary embolism. N. Engl. J. Med. 1998, 338, 1169–1173. [Google Scholar] [CrossRef]
- Baron, J.A.; Gridley, G.; Weiderpass, E.; Nyrén, O.; Linet, M. Venous thromboembolism and cancer. Lancet Lond. Engl. 1998, 351, 1077–1080. [Google Scholar] [CrossRef]
- Clayburgh, D.R.; Stott, W.; Cordiero, T.; Park, R.; Detwiller, K.; Buniel, M.; Flint, P.; Schindler, J.; Andersen, P.; Wax, M.K.; et al. Prospective study of venous thromboembolism in patients with head and neck cancer after surgery. JAMA Otolaryngol. Head Neck Surg. 2013, 139, 1143–1150. [Google Scholar] [CrossRef]
- Kakei, Y.; Akashi, M.; Hasegawa, T.; Minamikawa, T.; Usami, S.; Komori, T. Incidence of Venous Thromboembolism After Oral Oncologic Surgery With Simultaneous Reconstruction. J. Oral Maxillofac. Surg. 2016, 74, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Kosugi, T.; Takagi, I.; Ariga, Y.; Okada, S.; Morimitsu, T.; Mihara, H. Fibrinolysis-coagulation system in patients with cancer of the head and neck. Preliminary report. Arch. Otorhinolaryngol. 1982, 236, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Jagielska, B.; Symonides, M.; Stachurska, E.; Kawecki, A.; Kraszewska, E. Coagulation disorders in patients with locally advanced head and neck cancer—should they really be disregarded? Neoplasma 2011, 58, 129–134. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Christensen, A.; Kiss, K.; Lelkaitis, G.; Juhl, K.; Persson, M.; Charabi, B.W.; Mortensen, J.; Forman, J.L.; Sørensen, A.L.; Jensen, D.H.; et al. Urokinase-type plasminogen activator receptor (uPAR), tissue factor (TF) and epidermal growth factor receptor (EGFR): Tumor expression patterns and prognostic value in oral cancer. BMC Cancer 2017, 17, 572. [Google Scholar] [CrossRef]
- Chang, M.-C.; Chan, C.-P.; Ho, Y.-S.; Lee, J.-J.; Lin, P.-S.; Lin, B.-R.; Huang, Y.-L.; Hahn, L.-J.; Yeh, H.-W.; Wang, Y.-J.; et al. Signaling pathways for induction of platelet aggregation by SAS tongue cancer cells--a mechanism of hematogenous metastasis. J. Oral Pathol. Med. 2009, 38, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.G.; Man, Q.W.; Zhang, W.; Li, C.; Xiong, X.P.; Zhu, J.Y.; Wang, W.M.; Sun, Z.J.; Jia, J.; Zhang, W.F.; et al. Elevated Level of Circulating Platelet-derived Microparticles in Oral Cancer. J. Dent. Res. 2016, 95, 87–93. [Google Scholar] [CrossRef]
- Parks, R.R.; Yan, S.D.; Huang, C.C. Tumor necrosis factor-alpha production in human head and neck squamous cell carcinoma. Laryngoscope 1994, 104, 860–864. [Google Scholar] [CrossRef]
- Haen, P.; Mege, D.; Crescence, L.; Dignat-George, F.; Dubois, C.; Panicot-Dubois, L. Thrombosis Risk Associated with Head and Neck Cancer: A Review. Int. J. Mol. Sci. 2019, 20, 2838. [Google Scholar] [CrossRef]
- Hier, M.P.; Black, M.J.; Shenouda, G.; Sadeghi, N.; Karp, S.E. A murine model for the immunotherapy of head and neck squamous cell carcinoma. Laryngoscope 1995, 105, 1077–1080. [Google Scholar] [CrossRef]
- Paolini, F.; Massa, S.; Manni, I.; Franconi, R.; Venuti, A. Immunotherapy in new pre-clinical models of HPV-associated oral cancers. Hum. Vaccines Immunother. 2013, 9, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Mezouar, S.; Darbousset, R.; Dignat-George, F.; Panicot-Dubois, L.; Dubois, C. Inhibition of platelet activation prevents the P-selectin and integrin-dependent accumulation of cancer cell microparticles and reduces tumor growth and metastasis in vivo. Int. J. Cancer 2015, 136, 462–475. [Google Scholar] [CrossRef] [PubMed]
- Panicot-Dubois, L.; Thomas, G.M.; Furie, B.C.; Furie, B.; Lombardo, D.; Dubois, C. Bile salt-dependent lipase interacts with platelet CXCR4 and modulates thrombus formation in mice and humans. J. Clin. Investig. 2007, 117, 3708–3719. [Google Scholar] [CrossRef]
- Dubois, C.; Panicot-Dubois, L.; Merrill-Skoloff, G.; Furie, B.; Furie, B.C. Glycoprotein VI-dependent and -independent pathways of thrombus formation in vivo. Blood 2006, 107, 3902–3906. [Google Scholar] [CrossRef]
- Dubois, C.; Panicot-Dubois, L.; Gainor, J.F.; Furie, B.C.; Furie, B. Thrombin-initiated platelet activation in vivo is vWF independent during thrombus formation in a laser injury model. J. Clin. Investig. 2007, 117, 953–960. [Google Scholar] [CrossRef]
- Zarà, M.; Canobbio, I.; Visconte, C.; Canino, J.; Torti, M.; Guidetti, G.F. Molecular mechanisms of platelet activation and aggregation induced by breast cancer cells. Cell. Signal. 2018, 48, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Sousou, T.; Khorana, A.A. New insights into cancer-associated thrombosis. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 316–320. [Google Scholar] [CrossRef]
- Gerotziafas, G.T.; Galea, V.; Mbemba, E.; Khaterchi, A.; Sassi, M.; Baccouche, H.; Prengel, C.; van Dreden, P.; Hatmi, M.; Bernaudin, J.F.; et al. Tissue factor over-expression by human pancreatic cancer cells BXPC3 is related to higher prothrombotic potential as compared to breast cancer cells MCF7. Thromb. Res. 2012, 129, 779–786. [Google Scholar] [CrossRef] [PubMed]
- Abu Saadeh, F.; Norris, L.; O’Toole, S.; Mohamed, B.M.; Langhe, R.; O’Leary, J.; Gleeson, N. Tumour expresion of tissue factor and tissue factor pathway inhibitor in ovarian cancer- relationship with venous thrombosis risk. Thromb. Res. 2013, 132, 627–634. [Google Scholar] [CrossRef]
- Darbousset, R.; Thomas, G.M.; Mezouar, S.; Frère, C.; Bonier, R.; Mackman, N.; Renné, T.; Dignat-George, F.; Dubois, C.; Panicot-Dubois, L. Tissue factor-positive neutrophils bind to injured endothelial wall and initiate thrombus formation. Blood 2012, 120, 2133–2143. [Google Scholar] [CrossRef] [PubMed]
- Furie, B.; Furie, B.C. Mechanisms of thrombus formation. N. Engl. J. Med. 2008, 359, 938–949. [Google Scholar] [CrossRef]
- Darbousset, R.; Delierneux, C.; Mezouar, S.; Hego, A.; Lecut, C.; Guillaumat, I.; Riederer, M.A.; Evans, R.J.; Dignat-George, F.; Panicot-Dubois, L.; et al. P2 × 1 expressed on polymorphonuclear neutrophils and platelets is required for thrombosis in mice. Blood 2014, 124, 2575–2585. [Google Scholar] [CrossRef] [PubMed]
- Vandendries, E.R.; Hamilton, J.R.; Coughlin, S.R.; Furie, B.; Furie, B.C. Par4 is required for platelet thrombus propagation but not fibrin generation in a mouse model of thrombosis. Proc. Natl. Acad. Sci. USA 2007, 104, 288–292. [Google Scholar] [CrossRef]
- Dupuis, A.; Bordet, J.-C.; Eckly, A.; Gachet, C. Platelet δ-Storage Pool Disease: An Update. J. Clin. Med. 2020, 9, 2508. [Google Scholar] [CrossRef] [PubMed]
- Stein, P.D.; Beemath, A.; Meyers, F.A.; Skaf, E.; Sanchez, J.; Olson, R.E. Incidence of venous thromboembolism in patients hospitalized with cancer. Am. J. Med. 2006, 119, 60–68. [Google Scholar] [CrossRef]
- Gerrard, J.M.; McNicol, A. Platelet storage pool deficiency, leukemia, and myelodysplastic syndromes. Leuk. Lymphoma 1992, 8, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Carr, M.E.; Hines, S.; Carr, S.L.; Todd, W.M.; Taylor, T.L.; Mohanty, L. Storage pool disease in chronic lymphocytic leukemia: Abnormal aggregation and secretion without bleeding. Am. J. Med. Sci. 1997, 313, 176–181. [Google Scholar] [CrossRef]
- Franco-Moreno, A.I.; Santero-García, M.; Cabezón-Gutiérrez, L.; Martín-Díaz, R.M.; García-Navarro, M.J. Acquired hemophilia A in a patient with hepatocellular carcinoma: A case report and literature review. Ann. Hematol. 2016, 95, 2099–2100. [Google Scholar] [CrossRef]
- Shetty, S.; Kasatkar, P.; Ghosh, K. Pathophysiology of acquired von Willebrand disease: A concise review. Eur. J. Haematol. 2011, 87, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Ong, H.S.; Gokavarapu, S.; Al-Qamachi, L.; Yin, M.Y.; Su, L.X.; Ji, T.; Zhang, C.P. Justification of routine venous thromboembolism prophylaxis in head and neck cancer reconstructive surgery. Head Neck 2017, 39, 2450–2458. [Google Scholar] [CrossRef] [PubMed]
- Lodders, J.N.; Parmar, S.; Stienen, N.L.M.; Martin, T.J.; Karagozoglu, K.H.; Heymans, M.W.; Forouzanfar, T. Incidence of symptomatic venous thromboembolism in oncological oral and maxillofacial operations: Retrospective analysis. Br. J. Oral Maxillofac. Surg. 2015, 53, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Falanga, A.; Russo, L.; Milesi, V.; Vignoli, A. Mechanisms and risk factors of thrombosis in cancer. Crit. Rev. Oncol. Hematol. 2017, 118, 79–83. [Google Scholar] [CrossRef]
- Wakasaki, T.; Kiyohara, H.; Omori, H.; Nishi, K.; Taguchi, K.; Rikimaru, F.; Toh, S.; Higaki, Y.; Masuda, M. Massive internal jugular vein tumor thrombus derived from squamous cell carcinoma of the head and neck: Two case reports. Oral Maxillofac. Surg. 2017, 21, 69–74. [Google Scholar] [CrossRef]
- Aoyama, K.-I.; Tamura, M.; Uchibori, M.; Nakanishi, Y.; Arai, T.; Aoki, T.; Osawa, Y.; Kaneko, A.; Ota, Y. Trousseau syndrome in a patient with advanced oral squamous cell carcinoma: A case report. J. Med. Case Reports 2019, 13, 26. [Google Scholar] [CrossRef]
- Batman, B.; van Bladel, E.R.; van Hamersveld, M.; Pasker-de Jong, P.C.M.; Korporaal, S.J.A.; Urbanus, R.T.; Roest, M.; Boven, L.A.; Fijnheer, R. Agonist-induced platelet reactivity correlates with bleeding in haemato-oncological patients. Vox Sang. 2017, 112, 773–779. [Google Scholar] [CrossRef]
- Lager, T.W.; Roetman, J.J.; Kunkel, J.; Thacker, M.; Sheets, J.N.; Egland, K.A.; Miles, C.M.; Larson, M.K.; Gubbels, J.A.A. Sushi Domain Containing 2 (SUSD2) inhibits platelet activation and binding to high-grade serous ovarian carcinoma cells. Platelets 2018, 29, 834–837. [Google Scholar] [CrossRef]
- Selle, F.; James, C.; Tuffigo, M.; Pillois, X.; Viallard, J.-F.; Alessi, M.-C.; Fiore, M. Clinical and Laboratory Findings in Patients with δ-Storage Pool Disease: A Case Series. Semin. Thromb. Hemost. 2017, 43, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Falke, S.; Avenarius, H.J.; Deicher, H. [The induction of an acquired storage pool deficiency of the thrombocytes by IgG antibodies in systemic lupus erythematosus and chronic polyarthritis]. Z. Rheumatol. 1990, 49, 70–76. [Google Scholar]
- Gunning, W.T.; Karabin, B.L.; Blomquist, T.M.; Grubb, B.P. Postural orthostatic tachycardia syndrome is associated with platelet storage pool deficiency. Medicine 2016, 95, e4849. [Google Scholar] [CrossRef] [PubMed]
- Balduini, A.; Di Buduo, C.A.; Malara, A.; Lecchi, A.; Rebuzzini, P.; Currao, M.; Pallotta, I.; Jakubowski, J.A.; Cattaneo, M. Constitutively released adenosine diphosphate regulates proplatelet formation by human megakaryocytes. Haematologica 2012, 97, 1657–1665. [Google Scholar] [CrossRef] [PubMed]
- Kiran Gotru, S.; van Geffen, J.P.; Nagy, M.; Mammadova-Bach, E.; Eilenberger, J.; Volz, J.; Manukjan, G.; Schulze, H.; Wagner, L.; Eber, S.; et al. Defective Zn2+ homeostasis in mouse and human platelets with α- and δ-storage pool diseases. Sci. Rep. 2019, 9, 8333. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, A.; Napierala, M.; Golasik, M.; Herman, M.; Walas, S.; Piekoszewski, W.; Szyfter, W.; Szyfter, K.; Golusinski, W.; Baralkiewicz, D.; et al. Metal concentrations in hair of patients with various head and neck cancers as a diagnostic aid. Biometals 2016, 29, 81–93. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ressnerova, A.; Raudenska, M.; Holubova, M.; Svobodova, M.; Polanska, H.; Babula, P.; Masarik, M.; Gumulec, J. Zinc and Copper Homeostasis in Head and Neck Cancer: Review and Meta-Analysis. Curr. Med. Chem. 2016, 23, 1304–1330. [Google Scholar] [CrossRef] [PubMed]




| Tumor-Free Mice (WT) Mean (SD) | Tongue Tumor Mice (AT84) Mean (SD) | P (Wilcoxon) | |
|---|---|---|---|
| Platelets (nb × 103/μL) | 883.2 (399.6) | 846.5 (254) | 0.893 (NS) |
| Neutrophils (nb/μL) | 1238 (486) | 1381 (479) | 0.588 (NS) |
| Monocytes (nb/μL) | 159.8 (104.8) | 242.7 (185.5) | 0.168 (NS) |
| Lymphocytes (nb/μL) | 3095 (1851) | 2682 (1571) | 0.685 (NS) |
| Tail bleeding time (seconds) | 128.9 (28.48) | 146.2 (48.62) | 0.361 (NS) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haen, P.; Crescence, L.; Mege, D.; Altié, A.; Dubois, C.; Panicot-Dubois, L. Oral Squamous Cell Carcinoma Is Associated with a Low Thrombosis Risk Due to Storage Pool Deficiency in Platelets. Biomedicines 2021, 9, 228. https://doi.org/10.3390/biomedicines9030228
Haen P, Crescence L, Mege D, Altié A, Dubois C, Panicot-Dubois L. Oral Squamous Cell Carcinoma Is Associated with a Low Thrombosis Risk Due to Storage Pool Deficiency in Platelets. Biomedicines. 2021; 9(3):228. https://doi.org/10.3390/biomedicines9030228
Chicago/Turabian StyleHaen, Pierre, Lydie Crescence, Diane Mege, Alexandre Altié, Christophe Dubois, and Laurence Panicot-Dubois. 2021. "Oral Squamous Cell Carcinoma Is Associated with a Low Thrombosis Risk Due to Storage Pool Deficiency in Platelets" Biomedicines 9, no. 3: 228. https://doi.org/10.3390/biomedicines9030228
APA StyleHaen, P., Crescence, L., Mege, D., Altié, A., Dubois, C., & Panicot-Dubois, L. (2021). Oral Squamous Cell Carcinoma Is Associated with a Low Thrombosis Risk Due to Storage Pool Deficiency in Platelets. Biomedicines, 9(3), 228. https://doi.org/10.3390/biomedicines9030228







