The Neuropeptide VIP Limits Human Osteoclastogenesis: Clinical Associations with Bone Metabolism Markers in Patients with Early Arthritis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Isolation of PBMCs and Cell Culture
2.3. Quantification of Tartrate-Resistant Acid Phosphatse Positive (TRAP+) Stained Osteoclast
2.4. Resorption Assay
2.5. Real-Time PCR Analysis
2.6. Transcription Factor Activity Assay (TransAM)
2.7. Inmunofluorescence Microscopy
2.8. Statistical Analysis and New Variables
3. Results
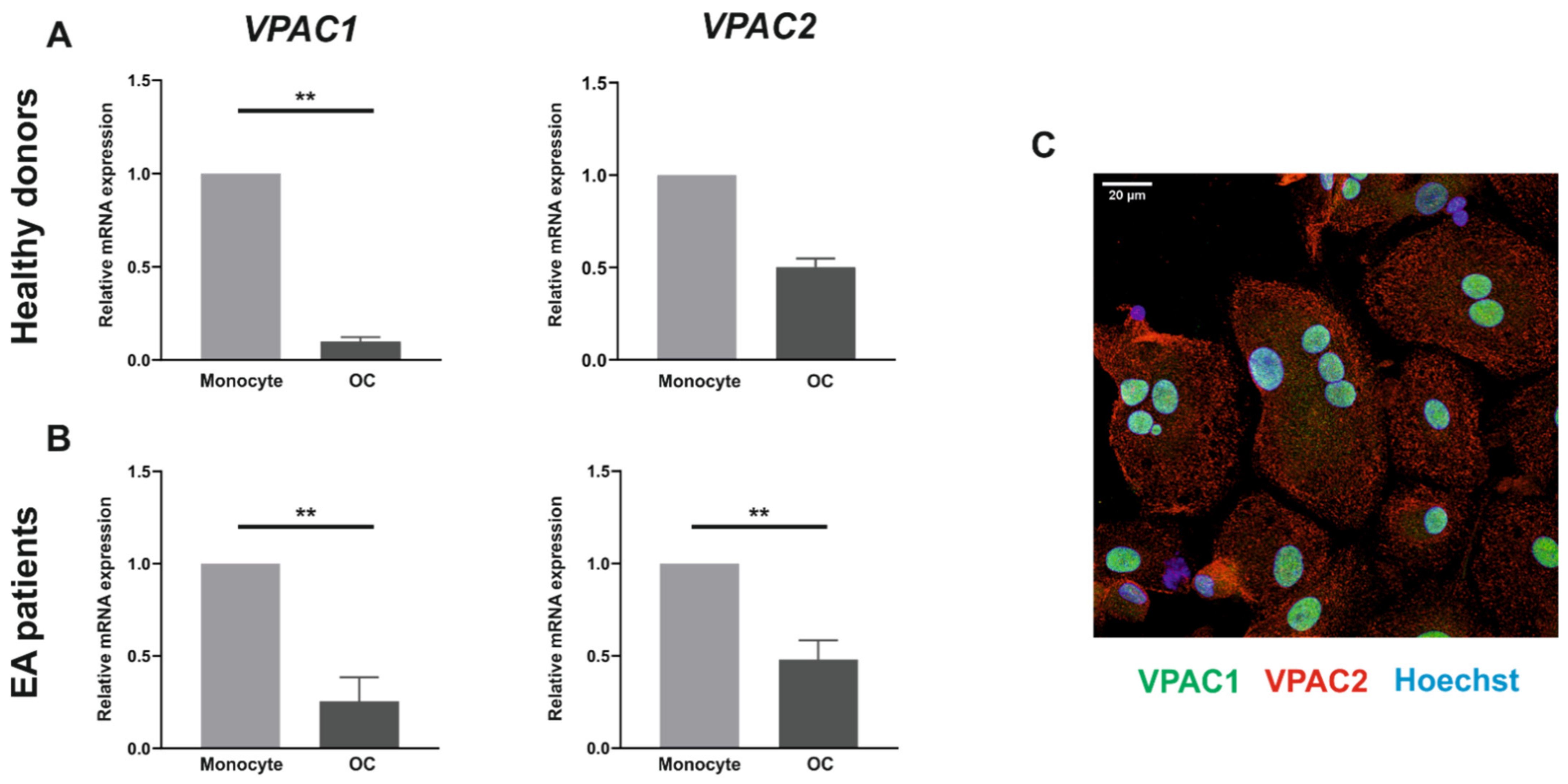
3.1. VPAC1 and VPAC2 Receptors Are Expressed by Both Human Monocytes and In Vitro Differentiated Osteoclasts
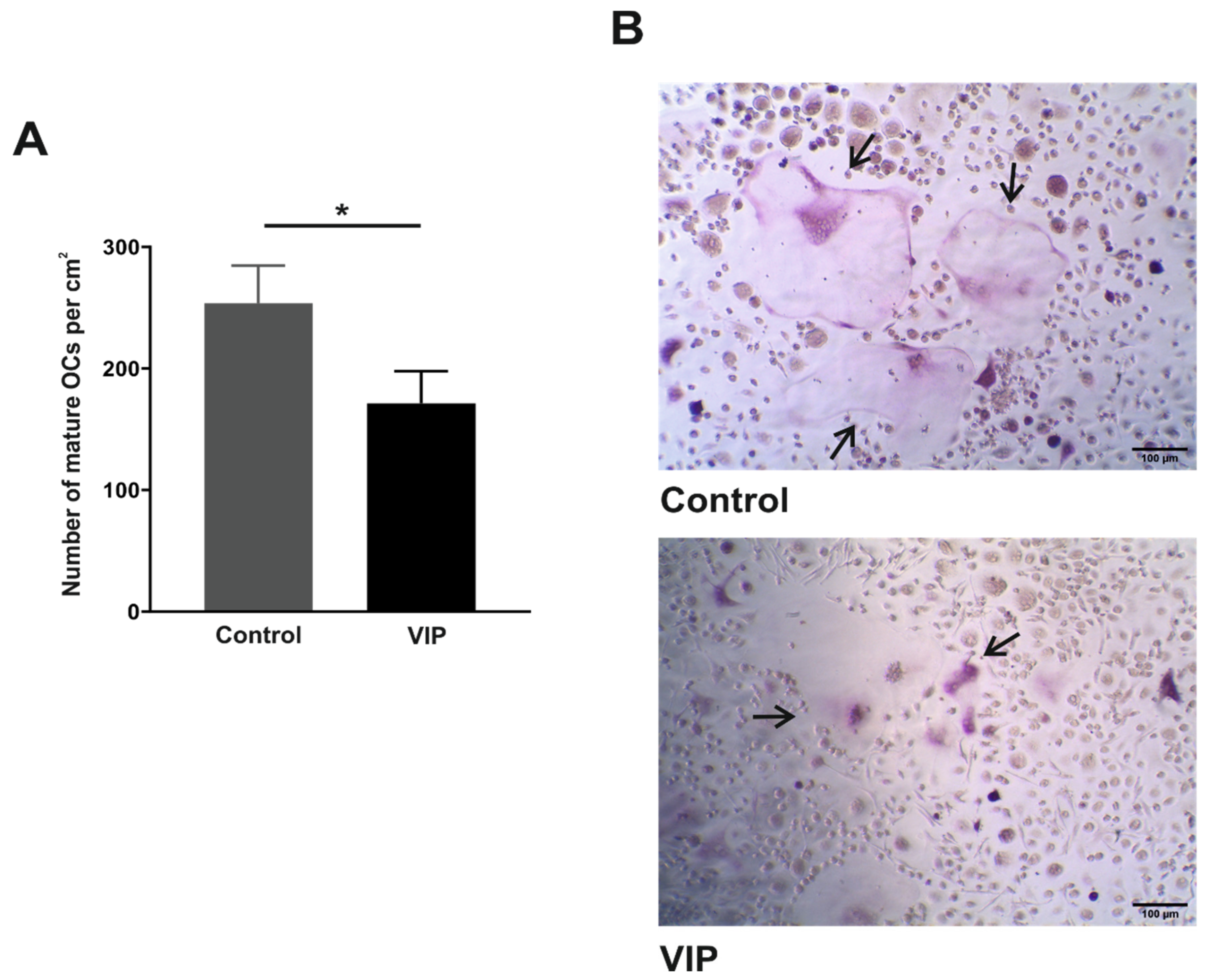
3.2. VIP Limits the In Vitro Osteoclast Differentiation
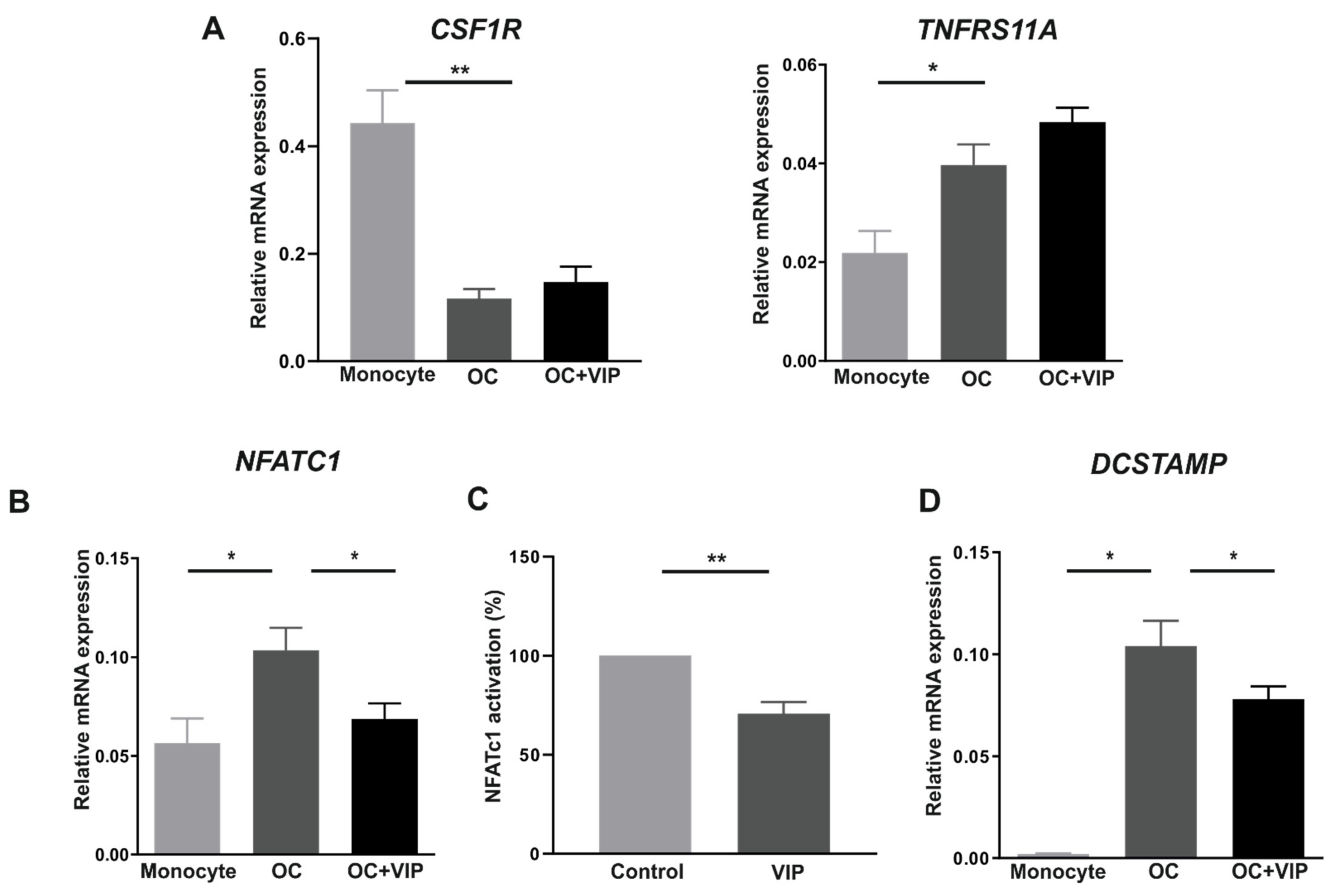
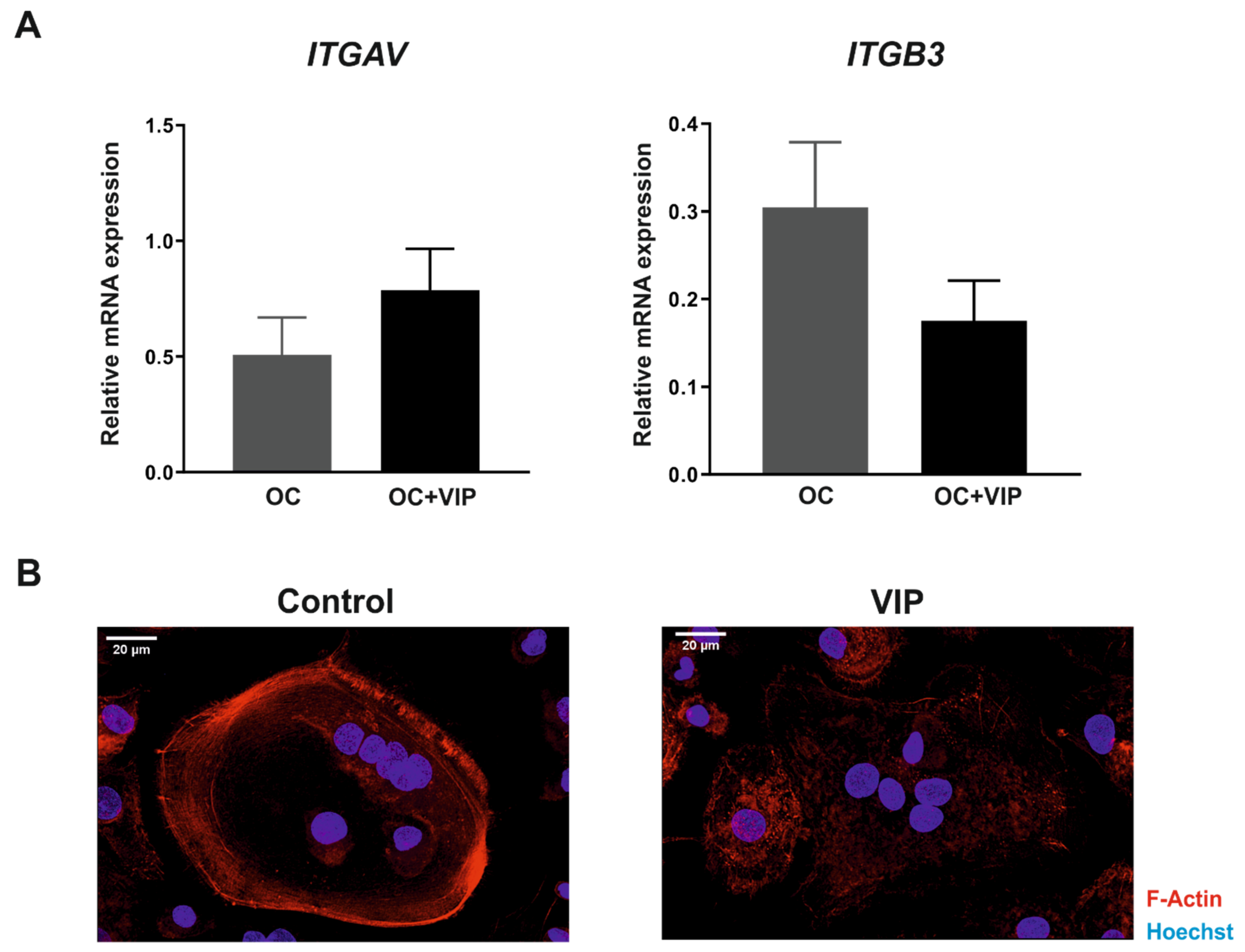
3.3. Osteoclast Specific Gene Markers Are Negatively Regulated by VIP
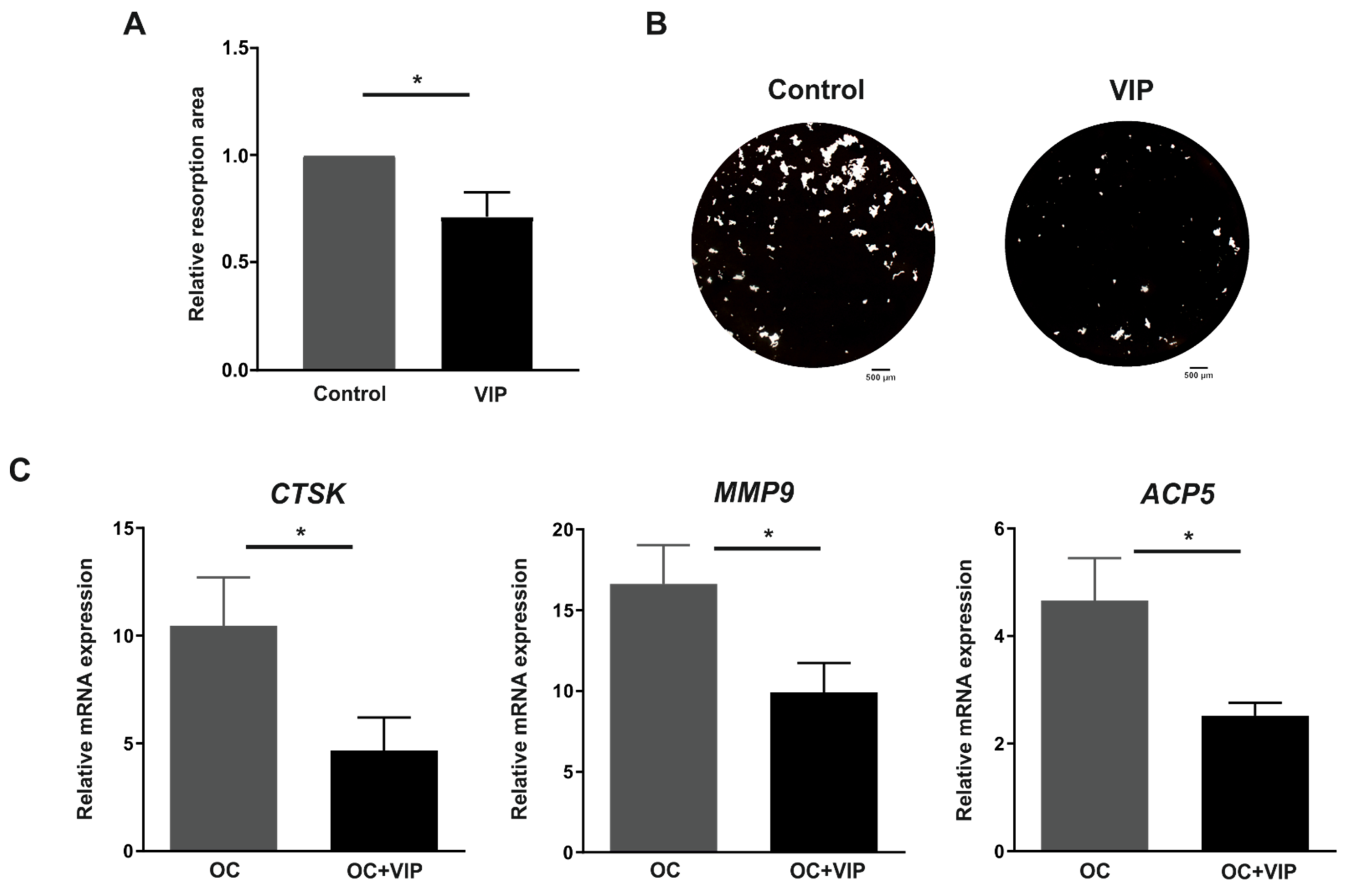
3.4. Osteoclast Resorptive Activity In Vitro Is Impaired in the Presence of VIP
3.5. The Formation of the Bone Resorption Lacuna Is Interfered by VIP
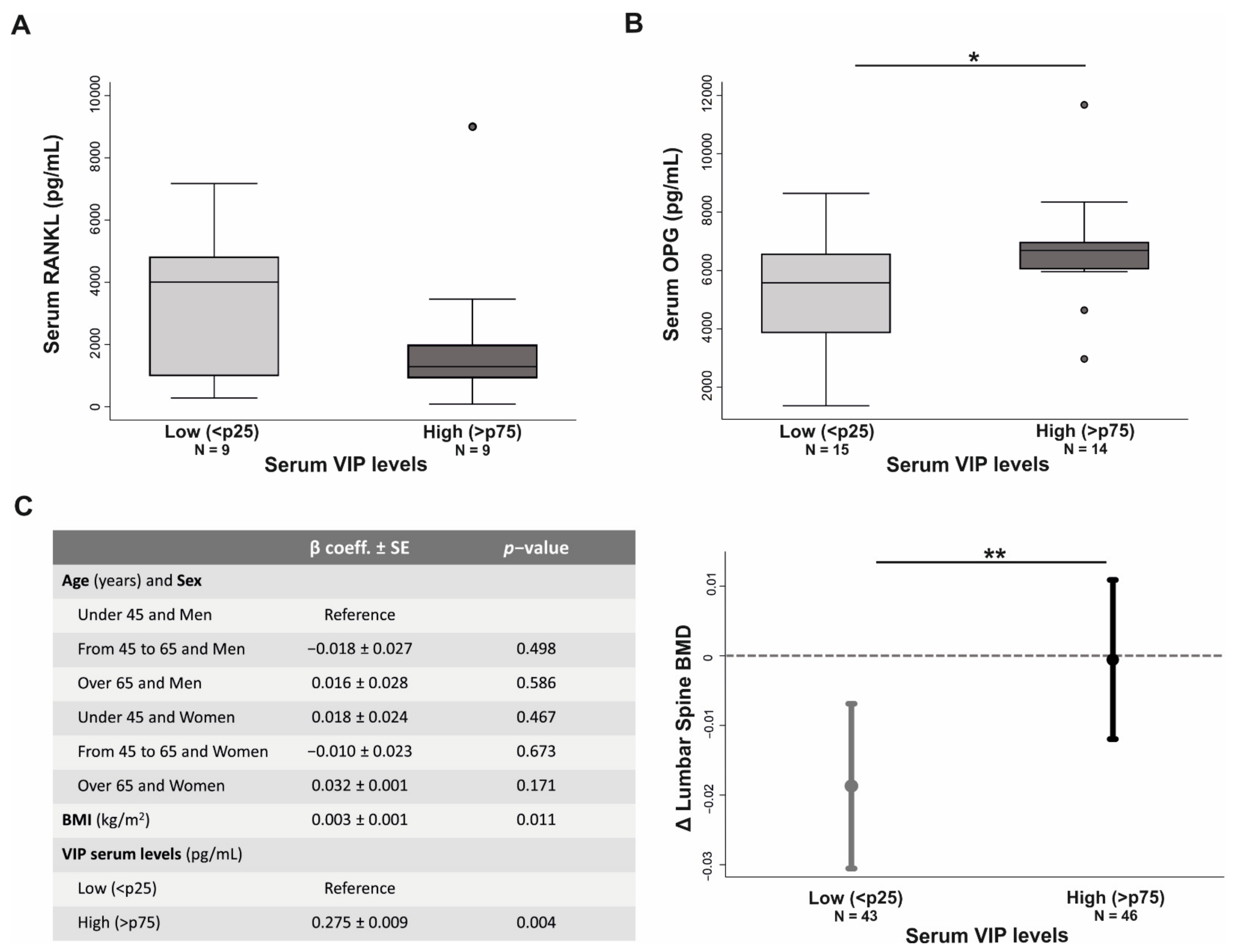
3.6. VIP Serum Levels Influences Bone Metabolism in Early Arthritis Patients
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fang, Q.; Zhou, C.; Nandakumar, K.S. Molecular and Cellular Pathways Contributing to Joint Damage in Rheumatoid Arthritis. Mediat. Inflamm. 2020, 2020, 3830212. [Google Scholar] [CrossRef]
- Gravallese, E.M.; Harada, Y.; Wang, J.T.; Gorn, A.H.; Thornhill, T.S.; Goldring, S.R. Identification of cell types responsible for bone resorption in rheumatoid arthritis and juvenile rheumatoid arthritis. Am. J. Pathol. 1998, 152, 943–951. [Google Scholar]
- Smolen, J.; Aletaha, D.; Barton, A. Rheumatoid arthritis. Nat. Rev. Dis. Primers 2018, 4, 18001. [Google Scholar] [CrossRef]
- Karmakar, S.; Kay, J.; Gravallese, E.M. Bone Damage in Rheumatoid Arthritis: Mechanistic Insights and Approaches to Prevention. Rheum. Dis. Clin. N. Am. 2010, 36, 385–404. [Google Scholar] [CrossRef] [PubMed]
- Shaw, A.T.; Gravallese, E.M. Mediators of inflammation and bone remodeling in rheumatic disease. Semin. Cell Dev. Biol. 2016, 49, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Blangy, A.; Bompard, G.; Guerit, D.; Marie, P.; Maurin, J.; Morel, A.; Vives, V. The osteoclast cytoskeleton—Current under-standing and therapeutic perspectives for osteoporosis. J. Cell Sci. 2020, 133, jcs244798. [Google Scholar] [CrossRef]
- Nakamura, I.; Pilkington, M.; Lakkakorpi, P.; Lipfert, L.; Sims, S.; Dixon, S.J.; Rodan, G.; Duong, L. Role of alpha(v)beta(3) integrin in osteoclast migration and formation of the sealing zone. J. Cell Sci. 1999, 112, 3985–3993. [Google Scholar] [CrossRef]
- Jacome-Galarza, C.E.; Percin, G.I.; Muller, J.T.; Mass, E.; Lazarov, T.; Eitler, J.; Rauner, M.; Yadav, V.K.; Crozet, L.; Bohm, M.; et al. Developmental origin, functional maintenance and genetic rescue of osteoclasts. Nat. Cell Biol. 2019, 568, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Takahashi, N.; Udagawa, N.; Tamura, T.; Akatsu, T.; Stanley, E. Mcsf is indispensable for both proliferation and differentiation of osteoclast progenitors. J. Clin. Investig. 1993, 91, 257–263. [Google Scholar] [CrossRef]
- Takayanagi, H.K.; Sunhwa, K.; Takako, N.; Hiroshi, I.; Masashi, Y.; Hiroki, S.; Akio, I.; Miho, Y.; Taeko, I.; Jun, I.; et al. Induction and activation of the transcription factor nfatc1 (nfat2) integrate rankl signaling in terminal differentiation of osteoclasts. Dev. Cell 2002, 3, 889–901. [Google Scholar] [CrossRef]
- Park, J.H.; Lee, N.K.; Lee, A.S.Y. Current Understanding of RANK Signaling in Osteoclast Differentiation and Maturation. Mol. Cells 2017, 40, 706–713. [Google Scholar] [CrossRef]
- Dodds, R.A.; James, I.E.; Rieman, D.; Ahern, R.; Hwang, S.M.; Connor, J.R.; Thompson, S.D.; Veber, D.F.; Drake, F.H.; Holmes, S.; et al. Human Osteoclast Cathepsin K Is Processed Intracellularly Prior to Attachment and Bone Resorption. J. Bone Miner. Res. 2001, 16, 478–486. [Google Scholar] [CrossRef]
- Zhu, L.; Tang, Y.; Li, X.-Y.; Keller, E.T.; Yang, J.; Cho, J.-S.; Feinberg, T.Y.; Weiss, S.J. Osteoclast-mediated bone resorption is controlled by a compensatory network of secreted and membrane-tethered metalloproteinases. Sci. Transl. Med. 2020, 12, 12. [Google Scholar] [CrossRef] [PubMed]
- Udagawa, N.; Koide, M.; Nakamura, M.; Nakamichi, Y.; Yamashita, T.; Uehara, S.; Kobayashi, Y.; Furuya, Y.; Yasuda, H.; Fukuda, C.; et al. Osteoclast differentiation by RANKL and OPG signaling pathways. J. Bone Miner. Metab. 2021, 39, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Walsh, M.; Choi, Y. Biology of the ranklrank-opg system in immunity, bone, and beyond. Front. Immunol. 2014, 5, 511. [Google Scholar] [CrossRef]
- Steffen, U.; Schett, G.; Bozec, A. How Autoantibodies Regulate Osteoclast Induced Bone Loss in Rheumatoid Arthritis. Front. Immunol. 2019, 10, 1483. [Google Scholar] [CrossRef]
- Neumann, E.; Gay, S.; Müller-Ladner, U. The RANK/RANKL/osteoprotegerin system in rheumatoid arthritis: New insights from animal models. Arthritis Rheum. 2005, 52, 2960–2967. [Google Scholar] [CrossRef] [PubMed]
- Walsh, N.C.; Crotti, T.N.; Goldring, S.R.; Gravallese, E.M. Rheumatic diseases: The effects of inflammation on bone. Immunol. Rev. 2005, 208, 228–251. [Google Scholar] [CrossRef] [PubMed]
- Guler-Yuksel, M.; Bijsterbosch, J.; Goekoop-Ruiterman, Y.P.M.; De Vries-Bouwstra, J.K.; Hulsmans, H.M.J.; De Beus, W.M.; Han, K.H.; Breedveld, F.C.; Dijkmans, B.A.C.; Allaart, C.F.; et al. Changes in bone mineral density in patients with recent onset, active rheumatoid arthritis. Ann. Rheum. Dis. 2008, 67, 823–828. [Google Scholar] [CrossRef] [PubMed]
- Devlin, J.; Lilley, J.; Gough, A.; Huissoon, A.; Holder, R.; Reece, R.; Perkins, P.; Emery, P. Clinical associations of dual-energy X-ray absorptiometry measurement of hand bone mass in rheumatoid arthritis. Br. J. Rheumatol. 1996, 35, 1256–1262. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Deodhar, A.A.; Brabyn, J.; Pande, I.; Scott, D.L.; Woolf, A.D. Hand bone densitometry in rheumatoid arthritis, a five year longitudinal study: An outcome measure and a prognostic marker. Ann. Rheum. Dis. 2003, 62, 767–770. [Google Scholar] [CrossRef]
- Llorente, I.; Garcia-Castaneda, N.; Valero, C.; Gonzalez-Alvaro, I.; Castaneda, S. Osteoporosis in rheumatoid arthritis: Dan-gerous liaisons. Front. Med. 2020, 7, 601–618. [Google Scholar] [CrossRef] [PubMed]
- Auréal, M.; Machuca-Gayet, I.; Coury, F. Rheumatoid Arthritis in the View of Osteoimmunology. Biomolecules 2020, 11, 48. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Li, Z.; Liu, Y.; Gao, D.; Zhang, X.; Hao, J.; Yang, F. Neural regulation of bone remodeling: Identifying novel neural molecules and pathways between brain and bone. J. Cell. Physiol. 2019, 234, 5466–5477. [Google Scholar] [CrossRef] [PubMed]
- Lerner, U.H. Neuropeptidergic regulation of bone resorption and bone formation. J. Musculoskelet. Neuronal Interact. 2002, 2, 440–447. [Google Scholar] [PubMed]
- Lerner, U.H.; Persson, E. Osteotropic effects by the neuropeptides calcitonin gene-related peptide, substance P and vasoactive intestinal peptide. J. Musculoskelet. Neuronal Interact. 2008, 8, 154–165. [Google Scholar]
- Lundberg, P.; Boström, I.; Mukohyama, H. Neuro-hormonal control of bone metabolism: Vasoactive intestinal peptide stimu-lates alkaline phosphatase activity and mrna expression in mouse calvarial osteoblasts as well as calcium accumulation min-eralized bone nodules. Regul. Pept. 1999, 85, 47–58. [Google Scholar] [CrossRef]
- Lundberg, P.; Lie, A.; Bjurholm, A.; Lehenkari, P.; Horton, M.; Lerner, U.; Ransjö, M. Vasoactive intestinal peptide regulates osteoclast activity via specific binding sites on both osteoclasts and osteoblasts. Bone 2000, 27, 803–810. [Google Scholar] [CrossRef]
- Mukohyama, H.; Ransjö, M.; Taniguchi, H.; Ohyama, T.; Lerner, U. The Inhibitory Effects of Vasoactive Intestinal Peptide and Pituitary Adenylate Cyclase-Activating Polypeptide on Osteoclast Formation Are Associated with Upregulation of Osteoprotegerin and Downregulation of RANKL and RANK. Biochem. Biophys. Res. Commun. 2000, 271, 158–163. [Google Scholar] [CrossRef]
- Qu, H.; Zhuang, Y.; Zhu, L.; Zhao, Z.; Wang, K. The effects of vasoactive intestinal peptide on rankl-induced osteoclast for-mation. Ann. Transl. Med. 2021, 9, 127. [Google Scholar] [CrossRef]
- Gozes, I. VIP, From Gene to Behavior and Back: Summarizing my 25 Years of Research. J. Mol. Neurosci. 2008, 36, 115–124. [Google Scholar] [CrossRef]
- Gomariz, R.P.; Juarranz, Y.; Carrión, M.; Pérez-García, S.; Romero, R.V.; González-Álvaro, I.; Gutiérrez-Cañas, I.; Lamana, A.; Martínez, M.D.C. An Overview of VPAC Receptors in Rheumatoid Arthritis: Biological Role and Clinical Significance. Front. Endocrinol. 2019, 10, 729. [Google Scholar] [CrossRef]
- Martínez, C.; Juarranz, Y.; Gutiérrez-Cañas, I.; Carrión, M.; Pérez-García, S.; Villanueva-Romero, R.; Castro, D.; Lamana, A.; Mellado, M.; González-Álvaro, I.; et al. A Clinical Approach for the Use of VIP Axis in Inflammatory and Autoimmune Diseases. Int. J. Mol. Sci. 2019, 21, 65. [Google Scholar] [CrossRef] [PubMed]
- Martínez, C.; Ortiz, A.M.; Juarranz, Y.; Lamana, A.; Seoane, I.V.; Leceta, J.; García-Vicuña, R.; Gomariz, R.P.; González-Álvaro, I. Serum Levels of Vasoactive Intestinal Peptide as a Prognostic Marker in Early Arthritis. PLoS ONE 2014, 9, e85248. [Google Scholar] [CrossRef] [PubMed]
- Seoane, I.V.; Martínez, M.D.C.; García-Vicuña, R.; Ortiz, A.M.; Juarranz, Y.; Talayero, V.C.; González-Álvaro, I.; Gomariz, R.P.; Lamana, A. Vasoactive intestinal peptide gene polymorphisms, associated with its serum levels, predict treatment requirements in early rheumatoid arthritis. Sci. Rep. 2018, 8, 2035. [Google Scholar] [CrossRef] [PubMed]
- Juarranz, Y.; Abad, C.; Martinez, C.; Arranz, A.; Gutierrez-Cañas, I.; Rosignoli, F.; Gomariz, R.P.; Leceta, J. Protective effect of vasoactive intestinal peptide on bone destruction in the collagen-induced arthritis model of rheumatoid arthritis. Arthritis Res. 2005, 7, R1034–R1045. [Google Scholar] [CrossRef]
- Delgado, M.; Abad, C.; Martinez, C.; Leceta, J.; Gomariz, R.P. Vasoactive intestinal peptide prevents experimental arthritis by downregulating both autoimmune and inflammatory components of the disease. Nat. Med. 2001, 7, 563–568. [Google Scholar] [CrossRef]
- Eger, M.; Liron, T.; Hiram-Bab, S.; Awida, Z.; Giladi, E.; Dangoor, D.; Fridkin, M.; Kohavi, D.; Gozes, I.; Gabet, Y. Therapeutic Potential of Vasoactive Intestinal Peptide and its Derivative Stearyl-Norleucine-VIP in Inflammation-Induced Osteolysis. Front. Pharmacol. 2021, 12, 638128. [Google Scholar] [CrossRef]
- Gonzalez-Alvaro, I.; Ortiz, A.M.; Álvaro-Gracia, J.M.; Castañeda, S.; Díaz-Sánchez, B.; Carvajal, I.; García-Vadillo, J.A.; Humbría, A.; López-Bote, J.P.; Patiño, E.; et al. Interleukin 15 Levels in Serum May Predict a Severe Disease Course in Patients with Early Arthritis. PLoS ONE 2011, 6, e29492. [Google Scholar] [CrossRef]
- Schett, G.; Gravallese, E. Bone erosion in rheumatoid arthritis: Mechanisms, diagnosis and treatment. Nat. Rev. Rheumatol. 2012, 8, 656–664. [Google Scholar] [CrossRef]
- Fardellone, P.; Salawati, E.; Le Monnier, L.; Goëb, V. Bone Loss, Osteoporosis, and Fractures in Patients with Rheumatoid Arthritis: A Review. J. Clin. Med. 2020, 9, 3361. [Google Scholar] [CrossRef]
- Muschter, D.; Schäfer, N.; Stangl, H.; Straub, R.H.; Grässel, S. Sympathetic Neurotransmitters Modulate Osteoclastogenesis and Osteoclast Activity in the Context of Collagen-Induced Arthritis. PLoS ONE 2015, 10, e0139726. [Google Scholar] [CrossRef]
- Ransjö, M.; Lie, A.; Mukohyama, H.; Lundberg, P.; Lerner, U. Microisolated mouse osteoclasts express vip-1 and pacap re-ceptors. Biochem. Biophys. Res. Commun. 2000, 274, 400–404. [Google Scholar] [CrossRef] [PubMed]
- Carrión, M.; Pérez-García, S.; Martínez, C.; Juarranz, Y.; Estrada-Capetillo, L.; Puig-Kröger, A.; Gomariz, R.P.; Gutiérrez-Cañas, I. VIP impairs acquisition of the macrophage proinflammatory polarization profile. J. Leukoc. Biol. 2016, 100, 1385–1393. [Google Scholar] [CrossRef]
- Paniagua, R.T. C-fms-mediated differentiation and priming of monocyte lineage cells play a central role in autoimmune ar-thritis. Arthritis Res. Ther. 2010, 12, R32. [Google Scholar] [CrossRef]
- Arai, F.; Miyamoto, T.; Ohneda, O.; Inada, T.; Sudo, T.; Brasel, K.; Miyata, T.; Anderson, D.; Dirk, M.; Suda, T. Commitment and differentiation of osteoclast precursor cells by the sequential expression of c-fms and receptor activator of nuclear factor kappab (rank) receptors. J. Exp. Med. 1999, 190, 1741–1754. [Google Scholar] [CrossRef] [PubMed]
- Takayanagi, H. RANKL as the master regulator of osteoclast differentiation. J. Bone Miner. Metab. 2021, 39, 13–18. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, N. Regulation of NFATc1 in Osteoclast Differentiation. J. Bone Metab. 2014, 21, 233–241. [Google Scholar] [CrossRef]
- Carrión, M.; Juarranz, Y.; Pérez-García, S.; Jimeno, R.; Pablos, J.L.; Gomariz, R.P.; Gutiérrez-Cañas, I. RNA sensors in human osteoarthritis and rheumatoid arthritis synovial fibroblasts: Immune regulation by vasoactive intestinal peptide. Arthritis Rheum. 2011, 63, 1626–1636. [Google Scholar] [CrossRef] [PubMed]
- Szema, A.M.; Forsyth, E.; Ying, B.; Hamidi, S.A.; Chen, J.J.; Hwang, S.; Li, J.C.; Dwyer, D.S.; Ramiro-Diaz, J.M.; Giermakowska, W.; et al. NFATc3 and VIP in Idiopathic Pulmonary Fibrosis and Chronic Obstructive Pulmonary Disease. PLoS ONE 2017, 12, e0170606. [Google Scholar] [CrossRef]
- Ishii, T.; Ruiz-Torruella, M.; Ikeda, A.; Shindo, S.; Movila, A.; Mawardi, H.; Albassam, A.; Kayal, R.A.; Al-Dharrab, A.A.; Egashira, K.; et al. OC-STAMP promotes osteoclast fusion for pathogenic bone resorption in periodontitis via up-regulation of permissive fusogen CD. FASEB J. 2018, 32, 4016–4030. [Google Scholar] [CrossRef] [PubMed]
- Qu, H.; Wang, K.; Zhuang, Y. Expression of neuropeptide y and vasoactive intestine peptide in pathological tissues of con-genital pseudarthrosis of the tibia and its clinical significance. Int. J. Clin. Exp. Pathol. 2016, 9, 8172–8180. [Google Scholar]
- Logar, D.B.; Komadina, R.; Preželj, J.; Ostanek, B.; Trošt, Z.; Marc, J. Expression of bone resorption genes in osteoarthritis and in osteoporosis. J. Bone Miner. Metab. 2007, 25, 219–225. [Google Scholar] [CrossRef]
- Matsumoto, M.; Kogawa, M.; Wada, S.; Takayanagi, H.; Tsujimoto, M.; Katayama, S.; Hisatake, K.; Nogi, Y. Essential Role of p38 Mitogen-activated Protein Kinase in Cathepsin K Gene Expression during Osteoclastogenesis through Association of NFATc1 and PU. J. Biol. Chem. 2004, 279, 45969–45979. [Google Scholar] [CrossRef]
- Song, I.; Kim, J.H.; Kim, K.; Jin, H.M.; Youn, B.U.; Kim, N. Regulatory mechanism of NFATc1 in RANKL-induced osteoclast activation. FEBS Lett. 2009, 583, 2435–2440. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Kim, J.; Lee, J.; Jin, H.; Lee, S.; Fisher, D.; Kook, H.; Kim, K.; Choi, Y.; Kim, N. Nuclear factor of activated t cells c1 induces osteoclast-associated receptor gene expression during tumor necrosis factor-related activation-induced cyto-kine-mediated osteoclastogenesis. J. Biol. Chem. 2005, 280, 35209–35216. [Google Scholar] [CrossRef]
- Christensen, J.; Shastri, V.P. Matrix-metalloproteinase-9 is cleaved and activated by cathepsin K. BMC Res. Notes 2015, 8, 322. [Google Scholar] [CrossRef]
- Ljusberg, J.; Wang, Y.; Lång, P.; Norgård, M.; Dodds, R.; Hultenby, K.; Ek-Rylander, B.; Andersson, G. Proteolytic Excision of a Repressive Loop Domain in Tartrate-resistant Acid Phosphatase by Cathepsin K in Osteoclasts. J. Biol. Chem. 2005, 280, 28370–28381. [Google Scholar] [CrossRef]
- Ganea, D.; Hooper, K.; Kong, W. The neuropeptide vasoactive intestinal peptide: Direct effects on immune cells and in-volvement in inflammatory and autoimmune diseases. Acta Physiol. 2015, 213, 442–452. [Google Scholar] [CrossRef]
- Liu, Y.-P.; Guan, C.-X.; Bai, H.-B.; Qin, X.-Q.; Liu, H.-J. Effects of vasoactive intestinal peptide on LPS-induced MMP-9 expression by alveolar macrophages in rats. Zhong Nan Da Xue Xue Bao Yi Xue Ban 2005, 30, 645–649. [Google Scholar] [PubMed]
- Pérez-García, S.; Carrión, M.; Jimeno, R.; Ortiz, A.M.; González-Álvaro, I.; Fernández, J.; Gomariz, R.P.; Juarranz, Y. Urokinase Plasminogen Activator System in Synovial Fibroblasts from Osteoarthritis Patients: Modulation by Inflammatory Mediators and Neuropeptides. J. Mol. Neurosci. 2013, 52, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Novack, D.V.; Faccio, R. Osteoclast motility: Putting the brakes on bone resorption. Ageing Res. Rev. 2011, 10, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Jurdic, P.; Saltel, F.; Chabadel, A.; Destaing, O. Podosome and sealing zone: Specificity of the osteoclast model. Eur. J. Cell Biol. 2006, 85, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Crotti, T.N.; Flannery, M.; Walsh, N.C.; Fleming, J.D.; Goldring, S.R.; McHugh, K.P. NFATc1 regulation of the human β3 integrin promoter in osteoclast differentiation. Gene 2006, 372, 92–102. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castro-Vazquez, D.; Lamana, A.; Arribas-Castaño, P.; Gutiérrez-Cañas, I.; Villanueva-Romero, R.; Pérez-García, S.; Martínez, C.; Juarranz, Y.; Fernández de Córdoba, S.; González-Álvaro, I.; et al. The Neuropeptide VIP Limits Human Osteoclastogenesis: Clinical Associations with Bone Metabolism Markers in Patients with Early Arthritis. Biomedicines 2021, 9, 1880. https://doi.org/10.3390/biomedicines9121880
Castro-Vazquez D, Lamana A, Arribas-Castaño P, Gutiérrez-Cañas I, Villanueva-Romero R, Pérez-García S, Martínez C, Juarranz Y, Fernández de Córdoba S, González-Álvaro I, et al. The Neuropeptide VIP Limits Human Osteoclastogenesis: Clinical Associations with Bone Metabolism Markers in Patients with Early Arthritis. Biomedicines. 2021; 9(12):1880. https://doi.org/10.3390/biomedicines9121880
Chicago/Turabian StyleCastro-Vazquez, David, Amalia Lamana, Paula Arribas-Castaño, Irene Gutiérrez-Cañas, Raúl Villanueva-Romero, Selene Pérez-García, Carmen Martínez, Yasmina Juarranz, Sara Fernández de Córdoba, Isidoro González-Álvaro, and et al. 2021. "The Neuropeptide VIP Limits Human Osteoclastogenesis: Clinical Associations with Bone Metabolism Markers in Patients with Early Arthritis" Biomedicines 9, no. 12: 1880. https://doi.org/10.3390/biomedicines9121880
APA StyleCastro-Vazquez, D., Lamana, A., Arribas-Castaño, P., Gutiérrez-Cañas, I., Villanueva-Romero, R., Pérez-García, S., Martínez, C., Juarranz, Y., Fernández de Córdoba, S., González-Álvaro, I., Gomariz, R. P., & Carrión, M. (2021). The Neuropeptide VIP Limits Human Osteoclastogenesis: Clinical Associations with Bone Metabolism Markers in Patients with Early Arthritis. Biomedicines, 9(12), 1880. https://doi.org/10.3390/biomedicines9121880








