Displacement of Gray Matter and Incidence of Seizures in Patients with Cerebral Cavernous Malformations
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patient Population
2.2. MRI Protocol
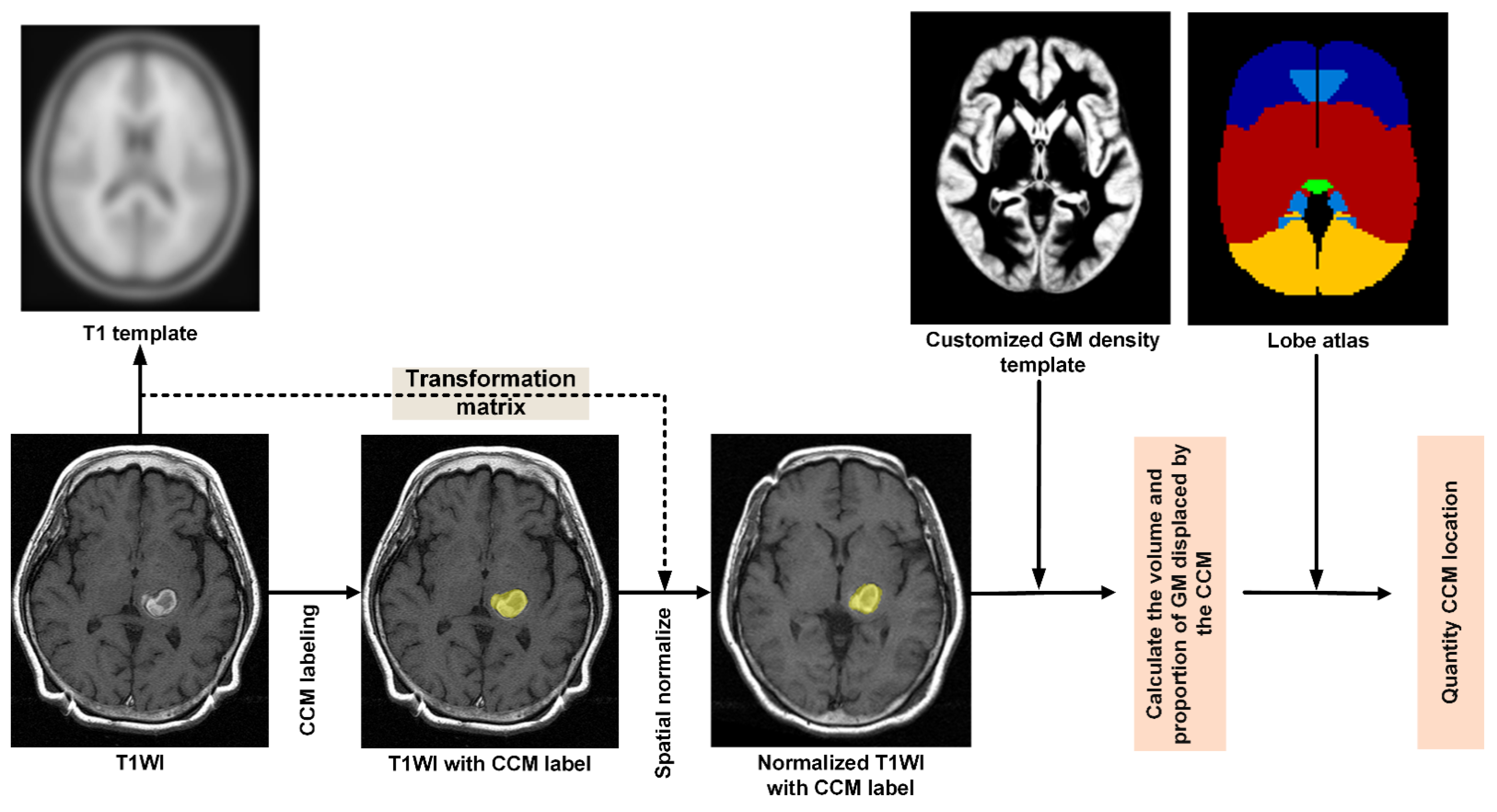
2.3. Quantitative CCM Evaluation
2.3.1. Stage 1–Preprocessing Control MPRAGE Images
- Step C1. Image format conversion
- Step C2. Resetting image orientation
- Step C3. Spatial normalization to the Montreal Neurological Institute (MNI) space
- Step C4. Establishing a customized GM density template in the MNI space
2.3.2. Stage 2—Preprocessing of T1WI Images from Patients
- Step P1. Conversion of DICOM to NIfTI format
- Step P2. Resetting the T1WI orientation
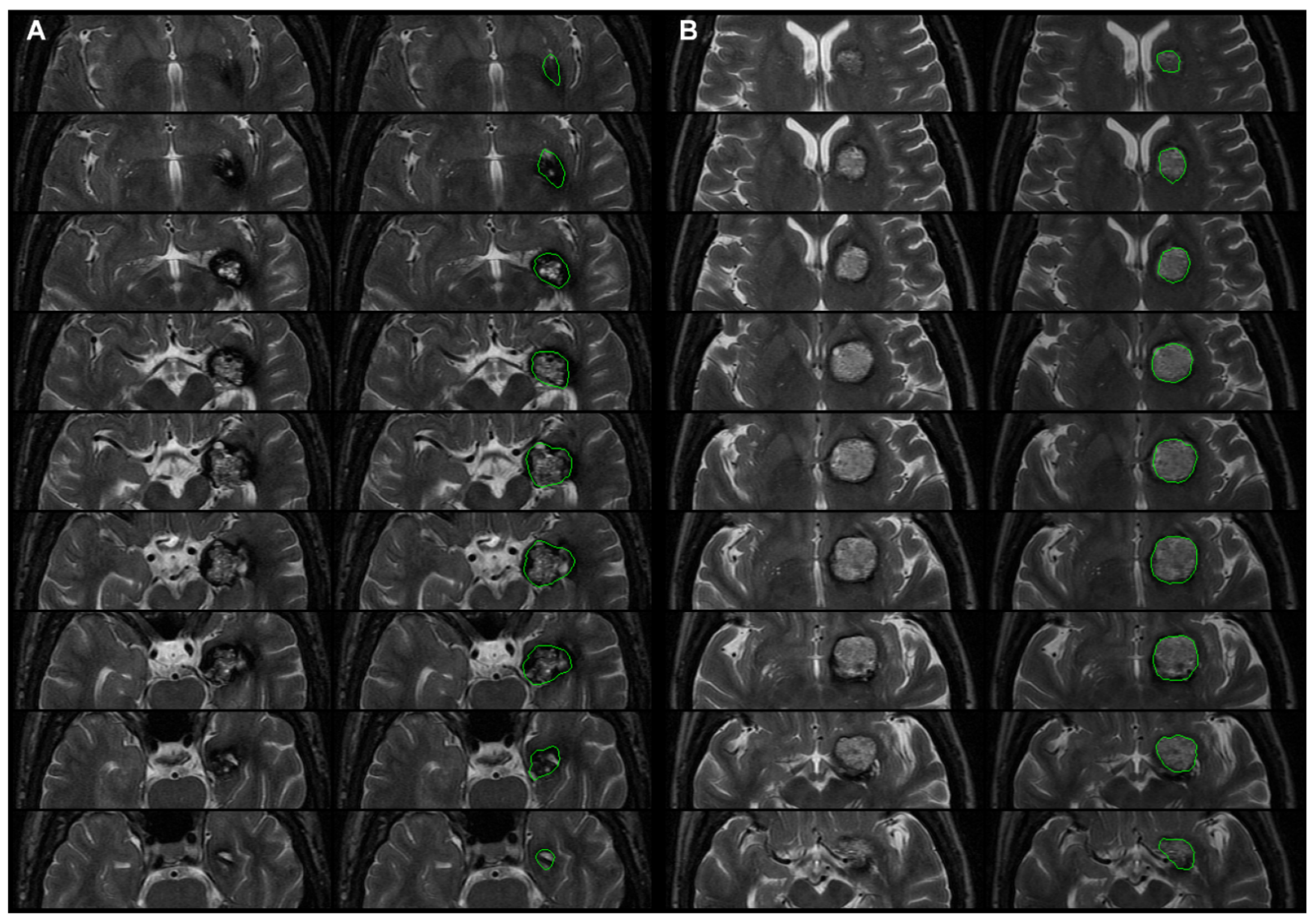
- Step P3. Manual delineation of the CCM
- Step P4. Normalizing T1WI and CCM image labels to the MNI space
- Step P5. Quantifying the displacement of GM by CCM
- Step P6. Quantifying lobe proportions within the CCM
2.4. Statistical Analysis
3. Results
3.1. Patient Population
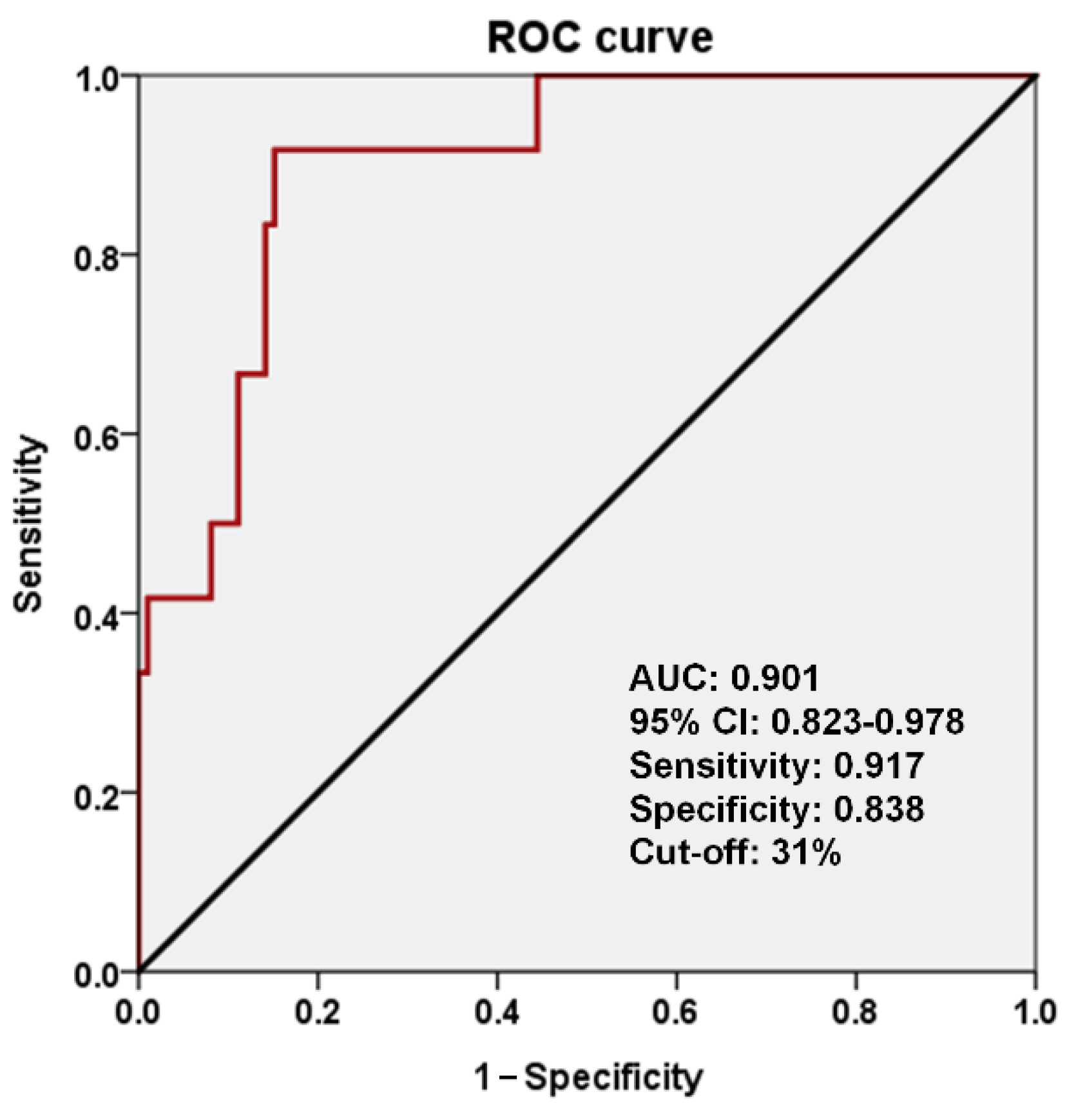
3.2. Volume and Proportion of GM Displaced by the CCM
3.3. Percentage of CCM Located within the Standard Brain Space
3.4. Subgroup Analysis of CCM Location and Relative Seizure Risk
4. Discussion
4.1. Volume and Proportion of Displaced GM as Predictors of Epileptogenicity
4.2. CCM Location and Epileptogenicity
4.3. Potential Discrepancies between CCM Images and Gray Matter Density Template
4.4. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rosenow, F.; Alonso-Vanegas, M.A.; Baumgartner, C.; Blümcke, I.; Carreño, M.; Gizewski, E.R.; Hamer, H.M.; Knake, S.; Kahane, P.; Lüders, H.O.; et al. Cavernoma-related epilepsy: Review and recommendations for management--report of the Surgical Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia 2013, 54, 2025–2035. [Google Scholar] [CrossRef] [PubMed]
- Menzler, K.; Chen, X.; Thiel, P.; Iwinska-Zelder, J.; Miller, D.; Reuss, A.; Hamer, H.M.; Reis, J.; Pagenstecher, A.; Knake, S.; et al. Epileptogenicity of cavernomas depends on (archi-) cortical localization. Neurosurgery 2010, 67, 918–924. [Google Scholar] [CrossRef] [PubMed]
- Casazza, M.; Broggi, G.; Franzini, A.; Avanzini, G.; Spreafico, R.; Bracchi, M.; Valentini, M.C. Supratentorial cavernous angiomas and epileptic seizures: Preoperative course and postoperative outcome. Neurosurgery 1996, 39, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, D.L.; Awad, I.A. Vascular malformations and epilepsy: Clinical considerations and basic mechanisms. Epilepsia 1994, 35 (Suppl. S6), S30–S43. [Google Scholar] [CrossRef] [PubMed]
- Ruan, D.; Yu, X.B.; Shrestha, S.; Wang, L.; Chen, G. The role of hemosiderin excision in seizure outcome in cerebral cavernous malformation surgery: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0136619. [Google Scholar]
- Agosti, E.; Flemming, K.D.; Lanzino, G. Symptomatic cavernous malformation presenting with seizure without hemorrhage: Analysis of factors influencing clinical presentation. World Neurosurg. 2019, 129, e387–e392. [Google Scholar] [CrossRef] [PubMed]
- Ashburner, J. A fast diffeomorphic image registration algorithm. NeuroImage 2007, 38, 95–113. [Google Scholar] [CrossRef]
- Lancaster, J.L.; Woldorff, M.G.; Parsons, L.M.; Liotti, M.; Freitas, C.S.; Rainey, L.; Kochunov, P.V.; Nickerson, D.; Mikiten, S.A.; Fox, P.T. Automated Talairach atlas labels for functional brain mapping. Human Brain Mapp. 2000, 10, 120–131. [Google Scholar] [CrossRef]
- Lancaster, J.L.; Summerlin, J.L.; Rainey, L.; Freitas, C.S.; Fox, P.T. The Talairach Daemon a database server for talairach atlas labels. NeuroImage 1997, 5, S633. [Google Scholar]
- Gross, B.A.; Lin, N.; Du, R.; Day, A.L. The natural history of intracranial cavernous malformations. Neurosurg. Focus 2011, 30, E24. [Google Scholar] [CrossRef] [PubMed]
- Josephson, C.B.; Leach, J.P.; Duncan, R.; Roberts, R.C.; Counsell, C.E.; Al-Shahi Salman, R. Seizure risk from cavernous or arteriovenous malformations: Prospective population-based study. Neurology 2011, 76, 1548–1554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harris, G.J.; Barta, P.E.; Peng, L.W.; Lee, S.; Brettschneider, P.D.; Shah, A.; Henderer, J.D.; Schlaepfer, T.E.; Pearlson, G.D. MR volume segmentation of gray matter and white matter using manual thresholding: Dependence on image brightness. AJNR Am. J. Neuroradiol. 1994, 15, 225–230. [Google Scholar] [PubMed]
- Ashburner, J.; Friston, K.J. Unified segmentation. NeuroImage 2005, 26, 839–851. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Brady, M.; Smith, S. Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans. Med. Imaging 2001, 20, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Moeskops, P.; de Bresser, J.; Kuijf, H.J.; Mendrik, A.M.; Biessels, G.J.; Pluim, J.P.W.; Išgum, I. Evaluation of a deep learning approach for the segmentation of brain tissues and white matter hyperintensities of presumed vascular origin in MRI. NeuroImage Clin. 2018, 17, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Scahill, R.I.; Frost, C.; Jenkins, R.; Whitwell, J.L.; Rossor, M.N.; Fox, N.C. A longitudinal study of brain volume changes in normal aging using serial registered magnetic resonance imaging. Arch. Neurol. 2003, 60, 989–994. [Google Scholar] [CrossRef] [Green Version]
- Benedetti, B.; Charil, A.; Rovaris, M.; Judica, E.; Valsasina, P.; Sormani, M.P.; Filippi, M. Influence of aging on brain gray and white matter changes assessed by conventional, MT, and DT MRI. Neurology 2006, 66, 535–539. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Total (n = 111) | Seizure (n = 12) | Nonseizure (n = 99) | p | OR (95%CI) |
|---|---|---|---|---|---|
| Age (range) | 42.6 (9.8–79.7) | 32.1 (9.8–52.9) | 43.9 (14.26–79.7) | 0.01 | 0.94 (0.89–0.99) |
| Gender | M:54 F:57 | M:6 F:6 | M:48 F:51 | 0.92 | 1.06 (0.32–3.52) |
| Volume of CCMs (mL) (with hemosiderin) | 2.79 | 6.09 | 2.39 | 0.001 | 1.27 (1.09–1.47) |
| Initial symptoms | |||||
| Seizure | 11 | 11 | 0 | <0.001 | ∞ (317.83–∞) |
| Headache/dizziness | 31 | 0 | 31 | 0.19 | 0 (0–0.73) |
| Paresis/paresthesia | 44 | 1 | 43 | 0.04 | 0.12 (0.02–0.95) |
| Others | 22 | 0 | 22 | 0.12 | 0 (0–1.17) |
| Incidental finding | 3 | 0 | 3 | 1 | 0 (0–11.28) |
| Location of CCMs | |||||
| Frontal lobe | 17 | 5 | 12 | 0.01 | 5.18 (1.42–18.94) |
| Temporal lobe | 2 | 1 | 1 | 0.13 | 8.9 (0.52–152.60) |
| Parietal lobe | 4 | 2 | 2 | 0.03 | 9.7 (1.23–76.50) |
| Occipital lobe | 2 | 1 | 1 | 0.13 | 8.9 (0.52–152.60) |
| Sublobar | 18 | 1 | 17 | 0.44 | 0.44 (0.05–3.63) |
| Limbic lobe | 3 | 2 | 1 | 0.02 | 19.6 (1.63–235.70) |
| Cerebellum | 11 | 0 | 11 | 0.61 | 0 (0–2.73) |
| Brainstem | 54 | 0 | 54 | <0.001 | 0 (0–0.28) |
| Gray Matter | Seizure (n = 12) | Nonseizure (n = 99) | p |
|---|---|---|---|
| Volume (mL) | 3.024 ± 2.415 | 0.521 ± 0.970 | <0.00001 * |
| Proportion (%) | 48.540 ± 22.076 | 15.938 ± 15.59 | <0.00001 * |
| Location Group | Seizure | Nonseizure | OR (95%CI) | p |
|---|---|---|---|---|
| * Total (n = 111) | (n = 12) | (n = 99) | ||
| Infratentorial (n = 65) | 0 | 65 | 1 | - |
| Supratentorial (n = 46) | 12 | 34 | - | - |
| ** Supratentorial (n = 46) | (n = 12) | (n = 34) | ||
| Extratemporal (n = 41) | 9 | 32 | 1 | - |
| Temporal (n = 5) | 3 | 2 | 5.3 (0.77–36.96) | 0.09 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chou, C.-J.; Lee, C.-C.; Chen, C.-J.; Yang, H.-C.; Peng, S.-J. Displacement of Gray Matter and Incidence of Seizures in Patients with Cerebral Cavernous Malformations. Biomedicines 2021, 9, 1872. https://doi.org/10.3390/biomedicines9121872
Chou C-J, Lee C-C, Chen C-J, Yang H-C, Peng S-J. Displacement of Gray Matter and Incidence of Seizures in Patients with Cerebral Cavernous Malformations. Biomedicines. 2021; 9(12):1872. https://doi.org/10.3390/biomedicines9121872
Chicago/Turabian StyleChou, Chi-Jen, Cheng-Chia Lee, Ching-Jen Chen, Huai-Che Yang, and Syu-Jyun Peng. 2021. "Displacement of Gray Matter and Incidence of Seizures in Patients with Cerebral Cavernous Malformations" Biomedicines 9, no. 12: 1872. https://doi.org/10.3390/biomedicines9121872
APA StyleChou, C.-J., Lee, C.-C., Chen, C.-J., Yang, H.-C., & Peng, S.-J. (2021). Displacement of Gray Matter and Incidence of Seizures in Patients with Cerebral Cavernous Malformations. Biomedicines, 9(12), 1872. https://doi.org/10.3390/biomedicines9121872







