Obesity-Associated Metabolic Disturbances Reverse the Antioxidant and Anti-Inflammatory Properties of High-Density Lipoproteins in Microglial Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects and Ethics
2.2. Clinical Measurements
2.3. Biochemical Measurements
2.4. HDL Isolation and Oxidized HDL Measurement
2.5. Paraoxonase-1 Levels and Activity Assay
2.6. BV2 Microglial Cell Culture and Treatments
2.7. RNA Isolation and RT-qPCR Analysis
2.8. Statistical Analysis
3. Results
3.1. Clinical and Biochemical Characteristics of the Study Subjects
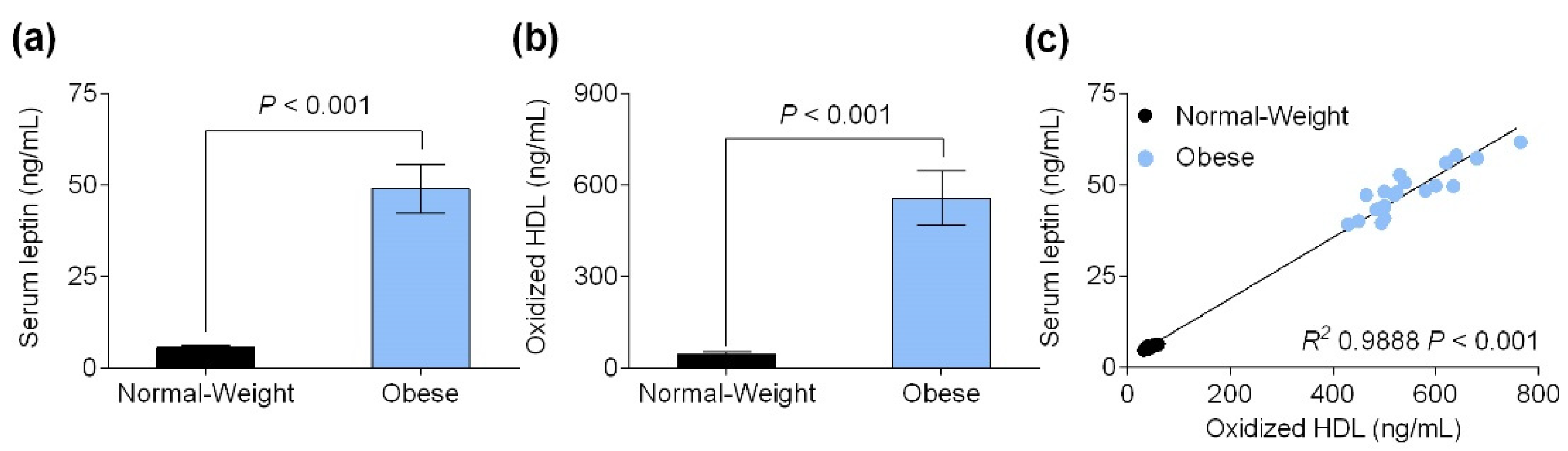
3.2. Serum Leptin Level Is Positively Correlated with Oxidized HDL Levels
3.3. Obesity-Associated Hyperleptinemia Reduced Paraoxonase-1 Levels and Activity
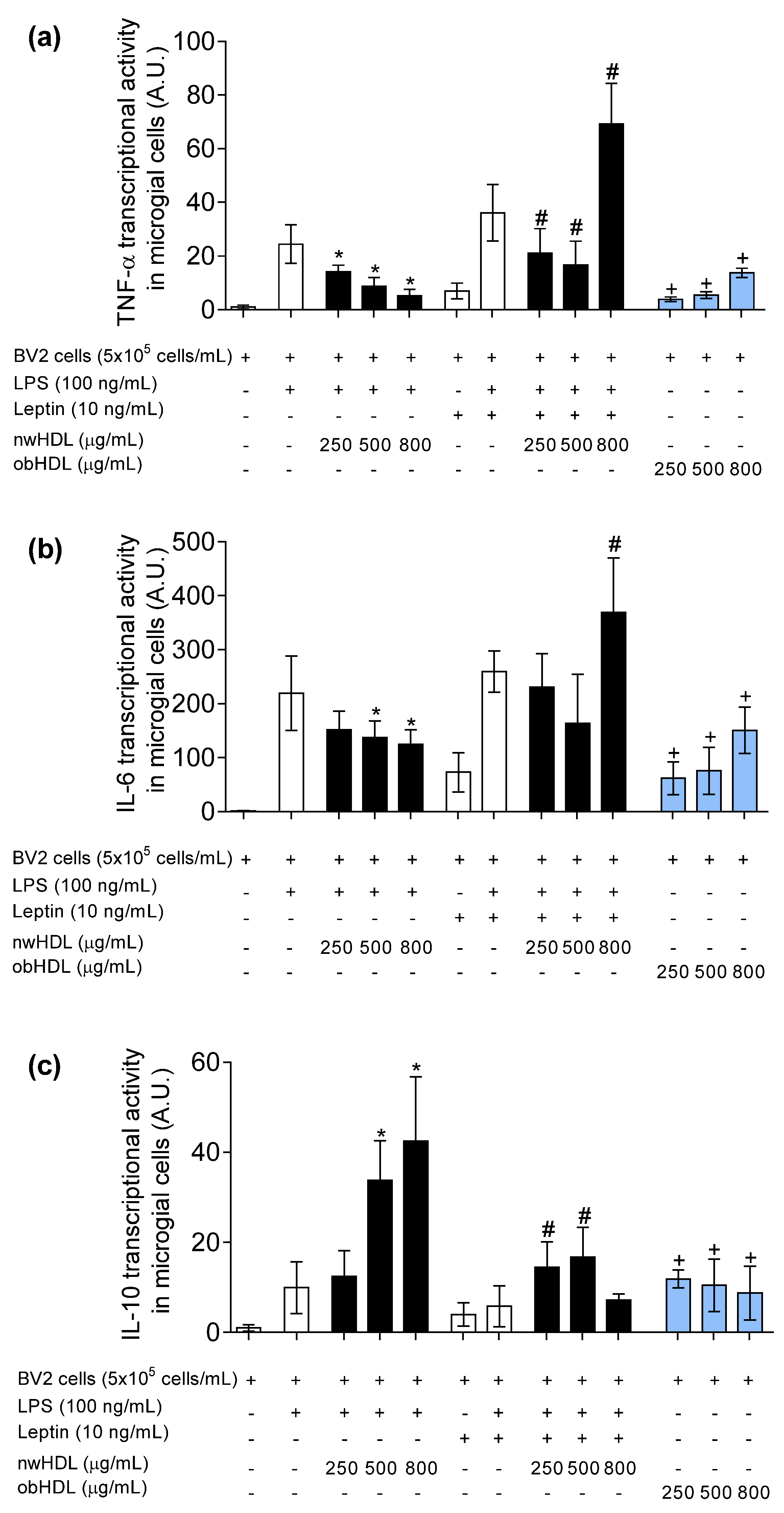
3.4. HDL Isolated from Hyperleptinemic Obese Subjects Increased Pro-Inflammatory Cytokine Expression in Microglial Cells
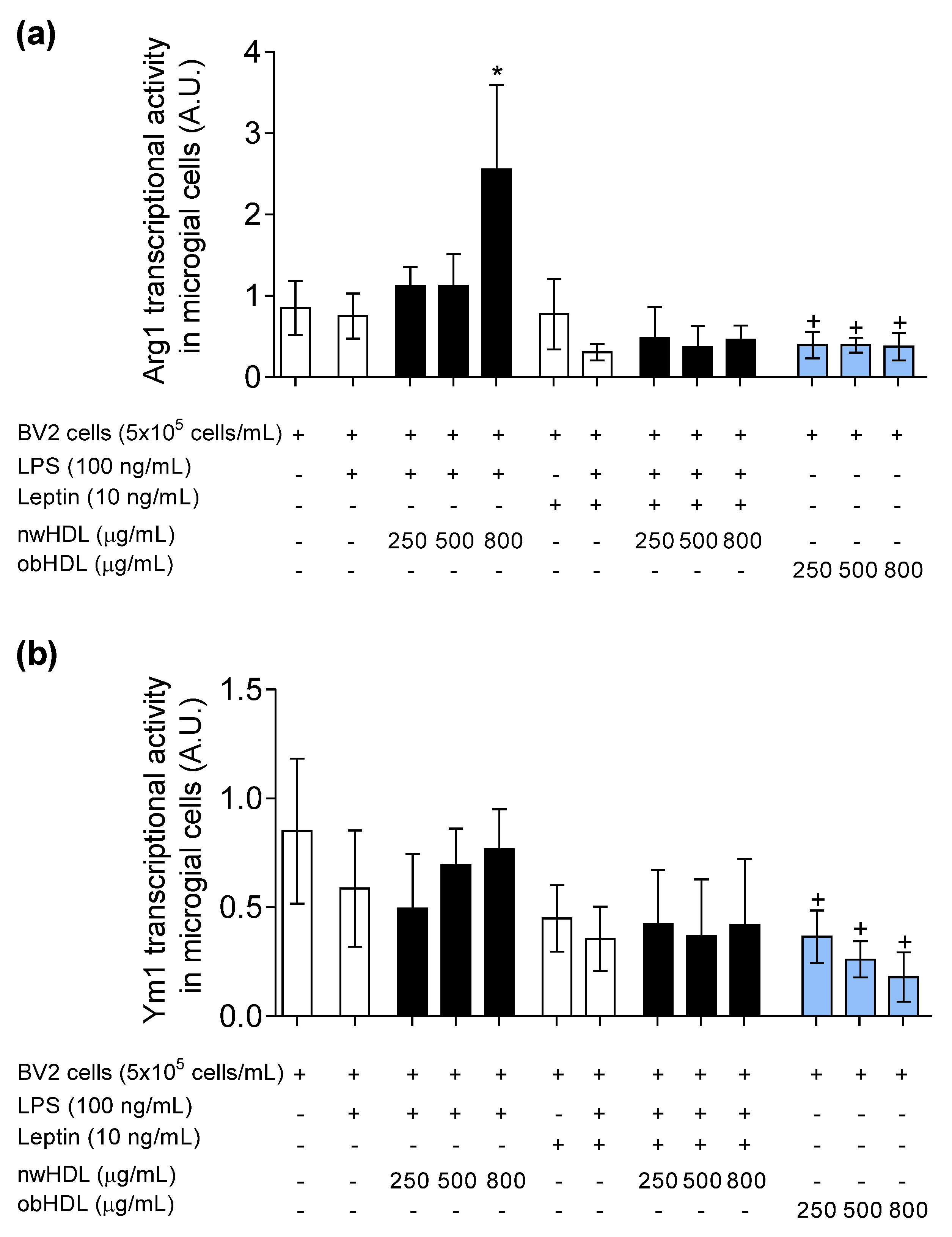
3.5. HDL Isolated from Hyperleptinemic Obese Subjects Promotes M1 Polarization in Microglial Cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Limitations of the Study
References
- Ben-Aicha, S.; Badimon, L.; Vilahur, G. Advances in HDL: Much More than Lipid Transporters. Int. J. Mol. Sci. 2020, 21, 732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fotakis, P.; Kothari, V.; Thomas, D.G.; Westerterp, M.; Molusky, M.M.; Altin, E.; Abramowicz, S.; Wang, N.; He, Y.; Heinecke, J.W.; et al. Anti-Inflammatory Effects of HDL (High-Density Lipoprotein) in Macrophages Predominate Over Proinflammatory Effects in Atherosclerotic Plaques. Arter. Thromb. Vasc. Biol. 2019, 39, e253–e272. [Google Scholar] [CrossRef]
- Mahrooz, A.; Shokri, Y.; Variji, A.; Zargari, M.; Alizadeh, A.; Mehtarian, E. Improved risk assessment of coronary artery disease by substituting paraoxonase 1 activity for HDL-C: Novel cardiometabolic biomarkers based on HDL functionality. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 1166–1176. [Google Scholar] [CrossRef] [PubMed]
- Sutter, I.; Velagapudi, S.; Othman, A.; Riwanto, M.; Manz, J.; Rohrer, L.; Rentsch, K.; Hornemann, T.; Landmesser, U.; von Eckardstein, A. Plasmalogens of high-density lipoproteins (HDL) are associated with coronary artery disease and anti-apoptotic activity of HDL. Atherosclerosis 2015, 241, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Shih, C.-M.; Lin, F.-Y.; Yeh, J.-S.; Lin, Y.-W.; Loh, S.-H.; Tsao, N.-W.; Nakagami, H.; Morishita, R.; Sawamura, T.; Li, C.-Y.; et al. Dysfunctional high density lipoprotein failed to rescue the function of oxidized low density lipoprotein-treated endothelial progenitor cells: A novel index for the prediction of HDL functionality. Transl. Res. 2019, 205, 17–32. [Google Scholar] [CrossRef]
- Gao, D.; Ashraf, M.Z.; Zhang, L.; Kar, N.; Byzova, T.V.; Podrez, E.A. Cross-linking modifications of HDL apoproteins by oxidized phospholipids: Structural characterization, in vivo detection, and functional implications. J. Biol. Chem. 2020, 295, 1973–1984. [Google Scholar] [CrossRef]
- Soria-Florido, M.T.; Castañer, O.; Lassale, C.; Estruch, R.; Salas-Salvadó, J.; Martínez-González, M.Á.; Corella, D.; Ros, E.; Arós, F.; Elosua, R.; et al. Dysfunctional High-density Lipoproteins Are Associated with a Greater Incidence of Acute Coronary Syndrome in a Population at High Cardiovascular Risk: A Nested Case-Control Study. Circulation 2020, 141, 444–453. [Google Scholar] [CrossRef]
- Li, B.-Q.; Zhong, Y.-C.; Wang, X. Plasma oxidized high-density lipoprotein and glycated apolipoprotein A-I concentrations in ST-segment elevation myocardial infarction patients with stress hyperglycaemia or high thrombus burden. Upsala J. Med Sci. 2018, 123, 158–166. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Zhou, C.; Xu, H.; Brook, R.D.; Liu, S.; Yi, T.; Wang, Y.; Feng, B.; Zhao, M.; Wang, X.; et al. Ambient Air Pollution Is Associated with HDL (High-Density Lipoprotein) Dysfunction in Healthy Adults. Arter. Thromb. Vasc. Biol. 2019, 39, 513–522. [Google Scholar] [CrossRef] [Green Version]
- Delbosc, S.; Diallo, D.; Dejouvencel, T.; Lamiral, Z.; Louedec, L.; Martin-Ventura, J.-L.; Rossignol, P.; Leseche, G.; Michel, J.-B.; Meilhac, O. Impaired high-density lipoprotein anti-oxidant capacity in human abdominal aortic aneurysm. Cardiovasc. Res. 2013, 100, 307–315. [Google Scholar] [CrossRef] [Green Version]
- Peterson, S.J.; Shapiro, J.I.; Thompson, E.; Singh, S.; Liu, L.; Weingarten, J.A.; O’Hanlon, K.; Bialczak, A.; Bhesania, S.R.; Abraham, N.G. Oxidized HDL, Adipokines, and Endothelial Dysfunction: A Potential Biomarker Profile for Cardiovascular Risk in Women with Obesity. Obesity 2018, 27, 87–93. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Arnal-Levron, M.; Hullin-Matsuda, F.; Knibbe, C.; Moulin, P.; Luquain-Costaz, C.; Delton, I. In vitro oxidized HDL and HDL from type 2 diabetes patients have reduced ability to efflux oxysterols from THP-1 macrophages. Biochimie 2018, 153, 232–237. [Google Scholar] [CrossRef]
- Perakakis, N.; Farr, O.M.; Mantzoros, C.S. Leptin in Leanness and Obesity: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2021, 77, 745–760. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Reed, F.; Herzog, H. Leptin signalling on arcuate NPY neurones controls adiposity independent of energy balance or diet composition. J. Neuroendocr. 2020, 32, 12898. [Google Scholar] [CrossRef] [PubMed]
- Tsiotra, P.C.; Boutati, E.; Dimitriadis, G.; Raptis, S.A. High Insulin and Leptin Increase Resistin and Inflammatory Cytokine Production from Human Mononuclear Cells. BioMed Res. Int. 2013, 2013, 487081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gerriets, V.A.; Danzaki, K.; Kishton, R.J.; Eisner, W.; Nichols, A.G.; Saucillo, D.C.; Shinohara, M.L.; MacIver, N.J. Leptin directly promotes T-cell glycolytic metabolism to drive effector T-cell differentiation in a mouse model of autoimmunity. Eur. J. Immunol. 2016, 46, 1970–1983. [Google Scholar] [CrossRef] [Green Version]
- Kiernan, K.; Maclver, N.J. The Role of the Adipokine Leptin in Immune Cells Function in Health and Disease. Front. Immunol. 2021, 11, 622468. [Google Scholar] [CrossRef]
- Agrawal, S.; Gollapudi, S.; Su, H.; Gupta, S. Leptin activates human B cells to secrete TNF-α, IL-6, and IL-10 via JAK2/STAT3 and p38MAPK/ERK1/2 signaling pathway. J. Clin. Immunol. 2011, 31, 472–478. [Google Scholar] [CrossRef] [Green Version]
- Chagas, L.; Sandre, P.C.; Ribeiro, N.C.A.R.E.; Marcondes, H.; Silva, P.O.; Savino, W.; Serfaty, C.A. Environmental Signals on Microglial Function during Brain Development, Neuroplasticity, and Disease. Int. J. Mol. Sci. 2020, 21, 2111. [Google Scholar] [CrossRef] [Green Version]
- Batista, C.R.A.; Gomes, G.F.; Candelario-Jalil, E.; Fiebich, B.L.; De Oliveira, A.C.P. Lipopolysaccharide-Induced Neuroinflammation as a Bridge to Understand Neurodegeneration. Int. J. Mol. Sci. 2019, 20, 2293. [Google Scholar] [CrossRef] [Green Version]
- Orihuela, R.; McPherson, C.A.; Harry, G.J. Microglial M1/M2 polarization and metabolic states. Br. J. Pharmacol. 2016, 173, 649–665. [Google Scholar] [CrossRef]
- Plastira, I.; Bernhart, E.; Goeritzer, M.; Reicher, H.; Kumble, V.B.; Kogelnik, N.; Wintersperger, A.; Hammer, A.; Schlager, S.; Jandl, K.; et al. 1-Oleyl-lysophosphatidic acid (LPA) promotes polarization of BV-2 and primary murine microglia towards and M1-like phenotype. J. Neuroinflamm. 2016, 13, 205. [Google Scholar] [CrossRef] [Green Version]
- Ilarregui, J.M.; Kooij, G.; Rodríguez, E.; Van Der Pol, S.M.A.; Koning, N.; Kalay, H.; Van Der Horst, J.C.; Van Vliet, S.J.; García-Vallejo, J.J.; De Vries, H.E.; et al. Macrophage galactose-type lectin (MGL) is induced on M2 microglia and participates in the resolution phase of autoimmune neuroinflammation. J. Neuroinflamm. 2019, 16, 130. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Dai, S.K.; Shi, R.X.; He, X.C.; Wang, Y.Y.; He, B.D.; Sun, X.W.; Du, H.Z.; Liu, C.M.; Teng, Z.Q. Transcriptional profiling of microglia in the injured brain reveals distinct molecular features underlying neurodegeneration. Glia 2021, in press. [Google Scholar] [CrossRef] [PubMed]
- Sobue, A.; Komine, O.; Hara, Y.; Endo, F.; Mizoguchi, H.; Watanabe, S.; Murayama, S.; Saito, T.; Saido, T.C.; Sahara, N.; et al. Microglial gene signature reveals loss of homeostatic microglia associated with neurodegeneration of Alzheimer’s disease. Acta Neuropathol. Commun. 2021, 9, 1. [Google Scholar] [CrossRef]
- Fung, K.Y.; Wang, C.; Nyegaard, S.; Heit, B.; Fairn, G.D.; Lee, W.L. SR-BI Mediated Transcytosis of HDL in Brain Microvascular Endothelial Cells Is Independent of Caveolin, Clathrin, and PDZK1. Front. Physiol. 2017, 8, 841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, J.L.; Slentz, C.A.; Duscha, B.D.; Samsa, G.P.; McCartney, J.S.; Houmard, J.A.; Kraus, W.E. Gender and racial differences in lipoprotein subclass distributions: The STRRIDE study. Atherosclerosis 2004, 176, 371–377. [Google Scholar] [CrossRef] [PubMed]


| Variables | Normal-Weight | Obese | p-Value |
|---|---|---|---|
| N | 20 | 20 | |
| Age (years) | 31.2 ± 6.7 | 38.5 ± 4.3 | 0.060 |
| Smoking (%) | 20 | 30 | 0.465 |
| Alcohol consumption (%) | 45 | 50 | 0.751 |
| Physical activity (hours/week) | 2.7 ± 2.5 | 2.4 ± 1.8 | 0.161 |
| BMI (kg/m2) | 23.2 ± 1.5 | 31.9 ± 4.1 | <0.001 |
| Waist circumference (cm) | 85.3 ± 7.0 | 124.5 ± 16.1 | <0.001 |
| Neck circumference (cm) | 29.3 ± 0.5 | 38.4 ± 2.1 | <0.001 |
| Percent body fat | 27.4 ± 0.4 | 39.6 ± 1.6 | <0.001 |
| Systolic blood pressure (mmHg) | 126.8 ± 5.2 | 133.8 ± 8.0 | 0.068 |
| Diastolic blood pressure (mmHg) | 82.6 ± 7.1 | 84.2 ± 6.9 | 0.902 |
| NEFAs (µmol/L) | 387 ± 42 | 433 ± 45 | 0.767 |
| Triglycerides (mmol/L) | 0.53 ± 0.17 | 1.29 ± 0.16 | 0.794 |
| Total Cholesterol (mmol/L) | 4.4 ± 0.4 | 5.2 ± 0.5 | 0.339 |
| LDL-C (mmol/L) | 2.41 ± 0.43 | 3.03 ± 0.32 | 0.207 |
| HDL-C (mmol/L) | 1.59 ± 0.16 | 1.11 ± 0.11 | 0.111 |
| Glucose (mmol/L) | 4.45 ± 0.49 | 5.27 ± 0.62 | 0.317 |
| HbA1c (%) | 5.1 ± 0.4 | 5.2 ± 0.5 | 0.339 |
| Insulin (pmol/L) | 43.9 ± 4.6 | 105 ± 18.6 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grao-Cruces, E.; Millan-Linares, M.C.; Martin-Rubio, M.E.; Toscano, R.; Barrientos-Trigo, S.; Bermudez, B.; Montserrat-de la Paz, S. Obesity-Associated Metabolic Disturbances Reverse the Antioxidant and Anti-Inflammatory Properties of High-Density Lipoproteins in Microglial Cells. Biomedicines 2021, 9, 1722. https://doi.org/10.3390/biomedicines9111722
Grao-Cruces E, Millan-Linares MC, Martin-Rubio ME, Toscano R, Barrientos-Trigo S, Bermudez B, Montserrat-de la Paz S. Obesity-Associated Metabolic Disturbances Reverse the Antioxidant and Anti-Inflammatory Properties of High-Density Lipoproteins in Microglial Cells. Biomedicines. 2021; 9(11):1722. https://doi.org/10.3390/biomedicines9111722
Chicago/Turabian StyleGrao-Cruces, Elena, Maria C. Millan-Linares, Maria E. Martin-Rubio, Rocio Toscano, Sergio Barrientos-Trigo, Beatriz Bermudez, and Sergio Montserrat-de la Paz. 2021. "Obesity-Associated Metabolic Disturbances Reverse the Antioxidant and Anti-Inflammatory Properties of High-Density Lipoproteins in Microglial Cells" Biomedicines 9, no. 11: 1722. https://doi.org/10.3390/biomedicines9111722
APA StyleGrao-Cruces, E., Millan-Linares, M. C., Martin-Rubio, M. E., Toscano, R., Barrientos-Trigo, S., Bermudez, B., & Montserrat-de la Paz, S. (2021). Obesity-Associated Metabolic Disturbances Reverse the Antioxidant and Anti-Inflammatory Properties of High-Density Lipoproteins in Microglial Cells. Biomedicines, 9(11), 1722. https://doi.org/10.3390/biomedicines9111722









