Mesenchymal Stromal Cells Isolated from Ectopic but Not Eutopic Endometrium Display Pronounced Immunomodulatory Activity In Vitro
Abstract
:1. Introduction
2. Experimental Section
2.1. Endometrial Tissue Samples and Mesenchymal Stromal Cell Cultures
2.2. Dendritic Cell Differentiation and Maturation in the MSC Co-Culture
2.3. T-Lymphocyte Proliferation in the MSC Co-Culture
2.4. Flow Cytometry
2.5. Multiplex Analysis of the Cytokine Secretion
2.6. Statistical Analysis
3. Results
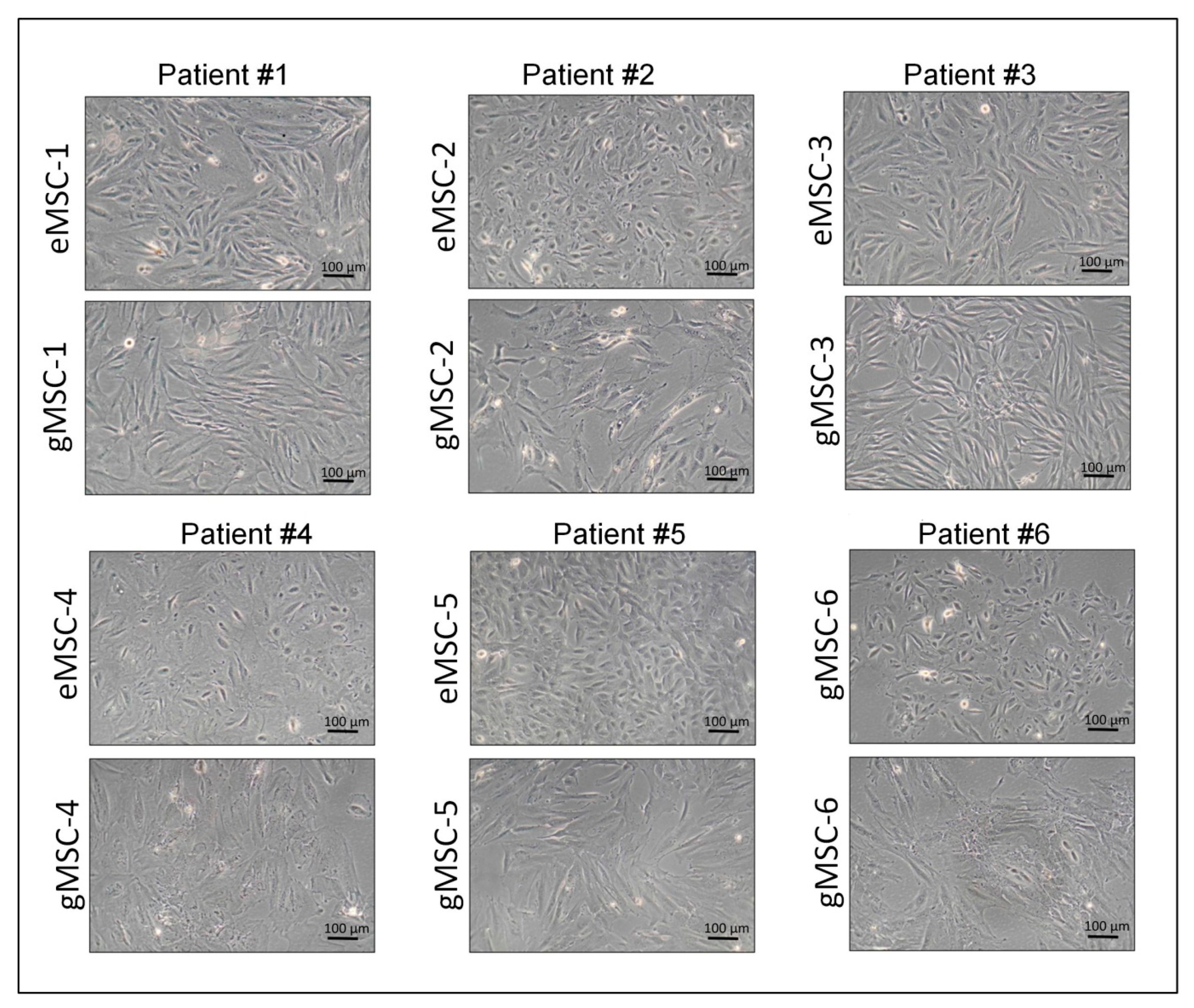
3.1. Isolation of Cell Cultures from Eutopic and Ectopic Endometrium
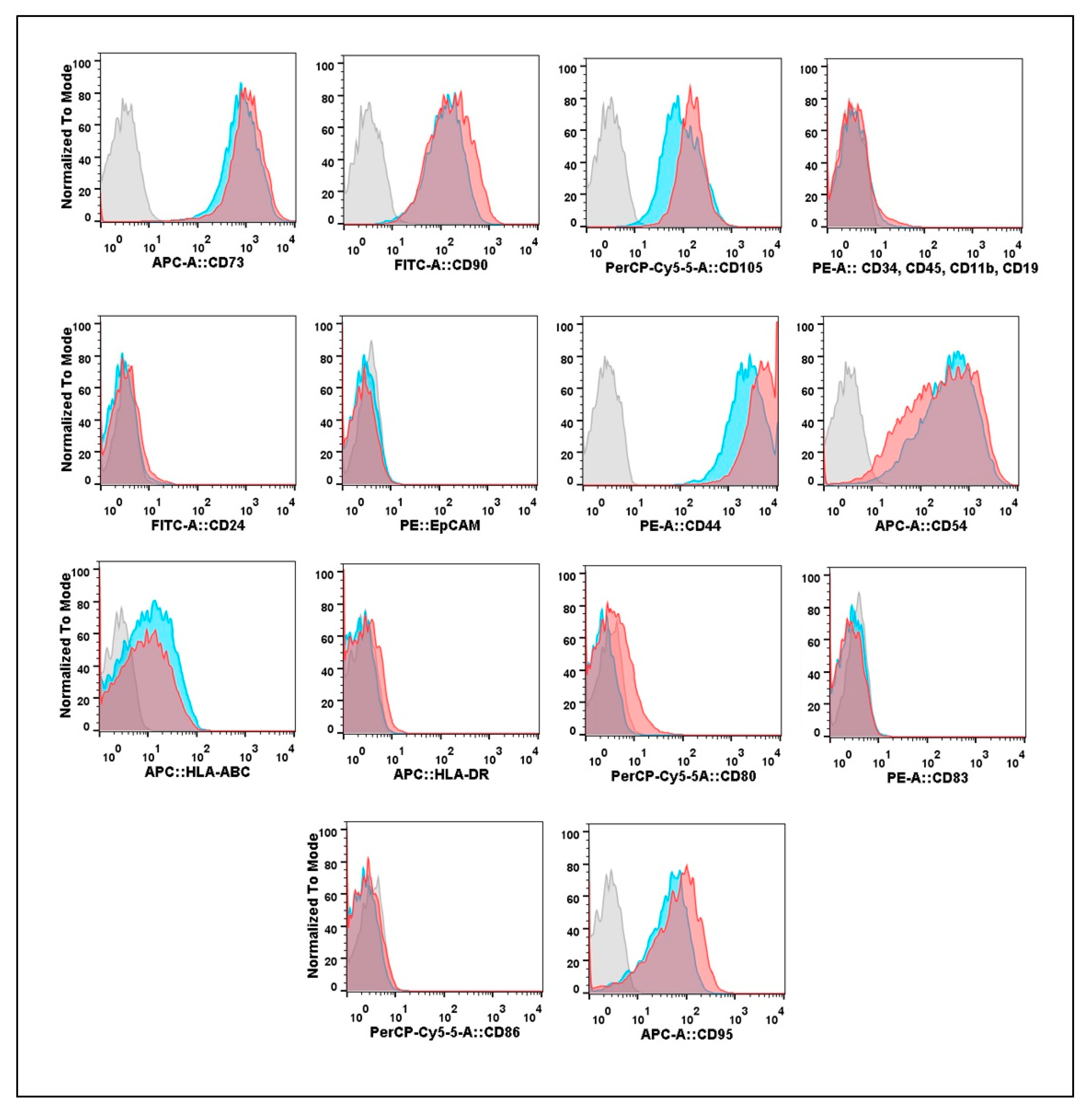
3.2. Expression of Cell Surface Molecular Markers by Cultures Isolated from Eutopic or Ectopic Endometrium
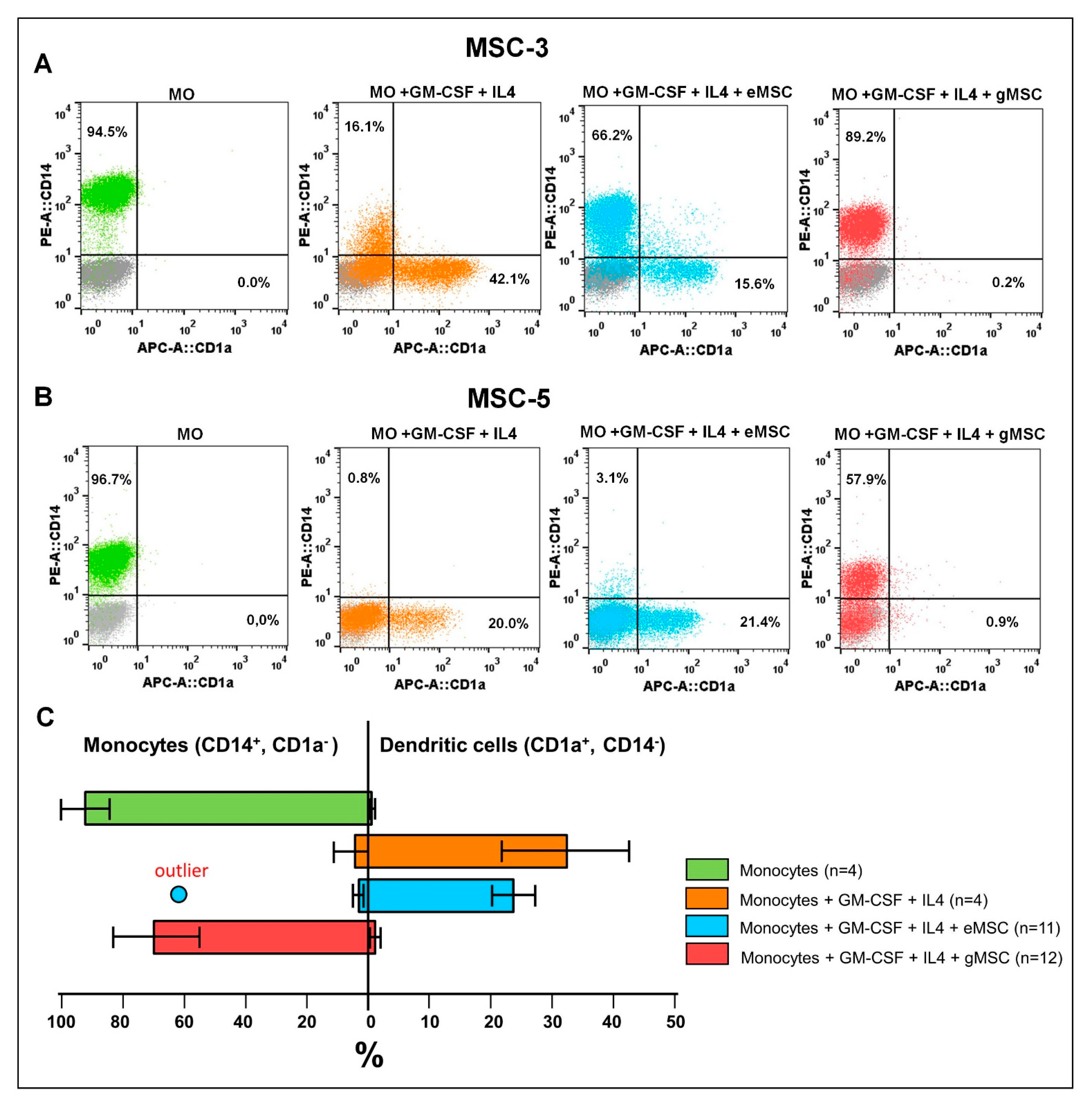
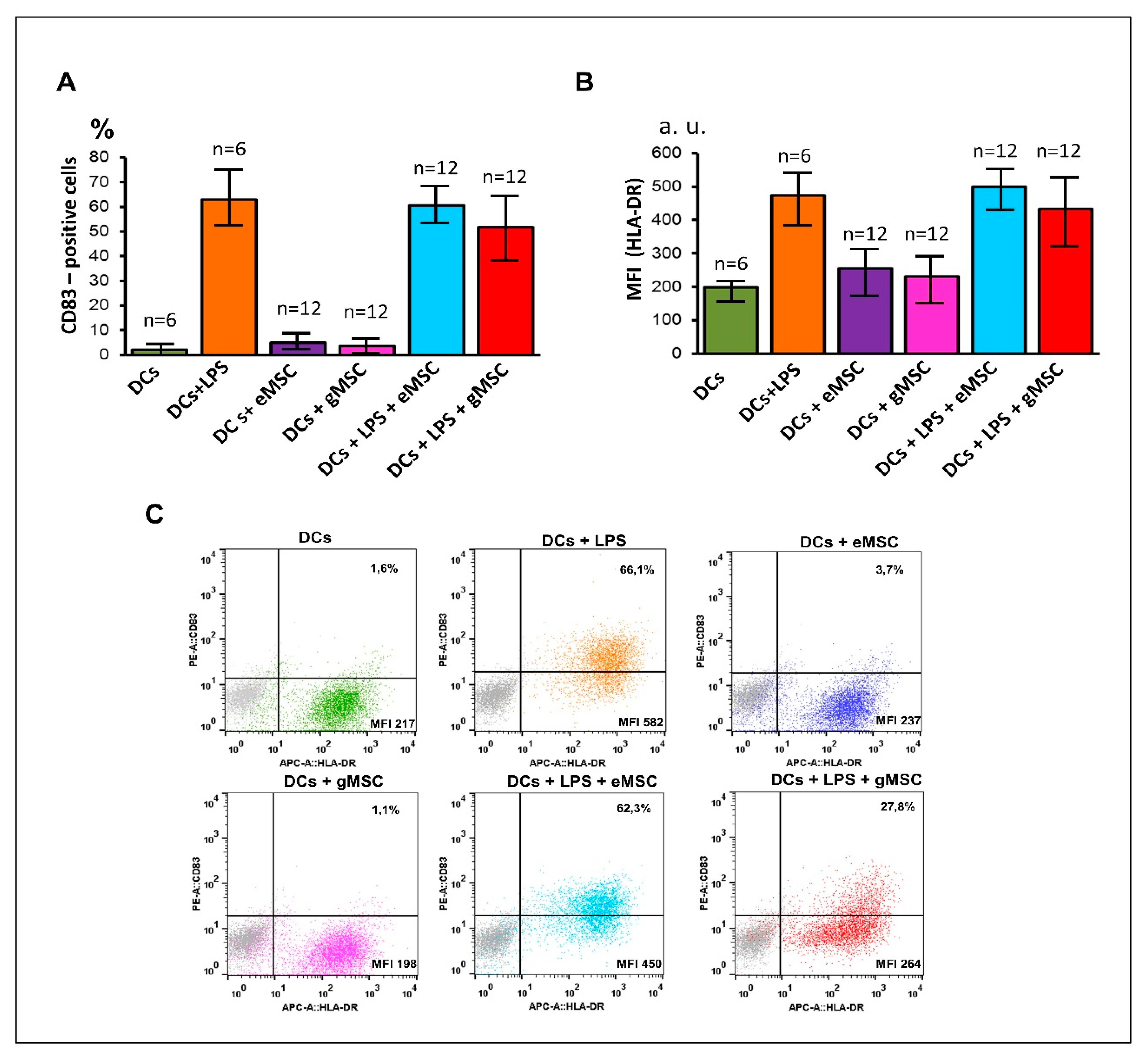
3.3. Influence of the MSC Cultures Isolated from the Eutopic or Ectopic Endometrium on the Differentiation and Maturation of DCs In Vitro
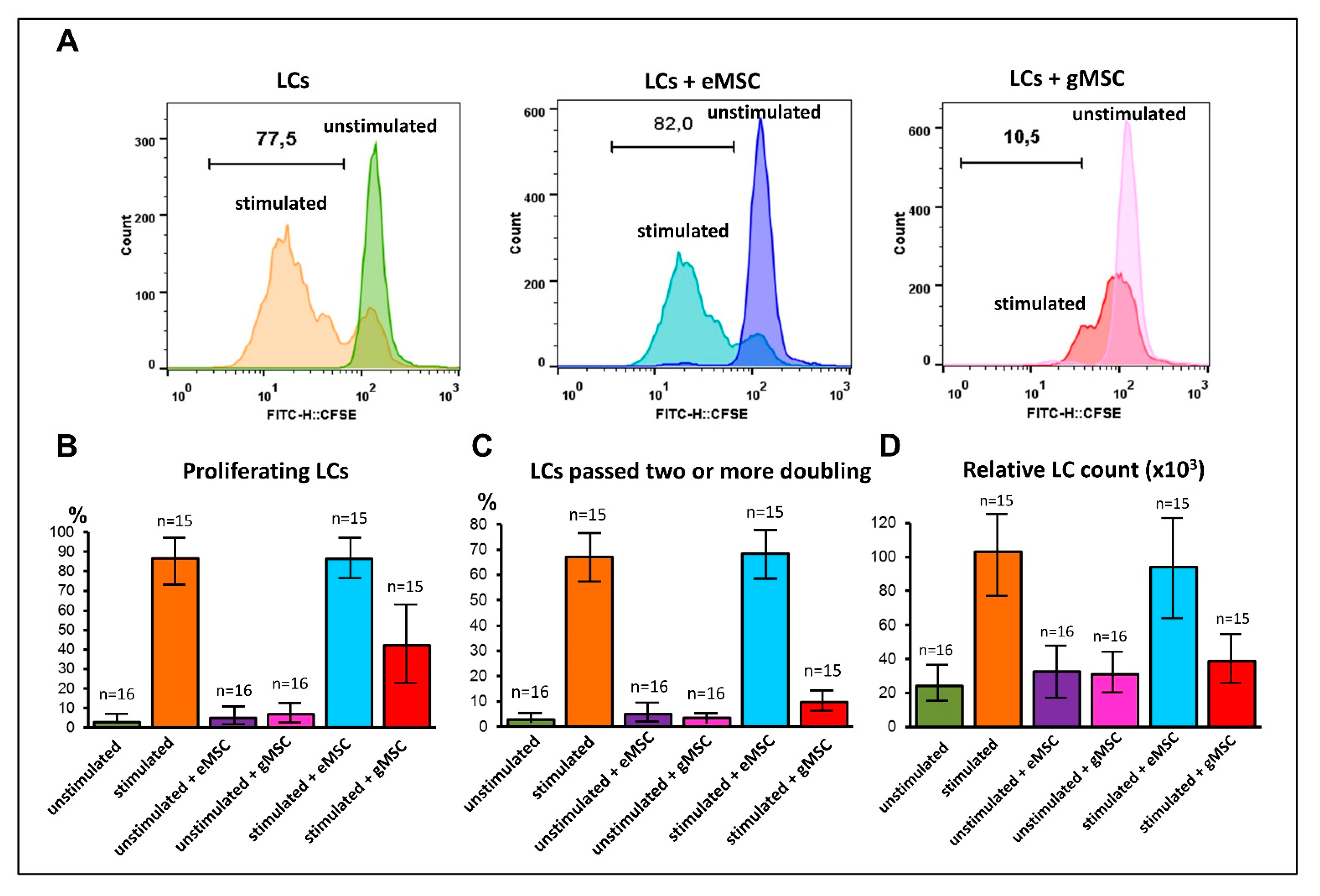
3.4. Influence of MSC Cultures Derived from Eutopic or Ectopic Endometrium on T-Cell Proliferation In Vitro
3.5. Comparative Profile of Cytokine Secretion by Cell Cultures of Eutopic and Ectopic Endometrium
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Johnson, N.P.; Hummelshoj, L.; Adamson, G.D.; Keckstein, J.; Taylor, H.S.; Abrao, M.S.; Bush, D.; Kiesel, L.; Tamimi, R.; Sharpe-Timms, K.L.; et al. World endometriosis society consensus on the classification of endometriosis. Hum. Reprod. 2017, 32, 315–324. [Google Scholar] [CrossRef]
- Sarria-Santamera, A.; Orazumbekova, B.; Terzic, M.; Issanov, A.; Chaowen, C.; Asúnsolo-Del-barco, A. Systematic review and meta-analysis of incidence and prevalence of endometriosis. Healthcare 2021, 9, 29. [Google Scholar] [CrossRef]
- Eisenberg, V.H.; Weil, C.; Chodick, G.; Shalev, V. Epidemiology of endometriosis: A large population-based database study from a healthcare provider with 2 million members. BJOG Int. J. Obstet. Gynaecol. 2018, 125, 55–62. [Google Scholar] [CrossRef] [Green Version]
- Meggyesy, M.; Friese, M.; Gottschalk, J.; Kehler, U. Case Report of Cerebellar Endometriosis. J. Neurol. Surg. Part A Cent. Eur. Neurosurg. 2020, 81, 372–376. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Li, C.; Zarogoulidis, P.; Darwiche, K.; Machairiotis, N.; Yang, L.; Simoff, M.; Celis, E.; Zhao, T.; Zarogoulidis, K.; et al. Endometriosis of the lung: Report of a case and literature review. Eur. J. Med. Res. 2013, 18, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pospisilova, E.; Kiss, I.; Souckova, H.; Tomes, P.; Spicka, J.; Matkowski, R.; Jedryka, M.; Ferrero, S.; Bobek, V.; Kolostova, K. Circulating Endometrial Cells: A New Source of Information on Endometriosis Dynamics. J. Clin. Med. 2019, 8, 1938. [Google Scholar] [CrossRef] [Green Version]
- Yachida, N.; Yoshihara, K.; Yamaguchi, M.; Suda, K.; Tamura, R.; Enomoto, T. How does endometriosis lead to ovarian cancer? The molecular mechanism of endometriosis-associated ovarian cancer development. Cancers 2021, 13, 1439. [Google Scholar] [CrossRef]
- Sapalidis, K.; Machairiotis, N.; Zarogoulidis, P.; Vasilakaki, S.; Sardeli, C.; Koimtzis, G.; Pavlidis, E.; Katsaounis, A.; Giannakidis, D.; Michalopoulos, N.; et al. Genes’ interactions: A major contributor to the malignant transformation of endometriosis. Int. J. Mol. Sci. 2019, 20, 1842. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Machairiotis, N.; Vasilakaki, S.; Thomakos, N. Inflammatory mediators and pain in endometriosis: A systematic review. Biomedicines 2021, 9, 54. [Google Scholar] [CrossRef]
- Warzecha, D.; Szymusik, I.; Wielgos, M.; Pietrzak, B. The impact of endometriosis on the quality of life and the incidence of depression—A cohort study. Int. J. Environ. Res. Public Health 2020, 17, 3641. [Google Scholar] [CrossRef] [PubMed]
- Dastur, A.E.; Tank, P.D. John A Sampson and the origins of Endometriosis. J. Obstet. Gynecol. India 2010, 60, 299–300. [Google Scholar] [CrossRef] [Green Version]
- Beckman, E.N.; Pintado, S.O.; Leonard, G.L.; Sternberg, W.H. Endometriosis of the prostate. Am. J. Surg. Pathol. 1985, 9, 374–379. [Google Scholar] [CrossRef]
- Sourial, S.; Tempest, N.; Hapangama, D.K. Theories on the Pathogenesis of Endometriosis. Int. J. Reprod. Med. 2014, 2014, 179515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koppolu, A.; Maksym, R.B.; Paskal, W.; Machnicki, M.; Rak, B.; Pępek, M.; Garbicz, F.; Pełka, K.; Kuśmierczyk, Z.; Jacko, J.; et al. Epithelial Cells of Deep Infiltrating Endometriosis Harbor Mutations in Cancer Driver Genes. Cells 2021, 10, 749. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Lang, J.H. Is abnormal eutopic endometrium the cause of endometriosis? The role of eutopic endometrium in pathogenesis of endometriosis. Med. Sci. Monit. 2011, 17, 92–99. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Zhang, Z.; Yang, F.; Wang, H.; Liang, S.; Wang, H.; Yang, J.; Lin, J. The role of endometrial stem cells in the pathogenesis of endometriosis and their application to its early diagnosis. Biol. Reprod. 2020, 102, 1153–1159. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Hirata, T.; Arakawa, T.; Sun, H.; Neriishi, K.; Fukuda, S.; Nakazawa, A.; Wang, Y.; Harada, M.; Hirota, Y.; et al. Expression of ALDH1A Isozymes in Human Endometrium with and without Endometriosis and in Ovarian Endometrioma. Reprod. Sci. 2020, 27, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Gargett, C.E.; Schwab, K.E.; Deane, J.A. Endometrial stem/progenitor cells: The first 10 years. Hum. Reprod. Update 2016, 22, 137–163. [Google Scholar] [CrossRef] [Green Version]
- Lupatov, A.Y.; Saryglar, R.Y.; Chuprynin, V.D.; Pavlovich, S.V.; Yarygin, K.N. Comparison of the expression profile of surface molecular markers on mesenchymal stromal cell cultures isolated from human endometrium and umbilical cord. Biomeditsinskaya Khimiya 2017, 63, 85–90. [Google Scholar] [CrossRef]
- Savilova, A.M.; Yushina, M.N.; Rudimova, Y.V.; Khil’kevich, E.G.; Chuprynin, V.D. Characteristics of Multipotent Mesenchymal Stromal Cells Isolated from Human Endometrium. Bull. Exp. Biol. Med. 2016, 160, 560–564. [Google Scholar] [CrossRef]
- Patel, A.N.; Park, E.; Kuzman, M.; Benetti, F.; Silva, F.J.; Allickson, J.G. Multipotent menstrual blood stromal stem cells: Isolation, characterization, and differentiation. Cell Transplant. 2008, 17, 303–311. [Google Scholar] [CrossRef] [Green Version]
- Vallvé-Juanico, J.; López-Gil, C.; Ballesteros, A.; Santamaria, X. Endometrial stromal cells circulate in the bloodstream of women with endometriosis: A pilot study. Int. J. Mol. Sci. 2019, 20, 3740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsuzaki, S.; Darcha, C. Epithelial to mesenchymal transition-like and mesenchymal to epithelial transition-like processes might be involved in the pathogenesis of pelvic endometriosis. Hum. Reprod. 2012, 27, 712–721. [Google Scholar] [CrossRef]
- Cervelló, I.; Gil-Sanchis, C.; Mas, A.; Delgado-Rosas, F.; Martínez-Conejero, J.A.; Galán, A.; Martínez-Romero, A.; Martínez, S.; Navarro, I.; Ferro, J.; et al. Human endometrial side population cells exhibit genotypic, phenotypic and functional features of somatic stem cells. PLoS ONE 2010, 5, e10964. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Liang, S.; Yang, F.; Sun, Y.; Niu, L.; Ren, Y.; Wang, H.; He, Y.; Du, J.; Yang, J.; et al. Biological characteristics of endometriotic mesenchymal stem cells isolated from ectopic lesions of patients with endometriosis. Stem Cell Res. Ther. 2020, 11, 346. [Google Scholar] [CrossRef] [PubMed]
- Konrad, L.; Dietze, R.; Riaz, M.A.; Scheiner-Bobis, G.; Behnke, J.; Horné, F.; Hoerscher, A.; Reising, C.; Meinhold-Heerlein, I. Epithelial—Mesenchymal Transition in Endometriosis—When Does It Happen? J. Clin. Med. 2020, 9, 1915. [Google Scholar] [CrossRef] [PubMed]
- Králíčková, M.; Vetvicka, V. Immunological aspects of endometriosis: A review. Ann. Transl. Med. 2015, 3, 153. [Google Scholar] [CrossRef]
- Ścieżyńska, A.; Komorowski, M.; Soszyńska, M.; Malejczyk, J. NK Cells as Potential Targets for Immunotherapy in Endometriosis. J. Clin. Med. 2019, 8, 1468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yarygin, K.N.; Lupatov, A.Y.; Sukhikh, G.T. Modulation of immune responses by mesenchymal stromal cells. Bull. Exp. Biol. Med. 2016, 161, 561–565. [Google Scholar] [CrossRef]
- Quah, B.J.C.; Warren, H.S.; Parish, C.R. Monitoring lymphocyte proliferation in vitro and in vivo with the intracellular fluorescent dye carboxyfluorescein diacetate succinimidyl ester. Nat. Protoc. 2007, 9, 2049–2056. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Mitsuhashi, N.; Klein, A.; Barsky, L.W.; Weinberg, K.; Barr, M.L.; Demetriou, A.; Wu, G.D. The role of the hyaluronan receptor CD44 in mesenchymal stem cell migration in the extracellular matrix. Stem Cells 2006, 24, 928–935. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Kaidina, A.M.; Chiang, J.-H.; Yarygin, K.N.; Lupatov, A.Y. Molecular markers of cancer stem cells verified in vivo. Biomeditsinskaya Khimiya 2016, 62, 228–238. [Google Scholar] [CrossRef]
- Lupatov, A.Y.; Vdovin, A.S.; Vakhrushev, I.V.; Poltavtseva, R.A.; Yarygin, K.N. Comparative analysis of the expression of surface markers on fibroblasts and fibroblast-like cells isolated from different human tissues. Bull. Exp. Biol. Med. 2015, 158, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Lazar, G.A.; Kortemme, T.; Shimaoka, M.; Desjarlais, J.R.; Baker, D.; Springer, T.A. Rational Design of Intercellular Adhesion Molecule-1 (ICAM-1) Variants for Antagonizing Integrin Lymphocyte Function-associated Antigen-1-dependent Adhesion. J. Biol. Chem. 2006, 281, 5042–5049. [Google Scholar] [CrossRef] [Green Version]
- Bousso, P. T-cell activation by dendritic cells in the lymph node: Lessons from the movies. Nat. Rev. Immunol. 2008, 8, 675–684. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, X.; Cao, W.; Shi, Y. Plasticity of mesenchymal stem cells in immunomodulation: Pathological and therapeutic implications. Nat. Immunol. 2014, 15, 1009–1016. [Google Scholar] [CrossRef]
- Harrell, C.R.; Fellabaum, C.; Jovicic, N.; Djonov, V.; Arsenijevic, N.; Volarevic, V. Molecular Mechanisms Responsible for Therapeutic Potential of Mesenchymal Stem Cell-Derived Secretome. Cells 2019, 8, 467. [Google Scholar] [CrossRef] [Green Version]
- Djouad, F.; Charbonnier, L.-M.; Bouffi, C.; Louis-Plence, P.; Bony, C.; Apparailly, F.; Cantos, C.; Jorgensen, C.; Noël, D. Mesenchymal Stem Cells Inhibit the Differentiation of Dendritic Cells Through an Interleukin-6-Dependent Mechanism. Stem Cells 2007, 5, 2025–2032. [Google Scholar] [CrossRef]
- Klinker, M.W.; Marklein, R.A.; Surdo, J.L.L.; Wei, C.H.; Bauer, S.R. Morphological features of IFN-γ-stimulated mesenchymal stromal cells predict overall immunosuppressive capacity. Proc. Natl. Acad. Sci. USA 2017, 114, 2598–2607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cuesta-Gomez, N.; Graham, G.J.; Campbell, J.D.M. Chemokines and their receptors: Predictors of the therapeutic potential of mesenchymal stromal cells. J. Transl. Med. 2021, 19, 156. [Google Scholar] [CrossRef]
- Akoum, A.; Kong, J.; Metz, C.; Beaumont, M.-C. Spontaneous and stimulated secretion of monocyte chemotactic protein-1 and macrophage migration inhibitory factor by peritoneal macrophages in women with and without endometriosis. Fertil. Steril. 2002, 77, 989–994. [Google Scholar] [CrossRef]
- Jakubowicz-Gil, J.; Kotarski, J.; Wertel, I. The Role of Myeloid-Derived Suppressor Cells (MDSCs) in the Development and/or Progression of Endometriosis-State of the Art. Cells 2021, 10, 677. [Google Scholar] [CrossRef]
- Berbic, M.; Hey-Cunningham, A.J.; Ng, C.; Tokushige, N.; Ganewatta, S.; Markham, R.; Russell, P.; Fraser, I.S. The role of Foxp3+ regulatory T-cells in endometriosis: A potential controlling mechanism for a complex, chronic immunological condition. Hum. Reprod. 2010, 25, 900–907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stanic, A.K.; Kim, M.; Styer, A.K.; Rueda, B.R. Dendritic cells attenuate the early establishment of endometriosis-like lesions in a murine model. Reprod. Sci. 2014, 21, 1228–1236. [Google Scholar] [CrossRef]
- Nauta, A.J.; Kruisselbrink, A.B.; Lurvink, E.; Willemze, R.; Fibbe, W.E. Mesenchymal stem cells inhibit generation and function of both CD34+-derived and monocytederived dendritic cells. J. Immunol. 2006, 177, 2080–2087. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, X.X.; Zhang, Y.; Liu, B.; Zhang, S.X.; Wu, Y.; Yu, X.D.; Mao, N. Human mesenchymal stem cells inhibit differentiation and function of monocyte-derived dendritic cells. Blood 2005, 105, 4120–4126. [Google Scholar] [CrossRef] [Green Version]
- Mareschi, K.; Castiglia, S.; Sanavio, F.; Rustichelli, D.; Muraro, M.; Defedele, D.; Bergallo, M.; Fagioliet, F. Immunoregulatory effects on T lymphocytes by human mesenchymal stromal cells isolated from bone marrow, amniotic fluid, and placenta. Exp. Hematol. 2016, 44, 138–150.e1. [Google Scholar] [CrossRef] [Green Version]
- Ren, G.; Zhang, L.; Zhao, X.; Xu, G.; Zhang, Y.; Roberts, A.I.; Zhao, R.C.; Shi, Y. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell 2008, 2, 141–150. [Google Scholar] [CrossRef] [Green Version]
- Krampera, M.; Cosmi, L.; Angeli, R.; Pasini, A.; Liotta, F.; Andreini, A.; Santarlasci, V.; Mazzinghi, B.; Pizzolo, G.; Vinante, F.; et al. Role for interferon-γ in the immunomodulatory activity of human bone marrow mesenchymal stem cells. Stem Cells 2006, 24, 386–398. [Google Scholar] [CrossRef]
- Ramasamy, R.; Fazekasova, H.; Lam, E.W.; Soeiro, I.; Lombardi, G.; Dazzi, F. Mesenchymal stem cells inhibit dendritic cell differentiation and function by preventing entry into the cell cycle. Transplantation 2007, 83, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Glennie, S.; Soeiro, I.; Dyson, P.J.; Lam, E.W.; Dazzi, F. Bone marrow mesenchymal stem cells induce division arrest anergy of activated T cells. Blood 2005, 105, 2821–2827. [Google Scholar] [CrossRef]
- Chomarat, P.; Banchereau, J.; Davoust, J.; Palucka, A.K. IL-6 switches the differentiation of monocytes from dendritic cells to macrophages. Nat. Immunol. 2000, 1, 510–514. [Google Scholar] [CrossRef]
- Laganà, A.S.; Salmeri, F.M.; Ban Frangež, H.; Ghezzi, F.; Vrtačnik-Bokal, E.; Granese, R. Evaluation of M1 and M2 macrophages in ovarian endometriomas from women affected by endometriosis at different stages of the disease. Gynecol. Endocrinol. 2020, 36, 441–444. [Google Scholar] [CrossRef]
- Na, Y.J.; Jin, J.O.; Lee, M.S.; Song, M.G.; Lee, K.S.; Kwak, J.Y. Peritoneal fluid from endometriosis patients switches differentiation of monocytes from dendritic cells to macrophages. Reprod. Immunol. 2008, 77, 63–74. [Google Scholar] [CrossRef]
- Pizzo, A.; Salmeri, F.M.; Ardita, F.V.; Sofo, V.; Tripepi, M.; Marsico, S. Behaviour of cytokine levels in serum and peritoneal fluid of women with endometriosis. Gynecol. Obstet. Investig. 2002, 54, 82–87. [Google Scholar] [CrossRef]
- Waterman, R.S.; Tomchuck, S.L.; Henkle, S.L.; Betancourt, A.M. A new mesenchymal stem cell (MSC) paradigm: Polarization into a pro-inflammatory MSC1 or an immunosuppressive MSC2 phenotype. PLoS ONE 2010, 5, e10088. [Google Scholar] [CrossRef]
- Abramiuk, M.; Bębnowska, D.; Hrynkiewicz, R.; Polak, P.N.G.; Kotarski, J.; Roliński, J.; Grywalska, E. CLTA-4 expression is associated with the maintenance of chronic inflammation in endometriosis and infertility. Cells 2021, 10, 487. [Google Scholar] [CrossRef] [PubMed]
- Najar, M.; Rouas, R.; Raicevic, G.; Boufker, H.I.; Lewalle, P.; Meuleman, N.; Bron, D.; Toungouz, M.; Martiat, P.; Lagneaux, L. Mesenchymal stromal cells promote or suppress the proliferation of T lymphocytes from cord blood and peripheral blood: The importance of low cell ratio and role of interleukin-6. Cytotherapy 2009, 11, 570–583. [Google Scholar] [CrossRef] [PubMed]
- Batlle, E.; Clevers, H. Cancer stem cells revisited. Nat. Med. 2017, 23, 1124–1134. [Google Scholar] [CrossRef] [PubMed]
- Laganà, A.S.; Salmeri, F.M.; Vitale, S.G.; Triolo, O.; Götte, M. Stem cell trafficking during endometriosis: May epigenetics play a pivotal role? Reprod. Sci. 2018, 25, 978–979. [Google Scholar] [CrossRef] [PubMed]
- Suda, K.; Nakaoka, H.; Yoshihara, K.; Ishiguro, T.; Adachi, S.; Kase, H.; Motoyama, T.; Inoue, I.; Enomoto, T. Different mutation profiles between epithelium and stroma in endometriosis and normal endometrium. Hum. Reprod. 2019, 34, 1899–1905. [Google Scholar] [CrossRef] [PubMed]
- Noë, M.; Ayhan, A.; Wang, T.L.; Shih, I.M. Independent development of endometrial epithelium and stroma within the same endometriosis. J. Pathol. 2018, 245, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Séguier, S.; Tartour, E.; Guérin, C.; Couty, L.; Lemitre, M.; Lallement, L.; Folliguet, M.; El Naderi, S.; Terme, M.; Badoual, C.; et al. Inhibition of the Differentiation of Monocyte-Derived Dendritic Cells by Human Gingival Fibroblasts. PLoS ONE 2013, 8, e70937. [Google Scholar] [CrossRef]
| Cytokine | Cell Cultures | Fold Change | p-Value | |||||
|---|---|---|---|---|---|---|---|---|
| eMSC | gMSC | |||||||
| Mean (pg/mL) | SD | SEM | Mean (pg/mL) | SD | SEM | |||
| IL1b | 0.77 | 1.38 | 0.56 | 8.02 | 16.14 | 6.59 | 10.4 | 0.18 |
| IL6 | 222.46 | 191.36 | 78.12 | 40,067.58 | 42,742.52 | 17,449.56 | 180.1 | 0.002 |
| IFN-γ | 3.45 | 4.32 | 1.76 | 20.26 | 11.07 | 4.52 | 5.9 | 0.004 |
| CTACK/CCL27 | 0.59 | 0.55 | 0.23 | 2.67 | 1.71 | 0.70 | 4.5 | 0.018 |
| GCP-2/CXCL6 | 2.23 | 2.96 | 1.21 | 180.61 | 208.40 | 85.08 | 81.0 | 0.002 |
| MCP-1/CCL2 | 157.60 | 226.60 | 92.51 | 2647.84 | 2037.01 | 831.60 | 16.8 | 0.002 |
| MCP-2/CCL8 | 0.73 | 1.07 | 0.44 | 16.98 | 2.42 | 11.19 | 23.2 | 0.026 |
| MCP-3/CCL7 | 13.49 | 10.82 | 4.41 | 166.18 | 118.37 | 48.32 | 12.3 | 0.002 |
| MIG/CXCL9 | 4.90 | 3.72 | 1.52 | 22.33 | 5.43 | 2.21 | 4.5 | <0.001 |
| TARC/CCL17 | 3.44 | 4.87 | 1.99 | 19.79 | 7.74 | 3.16 | 5.7 | 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lupatov, A.Y.; Saryglar, R.Y.; Vtorushina, V.V.; Poltavtseva, R.A.; Bystrykh, O.A.; Chuprynin, V.D.; Krechetova, L.V.; Pavlovich, S.V.; Yarygin, K.N.; Sukhikh, G.T. Mesenchymal Stromal Cells Isolated from Ectopic but Not Eutopic Endometrium Display Pronounced Immunomodulatory Activity In Vitro. Biomedicines 2021, 9, 1286. https://doi.org/10.3390/biomedicines9101286
Lupatov AY, Saryglar RY, Vtorushina VV, Poltavtseva RA, Bystrykh OA, Chuprynin VD, Krechetova LV, Pavlovich SV, Yarygin KN, Sukhikh GT. Mesenchymal Stromal Cells Isolated from Ectopic but Not Eutopic Endometrium Display Pronounced Immunomodulatory Activity In Vitro. Biomedicines. 2021; 9(10):1286. https://doi.org/10.3390/biomedicines9101286
Chicago/Turabian StyleLupatov, Alexey Yu., Roza Yu. Saryglar, Valentina V. Vtorushina, Rimma A. Poltavtseva, Oxana A. Bystrykh, Vladimir D. Chuprynin, Lyubov V. Krechetova, Stanislav V. Pavlovich, Konstantin N. Yarygin, and Gennady T. Sukhikh. 2021. "Mesenchymal Stromal Cells Isolated from Ectopic but Not Eutopic Endometrium Display Pronounced Immunomodulatory Activity In Vitro" Biomedicines 9, no. 10: 1286. https://doi.org/10.3390/biomedicines9101286
APA StyleLupatov, A. Y., Saryglar, R. Y., Vtorushina, V. V., Poltavtseva, R. A., Bystrykh, O. A., Chuprynin, V. D., Krechetova, L. V., Pavlovich, S. V., Yarygin, K. N., & Sukhikh, G. T. (2021). Mesenchymal Stromal Cells Isolated from Ectopic but Not Eutopic Endometrium Display Pronounced Immunomodulatory Activity In Vitro. Biomedicines, 9(10), 1286. https://doi.org/10.3390/biomedicines9101286







