Human VAMP3 Suppresses or Negatively Regulates Bax Induced Apoptosis in Yeast
Abstract
1. Introduction
2. Materials and Methods
2.1. Yeast Strains
2.2. Yeast Transformation
2.3. Detection of Dead Cells with Phloxine B Dye
2.4. Detection of Reactive Oxygen Species (ROS)
2.5. Quantifying Mitochondrial Membrane Potential (MMP) with the JC-10 Dye
2.6. Staining with Hoechst Dye for Monitoring Live Cells
2.7. Assessing Nuclear DNA Fragmentation via the TUNEL Assay
2.8. Western Blotting
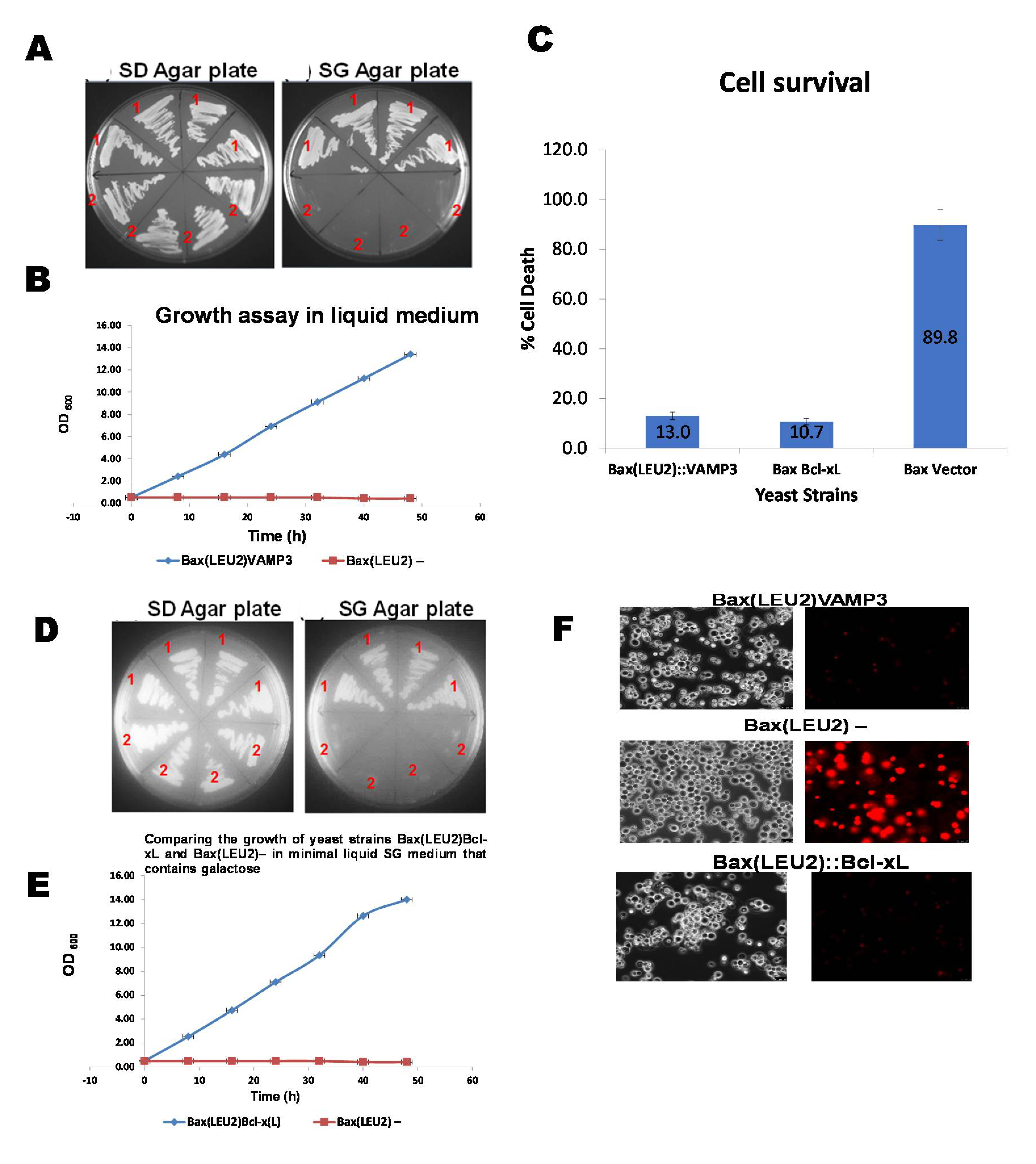
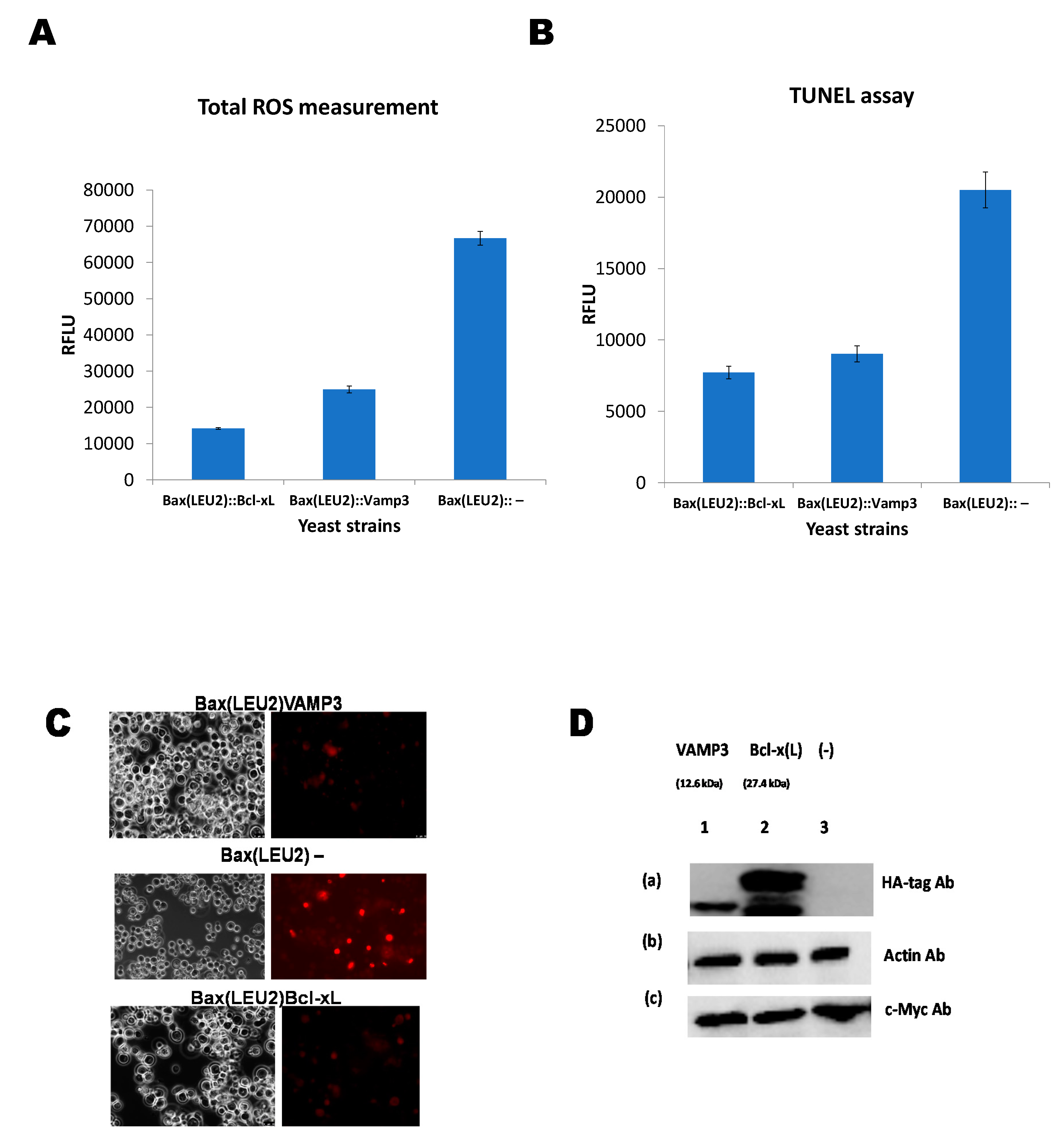
3. Results and Discussion
Coexpression of Human VAMP3 from PGK1 Promoter and Bax from GAL1p in Yeast along with Bcl xL on PGKp
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Coleman, M.; Olson, M. Rho GTPase signalling pathways in the morphological changes associated with apoptosis. Cell Death Differ. 2002, 9, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Polgár, J.; Chung, S.-H.; Reed, G.L. Vesicle-associated membrane protein 3 (VAMP-3) and VAMP-8 are present in human platelets and are required for granule secretion. Blood J. Am. Soc. Hematol. 2002, 100, 1081–1083. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.; Belenghi, B.; Damari-Weisler, H.; Granot, D. Vesicle-associated Membrane Protein ofArabidopsis Suppresses Bax-induced Apoptosis in Yeast Downstream of Oxidative Burst. J. Biol. Chem. 2001, 276, 46284–46289. [Google Scholar] [CrossRef] [PubMed]
- Ligr, M.; Madeo, F.; Fröhlich, E.; Hilt, W.; Fröhlich, K.-U.; Wolf, D.H. Mammalian Bax triggers apoptotic changes in yeast. FEBS Lett. 1998, 438, 61–65. [Google Scholar] [CrossRef]
- Yang, C.; Mora, S.; Ryder, J.W.; Coker, K.J.; Hansen, P.; Allen, L.-A.; Pessin, J.E. VAMP3 null mice display normal constitutive, insulin-and exercise-regulated vesicle trafficking. Mol. Cell. Biol. 2001, 21, 1573–1580. [Google Scholar] [CrossRef]
- Seguin, O.; Descoteaux, A. VAMP3 as a negative regulator of Leishmania amazonensis infection and antigen cross-presentation. Am. Assoc. Immnol. 2020, 204, 231. [Google Scholar]
- Chen, Y.; Sun, J.-X.; Chen, W.-K.; Wu, G.-C.; Wang, Y.-Q.; Zhu, K.; Wang, J. miR-124/VAMP3 is a novel therapeutic target for mitigation of surgical trauma-induced microglial activation. Signal Transduct. Target. Ther. 2019, 4, 1–11. [Google Scholar] [CrossRef]
- Lin, S.-J.; Austriaco, N. Aging and cell death in the other yeasts, Schizosaccharomyces pombe and Candida albicans. FEMS Yeast Res. 2014, 14, 119–135. [Google Scholar] [CrossRef]
- Ameisen, J.C. The origin of programmed cell death. Science 1996, 272, 1278–1279. [Google Scholar] [CrossRef]
- Madeo, F.; Herker, E.; Maldener, C.; Wissing, S.; Lächelt, S.; Herlan, M.; Fehr, M.; Lauber, K.; Sigrist, S.J.; Wesselborg, S.; et al. A caspase-related protease regulates apoptosis in yeast. Mol. Cell 2002, 9, 911–917. [Google Scholar] [CrossRef]
- O’Brien, I.E.; Reutelingsperger, C.P.; Holdaway, K.M. Annexin-V and TUNEL use in monitoring the progression of apoptosis in plants. Cytom. J. Int. Soc. Anal. Cytol. 1997, 29, 28–33. [Google Scholar] [CrossRef]
- Jović, M.; Kean, M.J.; Dubankova, A.; Boura, E.; Gingras, A.-C.; Brill, J.A.; Balla, T. Endosomal sorting of VAMP3 is regulated by PI4K2A. J. Cell Sci. 2014, 127, 3745–3756. [Google Scholar] [CrossRef] [PubMed]
- Hager, H.A.; Roberts, R.J.; Cross, E.E.; Proux-Gillardeaux, V.; Bader, D.M. Identification of a novel Bves function: Regulation of vesicular transport. EMBO J. 2010, 29, 532–545. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Hardee, D.; Minnear, F. Membrane fusion by VAMP3 and plasma membrane t-SNAREs. Exp. Cell Res. 2007, 313, 3198–3209. [Google Scholar] [CrossRef] [PubMed]
- Akintade, D.D.; Chaudhuri, B. Sensing the Generation of Intracellular Free Electrons Using the Inactive Catalytic Subunit of Cytochrome P450s as a Sink. Sensors 2020, 20, 4050. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, B. Apoptosis Methods, Genes and Proteins. U.S. Patent 20090258794, 15 October 2009. [Google Scholar]
- Kawai, S.; Hashimoto, W.; Murata, K. Transformation of Saccharomyces cerevisiae and other fungi: Methods and possible underlying mechanism. Bioeng. Bugs 2010, 1, 395–403. [Google Scholar] [CrossRef]
- Kwolek-Mirek, M.; Zadrag-Tecza, R. Comparison of methods used for assessing the viability and vitality of yeast cells. FEMS Yeast Res. 2014, 14, 1068–1079. [Google Scholar] [CrossRef]
- Derf, A.; Mudududdla, R.; Akintade, D.; Williams, I.S.; Abdullaha, M.; Chaudhuri, B.; Bharate, S.B. Nonantioxidant Tetramethoxystilbene Abrogates α-Synuclein-Induced Yeast Cell Death but Not That Triggered by the Bax or βA4 Peptide. ACS Omega 2018, 3, 9513–9532. [Google Scholar] [CrossRef]
- Von Der Haar, T. Optimized protein extraction for quantitative proteomics of yeasts. PLoS ONE 2007, 2, e1078. [Google Scholar] [CrossRef]
- Gerst, J.E.; Rodgers, L.; Riggs, M.; Wigler, M. SNC1, a yeast homolog of the synaptic vesicle-associated membrane protein/synaptobrevin gene family: Genetic interactions with the RAS and CAP genes. Proc. Natl. Acad. Sci. USA 1992, 89, 4338–4342. [Google Scholar] [CrossRef]
- Fleury, C.; Pampin, M.; Tarze, A.; Mignotte, B. Yeast as a model to study apoptosis? Biosci. Rep. 2002, 22, 59–79. [Google Scholar] [CrossRef] [PubMed]
- Attardi, G.; Schatz, G. Biogenesis of mitochondria. Annu. Rev. Cell Biol. 1988, 4, 289–331. [Google Scholar] [CrossRef]
- Akintade, D.D.; Chaudhuri, B. The effect of copy number on α-synuclein’s toxicity and its protective role in Bax-induced apoptosis, in yeast. Biosci. Rep. 2020, 40, BSR20201912. [Google Scholar] [CrossRef] [PubMed]
- Gordon, D.E.; Chia, J.; Jayawardena, K.; Antrobus, R.; Bard, F.; Peden, A.A. VAMP3/Syb and YKT6 are required for the fusion of constitutive secretory carriers with the plasma membrane. PLoS Genet. 2017, 13, e1006698. [Google Scholar] [CrossRef] [PubMed]
- Kean, M.J.; Williams, K.C.; Skalski, M.; Myers, D.; Burtnik, A.; Foster, D.; Coppolino, M.G. VAMP3, syntaxin-13 and SNAP23 are involved in secretion of matrix metalloproteinases, degradation of the extracellular matrix and cell invasion. J. Cell Sci. 2009, 122, 4089–4098. [Google Scholar] [CrossRef]
- Kumar, R.; Tang, Q.; Müller, S.A.; Gao, P.; Mahlstedt, D.; Zampagni, S.; Tan, Y.; Klingl, A.; Bötzel, K.; Lichtenthaler, S.F.; et al. Fibroblast Growth Factor 2-Mediated Regulation of Neuronal Exosome Release Depends on VAMP3/Cellubrevin in Hippocampal Neurons. Adv. Sci. 2020, 7, 1902372. [Google Scholar] [CrossRef]
- Nozawa, T.; Minowa-Nozawa, A.; Aikawa, C.; Nakagawa, I. The STX6-VTI1B-VAMP3 complex facilitates xenophagy by regulating the fusion between recycling endosomes and autophagosomes. Autophagy 2017, 13, 57–69. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akintade, D.D.; Chaudhuri, B. Human VAMP3 Suppresses or Negatively Regulates Bax Induced Apoptosis in Yeast. Biomedicines 2021, 9, 95. https://doi.org/10.3390/biomedicines9010095
Akintade DD, Chaudhuri B. Human VAMP3 Suppresses or Negatively Regulates Bax Induced Apoptosis in Yeast. Biomedicines. 2021; 9(1):95. https://doi.org/10.3390/biomedicines9010095
Chicago/Turabian StyleAkintade, Damilare D., and Bhabatosh Chaudhuri. 2021. "Human VAMP3 Suppresses or Negatively Regulates Bax Induced Apoptosis in Yeast" Biomedicines 9, no. 1: 95. https://doi.org/10.3390/biomedicines9010095
APA StyleAkintade, D. D., & Chaudhuri, B. (2021). Human VAMP3 Suppresses or Negatively Regulates Bax Induced Apoptosis in Yeast. Biomedicines, 9(1), 95. https://doi.org/10.3390/biomedicines9010095




