Mechanisms Underlying Resistance to FLT3 Inhibitors in Acute Myeloid Leukemia
Abstract
1. Introduction
2. Prognostic Impact of FLT3 Mutations
3. Classification of FLT3 Inhibitors by Its Pharmacodynamics
4. Current Clinical Role of FLT3 Inhibitors
5. Mechanisms of Resistance to FLT3 Inhibitors
5.1. Primary Resistance
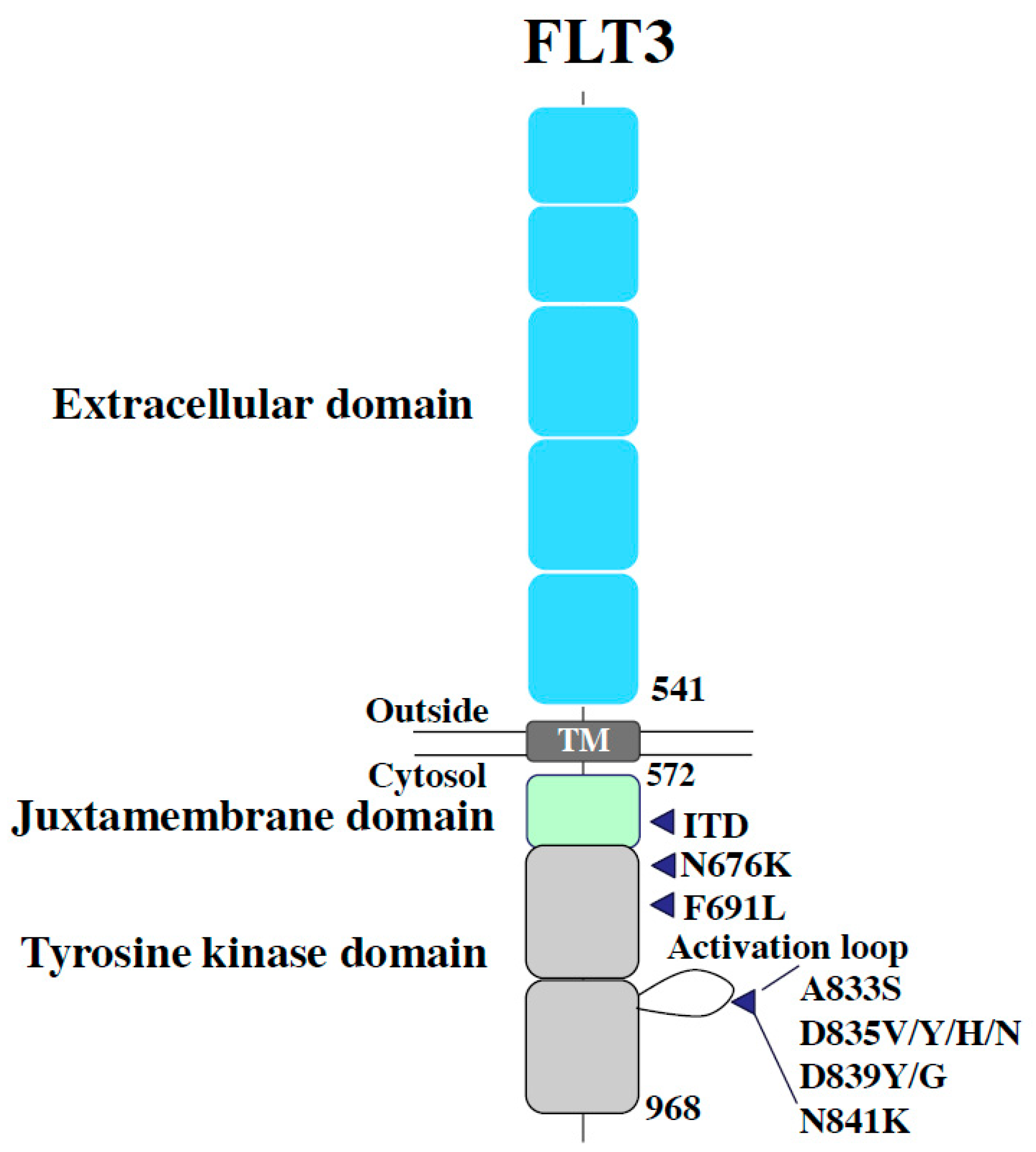
5.2. Secondary Resistance Due to Additional FLT3 Mutations (on-Target Resistance)
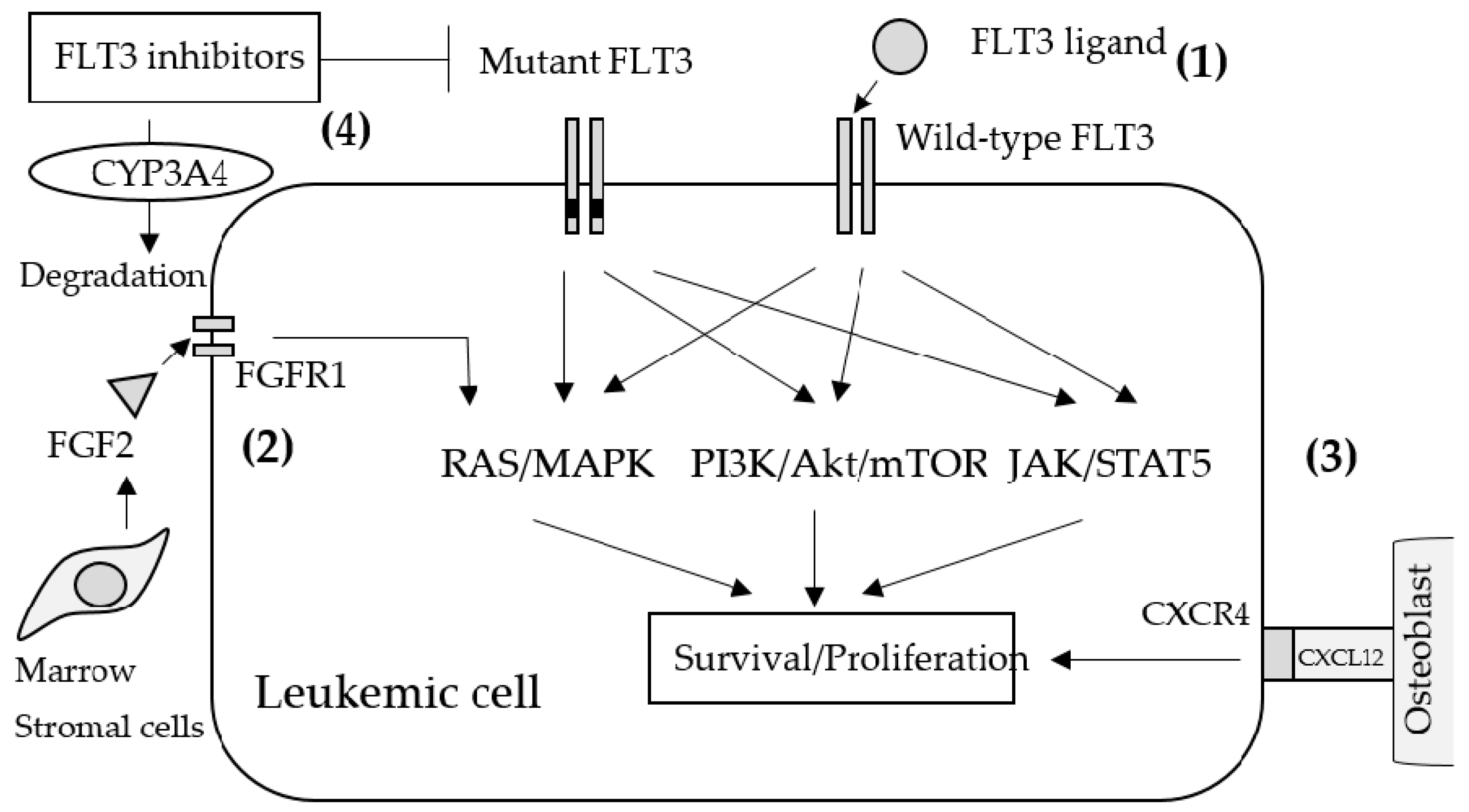
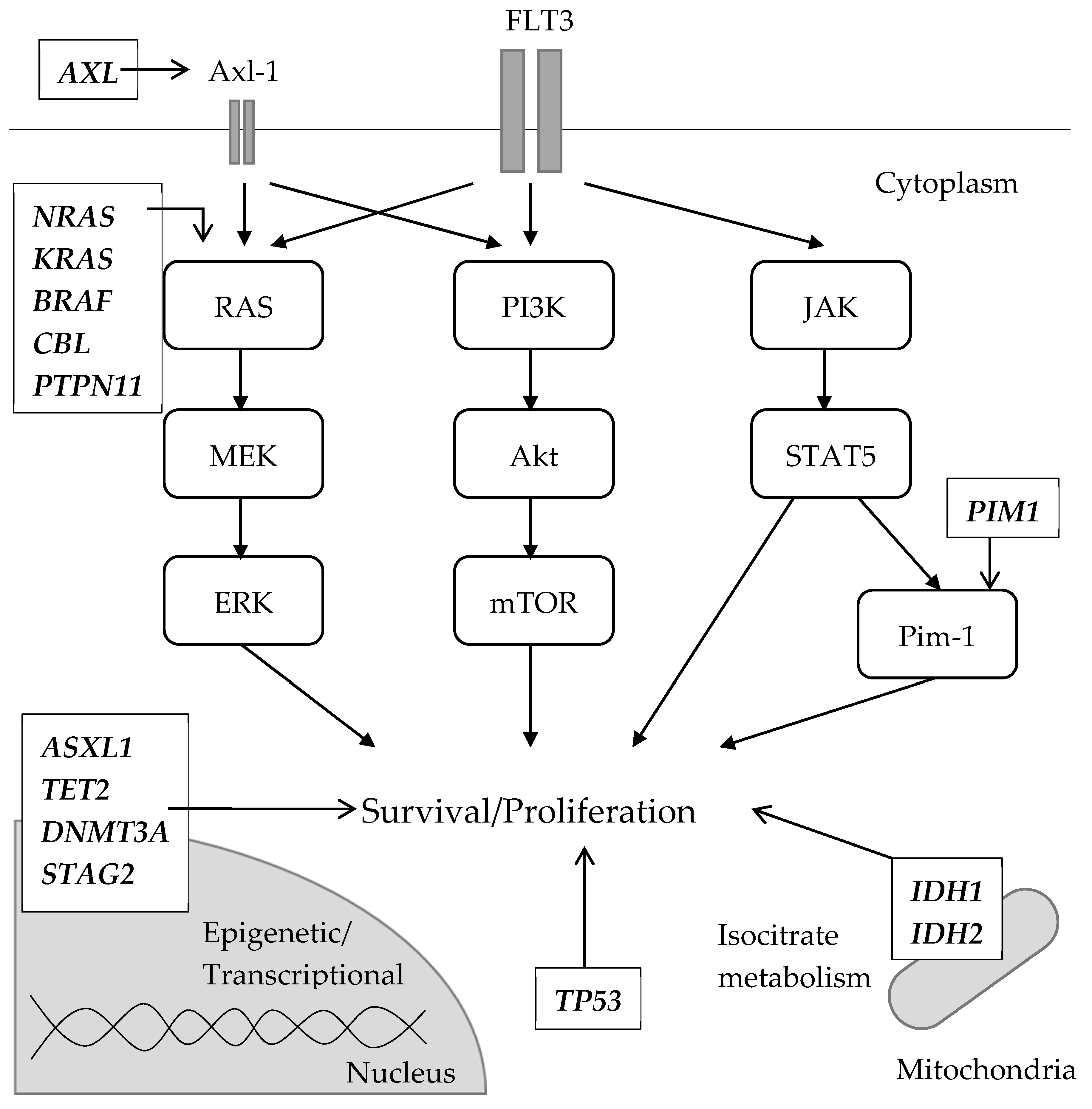
5.3. Secondary Resistance Due to Non-FLT3 Abnormalities (off-Target Resistance)
6. Strategies to Overcome Resistance to FLT3 Inhibitors
6.1. Development of Novel Agents
6.2. Combination with Different Class Agents
6.3. Genetic Mutation Analysis
7. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Rosnet, O.; Mattei, M.-G.; Marchetto, S.; Birnbaum, D. Isolation and chromosomal localization of a novel FMS-like tyrosine kinase gene. Genomics 1991, 9, 380–385. [Google Scholar] [CrossRef]
- Rosnet, O.; Schiff, C.; Pebusque, M.J.; Marchetto, S.; Tonnelle, C.; Toiron, Y.; Birg, F.; Birnbaum, D. Human FLT3/FLK2 gene: cDNA cloning and expression in hematopoietic cells. Blood 1993, 82, 1110–1119. [Google Scholar] [CrossRef] [PubMed]
- Lemmon, M.A.; Schlessinger, J. Cell Signaling by Receptor Tyrosine Kinases. Cell 2010, 141, 1117–1134. [Google Scholar] [CrossRef] [PubMed]
- Small, D.; Levenstein, M.; Kim, E.; Carow, C.; Amin, S.; Rockwell, P.; Witte, L.; Burrow, C.; Ratajczak, M.Z.; Gewirtz, A.M. STK-1, the human homolog of Flk-2/Flt-3, is selectively expressed in CD34+ human bone marrow cells and is involved in the proliferation of early progenitor/stem cells. Proc. Natl. Acad. Sci. USA 1994, 91, 459–463. [Google Scholar] [CrossRef] [PubMed]
- Hannum, C.; Culpepper, J.; Campbell, D.; McClanahan, T.; Zurawski, S.; Kastelein, R.; Bazan, J.F.; Hudak, S.; Wagner, J.; Mattson, J.; et al. Ligand for FLT3/FLK2 receptor tyrosine kinase regulates growth of haematopoietic stem cells and is encoded by variant RNAs. Nature 1994, 368, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Lyman, S.D.; James, L.; Bos, T.V.; De Vries, P.; Brasel, K.; Gliniak, B.; Hollingsworth, L.; Picha, K.S.; McKenna, H.J.; Splett, R.R.; et al. Molecular cloning of a ligand for the flt3flk-2 tyrosine kinase receptor: A proliferative factor for primitive hematopoietic cells. Cell 1993, 75, 1157–1167. [Google Scholar] [CrossRef]
- Hayakawa, F.; Towatari, M.; Kiyoi, H.; Tanimoto, M.; Kitamura, T.; Saito, H.; Naoe, T. Tandem-duplicated Flt3 constitutively activates STAT5 and MAP kinase and introduces autonomous cell growth in IL-3-dependent cell lines. Oncogene 2000, 19, 624–631. [Google Scholar] [CrossRef]
- Mizuki, M.; Fenski, R.; Halfter, H.; Matsumura, I.; Schmidt, R.; Müller, C.; Grüning, W.; Kratz-Albers, K.; Serve, S.; Steur, C.; et al. Flt3 mutations from patients with acute myeloid leukemia induce transformation of 32D cells mediated by the Ras and STAT5 pathways. Blood 2000, 96, 3907–3914. [Google Scholar] [CrossRef]
- Meshinchi, S.; Appelbaum, F.R. Structural and functional alterations of FLT3 in acute myeloid leukemia. Clin. Cancer Res. 2009, 15, 4263–4269. [Google Scholar] [CrossRef]
- Stirewalt, D.L.; Radich, J.P. The role of FLT3 in haematopoietic malignancies. Nat. Rev. Cancer 2003, 3, 650–665. [Google Scholar] [CrossRef]
- Drexler, H.G.; Meyer, C.; Quentmeier, H. Effects of FLT3 Ligand on Proliferation and Survival of Myeloid Leukemia Cells. Leuk. Lymphoma 1999, 33, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Nakao, M.; Yokota, S.; Iwai, T.; Kaneko, H.; Horiike, S.; Kashima, K.; Sonoda, Y.; Fujimoto, T.; Misawa, S. Internal tandem duplication of the flt3 gene found in acute myeloid leukemia. Leukemia 1996, 10, 1911–1918. [Google Scholar] [PubMed]
- Yamamoto, Y. Activating mutation of D835 within the activation loop of FLT3 in human hematologic malignancies. Blood 2001, 97, 2434–2439. [Google Scholar] [CrossRef] [PubMed]
- Kiyoi, H.; Naoe, T. Biology, Clinical Relevance, and Molecularly Targeted Therapy in Acute Leukemia with FLT3 Mutation. Int. J. Hematol. 2006, 83, 301–308. [Google Scholar] [CrossRef]
- Kihara, R.; Nagata, Y.; Kiyoi, H.; Kato, T.; Yamamoto, E.; Suzuki, K.; Chen, F.; Asou, N.; Ohtake, S.; Miyawaki, S.; et al. Comprehensive analysis of genetic alterations and their prognostic impacts in adult acute myeloid leukemia patients. Leukemia 2014, 28, 1586–1595. [Google Scholar] [CrossRef]
- Ley, T.J.; Miller, C.A.; Ding, L.; Raphael, B.J.; Mungall, A.J.; Robertson, A.G.; Hoadley, K.A.; Triche, T.J.; Laird, P.W.; Baty, J.D.; et al. The Cancer Genome Atlas Research Network; Cancer Genome Atlas Research Network; Genomic and Epigenomic Landscapes of Adult De Novo Acute Myeloid Leukemia. N. Engl. J. Med. 2013, 368, 2059–2074. [Google Scholar] [CrossRef]
- Janke, H.; Pastore, F.; Schumacher, D.; Herold, T.; Hopfner, K.-P.; Schneider, S.; Berdel, W.E.; Buchner, T.; Woermann, B.J.; Subklewe, M.; et al. Activating FLT3 Mutants Show Distinct Gain-of-Function Phenotypes In Vitro and a Characteristic Signaling Pathway Profile Associated with Prognosis in Acute Myeloid Leukemia. PLoS ONE 2014, 9, e89560. [Google Scholar] [CrossRef]
- Okada, K.; Nogami, A.; Ishida, S.; Akiyama, H.; Chen, C.; Umezawa, Y.; Miura, O. FLT3-ITD induces expression of Pim kinases through STAT5 to confer resistance to the PI3K/Akt pathway inhibitors on leukemic cells by enhancing the mTORC1/Mcl-1 pathway. Oncotarget 2017, 9, 8870–8886. [Google Scholar] [CrossRef]
- Solovey, M.; Wang, Y.; Michel, C.; Metzeler, K.H.; Herold, T.; Göthert, J.R.; Ellenrieder, V.; Hessmann, E.; Gattenlöhner, S.; Neubauer, A.; et al. Nuclear factor of activated T-cells, NFATC1, governs FLT3ITD-driven hematopoietic stem cell transformation and a poor prognosis in AML. J. Hematol. Oncol. 2019, 12, 72. [Google Scholar] [CrossRef]
- Sun, Y.-M.; Wang, W.-T.; Zeng, Z.-C.; Chen, T.-Q.; Han, C.; Pan, Q.; Huang, W.; Fang, K.; Sun, L.-Y.; Zhou, Y.-F.; et al. circMYBL2, a circRNA from MYBL2, regulates FLT3 translation by recruiting PTBP1 to promote FLT3-ITD AML progression. Blood 2019, 134, 1533–1546. [Google Scholar] [CrossRef]
- Zhang, Y.; Askenazi, M.; Jiang, J.; Luckey, C.J.; Griffin, J.D.; Marto, J.A. A Robust Error Model for iTRAQ Quantification Reveals Divergent Signaling between Oncogenic FLT3 Mutants in Acute Myeloid Leukemia. Mol. Cell. Proteom. 2009, 9, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Klingmüller, U.; Lorenz, U.; Cantley, L.C.; Neel, B.G.; Lodish, H.F. Specific recruitment of SH-PTP1 to the erythropoietin receptor causes inactivation of JAK2 and termination of proliferative signals. Cell 1995, 80, 729–738. [Google Scholar] [CrossRef]
- Grundler, R.; Miething, C.; Thiede, C.; Peschel, C.; Duyster, J. FLT3-ITD and tyrosine kinase domain mutants induce 2 distinct phenotypes in a murine bone marrow transplantation model. Blood 2005, 105, 4792–4799. [Google Scholar] [CrossRef] [PubMed]
- Bailey, E.; Li, L.; Duffield, A.S.; Ma, H.S.; Huso, D.L.; Small, N. FLT3/D835Y mutation knock-in mice display less aggressive disease compared with FLT3/internal tandem duplication (ITD) mice. Proc. Natl. Acad. Sci. USA 2013, 110, 21113–21118. [Google Scholar] [CrossRef] [PubMed]
- Bagrintseva, K.; Geisenhof, S.; Kern, R.; Eichenlaub, S.; Reindl, C.; Ellwart, J.W.; Hiddemann, W.; Spiekermann, K. FLT3-ITD-TKD dual mutants associated with AML confer resistance to FLT3 PTK inhibitors and cytotoxic agents by overexpression of Bcl-x(L). Blood 2005, 105, 3679–3685. [Google Scholar] [CrossRef] [PubMed]
- Kiyoi, H. FLT3 Inhibitors: Recent advances and problems for clinical application. Nagoya J. Med. Sci. 2015, 77, 7–17. [Google Scholar]
- Döhner, H.; Estey, E.; Grimwade, D.; Amadori, S.; Appelbaum, F.R.; Büchner, T.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Larson, R.A.; et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 2017, 129, 424–447. [Google Scholar] [CrossRef]
- Ghiaur, G.; Levis, M.J. Mechanisms of Resistance to FLT3 Inhibitors and the Role of the Bone Marrow Microenvironment. Hematol. Clin. N. Am. 2017, 31, 681–692. [Google Scholar] [CrossRef]
- Zhou, J.; Chng, W.-J. Resistance to FLT3 inhibitors in acute myeloid leukemia: Molecular mechanisms and resensitizing strategies. World J. Clin. Oncol. 2018, 9, 90–97. [Google Scholar] [CrossRef]
- Daver, N.; Schlenk, R.F.; Russell, N.H.; Levis, M.J. Targeting FLT3 mutations in AML: Review of current knowledge and evidence. Leukemia 2019, 33, 299–312. [Google Scholar] [CrossRef]
- Port, M.; Böttcher, M.; Thol, F.; Ganser, A.; Schlenk, R.; Wasem, J.; Neumann, A.; Pouryamout, L. Prognostic significance of FLT3 internal tandem duplication, nucleophosmin 1, and CEBPA gene mutations for acute myeloid leukemia patients with normal karyotype and younger than 60 years: A systematic review and meta-analysis. Ann. Hematol. 2014, 93, 1279–1286. [Google Scholar] [CrossRef] [PubMed]
- Thiede, C.; Steudel, C.; Mohr, B.; Schaich, M.; Schäkel, U.; Platzbecker, U.; Wermke, M.; Bornhäuser, M.; Ritter, M.; Neubauer, A.; et al. Analysis of FLT3-activating mutations in 979 patients with acute myelogenous leukemia: Association with FAB subtypes and identification of subgroups with poor prognosis. Blood 2002, 99, 4326–4335. [Google Scholar] [CrossRef] [PubMed]
- Schlenk, R.F.; Kayser, S.; Bullinger, L.; Kobbe, G.; Casper, J.; Ringhoffer, M.; Held, G.; Brossart, P.; Lübbert, M.; Salih, H.R.; et al. Differential impact of allelic ratio and insertion site in FLT3-ITD–positive AML with respect to allogeneic transplantation. Blood 2014, 124, 3441–3449. [Google Scholar] [CrossRef]
- Yalniz, F.F.; Dalle, I.A.; Kantarjian, H.M.; Borthakur, G.; Kadia, T.M.; Patel, K.; Loghavi, S.; Garcia-Manero, G.; Sasaki, K.; Daver, N.; et al. Prognostic significance of baselineFLT3-ITD mutant allele level in acute myeloid leukemia treated with intensive chemotherapy with/without sorafenib. Am. J. Hematol. 2019, 94, 984–991. [Google Scholar] [CrossRef]
- Pratcorona, M.; Brunet, S.; Nomdedéu, J.F.; Ribera, J.; Tormo, M.; Duarte, R.; Escoda, L.; Guàrdia, R.; De Llano, M.P.Q.; Salamero, O.; et al. Favorable outcome of patients with acute myeloid leukemia harboring a low-allelic burden FLT3-ITD mutation and concomitant NPM1 mutation: Relevance to post-remission therapy. Blood 2013, 121, 2734–2738. [Google Scholar] [CrossRef] [PubMed]
- Linch, D.C.; Hills, R.K.; Burnett, A.K.; Khwaja, A.; Gale, R.E. Impact of FLT3ITD mutant allele level on relapse risk in intermediate-risk acute myeloid leukemia. Blood 2014, 124, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Versluis, J.; Devillier, R.; Van Putten, W.L.J.; Manz, M.G.; Vekemans, M.-C.; Legdeur, M.-C.; Passweg, J.R.; Maertens, J.; Kuball, J.; Biemond, B.J.; et al. Comparative value of post-remission treatment in cytogenetically normal AML subclassified by NPM1 and FLT3-ITD allelic ratio. Leukemia 2016, 31, 26–33. [Google Scholar] [CrossRef]
- Sakaguchi, M.; Yamaguchi, H.; Najima, Y.; Usuki, K.; Ueki, T.; Oh, I.; Mori, S.; Kawata, E.; Uoshima, N.; Kobayashi, Y.; et al. Prognostic impact of low allelic ratio FLT3-ITD and NPM1 mutation in acute myeloid leukemia. Blood Adv. 2018, 2, 2744–2754. [Google Scholar] [CrossRef]
- Bacher, U.; Haferlach, C.; Kern, W.; Haferlach, T.; Schnittger, S. Prognostic relevance of FLT3-TKD mutations in AML: The combination matters—An analysis of 3082 patients. Blood 2008, 111, 2527–2537. [Google Scholar] [CrossRef]
- Zhang, H.; Savage, S.; Reister-Schultz, A.; Bottomly, D.; White, L.; Segerdell, E.; Wilmot, B.; McWeeney, S.K.; Eide, C.A.; Nechiporuk, T.; et al. Clinical resistance to crenolanib in acute myeloid leukemia due to diverse molecular mechanisms. Nat. Commun. 2019, 10, 244. [Google Scholar] [CrossRef]
- Kelly, L.M.; Yu, J.-C.; Boulton, C.L.; Apatira, M.; Li, J.; Sullivan, C.M.; Williams, I.; Amaral, S.M.; Curley, D.P.; Duclos, N.; et al. CT53518, a novel selective FLT3 antagonist for the treatment of acute myelogenous leukemia (AML). Cancer Cell 2002, 1, 421–432. [Google Scholar] [CrossRef]
- Smith, B.D.; Levis, M.; Beran, M.; Giles, F.; Kantarjian, H.; Berg, K.; Murphy, K.M.; Dauses, T.; Allebach, J.; Small, D. Single-agent CEP-701, a novel FLT3 inhibitor, shows biologic and clinical activity in patients with relapsed or refractory acute myeloid leukemia. Blood 2004, 103, 3669–3676. [Google Scholar] [CrossRef] [PubMed]
- Stone, R.M.; DeAngelo, D.J.; Klimek, V.; Galinsky, I.; Estey, E.; Nimer, S.D.; Grandin, W.; Lebwohl, D.; Wang, Y.; Cohen, P.; et al. Patients with acute myeloid leukemia and an activating mutation in FLT3 respond to a small-molecule FLT3 tyrosine kinase inhibitor, PKC412. Blood 2005, 105, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Fiedler, W.; Serve, H.; Döhner, H.; Schwittay, M.; Ottmann, O.G.; O’Farrell, A.-M.; Bello, C.L.; Allred, R.; Manning, W.C.; Cherrington, J.M.; et al. A phase 1 study of SU11248 in the treatment of patients with refractory or resistant acute myeloid leukemia (AML) or not amenable to conventional therapy for the disease. Blood 2005, 105, 986–993. [Google Scholar] [CrossRef] [PubMed]
- Ravandi, F.; Cortes, J.E.; Jones, D.; Faderl, S.; Garcia-Manero, G.; Konopleva, M.Y.; O’Brien, S.; Estrov, Z.; Borthakur, G.; Thomas, D.; et al. Phase I/II Study of Combination Therapy with Sorafenib, Idarubicin, and Cytarabine in Younger Patients with Acute Myeloid Leukemia. J. Clin. Oncol. 2010, 28, 1856–1862. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Kaneko, N.; Ueno, Y.; Yamada, M.; Tanaka, R.; Saito, R.; Shimada, I.; Mori, K.; Kuromitsu, S. Gilteritinib, a FLT3/AXL inhibitor, shows antileukemic activity in mouse models of FLT3 mutated acute myeloid leukemia. Investig. New Drugs 2017, 35, 556–565. [Google Scholar] [CrossRef]
- Lee, L.Y.; Hernandez, D.; Rajkhowa, T.; Smith, S.C.; Raman, J.; Nguyen, B.; Small, N.; Levis, M. Preclinical studies of gilteritinib, a next-generation FLT3 inhibitor. Blood 2017, 129, 257–260. [Google Scholar] [CrossRef]
- Zarrinkar, P.P.; Gunawardane, R.N.; Cramer, M.D.; Gardner, M.F.; Brigham, D.; Belli, B.; Karaman, M.W.; Pratz, K.W.; Pallares, G.; Chao, Q.; et al. AC220 is a uniquely potent and selective inhibitor of FLT3 for the treatment of acute myeloid leukemia (AML). Blood 2009, 114, 2984–2992. [Google Scholar] [CrossRef]
- Galanis, A.; Ma, H.; Rajkhowa, T.; Ramachandran, A.; Small, N.; Cortes, J.; Levis, M. Crenolanib is a potent inhibitor of FLT3 with activity against resistance-conferring point mutants. Blood 2014, 123, 94–100. [Google Scholar] [CrossRef]
- Smith, C.C.; Lasater, E.A.; Lin, K.C.; Wang, Q.; McCreery, M.Q.; Stewart, W.K.; Damon, L.E.; Perl, A.E.; Jeschke, G.; Sugita, M.; et al. Crenolanib is a selective type I pan-FLT3 inhibitor. Proc. Natl. Acad. Sci. USA 2014, 111, 5319–5324. [Google Scholar] [CrossRef]
- Angiolini, M. Targeting the DFG-in kinase conformation: A new trend emerging from a patent analysis. Future Med. Chem. 2011, 3, 309–337. [Google Scholar] [CrossRef] [PubMed]
- Ke, Y.-Y.; Singh, V.K.; Coumar, M.S.; Hsu, Y.C.; Wang, W.-C.; Song, J.-S.; Chen, C.-H.; Lin, W.-H.; Wu, S.-H.; Hsu, J.T.A.; et al. Homology modeling of DFG-in FMS-like tyrosine kinase 3 (FLT3) and structure-based virtual screening for inhibitor identification. Sci. Rep. 2015, 5, 11702. [Google Scholar] [CrossRef] [PubMed]
- Bhullar, K.S.; Lagarón, N.O.; McGowan, E.M.; Parmar, I.; Jha, A.; Hubbard, B.P.; Rupasinghe, H.P.V. Kinase-targeted cancer therapies: Progress, challenges and future directions. Mol. Cancer 2018, 17, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Kiyoi, H.; Kawashima, N.; Ishikawa, Y. FLT3 mutations in acute myeloid leukemia: Therapeutic paradigm beyond inhibitor development. Cancer Sci. 2019, 111, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.C.; Lin, K.; Stecula, A.; Sali, A.; Shah, N.P. FLT3 D835 mutations confer differential resistance to type II FLT3 inhibitors. Leukemia 2015, 29, 2390–2392. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.B.; Nguyen, B.; Li, L.; Brown, P.; Levis, M.; Leahy, D.; Small, N. Mutations of FLT3/ITD confer resistance to multiple tyrosine kinase inhibitors. Leukemia 2012, 27, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Cools, J. Prediction of Resistance to Small Molecule FLT3 Inhibitors: Implications for Molecularly Targeted Therapy of Acute Leukemia. Cancer Res. 2004, 64, 6385–6389. [Google Scholar] [CrossRef]
- McMahon, C.M.; Canaani, J.; Rea, B.; McMahon, C.M.; Canaani, J.; Rea, B.; Sargent, R.L.; Morrissette, J.J.; Lieberman, D.B.; Watt, C.; et al. Mechanisms of Acquired Resistance to Gilteritinib Therapy in Relapsed and Refractory FLT3 -Mutated Acute Myeloid Leukemia. Blood 2017, 130 (Suppl. 1), 295. [Google Scholar] [CrossRef]
- Staudt, D.; Murray, H.C.; McLachlan, T.; Alvaro, F.; Enjeti, A.K.; Verrills, N.M.; Dun, M.D. Targeting Oncogenic Signaling in Mutant FLT3 Acute Myeloid Leukemia: The Path to Least Resistance. Int. J. Mol. Sci. 2018, 19, 3198. [Google Scholar] [CrossRef]
- Eid, S.; Turk, S.; Volkamer, A.; Rippmann, F.; Fulle, S. KinMap: A web-based tool for interactive navigation through human kinome data. BMC Bioinform. 2017, 18, 16. [Google Scholar] [CrossRef]
- Klaeger, S.; Heinzlmeir, S.; Wilhelm, M.; Polzer, H.; Vick, B.; Koenig, P.-A.; Reinecke, M.; Ruprecht, B.; Wiechmann, S.; Meng, C.; et al. The target landscape of clinical kinase drugs. Science 2017, 358, 4368. [Google Scholar] [CrossRef]
- Karaman, M.W.; Herrgard, S.; Treiber, D.K.; Gallant, P.; Atteridge, C.E.; Campbell, B.T.; Chan, K.W.; Ciceri, P.; Davis, M.I.; Edeen, P.T.; et al. A quantitative analysis of kinase inhibitor selectivity. Nat. Biotechnol. 2008, 26, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Weisberg, E.; Puissant, A.; Stone, R.; Sattler, M.; Buhrlage, S.J.; Yang, J.; Manley, P.W.; Meng, C.; Buonopane, M.; Daley, J.F.; et al. Characterization of midostaurin as a dual inhibitor of FLT3 and SYK and potentiation of FLT3 inhibition against FLT3-ITD-driven leukemia harboring activated SYK kinase. Oncotarget 2017, 8, 52026–52044. [Google Scholar] [CrossRef] [PubMed]
- O’Farrell, A.-M.; Abrams, T.J.; Yuen, H.A.; Ngai, T.J.; Louie, S.G.; Yee, K.W.H.; Wong, L.M.; Hong, W.; Lee, L.B.; Town, A.; et al. SU11248 is a novel FLT3 tyrosine kinase inhibitor with potent activity in vitro and in vivo. Blood 2003, 101, 3597–3605. [Google Scholar] [CrossRef] [PubMed]
- Levis, M.J.; Allebach, J.; Tse, K.-F.; Zheng, R.; Baldwin, B.R.; Smith, B.D.; Jones-Bolin, S.; Ruggeri, B.; Dionne, C.; Small, N. A FLT3-targeted tyrosine kinase inhibitor is cytotoxic to leukemia cells in vitro and in vivo. Blood 2002, 99, 3885–3891. [Google Scholar] [CrossRef] [PubMed]
- Mori, S.; Cortes, J.; Kantarjian, H.; Zhang, W.; Andreef, M.; Ravandi, F. Potential role of sorafenib in the treatment of acute myeloid leukemia. Leuk. Lymphoma 2008, 49, 2246–2255. [Google Scholar] [CrossRef]
- Schittenhelm, M.M.; Kampa, K.M.; Yee, K.W.H.; Heinrich, M.C. The FLT3 inhibitor tandutinib (formerly MLN518) has sequence-independent synergistic effects with cytarabine and daunorubicin. Cell Cycle 2009, 8, 2621–2630. [Google Scholar] [CrossRef]
- Aikawa, T.; Togashi, N.; Iwanaga, K.; Okada, H.; Nishiya, Y.; Inoue, S.; Levis, M.J.; Isoyama, T. Quizartinib, a selective FLT3 inhibitor, maintains antileukemic activity in preclinical models of RAS-mediated midostaurin-resistant acute myeloid leukemia cells. Oncotarget 2020, 11, 943–955. [Google Scholar] [CrossRef]
- Nguyen, B.; Williams, A.B.; Young, D.J.; Ma, H.; Li, L.; Levis, M.; Brown, P.; Small, N. FLT3 activating mutations display differential sensitivity to multiple tyrosine kinase inhibitors. Oncotarget 2017, 8, 10931–10944. [Google Scholar] [CrossRef]
- Barry, E.V.; Clark, J.J.; Cools, J.; Roesel, J.; Gilliland, D.G. Uniform sensitivity of FLT3 activation loop mutants to the tyrosine kinase inhibitor midostaurin. Blood 2007, 110, 4476–4479. [Google Scholar] [CrossRef]
- Yee, K.W.H.; Schittenhelm, M.; O’Farrell, A.-M.; Town, A.R.; McGreevey, L.; Bainbridge, T.; Cherrington, J.M.; Heinrich, M.C. Synergistic effect of SU11248 with cytarabine or daunorubicin on FLT3 ITD–positive leukemic cells. Blood 2004, 104, 4202–4209. [Google Scholar] [CrossRef] [PubMed]
- Schittenhelm, M.M.; Heinrich, M.C.; Akmut, F.; Döhner, H.; Döhner, K.; Schittenhelm, M.M. Quizartinib (AC220) is a potent second generation class III tyrosine kinase inhibitor that displays a distinct inhibition profile against mutant-FLT3, -PDGFRA and -KIT isoforms. Mol. Cancer 2013, 12, 19. [Google Scholar] [CrossRef] [PubMed]
- Stone, R.M.; Mandrekar, S.J.; Sanford, B.L.; Laumann, K.; Geyer, S.; Bloomfield, C.D.; Thiede, C.; Prior, T.W.; Döhner, K.; Marcucci, G.; et al. Midostaurin plus Chemotherapy for Acute Myeloid Leukemia with a FLT3 Mutation. N. Engl. J. Med. 2017, 377, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Schlenk, R.F.; Weber, D.; Fiedler, W.; Salih, H.R.; Wulf, G.; Salwender, H.; Schroeder, T.; Kindler, T.; Lübbert, M.; Wolf, D.; et al. Midostaurin added to chemotherapy and continued single-agent maintenance therapy in acute myeloid leukemia with FLT3-ITD. Blood 2019, 133, 840–851. [Google Scholar] [CrossRef]
- Perl, A.E.; Martinelli, G.; Cortes, J.E.; Neubauer, A.; Berman, E.; Paolini, S.; Montesinos, P.; Baer, M.R.; Larson, R.A.; Ustun, C.; et al. Gilteritinib or Chemotherapy for Relapsed or Refractory FLT3-Mutated AML. N. Engl. J. Med. 2019, 381, 1728–1740. [Google Scholar] [CrossRef]
- Cortes, J.; Khaled, S.; Martinelli, G.; Perl, A.E.; Ganguly, S.; Russell, N.; Krämer, A.; Dombret, H.; Hogge, D.; Jonas, B.A.; et al. Quizartinib versus salvage chemotherapy in relapsed or refractory FLT3-ITD acute myeloid leukaemia (QuANTUM-R): A multicentre, randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2019, 20, 984–997. [Google Scholar] [CrossRef]
- Altman, J.K.; Foran, J.M.; Pratz, K.W.; Trone, D.; Cortes, J.E.; Tallman, M.S. Phase 1 study of quizartinib in combination with induction and consolidation chemotherapy in patients with newly diagnosed acute myeloid leukemia. Am. J. Hematol. 2017, 93, 213–221. [Google Scholar] [CrossRef]
- Röllig, C.; Serve, H.; Hüttmann, A.; Noppeney, R.; Mueller-Tidow, C.; Krug, U.; Baldus, C.D.; Brandts, C.H.; Kunzmann, V.; Einsele, H.; et al. Addition of sorafenib versus placebo to standard therapy in patients aged 60 years or younger with newly diagnosed acute myeloid leukaemia (SORAML): A multicentre, phase 2, randomised controlled trial. Lancet Oncol. 2015, 16, 1691–1699. [Google Scholar] [CrossRef]
- Uy, G.L.; Mandrekar, S.J.; Laumann, K.; Marcucci, G.; Zhao, W.; Levis, M.J.; Klepin, H.D.; Baer, M.R.; Powell, B.L.; Westervelt, P.; et al. A phase 2 study incorporating sorafenib into the chemotherapy for older adults with FLT3-mutated acute myeloid leukemia: CALGB 11001. Blood Adv. 2017, 1, 331–340. [Google Scholar] [CrossRef]
- Ohanian, M.; Garcia-Manero, G.; Levis, M.J.; Jabbour, E.; Daver, N.; Borthakur, G.; Kadia, T.; Pierce, S.; Burger, J.; Richie, M.A.; et al. Sorafenib Combined with 5-azacytidine in Older Patients with Untreated FLT3-ITD Mutated Acute Myeloid Leukemia. Am. J. Hematol. 2018, 93, 1136–1141. [Google Scholar] [CrossRef]
- Sasaki, K.; Kantarjian, H.M.; Kadia, T.M.; Patel, K.; Loghavi, S.; Garcia-Manero, G.; Jabbour, E.J.; Dinardo, C.D.; Pemmaraju, N.; Daver, N.; et al. Sorafenib plus intensive chemotherapy improves survival in patients with newly diagnosed, FLT3-internal tandem duplication mutation–positive acute myeloid leukemia. Cancer 2019, 125, 3755–3766. [Google Scholar] [CrossRef]
- Muppidi, M.R.; Portwood, S.; Griffiths, E.A.; Thompson, J.E.; Ford, L.A.; Freyer, C.W.; Wetzler, M.; Wang, E.S. Decitabine and Sorafenib Therapy in FLT-3 ITD-Mutant Acute Myeloid Leukemia. Clin. Lymphoma Myeloma Leuk. 2015, 15, S73–S79. [Google Scholar] [CrossRef]
- Ravandi, F.; Alattar, M.L.; Grunwald, M.R.; Rudek, M.A.; Rajkhowa, T.; Richie, M.A.; Pierce, S.; Daver, N.; Garcia-Manero, G.; Faderl, S.; et al. Phase 2 study of azacytidine plus sorafenib in patients with acute myeloid leukemia and FLT-3 internal tandem duplication mutation. Blood 2013, 121, 4655–4662. [Google Scholar] [CrossRef]
- Bazarbachi, A.; Labopin, M.; Battipaglia, G.; Djabali, A.; Passweg, J.; Socié, G.; Forcade, E.; Blaise, D.; Chevallier, P.; Orvain, C.; et al. Sorafenib improves survival of FLT3-mutated acute myeloid leukemia in relapse after allogeneic stem cell transplantation: A report of the EBMT Acute Leukemia Working Party. Haematologica 2019, 104, e398–e401. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.S.; Stone, R.M.; Tallman, M.S.; Walter, R.B.; Eckardt, J.R.; Collins, R. Crenolanib, a Type I FLT3 TKI, Can be Safely Combined with Cytarabine and Anthracycline Induction Chemotherapy and Results in High Response Rates in Patients with Newly Diagnosed FLT3 Mutant Acute Myeloid Leukemia (AML). Blood 2016, 128, 1071. [Google Scholar] [CrossRef]
- Iyer, S.P.; Jethava, Y.; Karanes, C.; Eckardt, J.R.; Collins, R. Safety Study of Salvage Chemotherapy High-Dose Ara-C/Mitoxantrone (HAM) and Type I FLT3-TKI Crenolanib in First Relapsed/Primary Refractory AML. Blood 2016, 128, 3983. [Google Scholar] [CrossRef]
- Ohanian, M.; Kantarjian, H.M.; Borthakur, G.; Kadia, T.M.; Konopleva, M.; Garcia-Manero, G.; Estrov, Z.; Ferrajoli, A.; Takahashi, K.; Jabbour, E.J.; et al. Efficacy of a Type I FLT3 Inhibitor, Crenolanib, with Idarubicin and High-Dose Ara-C in Multiply Relapsed/Refractory FLT3+ AML. Blood 2016, 128, 2744. [Google Scholar] [CrossRef]
- Levis, M.J.; Ravandi, F.; Wang, E.S.; Baer, M.R.; Perl, A.; Coutre, S.; Erba, H.; Stuart, R.K.; Baccarani, M.; Cripe, L.D.; et al. Results from a randomized trial of salvage chemotherapy followed by lestaurtinib for patients with FLT3 mutant AML in first relapse. Blood 2011, 117, 3294–3301. [Google Scholar] [CrossRef]
- Knapper, S.; Russell, N.; Gilkes, A.; Hills, R.K.; Gale, R.E.; Cavenagh, J.D.; Jones, G.; Kjeldsen, L.; Grunwald, M.R.; Thomas, I.; et al. A randomized assessment of adding the kinase inhibitor lestaurtinib to first-line chemotherapy for FLT3-mutated AML. Blood 2017, 129, 1143–1154. [Google Scholar] [CrossRef]
- Sato, T.; Yang, X.; Knapper, S.; White, P.; Smith, B.D.; Galkin, S.; Small, D.; Burnett, A.; Levis, M.J. FLT3 ligand impedes the efficacy of FLT3 inhibitors in vitro and in vivo. Blood 2011, 117, 3286–3293. [Google Scholar] [CrossRef]
- Chen, F.; Ishikawa, Y.; Akashi, A.; Naoe, T.; Kiyoi, H. Co-expression of wild-type FLT3 attenuates the inhibitory effect of FLT3 inhibitor on FLT3 mutated leukemia cells. Oncotarget 2016, 7, 47018–47032. [Google Scholar] [CrossRef] [PubMed]
- Traer, E.; Martinez, J.; Javidi-Sharifi, N.; Agarwal, A.; Dunlap, J.; English, I.; Kovacsovics, T.; Tyner, J.W.; Wong, M.; Druker, B.J. FGF2 from Marrow Microenvironment Promotes Resistance to FLT3 Inhibitors in Acute Myeloid Leukemia. Cancer Res. 2016, 76, 6471–6482. [Google Scholar] [CrossRef] [PubMed]
- Jacobi, A.; Thieme, S.; Lehmann, R.; Ugarte, F.; Malech, H.L.; Koch, S.; Thiede, C.; Müller, K.; Bornhäuser, M.; Ryser, M.; et al. Impact of CXCR4 inhibition on FLT3-ITD-positive human AML blasts. Exp. Hematol. 2009, 38, 180–190. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zeng, Z.; Shi, Y.X.; Samudio, I.J.; Wang, R.-Y.; Ling, X.; Frolova, O.; Levis, M.; Rubin, J.B.; Negrin, R.R.; Estey, E.H.; et al. Targeting the leukemia microenvironment by CXCR4 inhibition overcomes resistance to kinase inhibitors and chemotherapy in AML. Blood 2009, 113, 6215–6224. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-T.; Hernandez, D.; Alonso, S.; Gao, M.; Su, M.; Ghiaur, G.; Levis, M.J.; Jones, R.J. Role of CYP3A4 in bone marrow microenvironment–mediated protection of FLT3/ITD AML from tyrosine kinase inhibitors. Blood Adv. 2019, 3, 908–916. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.C.; Wang, Q.; Chin, C.-S.; Salerno, S.; Damon, L.E.; Levis, M.J.; Perl, A.E.; Travers, K.J.; Wang, S.; Hunt, J.P.; et al. Validation of ITD mutations in FLT3 as a therapeutic target in human acute myeloid leukaemia. Nature 2012, 485, 260–263. [Google Scholar] [CrossRef]
- McMahon, C.M.; Ferng, T.; Canaani, J.; Wang, E.S.; Morrissette, J.J.; Eastburn, D.J.; Pellegrino, M.; Durruthy-Durruthy, R.; Watt, C.D.; Asthana, S.; et al. Clonal Selection with RAS Pathway Activation Mediates Secondary Clinical Resistance to Selective FLT3 Inhibition in Acute Myeloid Leukemia. Cancer Discov. 2019, 9, 1050–1063. [Google Scholar] [CrossRef]
- Piloto, O.; Wright, M.; Brown, P.; Kim, K.-T.; Levis, M.; Small, D. Prolonged exposure to FLT3 inhibitors leads to resistance via activation of parallel signaling pathways. Blood 2007, 109, 1643–1652. [Google Scholar] [CrossRef]
- Park, I.-K.; Mundy-Bosse, B.; Whitman, S.P.; Zhang, X.; Warner, S.L.; Bearss, D.; Blum, W.; Marcucci, G.; Caligiuri, M.A. Receptor tyrosine kinase Axl is required for resistance of leukemic cells to FLT3-targeted therapy in acute myeloid leukemia. Leukemia 2015, 29, 2382–2389. [Google Scholar] [CrossRef]
- Dumas, P.-Y.; Naudin, C.; Martin-Lannerée, S.; Izac, B.; Casetti, L.; Mansier, O.; Rousseau, B.; Artus, A.; Dufossée, M.; Giese, A.; et al. Hematopoietic niche drives FLT3-ITD acute myeloid leukemia resistance to quizartinib via STAT5-and hypoxia-dependent upregulation of AXL. Haematologica 2019, 104, 2017–2027. [Google Scholar] [CrossRef]
- Dinardo, C.D.; Stein, E.M.; De Botton, S.; Roboz, G.J.; Altman, J.K.; Mims, A.S.; Swords, R.; Collins, R.H.; Mannis, G.N.; Pollyea, D.A.; et al. Durable Remissions with Ivosidenib inIDH1-Mutated Relapsed or Refractory AML. N. Engl. J. Med. 2018, 378, 2386–2398. [Google Scholar] [CrossRef] [PubMed]
- Stein, E.M.; Dinardo, C.D.; Fathi, A.T.; Pollyea, D.A.; Stone, R.M.; Altman, J.K.; Roboz, G.J.; Patel, M.R.; Collins, R.; Flinn, I.W.; et al. Molecular remission and response patterns in patients with mutant-IDH2 acute myeloid leukemia treated with enasidenib. Blood 2019, 133, 676–687. [Google Scholar] [CrossRef] [PubMed]
- Stein, E.M.; Dinardo, C.D.; Pollyea, D.A.; Fathi, A.T.; Roboz, G.J.; Altman, J.K.; Stone, R.M.; DeAngelo, D.J.; Levine, R.L.; Flinn, I.W.; et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood 2017, 130, 722–731. [Google Scholar] [CrossRef] [PubMed]
- Lindblad, O.; Cordero, E.; Puissant, A.; Macaulay, L.; Ramos, A.; Kabir, N.N.; Sun, J.; Vallon-Christersson, J.; Haraldsson, K.; Hemann, M.T.; et al. Aberrant activation of the PI3K/mTOR pathway promotes resistance to sorafenib in AML. Oncogene 2016, 35, 5119–5131. [Google Scholar] [CrossRef]
- Tursynbay, Y.; Zhang, J.; Li, Z.; Tokay, T.; Zhumadilov, Z.; Wu, D.; Xie, Y. Pim-1 kinase as cancer drug target: An update. Biomed. Rep. 2015, 4, 140–146. [Google Scholar] [CrossRef]
- Kim, K.-T.; Baird, K.; Ahn, J.-Y.; Meltzer, P.; Lilly, M.; Levis, M.J.; Small, N. Pim-1 is up-regulated by constitutively activated FLT3 and plays a role in FLT3-mediated cell survival. Blood 2005, 105, 1759–1767. [Google Scholar] [CrossRef]
- Green, A.S.; Maciel, T.; Hospital, M.-A.; Yin, C.; Mazed, F.; Townsend, E.C.; Pilorge, S.; Lambert, M.; Paubelle, E.; Jacquel, A.; et al. Pim kinases modulate resistance to FLT3 tyrosine kinase inhibitors in FLT3-ITD acute myeloid leukemia. Sci. Adv. 2015, 1, e1500221. [Google Scholar] [CrossRef]
- Kohl, T.M.; Hellinger, C.; Ahmed, F.; Buske, C.; Hiddemann, W.; Bohlander, S.K.; Spiekermann, K. BH3 mimetic ABT-737 neutralizes resistance to FLT3 inhibitor treatment mediated by FLT3-independent expression of BCL2 in primary AML blasts. Leukemia 2007, 21, 1763–1772. [Google Scholar] [CrossRef]
- Breitenbuecher, F.; Markova, B.; Kasper, S.; Carius, B.; Stauder, T.; Böhmer, F.D.; Masson, K.; Rönnstrand, L.; Huber, C.; Kindler, T.; et al. A novel molecular mechanism of primary resistance to FLT3-kinase inhibitors in AML. Blood 2009, 113, 4063–4073. [Google Scholar] [CrossRef]
- Yoshimoto, G.; Miyamoto, T.; Jabbarzadeh-Tabrizi, S.; Iino, T.; Rocnik, J.L.; Kikushige, Y.; Mori, Y.; Shima, T.; Iwasaki, H.; Takenaka, K.; et al. FLT3-ITD up-regulates MCL-1 to promote survival of stem cells in acute myeloid leukemia via FLT3-ITD–specific STAT5 activation. Blood 2009, 114, 5034–5043. [Google Scholar] [CrossRef]
- Yamaura, T.; Nakatani, T.; Uda, K.; Ogura, H.; Shin, W.; Kurokawa, N.; Saito, K.; Fujikawa, N.; Date, T.; Takasaki, M.; et al. A novel irreversible FLT3 inhibitor, FF-10101, shows excellent efficacy against AML cells with FLT3 mutations. Blood 2018, 131, 426–438. [Google Scholar] [CrossRef] [PubMed]
- Czardybon, W.; Windak, R.; Gołas, A.; Gałezowski, M.; Sabiniarz, A.; Dolata, I.; Salwińska, M.; Guzik, P.; Zawadzka, M.; Gabor-Worwa, E.; et al. A novel, dual pan-PIM/FLT3 inhibitor SEL24 exhibits broad therapeutic potential in acute myeloid leukemia. Oncotarget 2018, 9, 16917–16931. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Zhao, Y.; Wang, X.; Gong, P.; Ge, W. MZH29 is a novel potent inhibitor that overcomes drug resistance FLT3 mutations in acute myeloid leukemia. Leukemia 2016, 31, 913–921. [Google Scholar] [CrossRef] [PubMed]
- Minson, K.A.; Smith, C.C.; DeRyckere, D.; Libbrecht, C.; Sherick, A.L.; Huey, M.G.; Lasater, E.A.; Kirkpatrick, G.D.; Stashko, M.A.; Zhang, W.; et al. The MERTK/FLT3 inhibitor MRX-2843 overcomes resistance-conferring FLT3 mutations in acute myeloid leukemia. JCI Insight 2016, 1. [Google Scholar] [CrossRef]
- Zirm, E.; Spies-Weisshart, B.; Heidel, F.H.; Schnetzke, U.; Böhmer, F.-D.; Hochhaus, A.; Fischer, T.; Scholl, S. Ponatinib may overcome resistance of FLT3-ITD harbouring additional point mutations, notably the previously refractory F691I mutation. Br. J. Haematol. 2012, 157, 483–492. [Google Scholar] [CrossRef]
- Sung, L.; Blonquist, T.M.; Hernandez, D.; Amrein, P.C.; Ballen, K.K.; McMasters, M.; Avigan, D.; Joyce, R.; Logan, E.K.; Hobbs, G.S.; et al. Cabozantinib is well tolerated in acute myeloid leukemia and effectively inhibits the resistance-conferring FLT3/tyrosine kinase domain/F691 mutation. Cancer 2017, 124, 306–314. [Google Scholar] [CrossRef]
- Wang, A.; Wu, H.; Chen, C.; Hu, C.; Qi, Z.; Wang, W.; Yu, K.; Liu, X.; Zou, F.; Zhao, Z.; et al. Dual inhibition of AKT/FLT3-ITD by A674563 overcomes FLT3 ligand-induced drug resistance in FLT3-ITD positive AML. Oncotarget 2016, 7, 29131–29142. [Google Scholar] [CrossRef]
- Zhang, W.; Borthakur, G.; Gao, C.; Chen, Y.; Mu, H.; Ruvolo, V.R.; Nomoto, K.; Zhao, N.; Konopleva, M.; Andreeff, M. The Dual MEK/FLT3 Inhibitor E6201 Exerts Cytotoxic Activity against Acute Myeloid Leukemia Cells Harboring Resistance-Conferring FLT3 Mutations. Cancer Res. 2016, 76, 1528–1537. [Google Scholar] [CrossRef]
- Larrue, C.; Saland, E.; Boutzen, H.; Vergez, F.; David, M.; Joffre, C.; Hospital, M.-A.; Tamburini, J.; Delabesse, E.; Manenti, S.; et al. Proteasome inhibitors induce FLT3-ITD degradation through autophagy in AML cells. Blood 2016, 127, 882–892. [Google Scholar] [CrossRef]
- Nagai, K.; Hou, L.; Li, L.; Nguyen, B.; Seale, T.; Shirley, C.; Ma, H.; Levis, M.; Ghiaur, G.; Duffield, A.; et al. Combination of ATO with FLT3 TKIs eliminates FLT3/ITD+ leukemia cells through reduced expression of FLT3. Oncotarget 2018, 9, 32885–32899. [Google Scholar] [CrossRef]
- Uras, I.Z.; Walter, G.J.; Scheicher, R.; Bellutti, F.; Prchal-Murphy, M.; Tigan, A.S.; Valent, P.; Heidel, F.H.; Kubicek, S.; Scholl, C.; et al. Palbociclib treatment of FLT3-ITD+ AML cells uncovers a kinase-dependent transcriptional regulation of FLT3 and PIM1 by CDK6. Blood 2016, 127, 2890–2902. [Google Scholar] [CrossRef]
- Gregory, M.; D’Alessandro, A.; Alvarez-Calderon, F.; Kim, J.; Nemkov, T.; Adane, B.; Rozhok, A.I.; Kumar, A.; Kumar, V.; Pollyea, D.A.; et al. ATM/G6PD-driven redox metabolism promotes FLT3 inhibitor resistance in acute myeloid leukemia. Proc. Natl. Acad. Sci. USA 2016, 113, E6669–E6678. [Google Scholar] [CrossRef]
- Dickson, G.J.; Bustraan, S.; Hills, R.K.; Ali, A.; Goldstone, A.H.; Burnett, A.K.; Linch, D.C.; Gale, R.E. The value of molecular stratification for CEBPA DM and NPM1 MUT FLT3 WT genotypes in older patients with acute myeloid leukaemia. Br. J. Haematol. 2015, 172, 573–580. [Google Scholar] [CrossRef]
- Rothenberg-Thurley, M.; Amler, S.; Goerlich, D.; Kohnke, T.; Konstandin, N.P.; Schneider, S.; Sauerland, M.C.; Herold, T.; Hubmann, M.; Ksienzyk, B.; et al. Persistence of pre-leukemic clones during first remission and risk of relapse in acute myeloid leukemia. Leukemia 2018, 32, 1598–1608. [Google Scholar] [CrossRef] [PubMed]
- Canaani, J.; Labopin, M.; Huang, X.-J.; Arcese, W.; Ciceri, F.; Blaise, D.; Irrera, G.; Corral, L.L.; Bruno, B.; Santarone, S.; et al. T-cell replete haploidentical stem cell transplantation attenuates the prognostic impact of FLT3-ITD in acute myeloid leukemia: A report from the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation. Am. J. Hematol. 2018, 93, 736–744. [Google Scholar] [CrossRef]
- Hou, P.; Wu, C.; Wang, Y.; Qi, R.; Bhavanasi, D.; Zuo, Z.; Dos Santos, C.; Chen, S.; Chen, Y.; Zheng, H.; et al. A Genome-Wide CRISPR Screen Identifies Genes Critical for Resistance to FLT3 Inhibitor AC220. Cancer Res. 2017, 77, 4402–4413. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, K.; Minami, Y. Precision medicine and novel molecular target therapies in acute myeloid leukemia: The background of hematologic malignancies (HM)-SCREEN-Japan 01. Int. J. Clin. Oncol. 2019, 24, 893–898. [Google Scholar] [CrossRef] [PubMed]
| Agent | Generation | Type | Selectivity | IC50 (nM) | Drug Sensitivity | ||||
|---|---|---|---|---|---|---|---|---|---|
| ITD | D835Y | ITD-D835Y | F691L | ||||||
| Midostaurin | (PKC412) | First | I | Low | 139 | S | S | R | R |
| Sunitinib | (SU11248) | First | I | Low | 250 | S | R | R | R |
| Lestaurtinib | (CEP701) | First | I | Low | 5 | S | Int | S | − |
| Gilteritinib | (ASP2215) | Second | I | Moderate | 1.6 | S | S | Int | R |
| Crenolanib | (CP868596) | Second | I | Moderate | 2 | S | S | Int | R |
| Sorafenib | (BAY43-9006) | First | II | Moderate | 58 | S | R | R | R |
| Tandutinib | (CT53518) | First | II | High | 100 | S | R | − | − |
| Quizartinib | (AC220) | Second | II | High | <1.0 | S | R | R | R |
| SORAFENIB (BAY 43-9006) | ||||||||||
| Authors and Jounals | Trial Name | Objectives | Disease Status | Agents * Controls Not Shown | Phase/Design | Response Rate | Median PFS | Median OS | Sequential allo-SCT | |
| Rolling, et al. Lancet Oncol 2015 | SORAML | AML (age < 60) | Newly diagnosed | Sorafenib + Standard therapy | II | CR | 60% (81/134) | 21 mo. [9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32] | Not Reached (3-yr OS 63%) | 31% (42/132) |
| Uy, et al. Blood Advances 2016 | CALGB 11001 | AML (age > 60) with FLT3-ITD and/or TKD | Newly diagnosed | Sorafenib + Standard therapy | II | CR | 74% (40/54) | 8.8 mo. (FLT3-ITD) 7.8 mo. (FLT3-TKD) | 15.0 mo. (FLT3-ITD) 16.2 mo. (FLT3-TKD) | 53% (22/54) |
| Ohanian, et al. Am J Hematol 2018 | − | AML (age > 60) with FLT3-ITD | Newly diagnosed | Sorafenib + Azacitidine | I/II | CR/Cri PR | 70% (19/27) 7% (2/27) | 7.1 mo. (only in responders) | 8.3 mo. (in all participants) | 11% (3/27) |
| Sasaki, et al. Cancer 2019 | − | AML with FLT3-ITD | Newly diagnosed | Soragenib + Standard therapy | Retrospective | CR/CRi | 99% (78/79) | 31 mo. [5.7–56.8] | 17 mo. [11.1–22.4] | 67% (53/79) |
| Muppidi, et al. Clinical Lymphoma Myeloma and Leukemia 2015 | − | AML with FLT3-ITD | Newly diagnosed or relapsed | Sorafenib + Decitabine | Case Series | CR/CRi | 83% (5/6) | Not Reported | 5.1 mo. [1.9–14.5] | 33% (2/6) |
| Ravandi, et al. Blood 2013 | − | AML with FLT3-ITD | Relapsed or refractory (including prior allo-SCT) | Sorafenib + Azacitidine | II | CR/Cri PR | 43% (16/37) 3% (1/37) | 3.8 mo. [1.0–16.4] | 6.2 mo. | 16% (6/37) |
| Bazarbachi, et al. Heamatologica 2019 | − | AML with FLT3-ITD | Relapsed ater allo-SCT | Sorafenib as part of/after salvage | Retrospective | CR | 39% (10/26) | Not Reported | (2-yr. OS 38%) | 13% (3/26) |
| MIDOSTAURIN (PKC412) | ||||||||||
| Authorsand Jounals | Trial Name | Objectives | Disease Status | Agents * Controls Not Shown | Phase/Design | Response Rate | Median PFS | Median OS | Sequential allo-SCT | |
| Stone, et al. N Engl J Med 2017 | RATIFY | AML with FLT3-ITD and/or TKD | Newly diagnosed | Midostaurin + Standard induction/consolidation | III | CR | 70% (504/717) | 8.2 mo. [5.4–10.7] | 74.7 mo. [31.5–inf.] | 57% (287/504) |
| Schlenk, et al. Blood 2019 | AMLSG 16-10 | AML with FLT3-ITD and/or TKD | Newly diagnosed | Midostaurin + Standard induction/consolidation f/b Midostaurin maitenance | II | CR/CRi | 76% (217/292) | 13.2 mo. [10.0–18.3] | 26.0 mo. [18.9–37.0] | 62% (134/217) |
| Fischer, et al. J Clin Oncol 2010 | − | AML or high-risk MDS | Relapsed or refractory or ineligible to standard therapy | Midostaurin | IIB | PR HI Blast | 1% (1/97) 46% (16/35 *) 71% (25/35 *) * only in FLT3-mt | Not Reported | 4.3 mo. [3.5–5.2] | 31% (42/132) |
| Strati, et al. Am J Hematol 2015 | − | AML or high-risk MDS | Relapsed or refractory or ineligible to standard therapy | Midostaurin + Azacitidine | I/II | CR/Cri PR/MLFS | 15% (8/54) 13% (7/54) | 4.6 mo. [2.3–6.9] * Duration of Response | 5.1 mo. [3.5–6.7] | 0% (0/8) |
| Walker, et al. Leukemia & Lymphoma 2016 | − | AML | Relapsed or refractory (including prior allo-SCT) | Midostaurin + Bortezomib + Chemotherapy(MEC) | I | CR/CRi | 83% (19/23) | Not Reported | 10.8 mo. | 63% (12/19) |
| Maziarz, et al. Blood 2018 | RADIUS | AML with FLT3-ITD | in 1st CR after allo-SCT | Midostaurin + Standard of care | II | Not Applicable | (18mo.-PFS 89%) | Not Reported | Not Applicable | |
| GILTERITINIB (ASP2215) | ||||||||||
| Authors and Jounals | Trial Name | Objectives | Disease Status | Agents * Controls Not Shown | Phase/Design | Response Rate | Median PFS | Median OS | Sequential allo-SCT | |
| Perl, et al. N Engl J Med 2019 | ADMIRAL | AML with FLT3-ITD and/or TKD | Relapsed or refractory | Gilteritinib | III | CR/Cri PR | 54% (134/247) 13% (33/247) | 2.8 mo. [1.4–3.7] | 9.3 mo. [7.7–10.7] | 26% (63/247) |
| Perl, et al. Lancet Oncol 2017 | − | AML with FLT3-ITD and/or TKD | Relapsed or refractory | Gilteritinib | I/II | CR/Cri PR | 41% (69/169) 11% (19/169) | 4.6 mo. * Duration of Response | 7.1 mo. | 22% (37/169) |
| Usuki, et al. Cancer Science 2018 | − | AML | Relapsed or refractory | Gilteritinib | I | CR/Cri PR | 60% (3/5 *) 20% (1/5 *) * only in FLT3-mt. | Not Reported | Not Reported | Not Reported |
| QUIZARTINIB (AC220) | ||||||||||
| Authors and Jounals | Trial Name | Objectives | Disease Status | Agents * Controls Not Shown | Phase/Design | Response Rate | Median PFS | Median OS | Sequential allo-SCT | |
| Altman, et al. Blood 2018 | − | AML | Newly diagnosed | Quizartinib + Standard induction/consolidation f/b Quizartinib maitenance | I | CR/CRi | 74% (14/19) | (Maximum 16.3 mo.) | Not Reported | 47% (9/19) |
| Cortes, et al. Blood 2019 | QuANTUM-R | AML with FLT3-ITD | Relapsed or refractory | Quizartinib | III | CR/Cri | 48% (118/245) | 1.4 mo. [0.0–1.9] | 6.2 mo. [5.3–7.2] | 32% (78/245) |
| Cortes, et al. Blood 2018 | − | AML with FLT3-ITD | Relapsed or refractory | Quizartinib | IIB | CR/Cri PR | 47% (36/76) 18% (14/76) | 12.3 mo. [9.7–16.1] | 22.6 mo. [19.9–28.3] | 37% (28/76) |
| Cortes, et al. Lancet Oncol 2018 | − | AML | Relapsed or refractory | Quizartinib | II | CR/Cri PR | 50% (125/248) 25% (62/248) * only in ITD-mt. | 2.8 mo. [1.4–3.6] * duration of CR, only in ITD-mt. | 5.8 mo. [4.9–6.8] * only in ITD-mt. | 35% (61/176) |
| Sandmaier, et al. Am J Hematol 2017 | − | AML with FLT3-ITD | in 1st CR after allo-SCT | Quizartinib maintenance | I | Not Applicable | (0.4–22.8 mo.) * duration of maitenance | (3.0–32.7 mo.) | Not Applicable | |
| CRENOLANIB (CP868596) | ||||||||||
| Authors and Jounals | Trial Name | Objectives | Disease Status | Agents * Controls Not Shown | Phase/Design | Response Rate | Median PFS | Median OS | Sequential allo-SCT | |
| Wang, et al. Blood 2016 | − | AML with FLT3-ITD and/or TKD | Newly diagnosed | Crenolanib + Standard induction/consolidation f/b Crenolanib maintenance | II | CR/CRi | 96% (24/25) | Not Reported | Not Reached (6 mo. OS 85%) | 50% (12/24) |
| Randhawa, et al. Blood 2014 | − | AML with FLT3-ITD and/or TKD | Relapsed or refractory | Crenolanib | II | CR/Cri MLFS HI | 23% (3/13) 8% (1/13) 31% (4/13) * only in TKI-naïve | 3.0 mo. * only in TKI-naïve | 12.7 mo. * only in TKI-naïve | 26% (9/34) |
| Ohanian, et al. Blood 2016 | − | AML with FLT3-ITD and/or TKD | Relapsed or refractory | Crenolanib + Salvage chemotherapy (IDA/AraC) | I | CR/CRi | 36% (4/11) | Not Reported | 8.5 mo. | 75% (3/4) |
| Iyer, et al. Blood 2016 | − | AML | Relapsed or refractory | Crenolanib + Chemotherapy (HAM) | I | CR/CRi | 67% (4/6) | Not Reported | Not Repoted | 25% (1/4) |
| LESTAURTINIB (CEP701) | ||||||||||
| Authors and Jounals | Trial Name | Objectives | Disease Status | Agents * Controls Not Shown | Phase/Design | Response Rate | Median PFS | Median OS | Sequential allo-SCT | |
| Levis, et al. Blood 2017 | − | AML with FLT3-ITD and/or TKD | Relapsed | Slavage chemotherapy (MEC) f/b Lestaurtinib maintenance | II | CR/CRi | 26% (29/112) | Not Reported | 5.2 mo. | 20% (22/112) |
| Knapper, et al. Blood 2017 | − | AML with FLT3-ITD and/or TKD | Newly diagnosed | Standard induction/consolidation f/b Lestaurtinib maintenance | III | CR/CRi | 92% (277/300) | (5-yr. PFS 39–40%) | (5-yr. OS 43–50%) | 21% (58/277) |
| SORAFENIB (BAY 43-9006) | ||||
| Trial Number | Objectives | Disease Status | Agents * Controls Not Shown | Phase/Design |
| NCT01371981 | AML with FLT3-ITD (high allelic ratio) | Newly diagnosed | Sorafenib + Bertezomib | III |
| NCT03170895 | AML with FLT3-ITD | Newly diagnosed or relapsed | Sorafenib + Homoharringtonine (STAT inhibitor) | II |
| MIDOSTAURIN (PKC412) | ||||
| NCT03686345 | Core binding factor AML | Newly diagnosed | Midostaurin + Standard induction | II |
| NCT03280030 | AML with FLT3-ITD and/or TKD | Newly diagnosed | Midostaurin + Standard induction/consolidation f/b Midostaurin maitenance | II |
| NCT03512197 | AML with FLT3-ITD and/or TKD | Newly diagnosed | Midostaurin + Standard induction/consolidation f/b Midostaurin maitenance | III |
| NCT03379727 | AML with FLT3-ITD and/or TKD | Newly diagnosed | Midostaurin + Standard induction/consolidation f/b Midostaurin maitenance | III |
| GILTERITINIB (ASP2215) | ||||
| NCT02236013 | AML | Newly diagnosed | Gilteritinib + Standard induction/consolidation | I |
| NCT02752035 | AML with FLT3-ITD and/or TKD | Newly diagnosed and ineligible to standard therapy | Gilteritinib + Azacitidine | III |
| NCT03730012 | AML with FLT3-ITD and/or TKD | Relapsed or refractory | Gilteritinib + Atezolizumab (anti-PD-L1 antibody) | I/II |
| NCT02310321 | AML | Newly diagnosed | Gilteritinib + Standard induction/consolidation | I/II |
| NCT03182244 | AML with FLT3-ITD and/or TKD | Relapsed or refractory | Gilteritinib + Salvage chemotherapy | III |
| QUIZARTINIB (AC220) | ||||
| NCT02668653 | AML with FLT3-ITD | Newly diagnosed | Quizartinib + Standard induction/consolidation | III |
| NCT03723681 | AML | Newly diagnosed | Quizartinib + Standard induction/consolidation | I |
| NCT02834390 | AML | Newly diagnosed | Quizartinib + Standard induction/consolidation | IB |
| NCT03552029 | AML with FLT3-ITD | Relapsed or refractory or ineligible to standard therapy | Quizartinib + Milademetan (MDM2 inhibitor) | I |
| NCT03135054 | AML with FLT3-ITD | Newly diagnosed or relapsed | Quizartinib + Homoharringtonine (STAT inihibitor) | II |
| NCT03661307 | AML with FLT3-ITD | Newly diagnosed or relapsed | Quizartinib + Decitabine + Venetoclax (BCL-2 inhibitor) | I/II |
| NCT03735875 | AML with FLT3-ITD | Relapsed or refractory | Quizartinib + Venetoclax (BCL-2 inhibitor) | IB/II |
| CRENOLANIB (CP868596) | ||||
| NCT03258931 | AML with FLT3-ITD and/or TKD | Newly diagnosed | Crenolanib + Standard induction/consolidation | III |
| NCT02283177 | AML with FLT3-ITD and/or TKD | Newly diagnosed | Crenolanib + Standard induction | II |
| NCT03250338 | AML with FLT3-ITD and/or TKD | Relapsed or refractory | Crenolanib + Salvage chemotherapy | III |
| NCT02400281 | AML with FLT3-ITD and/or TKD | Relapsed or refractory | Crenolanib + Salvage chemo. or Azacitidine | I/II |
| NCT02626338 | AML | Relapsed or refractory | Crenolanib + Salvage chemotherapy | I/II |
| NCT01522469 | AML with FLT3-ITD and/or TKD | Relapsed or refractory | Crenolanib + Standard induction/consolidation | IB |
| OTHERS | ||||
| NCT00783653 | AML with FLT3-ITD and/or TKD | Newly diagnosed | Sunitinib + Standard induction | I/II |
| NCT00469859 | AML with FLT3-ITD and/or TKD | Relapsed or refractory | Lestaurtinib + Salvage chemotherapy | I/II |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eguchi, M.; Minami, Y.; Kuzume, A.; Chi, S. Mechanisms Underlying Resistance to FLT3 Inhibitors in Acute Myeloid Leukemia. Biomedicines 2020, 8, 245. https://doi.org/10.3390/biomedicines8080245
Eguchi M, Minami Y, Kuzume A, Chi S. Mechanisms Underlying Resistance to FLT3 Inhibitors in Acute Myeloid Leukemia. Biomedicines. 2020; 8(8):245. https://doi.org/10.3390/biomedicines8080245
Chicago/Turabian StyleEguchi, Motoki, Yosuke Minami, Ayumi Kuzume, and SungGi Chi. 2020. "Mechanisms Underlying Resistance to FLT3 Inhibitors in Acute Myeloid Leukemia" Biomedicines 8, no. 8: 245. https://doi.org/10.3390/biomedicines8080245
APA StyleEguchi, M., Minami, Y., Kuzume, A., & Chi, S. (2020). Mechanisms Underlying Resistance to FLT3 Inhibitors in Acute Myeloid Leukemia. Biomedicines, 8(8), 245. https://doi.org/10.3390/biomedicines8080245





