Advanced Glycation End Products (AGE) and Soluble Forms of AGE Receptor: Emerging Role as Mortality Risk Factors in CKD
Abstract
1. Introduction
2. Experimental Section
2.1. Patients and Study Design
2.2. Malnutrition Inflammation Score
2.3. sRAGE, esRAGE and cRAGE Quantification
2.4. Glycated Albumin Quantification
2.5. AGE Quantification
2.6. Statistical Analysis
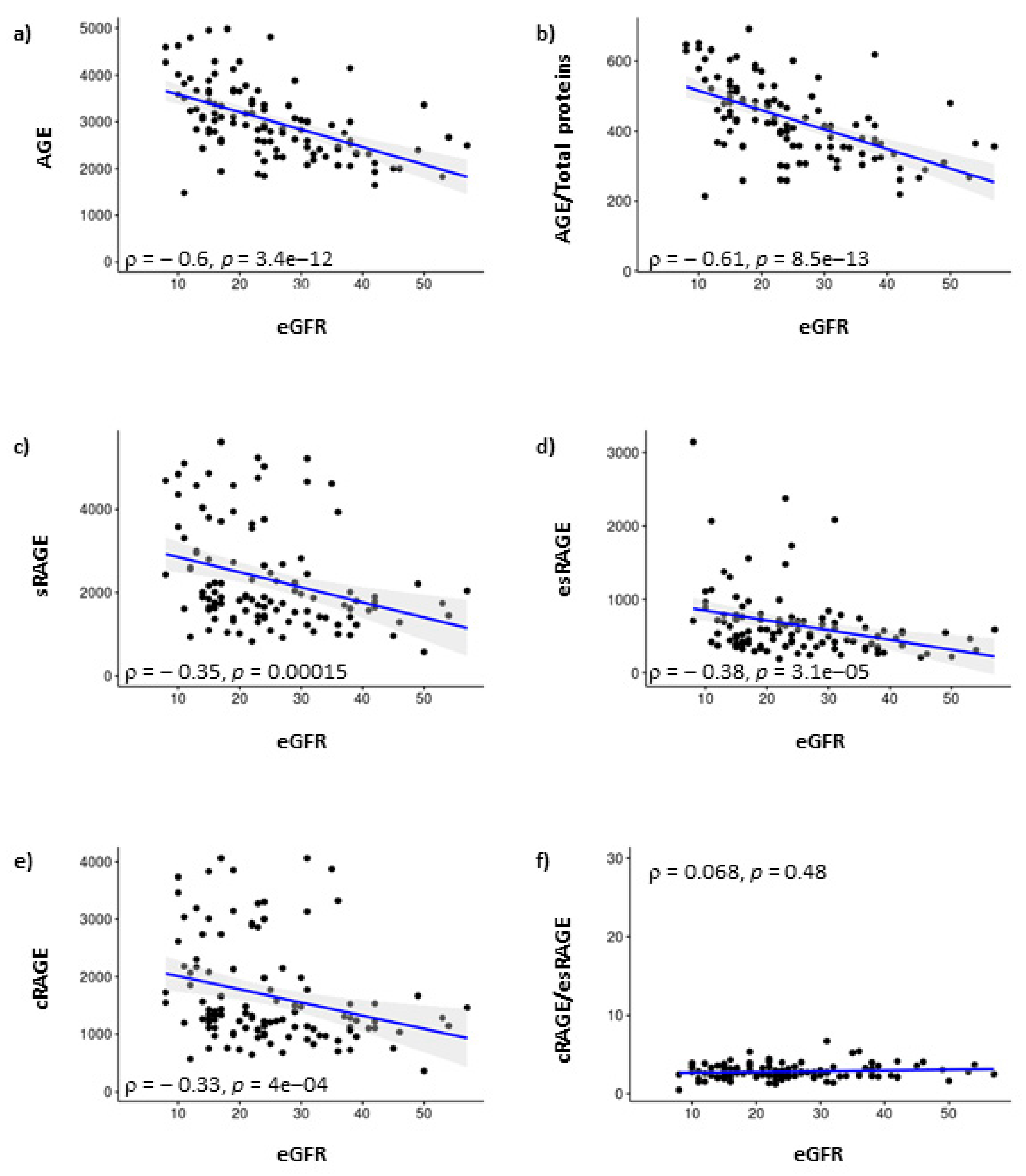
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Paoletti, E.; De Nicola, L.; Gabbai, F.B.; Chiodini, P.; Ravera, M.; Pieracci, L.; Marre, S.; Cassottana, P.; Luca, S.; Vettoretti, S.; et al. Associations of Left Ventricular Hypertrophy and Geometry with Adverse Outcomes in Patients with CKD and Hypertension. Clin. J. Am. Soc. Nephrol. 2016, 11, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Kooman, J.P.; Dekker, M.J.; Usvyat, L.A.; Kotanko, P.; van der Sande, F.M.; Schalkwijk, C.G.; Shiels, P.G.; Stenvinkel, P. Inflammation and premature aging in advanced chronic kidney disease. Am. J. Physiol. Renal Physiol. 2017, 313, F938–F950. [Google Scholar] [CrossRef] [PubMed]
- Guenzani, D.; Buoli, M.; Caldiroli, L.; Carnevali, G.S.; Serati, M.; Vezza, C.; Armelloni, S.; Messa, P.; Vettoretti, S.; Group, D.P. Malnutrition and inflammation are associated with severity of depressive and cognitive symptoms of old patients affected by chronic kidney disease. J. Psychosom. Res. 2019, 124, 109783. [Google Scholar] [CrossRef] [PubMed]
- Vettoretti, S.; Caldiroli, L.; Armelloni, S.; Ferrari, C.; Cesari, M.; Messa, P. Sarcopenia is Associated with Malnutrition but Not with Systemic Inflammation in Older Persons with Advanced CKD. Nutrients 2019, 11, 1378. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N. Advanced glycation endproducts–role in pathology of diabetic complications. Diabetes Res. Clin. Pract. 2005, 67, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Goldin, A.; Beckman, J.A.; Schmidt, A.M.; Creager, M.A. Advanced glycation end products: Sparking the development of diabetic vascular injury. Circulation 2006, 114, 597–605. [Google Scholar] [CrossRef]
- Tan, A.L.; Forbes, J.M.; Cooper, M.E. AGE, RAGE, and ROS in diabetic nephropathy. Semin. Nephrol. 2007, 27, 130–143. [Google Scholar] [CrossRef]
- Vlassara, H.; Striker, L.J.; Teichberg, S.; Fuh, H.; Li, Y.M.; Steffes, M. Advanced glycation end products induce glomerular sclerosis and albuminuria in normal rats. Proc. Natl. Acad. Sci. USA 1994, 91, 11704–11708. [Google Scholar] [CrossRef]
- Rojas, A.; Gonzalez, I.; Morales, M.A. SARS-CoV-2-mediated inflammatory response in lungs: Should we look at RAGE? Inflamm. Res. 2020, 69, 641–643. [Google Scholar] [CrossRef]
- Bucala, R.; Vlassara, H. Advanced glycosylation end products in diabetic renal and vascular disease. Am. J. Kidney Dis. 1995, 26, 875–888. [Google Scholar] [CrossRef]
- Shimoike, T.; Inoguchi, T.; Umeda, F.; Nawata, H.; Kawano, K.; Ochi, H. The meaning of serum levels of advanced glycosylation end products in diabetic nephropathy. Metabolism 2000, 49, 1030–1035. [Google Scholar] [CrossRef] [PubMed]
- Calvino, J.; Cigarran, S.; Gonzalez-Tabares, L.; Menendez, N.; Latorre, J.; Cillero, S.; Millan, B.; Cobelo, C.; Sanjurjo-Amado, A.; Quispe, J.; et al. Advanced glycation end products (AGEs) estimated by skin autofluorescence are related with cardiovascular risk in renal transplant. PLoS ONE 2018, 13, e0201118. [Google Scholar] [CrossRef] [PubMed]
- Tezuka, Y.; Nakaya, I.; Nakayama, K.; Nakayama, M.; Yahata, M.; Soma, J. Methylglyoxal as a prognostic factor in patients with chronic kidney disease. Nephrology 2019, 24, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Lazo, M.; Halushka, M.K.; Shen, L.; Maruthur, N.; Rebholz, C.M.; Rawlings, A.M.; Hoogeveen, R.C.; Brinkley, T.E.; Ballantyne, C.M.; Astor, B.C.; et al. Soluble receptor for advanced glycation end products and the risk for incident heart failure: The Atherosclerosis Risk in Communities Study. Am. Heart J. 2015, 170, 961–967. [Google Scholar] [CrossRef] [PubMed]
- Dozio, E.; Ambrogi, F.; de Cal, M.; Vianello, E.; Ronco, C.; Corsi Romanelli, M.M. Role of the Soluble Receptor for Advanced Glycation End Products (sRAGE) as a Prognostic Factor for Mortality in Hemodialysis and Peritoneal Dialysis Patients. Mediat. Inflamm. 2018, 2018, 1347432. [Google Scholar] [CrossRef]
- Rebholz, C.M.; Astor, B.C.; Grams, M.E.; Halushka, M.K.; Lazo, M.; Hoogeveen, R.C.; Ballantyne, C.M.; Coresh, J.; Selvin, E. Association of plasma levels of soluble receptor for advanced glycation end products and risk of kidney disease: The Atherosclerosis Risk in Communities study. Nephrol. Dial. Transplant. 2015, 30, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Dozio, E.; Corradi, V.; Vianello, E.; Scalzotto, E.; de Cal, M.; Corsi Romanelli, M.M.; Ronco, C. Increased Levels of sRAGE in Diabetic CKD-G5D Patients: A Potential Protective Mechanism against AGE-Related Upregulation of Fibroblast Growth Factor 23 and Inflammation. Mediat. Inflamm. 2017, 2017, 9845175. [Google Scholar] [CrossRef]
- Dozio, E.; Vianello, E.; Bandera, F.; Longhi, E.; Brizzola, S.; Nebuloni, M.; Corsi Romanelli, M.M. Soluble Receptor for Advanced Glycation End Products: A Protective Molecule against Intramyocardial Lipid Accumulation in Obese Zucker Rats? Mediat. Inflamm. 2019, 2019, 2712376. [Google Scholar] [CrossRef]
- Schlueter, C.; Hauke, S.; Flohr, A.M.; Rogalla, P.; Bullerdiek, J. Tissue-specific expression patterns of the RAGE receptor and its soluble forms—A result of regulated alternative splicing? Biochim. Biophys. Acta 2003, 1630, 1–6. [Google Scholar] [CrossRef]
- Raucci, A.; Cugusi, S.; Antonelli, A.; Barabino, S.M.; Monti, L.; Bierhaus, A.; Reiss, K.; Saftig, P.; Bianchi, M.E. A soluble form of the receptor for advanced glycation endproducts (RAGE) is produced by proteolytic cleavage of the membrane-bound form by the sheddase a disintegrin and metalloprotease 10 (ADAM10). FASEB J. 2008, 22, 3716–3727. [Google Scholar] [CrossRef]
- Zhao, D.; Wang, Y.; Xu, Y. Decreased serum endogenous secretory receptor for advanced glycation endproducts and increased cleaved receptor for advanced glycation endproducts levels in patients with atrial fibrillation. Int. J. Cardiol. 2012, 158, 471–472. [Google Scholar] [CrossRef] [PubMed]
- Dozio, E.; Corradi, V.; Proglio, M.; Vianello, E.; Menicanti, L.; Rigolini, R.; Caprara, C.; de Cal, M.; Corsi Romanelli, M.M.; Ronco, C. Usefulness of glycated albumin as a biomarker for glucose control and prognostic factor in chronic kidney disease patients on dialysis (CKD-G5D). Diabetes Res. Clin. Pract. 2018, 140, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Kalantar-Zadeh, K.; Kopple, J.D.; Block, G.; Humphreys, M.H. A malnutrition-inflammation score is correlated with morbidity and mortality in maintenance hemodialysis patients. Am. J. Kidney Dis. 2001, 38, 1251–1263. [Google Scholar] [CrossRef] [PubMed]
- Afsar, B.; Sezer, S.; Ozdemir, F.N.; Celik, H.; Elsurer, R.; Haberal, M. Malnutrition-inflammation score is a useful tool in peritoneal dialysis patients. Perit. Dial. Int. 2006, 26, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Yanagisawa, K.; Makita, Z.; Shiroshita, K.; Ueda, T.; Fusegawa, T.; Kuwajima, S.; Takeuchi, M.; Koike, T. Specific fluorescence assay for advanced glycation end products in blood and urine of diabetic patients. Metabolism 1998, 47, 1348–1353. [Google Scholar] [CrossRef]
- Guerin-Dubourg, A.; Cournot, M.; Planesse, C.; Debussche, X.; Meilhac, O.; Rondeau, P.; Bourdon, E. Association between Fluorescent Advanced Glycation End-Products and Vascular Complications in Type 2 Diabetic Patients. Biomed. Res. Int. 2017, 2017, 7989180. [Google Scholar] [CrossRef]
- Collaboration, G.B.D.C.K.D. Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef]
- Kim, J.K.; Park, S.; Lee, M.J.; Song, Y.R.; Han, S.H.; Kim, S.G.; Kang, S.W.; Choi, K.H.; Kim, H.J.; Yoo, T.H. Plasma levels of soluble receptor for advanced glycation end products (sRAGE) and proinflammatory ligand for RAGE (EN-RAGE) are associated with carotid atherosclerosis in patients with peritoneal dialysis. Atherosclerosis 2012, 220, 208–214. [Google Scholar] [CrossRef]
- Rabbani, N.; Thornalley, P.J. Advanced glycation end products in the pathogenesis of chronic kidney disease. Kidney Int. 2018, 93, 803–813. [Google Scholar] [CrossRef]
- Bai, Y.X.; Xu, Y.H.; Wang, X.; Sun, C.; Guo, Y.; Qiu, S.; Ma, K.W. Advances in SARS-CoV-2: A systematic review. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 9208–9215. [Google Scholar] [CrossRef]
- Kim, H.S.; Chung, W.; Kim, A.J.; Ro, H.; Chang, J.H.; Lee, H.H.; Jung, J.Y. Circulating levels of soluble receptor for advanced glycation end product are inversely associated with vascular calcification in patients on haemodialysis independent of S100A12 (EN-RAGE) levels. Nephrology 2013, 18, 777–782. [Google Scholar] [CrossRef] [PubMed]
- Nazratun, N.; Mahmood, A.A.; Kuppusamy, U.R.; Ahmad, T.S.; Tan, S.Y. Diabetes mellitus exacerbates advanced glycation end product accumulation in the veins of end-stage renal failure patients. Vasc. Med. 2006, 11, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Dozio, E.; Di Gaetano, N.; Findeisen, P.; Corsi Romanelli, M.M. Glycated albumin: From biochemistry and laboratory medicine to clinical practice. Endocrine 2017, 55, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Janda, K.; Krzanowski, M.; Gajda, M.; Dumnicka, P.; Jasek, E.; Fedak, D.; Pietrzycka, A.; Kuzniewski, M.; Litwin, J.A.; Sulowicz, W. Vascular effects of advanced glycation end-products: Content of immunohistochemically detected AGEs in radial artery samples as a predictor for arterial calcification and cardiovascular risk in asymptomatic patients with chronic kidney disease. Dis. Markers 2015, 2015, 153978. [Google Scholar] [CrossRef] [PubMed]
- Machowska, A.; Sun, J.; Qureshi, A.R.; Isoyama, N.; Leurs, P.; Anderstam, B.; Heimburger, O.; Barany, P.; Stenvinkel, P.; Lindholm, B. Plasma Pentosidine and Its Association with Mortality in Patients with Chronic Kidney Disease. PLoS ONE 2016, 11, e0163826. [Google Scholar] [CrossRef]
- Masola, V.; Gambaro, G.; Tibaldi, E.; Onisto, M.; Abaterusso, C.; Lupo, A. Regulation of heparanase by albumin and advanced glycation end products in proximal tubular cells. Biochim. Biophys. Acta 2011, 1813, 1475–1482. [Google Scholar] [CrossRef]
- Al-Hussaini, H.; Kilarkaje, N. Trans-resveratrol mitigates type 1 diabetes-induced oxidative DNA damage and accumulation of advanced glycation end products in glomeruli and tubules of rat kidneys. Toxicol. Appl. Pharmacol. 2018, 339, 97–109. [Google Scholar] [CrossRef]
- Lee, J.; Hyon, J.Y.; Min, J.Y.; Huh, Y.H.; Kim, H.J.; Lee, H.; Yun, S.H.; Choi, C.W.; Jeong Ha, S.; Park, J.; et al. Mitochondrial carnitine palmitoyltransferase 2 is involved in N(epsilon)-(carboxymethyl)-lysine-mediated diabetic nephropathy. Pharmacol. Res. 2020, 152, 104600. [Google Scholar] [CrossRef]
- Sotokawauchi, A.; Nakamura, N.; Matsui, T.; Higashimoto, Y.; Yamagishi, S.I. Glyceraldehyde-Derived Pyridinium Evokes Renal Tubular Cell Damage via RAGE Interaction. Int. J. Mol. Sci. 2020, 21, 2604. [Google Scholar] [CrossRef]
- Hudson, B.I.; Carter, A.M.; Harja, E.; Kalea, A.Z.; Arriero, M.; Yang, H.; Grant, P.J.; Schmidt, A.M. Identification, classification, and expression of RAGE gene splice variants. FASEB J. 2008, 22, 1572–1580. [Google Scholar] [CrossRef]
- Kalousova, M.; Hodkova, M.; Kazderova, M.; Fialova, J.; Tesar, V.; Dusilova-Sulkova, S.; Zima, T. Soluble receptor for advanced glycation end products in patients with decreased renal function. Am. J. Kidney Dis. 2006, 47, 406–411. [Google Scholar] [CrossRef] [PubMed]
- Basta, G.; Leonardis, D.; Mallamaci, F.; Cutrupi, S.; Pizzini, P.; Gaetano, L.; Tripepi, R.; Tripepi, G.; De Caterina, R.; Zoccali, C. Circulating soluble receptor of advanced glycation end product inversely correlates with atherosclerosis in patients with chronic kidney disease. Kidney Int. 2010, 77, 225–231. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Maillard-Lefebvre, H.; Boulanger, E.; Daroux, M.; Gaxatte, C.; Hudson, B.I.; Lambert, M. Soluble receptor for advanced glycation end products: A new biomarker in diagnosis and prognosis of chronic inflammatory diseases. Rheumatology 2009, 48, 1190–1196. [Google Scholar] [CrossRef] [PubMed]
- Dozio, E.; Briganti, S.; Delnevo, A.; Vianello, E.; Ermetici, F.; Secchi, F.; Sardanelli, F.; Morricone, L.; Malavazos, A.E.; Corsi Romanelli, M.M. Relationship between soluble receptor for advanced glycation end products (sRAGE), body composition and fat distribution in healthy women. Eur. J. Nutr. 2016, 56, 2557–2564. [Google Scholar] [CrossRef] [PubMed]
- Heier, M.; Margeirsdottir, H.D.; Gaarder, M.; Stensaeth, K.H.; Brunborg, C.; Torjesen, P.A.; Seljeflot, I.; Hanssen, K.F.; Dahl-Jorgensen, K. Soluble RAGE and atherosclerosis in youth with type 1 diabetes: A 5-year follow-up study. Cardiovasc. Diabetol. 2015, 14, 126. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.C.; Woodward, M.; Neal, B.; Li, Q.; Pickering, R.; Marre, M.; Williams, B.; Perkovic, V.; Cooper, M.E.; Zoungas, S.; et al. Relationship between levels of advanced glycation end products and their soluble receptor and adverse outcomes in adults with type 2 diabetes. Diabetes Care 2015, 38, 1891–1897. [Google Scholar] [CrossRef]
- Wong, F.N.; Chua, K.H.; Tan, J.; Wong, C.M.; Kuppusamy, U.R. Glycaemic control in type 2 diabetic patients with chronic kidney disease: The impacts on enzymatic antioxidants and soluble RAGE. PeerJ 2018, 6, e4421. [Google Scholar] [CrossRef]
- Lee, A.C.; Lam, J.K.; Shiu, S.W.; Wong, Y.; Betteridge, D.J.; Tan, K.C. Serum Level of Soluble Receptor for Advanced Glycation End Products Is Associated with A Disintegrin And Metalloproteinase 10 in Type 1 Diabetes. PLoS ONE 2015, 10, e0137330. [Google Scholar] [CrossRef]
- Vollmer, W.M.; Wahl, P.W.; Blagg, C.R. Survival with dialysis and transplantation in patients with end-stage renal disease. N. Engl. J. Med. 1983, 308, 1553–1558. [Google Scholar] [CrossRef]
- Held, P.J.; Brunner, F.; Odaka, M.; Garcia, J.R.; Port, F.K.; Gaylin, D.S. Five-year survival for end-stage renal disease patients in the United States, Europe, and Japan, 1982 to 1987. Am. J. Kidney Dis. 1990, 15, 451–457. [Google Scholar] [CrossRef]

| Overall Cohort | |
|---|---|
| N | 111 |
| Age, years | 78 (10) |
| Males, % | 71 |
| Diabetes, % | 54 |
| Previous cardiovascular events, % | 16 |
| Malnourished at MIS, % | 69 |
| eGFR, mL/min/1.73 m2 | 25 (11) |
| Fasting blood glucose, mg/dL | 116 (39) |
| Uric acid, mg/dL | 6.2 (1.5) |
| Hemoglobin, gr/dL | 12.4 (1.5) |
| Proteinuria, mg/24 h | 1070 (1464) |
| Overall Cohort | |
|---|---|
| MIS | 6.0 (4.7) |
| Albumin, gr/dL | 4.1 (0.4) |
| Prealbumin, mg/dL | 28.7 (5.7) |
| Total cholesterol, mg/dL | 168 (37) |
| Transferrin, mg/dL | 230 (40) |
| CRP, mg/dL | 0.46 (0.76) |
| nPCR, mg/kg/24 h | 759 (239) |
| BMI, kg/m2 | 27.9 (4.9) |
| Variable | Hazard Ratio | 95% CI | p |
|---|---|---|---|
| Age | 1.100 | 1.041 to 1.163 | <0.001 |
| AGE | 1.051 | 1.002 to 1.103 | 0.04 |
| AGE/total proteins | 1.004 | 1.000 to 1.007 | 0.03 |
| esRAGE | 1.083 | 1.027 to 1.141 | 0.003 |
| cRAGE/esRAGE | 0.539 | 0.331 to 0.878 | 0.013 |
| MIS | 1.079 | 1.025 to 1.137 | 0.004 |
| sRAGE | 1.025 | 0.997 to 1.055 | 0.084 |
| cRAGE | 1.018 | 0.979 to 1.059 | 0.373 |
| GA% | 0.971 | 0.909 to 1.037 | 0.381 |
| eGFR | 0.969 | 0.932 to 1.008 | 0.123 |
| Dialysis | 1.745 | 0.715 to 4.260 | 0.222 |
| Variable | Hazard Ratio | 95% CI | p |
|---|---|---|---|
| esRAGE | 1.087 | 1.023 to 1.156 | 0.007 |
| cRAGE/esRAGE | 0.572 | 0.347 to 0.943 | 0.029 |
| AGE | 1.062 | 1.005 to 1.121 | 0.031 |
| AGE/total proteins | 1.004 | 1.000 to 1.007 | 0.073 |
| sRAGE | 1.020 | 0.987 to 1.054 | 0.244 |
| cRAGE | 1.007 | 0.965 to 1.052 | 0.745 |
| GA% | 0.924 | 0.849 to 1.006 | 0.068 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dozio, E.; Vettoretti, S.; Caldiroli, L.; Nerini-Molteni, S.; Tacchini, L.; Ambrogi, F.; Messa, P.; Corsi Romanelli, M.M. Advanced Glycation End Products (AGE) and Soluble Forms of AGE Receptor: Emerging Role as Mortality Risk Factors in CKD. Biomedicines 2020, 8, 638. https://doi.org/10.3390/biomedicines8120638
Dozio E, Vettoretti S, Caldiroli L, Nerini-Molteni S, Tacchini L, Ambrogi F, Messa P, Corsi Romanelli MM. Advanced Glycation End Products (AGE) and Soluble Forms of AGE Receptor: Emerging Role as Mortality Risk Factors in CKD. Biomedicines. 2020; 8(12):638. https://doi.org/10.3390/biomedicines8120638
Chicago/Turabian StyleDozio, Elena, Simone Vettoretti, Lara Caldiroli, Silvia Nerini-Molteni, Lorenza Tacchini, Federico Ambrogi, Piergiorgio Messa, and Massimiliano M. Corsi Romanelli. 2020. "Advanced Glycation End Products (AGE) and Soluble Forms of AGE Receptor: Emerging Role as Mortality Risk Factors in CKD" Biomedicines 8, no. 12: 638. https://doi.org/10.3390/biomedicines8120638
APA StyleDozio, E., Vettoretti, S., Caldiroli, L., Nerini-Molteni, S., Tacchini, L., Ambrogi, F., Messa, P., & Corsi Romanelli, M. M. (2020). Advanced Glycation End Products (AGE) and Soluble Forms of AGE Receptor: Emerging Role as Mortality Risk Factors in CKD. Biomedicines, 8(12), 638. https://doi.org/10.3390/biomedicines8120638







