Objective Biomarkers of Outdoor Activity (Vitamin D and CUVAF) in Young Adults with Myopia During and After the SARS-CoV-2 Pandemic
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Protocol
2.3. Statistical Analysis
3. Results
3.1. Sun Exposure and Skin Protection Habits
3.2. Demographic Characteristics, Skin Care Habits, and Vitamin D Levels
3.3. Myopia and Vitamin D Levels During and After the Pandemic
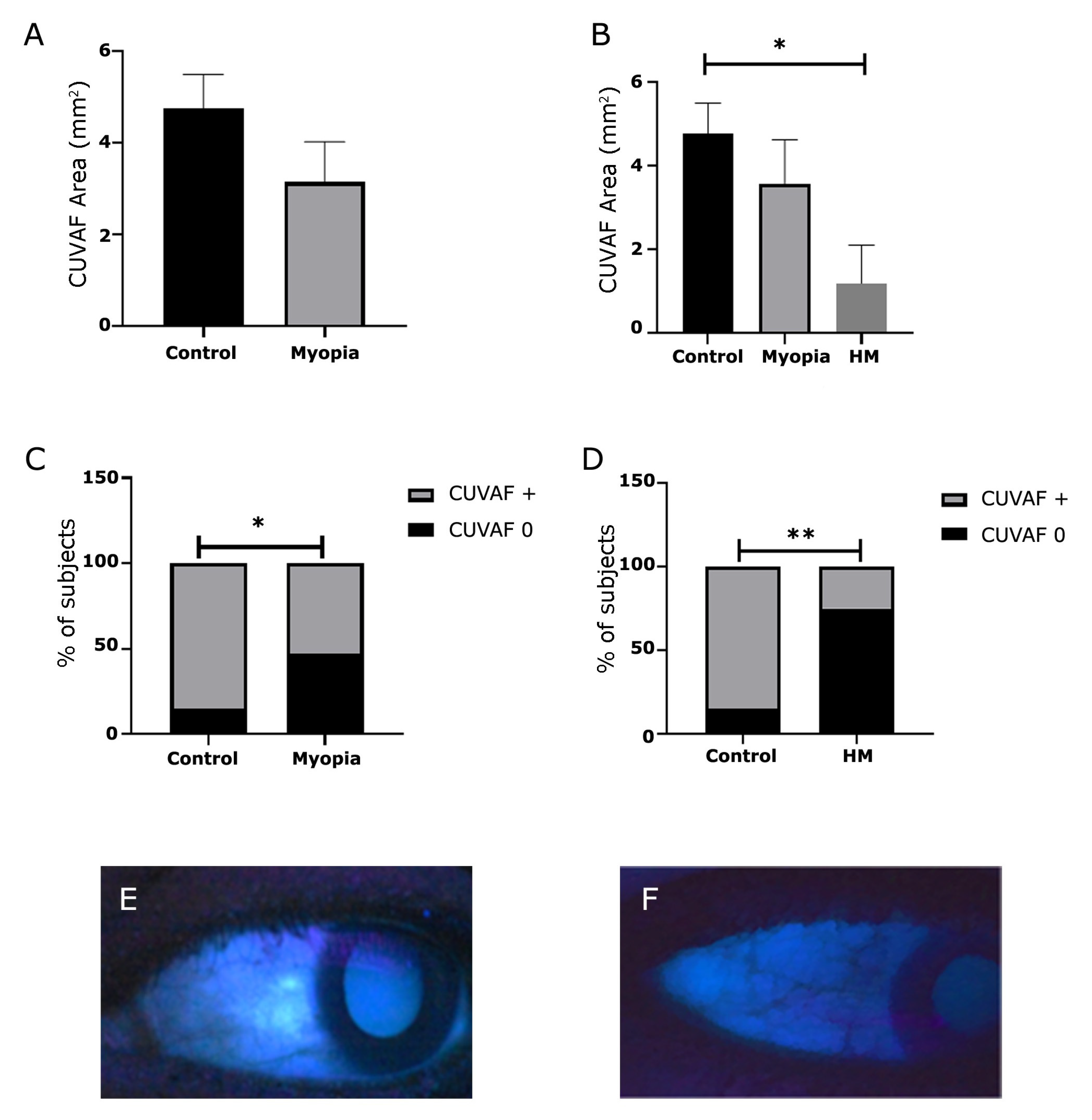
3.4. Myopia and CUVAF Area After the Pandemic
4. Discussion
- Changes in intracellular calcium levels may impair ciliary muscle contraction and relaxation, promoting myopia development [35].
- Serum vitamin D might influence retina–scleral signaling pathways mediated by dopamine and retinoic acid, playing a role in ocular elongation [36].
- Vitamin D levels could affect scleral metalloproteinase (MMP) activity, influencing ocular morphology and refraction [37].
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CUVAF | Conjunctival Ultraviolet Autofluorescence |
| WHO | World Health Organization |
| GPS | Global Positioning System |
| UV-B | Ultraviolet B |
| UV | Ultraviolet |
| MMP | Metalloproteinase |
| SE | Spherical Equivalent |
| SD | Standard Deviation |
| OR | Odds Ratio |
| CI | Confidence Interval |
References
- Wong, C.-W.; Tsai, A.; Jonas, J.; Mtasui, K.; Chen, J.; Ang, M.; Ting, D. Digital Screen Time During the COVID-19 Pandemic: Risk for a Further Myopia Boom? Am. J. Ophthalmol. 2021, 7, 333–348. [Google Scholar] [CrossRef]
- Wang, G.H.; Zhang, Y.T.; Zhao, J.; Zhang, J.; Jiang, F. Mitigate the Effects of Home Confinement on Children During the COVID-19 Outbreak. J. Shanghai Jiaotong Univ. (Med. Sci.) 2020, 40, 279–281. [Google Scholar] [CrossRef]
- Zhang, X.; Cheung, S.S.L.; Chan, H.N.; Zhang, Y.; Wang, Y.M.; Yip, B.H.; Kam, K.W.; Yu, M.; Cheng, C.Y.; Young, A.L.; et al. Myopia Incidence and Lifestyle Changes Among School Children During the COVID-19 Pandemic: A Population-Based Prospective Study. Br. J. Ophthalmol. 2021, 106, 1772–1778. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, Y.; Musch, D.C.; Wei, N.; Qi, X.; Ding, G.; Li, X.; Li, J.; Song, L.; Zhang, Y.; et al. Progression of Myopia in School-Aged Children After COVID-19 Home Confinement. JAMA Ophthalmol. 2021, 139, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Cao, K.; Yue, W.; Yusufu, M.; Wang, N. Significance of Outdoor Time for Myopia Prevention: A Systematic Review and Meta-Analysis Based on Randomized Controlled Trials. Ophthalmic Res. 2019, 63, 97–105. [Google Scholar] [CrossRef]
- Deng, L.; Pang, Y. Effect of Outdoor Activities in Myopia Control: Meta-Analysis of Clinical Studies. Optom. Vis. Sci. 2019, 96, 276–282. [Google Scholar] [CrossRef]
- Lee, S.S.Y.; Mackey, D.A. Prevalence and Risk Factors of Myopia in Young Adults: Review of Findings from the Raine Study. Front. Public Health 2022, 10, 861044. [Google Scholar] [CrossRef]
- Wang, J.; He, X.; Xu, X. The Measurement of Time Spent Outdoors in Child Myopia Research: A Systematic Review. Int. J. Ophthalmol. 2018, 11, 1045–1052. [Google Scholar] [CrossRef]
- Mutti, D.O.; Marks, A.R. Blood Levels of Vitamin D in Teens and Young Adults with Myopia. Optom. Vis. Sci. 2011, 88, 377–382. [Google Scholar] [CrossRef]
- Hoel, D.G.; Berwick, M.; de Gruijl, F.R.; Holick, M.F. The Risks and Benefits of Sun Exposure 2016. Dermato-Endocrinology 2016, 8, e1248325. [Google Scholar] [CrossRef]
- Choi, J.A.; Han, K.; Park, Y.-M.M.; La, T.Y. Low Serum 25-Hydroxyvitamin D Is Associated with Myopia in Korean Adolescents. Investing Ophthalmol. Vis. Sci. 2014, 55, 2041–2047. [Google Scholar] [CrossRef]
- Tideman, J.W.L.; Polling, J.R.; Voortman, T.; Jaddoe, V.W.V.; Uitterlinden, A.G.A.G.; Hofman, A.; Vingerling, J.R.; Franco, O.H.; Klaver, C.C.W. Low Serum Vitamin D Is Associated with Axial Length and Risk of Myopia in Young Children. Eur. J. Epidemiol. 2016, 31, 491–499. [Google Scholar] [CrossRef]
- Yazar, S.; Hewitt, A.W.; Black, L.J.; McKnight, C.M.; Mountain, J.A.; Sherwin, J.C.; Oddy, W.H.; Coroneo, M.T.; Lucas, R.M.; Mackey, D.A. Myopia Is Associated with Lower Vitamin D Status in Young Adults. Investig. Ophthalmol. Vis. Sci. 2014, 55, 4552–4559. [Google Scholar] [CrossRef] [PubMed]
- Chan, H.; Zhang, X.; Ling, X.; Bui, C.H.; Wang, Y.; Ip, P.; Chu, W.; Chen, L.; Tham, C.C.; Yam, J.C.; et al. Vitamin D and Ocular Diseases: A Systematic Review. Int. J. Mol. Sci. 2022, 23, 4226. [Google Scholar] [CrossRef]
- Li, X.; Lin, H.; Jiang, L.; Chen, X.; Chen, J.; Lu, F. Low Serum Vitamin D Is Not Correlated with Myopia in Chinese Children and Adolescents. Front. Med. 2022, 9, 809787. [Google Scholar] [CrossRef]
- Cuellar-Partida, G.; Williams, K.M.; Yazar, S.; Guggenheim, J.A.; Hewitt, A.W.; Williams, C.; Wang, J.J.; Kho, P.-F.; Saw, S.M.; Cheng, C.-Y.; et al. Genetically Low Vitamin D Concentrations and Myopic Refractive Error: A Mendelian Randomization Study. Int. J. Epidemiol. 2017, 46, 1882–1890. [Google Scholar] [CrossRef]
- Czepita, M.; Czepita, D. Conjunctival Ultraviolet Autofluorescence Imaging: An Emerging Diagnostic Tool for Monitoring the Effects of Ocular Sun Exposure. Ophthalmol. J. 2024, 9, 127–132. [Google Scholar] [CrossRef]
- Rodriguez, N.G.; Claici, A.O.; Ramos-Castaneda, J.A.; González-Zamora, J.; Bilbao-Malavé, V.; de la Puente, M.; Fernandez-Robredo, P.; Garzón-Parra, S.J.; Garza-Leon, M.; Recalde, S. Conjunctival Ultraviolet Autofluorescence as a Biomarker of Outdoor Exposure in Myopia: A Systematic Review and Meta-Analysis. Sci. Rep. 2024, 14, 1097. [Google Scholar] [CrossRef]
- De la Puente, M.; Bilbao-malavé, V.; González-zamora, J.; Claici, A.O.; Gomez-arteta, L.; Alonso, E.; Fernández-robredo, P.; García-layana, A.; Barrio-barrio, J.; Recalde, S. Conjunctival Ultraviolet Autofluorescence as a Biomarker of Outdoor Time in Myopic Children. Front. Med. 2024, 52, 1492180. [Google Scholar] [CrossRef] [PubMed]
- De la Puente, M.; Irigoyen-Bañegil, C.; Ortega Claici, A.; González-Zamora, J.; Bilbao-Malavé, V.; Fernandez-Robredo, P.; Hernández, M.; Barrio, J.; García-Layana, A.; Recalde, S. Could Children’s Myopization Have Been Avoided During the Pandemic Confinement? The Conjunctival Ultraviolet Autofluorescence (CUVAF) Biomarker as an Answer. Biomedicines 2024, 12, 347. [Google Scholar] [CrossRef]
- Galvis, V.; Tello, A.; Otero, J.; Serrano, A.A.; Gómez, L.M.; Camacho, P.A.; López-Jaramillo, J.P. Prevalence of Refractive Errors in Colombia: MIOPUR Study. Br. J. Ophthalmol. 2018, 102, 1320–1323. [Google Scholar] [CrossRef]
- Flitcroft, D.I.; He, M.; Jonas, J.B.; Jong, M.; Naidoo, K.; Ohno-Matsui, K.; Rahi, J.; Resnikoff, S.; Vitale, S.; Yannuzzi, L. IMI—Defining and Classifying Myopia: A Proposed Set of Standards for Clinical and Epidemiologic Studies. Investing Ophthalmol. Vis. Sci. 2019, 60, M20–M30. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.; Wu, W.; Liou, Y.M. Dose—Response Relationship of Outdoor Exposure and Myopia Indicators: A Systematic Review and Meta-Analysis of Various Research Methods. Int. J. Environ. Res. Public Health 2019, 16, 2595. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, W.; Dong, X.; Zhao, L.; Peng, J.; Wang, R. Association Between Time Spent Outdoors and Myopia Among Junior High School Students. Medicine 2020, 99, e23462. [Google Scholar] [CrossRef] [PubMed]
- Delic, N.C.; Lyons, J.G.; Di Girolamo, N.; Halliday, G.M. Damaging Effects of Ultraviolet Radiation on the Cornea. Photochem. Photobiol. 2017, 93, 920–929. [Google Scholar] [CrossRef] [PubMed]
- Wolf, A.T.; Klawe, J.; Liu, B.; Ahmad, S. Association Between Serum Vitamin D Levels and Myopia in the National Health and Nutrition Examination Survey (2001–2006). Ophthalmic Epidemiol. 2024, 31, 229–239. [Google Scholar] [CrossRef]
- Pannu, A.; Vichare, N.; Pushkar, K.; Kumar, A.; Gupta, S. Parallelism Between Hypovitaminosis D3 and Recently Detected Myopia in Children with Amplified Screen Use in the COVID-19 Era—A Preliminary Study. Indian J. Ophthalmol. 2022, 17, 229–234. [Google Scholar] [CrossRef]
- Tang, S.M.; Lau, T.; Rong, S.S.; Yazar, S.; Chen, L.J.; Mackey, D.A.; Lucas, R.M.; Pang, C.P.; Yam, J.C. Vitamin D and Its Pathway Genes in Myopia: Systematic Review and Meta-Analysis. Br. J. Ophthalmol. 2018, 103, 8–17. [Google Scholar] [CrossRef]
- Laan, D.; Tan, E.T.C.; Huis in het Veld, P.I.; Jellema, H.M.; Jenniskens, K. Myopia progression in children during home confinement in the COVID-19 pandemic: A systematic review and meta-analysis. J. Optom. 2024, 17, 100493. [Google Scholar] [CrossRef]
- Lingham, G.; Mackey, D.A.; Zhu, K.; Lucas, R.M.; Black, L.J.; Oddy, W.H.; Holt, P.; Walsh, J.P.; Sanfilippo, P.G.; Ping-Delfos, W.C.S.; et al. Time spent outdoors through childhood and adolescence—Assessed by 25-hydroxyvitamin D concentration—And risk of myopia at 20 years. Acta Ophthalmol. 2021, 99, 679–687. [Google Scholar] [CrossRef]
- Kwon, J.W.; Choi, J.A.; La, T.Y. Serum 25-Hydroxyvitamin D Level Is Associated with Myopia in the Korea National Health and Nutrition Examination Survey. Medicine 2016, 95, e5012. [Google Scholar] [CrossRef]
- Malik, Y.; Malik, A.; Shabbir, R.; Zaman, A.; Talib, M. Role of Vitamin D in Near Sightedness. Pak. J. Ophthalmol. 2020, 36, 137–141. [Google Scholar] [CrossRef]
- Williams, K.M.; Bentham, G.C.G.; Young, I.S.; Mcginty, A.; Mckay, G.J.; Hogg, R.; Hammond, C.J.; Chakravarthy, U.; Rahu, M.; Seland, J.; et al. Association Between Myopia, Ultraviolet B Radiation Exposure, Serum Vitamin D Concentrations, and Genetic Polymorphisms in Vitamin D Metabolic Pathways in a Multicountry European Study. JAMA Ophthalmol. 2016, 135, 47–53. [Google Scholar] [CrossRef]
- Guggenheim, J.A.; Williams, C.; Northstone, K.; Howe, L.D.; Tilling, K.; St Pourcain, B.; McMahon, G.; Lawlor, D.A. Does Vitamin D Mediate the Protective Effects of Time Outdoors on Myopia? Findings from a Prospective Birth Cohort. Investing Ophthalmol. Vis. Sci. 2014, 55, 8550–8558. [Google Scholar] [CrossRef]
- Gao, F.; Li, P.; Liu, Y.-Q.; Chen, Y. Association Study of the Serum 25(OH)D Concentration and Myopia in Chinese Children. Medicine 2021, 100, e26570. [Google Scholar] [CrossRef] [PubMed]
- Lepple-Wienhues, A.; Stahl, F.; Willner, U.; Schäfer, R.W.M. Endothelin-Evoked Contractions in Bovine Ciliary Muscle and Trabecular Meshwork: Interaction with Calcium, Nifedipine and Nickel. Curr. Eye Res. 1991, 10, 983–989. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.; Wood, C.L.; Wood, A.M.; Timms, P.; Allsopp, A.J. Changes in Vitamin D and Matrix Metalloproteinase-9 in Submariners During a Submerged Patrol. Occup. Environ. Med. 2014, 71, 104–108. [Google Scholar] [CrossRef]
- Amrein, K.; Scherkl, M.; Hoffmann, M.; Neuwersch-Sommeregger, S.; Köstenberger, M.; Tmava Berisha, A.; Martucci, G.; Pilz, S.; Malle, O. Vitamin D Deficiency 2.0: An Update on the Current Status Worldwide. Eur. J. Clin. Nutr. 2020, 74, 1498–1513. [Google Scholar] [CrossRef] [PubMed]
- Kearney, S.; O’Donoghue, L.; Pourshahidi, L.K.; Richardson, P.; Laird, E.; Healy, M.; Saunders, K.J. Conjunctival Ultraviolet Autofluorescence Area, but Not Intensity, Is Associated with Myopia. Clin. Exp. Optom. 2019, 102, 43–50. [Google Scholar] [CrossRef]
- Kumar, S.; Gupta, N.; Velpandian, T.; Gupta, V.; Vanathi, M.; Vashist, P.; Gowtham, L.; Tandon, R. Myopia, Melatonin and Conjunctival Ultraviolet Autofluorescence: A Comparative Cross-Sectional Study in Indian Myopes. Curr. Eye Res. 2021, 46, 1474–1481. [Google Scholar] [CrossRef]
- Bilbao, V.; Gonzalez, J.; Gandana, E.; Puente, M.; Escriche, E.; Bezunartea, J.; Marizkurrena, A.; Recalde, S. A Cross-Sectional Observational Study of the Relationship Between Outdoor Exposure and Myopia in University Students, Measured by Conjunctival Ultraviolet Autofluorescence (CUVAF). J. Clin. Med. 2022, 11, 4264. [Google Scholar] [CrossRef] [PubMed]
- Mcknight, C.M.; Sherwin, J.C.; Yazar, S.; Forward, H.; Tan, A.X.; Hewitt, A.W.; Pennell, C.E.; Mcallister, I.A.N.L.; Terri, L.; Coroneo, M.T.; et al. Myopia in Young Adults Is Inversely Related to an Objective Marker of Ocular Sun Exposure: The Western Australian Raine Cohort Study. Am. J. Ophthalmol. 2014, 158, 1079–1085. [Google Scholar] [CrossRef] [PubMed]
- Haworth, K.; Chandler, H. Seasonal Effect on Ocular Sun Exposure and Conjunctival UV Autofluorescence. Optom. Vis. Sci. 2017, 94, 219–228. [Google Scholar] [CrossRef] [PubMed]

| During the Pandemic | After the Pandemic | |||||||
|---|---|---|---|---|---|---|---|---|
| Insufficient | Sufficient | OR (CI) | p Value * | Insufficient | Sufficient | OR (CI) | p Value * | |
| Characteristics | n (%) | n (%) | ||||||
| Sex | ||||||||
| Female | 46 (97.9) | 1 (2.1) | 9.2 (0.75–111.62) | 0.041 | 46 (97.9) | 1 (2.1) | 15.3 (1.42–164.56) | 0.005 |
| Male | 10 (83.3) | 2 (16.7) | 9 (75.0) | 3 (25.0) | ||||
| Age | ||||||||
| Under 25 years old | 32 (94.1) | 2 (5.9) | 0.66 (0.05–7.77) | 0.615 | 32 (94.1) | 2 (5.9) | 1.39 (0.18–10.61) | 0.569 |
| From 26 to 38 years old | 24 (96.0) | 1 (4.0) | 23 (92.0) | 2 (8.0) | ||||
| Skin Color | ||||||||
| White | 32 (97.0) | 1 (3.0) | 2.67 (0.22–31.15) | 0.578 | 31 (93.9) | 2 (6.1) | 1.29 (0.16–9.84) | 0.598 |
| Brunette | 24 (92.3) | 2 (7.7) | 24 (92.3) | 2 (7.7) | ||||
| Using Sunscreen | ||||||||
| No | 34 (97.1) | 1 (2.9) | 3.09 (0.26–36.16) | 0.359 | 26 (96.3) | 1 (3.7) | 2.69 (0.26–27.48) | 0.373 |
| Yes | 22 (91.7) | 2 (8.3) | 29 (90.6) | 3 (9.4) | ||||
| Using Sunscreen Outdoors | ||||||||
| No | 11 (91.7) | 1 (8.3) | 0.48 (0.04–5.89) | 0.501 | 10 (90.9) | 1 (9.1) | 0.66 (0.06–7.09) | 0.572 |
| Yes | 45 (95.7) | 2 (4.3) | 45 (93.8) | 3 (6.3) | ||||
| Sunscreen Application Zones | ||||||||
| Face. neck and forearm | 12 (92.3) | 1 (7.7) | - | 0.683 | 7 (87.5) | 1 (12.5) | 1.71 (0.09–31.92) | 0.715 |
| Face and neck | 16 (94.1) | 1 (5.9) | - | 0.619 | 19 (95.0) | 1 (5.0) | 2.28 (0.12–41.98) | 0.569 |
| Face | 24 (96.0) | 1 (4.0) | - | 0.683 | 22 (95.7) | 1(4.3) | 3.14 (0.17–57.07) | 0.418 |
| Do not apply | 4 (100) | 0 | Ref | - | 7 (87.5) | 1 (12.5) | Ref | - |
| Hours of Sun Exposure | ||||||||
| Less than 2 h | 16 (100) | 0 | 1.07 (0.99–1.16) | 0.278 | 20 (95.2) | 1 (4.8) | 1.71 (0.16–17.60) | 0.551 |
| More than 2 h | 40 (93.0) | 3 (7.0) | 35 (92.1) | 3 (7.9) | ||||
| Daily Device Usage Time | ||||||||
| Less than 8 h | 31 (93.9) | 2 (6.1) | 0.62 (0.05–7.24) | 0.590 | 30 (93.8) | 2 (6.3) | 1.20 (0.15–9.14) | 0.627 |
| More than 8 h | 25 (96.2) | 1 (3.8) | 25 (92.6) | 2 (7.4) | ||||
| Type of Lighting | ||||||||
| Artificial | 20 (90.9) | 2 (9.1) | 30 (2.03–441.8) | 0.002 | 19 (90.5) | 2 (9.5) | - | 0.472 |
| Solar and Artificial | 33 (100) | 0 | - | <0.001 | 31 (93.9) | 2 (6.1) | - | 0.571 |
| Solar | 3 (75.0) | 1 (25.0) | Ref | - | 5 (100) | 0 | Ref | - |
| Work/Study Mode | ||||||||
| Virtual | 32 (100) | 0 | - | 0.009 | 10 (83.3) | 2 (16.7) | 0.2 (0.01–2.46) | 0.173 |
| Hybrid | 16 (94.1) | 1 (5.9) | 4 (0.31–51.02) | 0.260 | 20 (95.2) | 1 (4.8) | 0.8 (0.04–13.6) | 0.877 |
| Presential | 8 (80.0) | 2 (20.0) | Ref | - | 25 (96.2) | 1 (3.8) | Ref | - |
| Classification of myopia | ||||||||
| High Myopia | 6 (100) | 0 | - | 0.529 | 6 (100) | 0 | - | 0.491 |
| Normal myopia | 24 (92.3) | 2 (7.7) | 0.46 (0.03–5.42) | 0.632 | 24 (92.3) | 2 (7.7) | 0.96 (0.12–7.37) | 0.968 |
| Control | 26 (96.39) | 1 (3.7) | Ref | - | 25 (96.2) | 2 (7.4) | Ref | - |
| Refractive Data Right Eye | Refractive Data Left Eye | Vitamin D Levels | |||
|---|---|---|---|---|---|
| During | After | ||||
| Mean (Min/Max) | Mean (Min/Max) | Mean ± SD | Mean ± SD | p Value * | |
| Normal myopia | * −2.35 (−5.75, 1.75) | * −2.08 (−5.00, 1.25) | 21.35 ± 6.73 | 21.38 ± 6.90 | 0.334 |
| High myopia | * −8.12 (−12.00, −3.25) | * −7.50 (−13.00, −2.75) | 17.70 ± 7.17 | 18.55 ± 5.18 | 0.463 |
| Control | * 0.91 (−0.50, 5.00) | * 1.16 (−0.25, 4.75) | 20.35 ± 5.86 | 20.29 ± 5.18 | 0.859 |
| Vitamin D levels all groups | * −1.44 (−12.00, 5.00) | * −1.14 (−13.00, 4.75) | 20.52 ± 6.36 | 20.59 ± 6.23 | 0.739 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gutierrez-Rodriguez, N.; de la Puente-Carabot, M.; Rodriguez-Hilarion, J.A.; Ramos-Castaneda, J.A.; Bilbao-Malavé, V.; Avendaño-Vasquez, C.J.; Gonzalez-Zamora, J.; Garzón-Parra, S.J.; Recalde, S. Objective Biomarkers of Outdoor Activity (Vitamin D and CUVAF) in Young Adults with Myopia During and After the SARS-CoV-2 Pandemic. Biomedicines 2025, 13, 2042. https://doi.org/10.3390/biomedicines13082042
Gutierrez-Rodriguez N, de la Puente-Carabot M, Rodriguez-Hilarion JA, Ramos-Castaneda JA, Bilbao-Malavé V, Avendaño-Vasquez CJ, Gonzalez-Zamora J, Garzón-Parra SJ, Recalde S. Objective Biomarkers of Outdoor Activity (Vitamin D and CUVAF) in Young Adults with Myopia During and After the SARS-CoV-2 Pandemic. Biomedicines. 2025; 13(8):2042. https://doi.org/10.3390/biomedicines13082042
Chicago/Turabian StyleGutierrez-Rodriguez, Natali, Miriam de la Puente-Carabot, Javier Andres Rodriguez-Hilarion, Jorge A. Ramos-Castaneda, Valentina Bilbao-Malavé, Carlos Javier Avendaño-Vasquez, Jorge Gonzalez-Zamora, Sandra Johanna Garzón-Parra, and Sergio Recalde. 2025. "Objective Biomarkers of Outdoor Activity (Vitamin D and CUVAF) in Young Adults with Myopia During and After the SARS-CoV-2 Pandemic" Biomedicines 13, no. 8: 2042. https://doi.org/10.3390/biomedicines13082042
APA StyleGutierrez-Rodriguez, N., de la Puente-Carabot, M., Rodriguez-Hilarion, J. A., Ramos-Castaneda, J. A., Bilbao-Malavé, V., Avendaño-Vasquez, C. J., Gonzalez-Zamora, J., Garzón-Parra, S. J., & Recalde, S. (2025). Objective Biomarkers of Outdoor Activity (Vitamin D and CUVAF) in Young Adults with Myopia During and After the SARS-CoV-2 Pandemic. Biomedicines, 13(8), 2042. https://doi.org/10.3390/biomedicines13082042










