Immunohistochemical Demonstration of Tuft Cells in Human Acinar-to-Ductal Metaplasia and Pancreatic Intraepithelial Neoplasia
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. Histopathological Analysis
2.3. Immunohistochemical Analyses
2.4. Statistical Analyses
3. Results
3.1. Patient Characteristics
3.2. Histopathological Characteristics
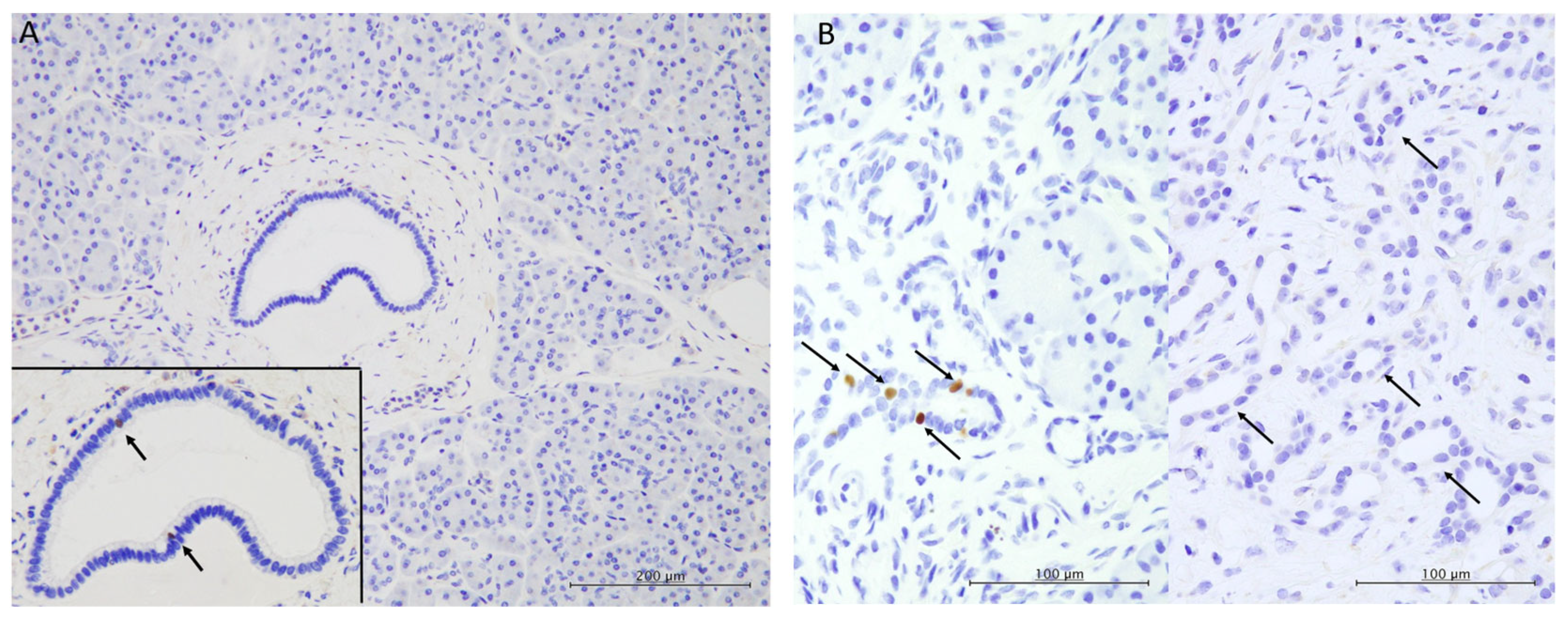
3.3. Immunohistochemical Characteristics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADM | Acinar-to-ductal metaplasia |
| DCLK1 | Double cortin-like kinase 1 |
| IPMN | Intraductal papillary mucinous neoplasm |
| PanIN | Pancreatic intraepithelial neoplasia |
| PDAC | Pancreatic ductal adenocarcinoma |
| POU2F3 | POU domain class 2 transcription factor 3 |
References
- Basturk, O.; Esposito, I.; Fukushima, N.; Furukawa, T.; Hong, S.M.; Klöppel, G. Pancreatic intraepithelial neoplasia. In Digestive System Tumours, 5th ed.; WHO Classification of Tumours; IARC: Lyon, France, 2019; pp. 307–309. [Google Scholar]
- Rodríguez, G.Y.; Jiménez, S.P.; Muñoz Velasco, R.; García, G.A.; Sánchez-Arévalo, L.V.J. Molecular alterations in pancreatic cancer: Transfer to the clinic. Int. J. Mol. Sci. 2021, 22, 2077. [Google Scholar] [CrossRef]
- Backx, E.; Coolens, K.; Van den Bossche, J.-L.; Houbracken, I.; Espinet, E.; Rooman, I. On the origin of pancreatic cancer: Molecular tumor subtypes in perspective of exocrine cell plasticity. Cell. Mol. Gastroenterol. Hepatol. 2022, 13, 1243–1253. [Google Scholar] [CrossRef] [PubMed]
- Storz, P. Acinar cell plasticity and development of pancreatic ductal adenocarcinoma. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 296–304. [Google Scholar] [CrossRef]
- Pinho, A.V.; Rooman, I.; Reichert, M.; De Medts, N.; Bouwens, L.; Rustgi, A.K.; Real, F.X. Adult pancreatic acinar cells dedifferentiate to an embryonic progenitor phenotype with concomitant activation of a senescence programme that is present in chronic pancreatitis. Gut 2010, 60, 958–966. [Google Scholar] [CrossRef] [PubMed]
- Willemer, S.; Adler, G. Histochemical and ultrastructural characteristics of tubular complexes in human acute pancreatitis. Dig. Dis. Sci. 1989, 34, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Parsa, I.; Longnecker, D.S.; Scarpelli, D.G.; Pour, P.; Reddy, J.K.; Lefkowitz, M. Ductal metaplasia of human exocrine pancreas and its association with carcinoma. Cancer Res. 1985, 45, 1285–1290. [Google Scholar]
- Liou, G.-Y.; Döppler, H.; Necela, B.; Krishna, M.; Crawford, H.C.; Raimondo, M.; Storz, P. Macrophage-secreted cytokines drive pancreatic acinar-to-ductal metaplasia through NF-ΚB and mmps. J. Cell Biol. 2013, 202, 563–577. [Google Scholar] [CrossRef]
- Strobel, O.; Dor, Y.; Alsina, J.; Stirman, A.; Lauwers, G.; Trainor, A.; Castillo, C.F.D.; Warshaw, A.L.; Thayer, S.P. In vivo lineage tracing defines the role of acinar-to-ductal transdifferentiation in inflammatory ductal metaplasia. Gastroenterology 2007, 133, 1999–2009. [Google Scholar] [CrossRef]
- Kotas, M.E.; O’Leary, C.E.; Locksley, R.M. Tuft cells: Context- and tissue-specific programming for a conserved cell lineage. Annu. Rev. Pathol. Mech. Dis. 2023, 18, 311–335. [Google Scholar] [CrossRef]
- Wu, X.S.; He, X.-Y.; Ipsaro, J.J.; Huang, Y.-H.; Preall, J.B.; Ng, D.; Shue, Y.-T.; Sage, J.; Egeblad, M.; Joshua-Tor, L.; et al. Oca-T1 and Oca-T2 are coactivators of POU2F3 in the tuft cell lineage. Nature 2022, 607, 169–175. [Google Scholar] [CrossRef]
- Ma, Z.; Lytle, N.K.; Chen, B.; Jyotsana, N.; Novak, S.W.; Cho, C.J.; Caplan, L.; Ben-Levy, O.; Neininger, A.C.; Burnette, D.T.; et al. Single-cell transcriptomics reveals a conserved metaplasia program in pancreatic injury. Gastroenterology 2022, 162, 604–620.e20. [Google Scholar] [CrossRef] [PubMed]
- DelGiorno, K.E.; Naeem, R.F.; Fang, L.; Chung, C.-Y.; Ramos, C.; Luhtala, N.; O’Connor, C.; Hunter, T.; Manor, U.; Wahl, G.M. Tuft cell formation reflects epithelial plasticity in pancreatic injury: Implications for modeling human pancreatitis. Front. Physiol. 2020, 11, 88. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Zhang, W.; Wu, S.; Duan, W.; Wang, Z. Advances in tuft cells, a chemosensory cell in sequential diseases of the pancreas. Biochim. Biophys. Acta Rev. Cancer 2023, 1878, 188911. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ding, L.; Cao, Z.; Qu, D.; Weygant, N. Tuft cells: An emerging therapeutic target for pancreatitis and pancreatic cancer? Expert Opin. Ther. Targets 2020, 24, 1183–1186. [Google Scholar] [CrossRef]
- DelGiorno, K.E.; Chung, C.-Y.; Vavinskaya, V.; Maurer, H.C.; Novak, S.W.; Lytle, N.K.; Ma, Z.; Giraddi, R.R.; Wang, D.; Fang, L.; et al. Tuft cells inhibit pancreatic tumorigenesis in mice by producing prostaglandin D2. Gastroenterology 2020, 159, 1866–1881.e8. [Google Scholar] [CrossRef]
- Schütz, B.; Ruppert, A.-L.; Strobel, O.; Lazarus, M.; Urade, Y.; Büchler, M.W.; Weihe, E. Distribution pattern and molecular signature of cholinergic tuft cells in human gastro-intestinal and pancreatic-biliary tract. Sci. Rep. 2019, 9, 1746. [Google Scholar] [CrossRef]
- Tosti, L.; Hang, Y.; Debnath, O.; Tiesmeyer, S.; Trefzer, T.; Steiger, K.; Ten, F.W.; Lukassen, S.; Ballke, S.; Kühl, A.A.; et al. Single-nucleus and in situ RNA–sequencing reveal cell topographies in the human pancreas. Gastroenterology 2021, 160, 1330–1344.e11. [Google Scholar] [CrossRef]
- Bang, U.C.; Benfield, T.; Hyldstrup, L.; Bendtsen, F.; Beck, J.J. Mortality, cancer, and comorbidities associated with chronic pancreatitis: A Danish nationwide matched-cohort study. Gastroenterology 2014, 146, 989–994. [Google Scholar] [CrossRef]
- Feldmann, G.; Beaty, R.; Hruban, R.H.; Maitra, A. Molecular genetics of pancreatic intraepithelial neoplasia. J. Hepatobil. Pancreat. Surg. 2007, 14, 224–232. [Google Scholar] [CrossRef]
- Hosoda, W.; Chianchiano, P.; Griffin, J.F.; Pittman, M.E.; Brosens, L.A.; Noë, M.; Yu, J.; Shindo, K.; Suenaga, M.; Rezaee, N.; et al. Genetic analyses of isolated high-grade pancreatic intraepithelial neoplasia (Hg-Panin) reveal paucity of alterations in TP53 and SMAD4. J. Pathol. 2017, 242, 16–23. [Google Scholar] [CrossRef]
- Andea, A.; Sarkar, F.; Adsay, V.N. Clinicopathological correlates of pancreatic intraepithelial neoplasia: A comparative analysis of 82 cases with and 152 cases without pancreatic ductal adenocarcinoma. Mod. Pathol. 2003, 16, 996–1006. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, Y.; Furukawa, T.; Yachida, S.; Nishimura, M.; Seki, A.; Nonaka, K.; Aida, J.; Takubo, K.; Ishiwata, T.; Kimura, W.; et al. The prevalence and clinicopathological characteristics of high-grade pancreatic intraepithelial neoplasia. Pancreas 2017, 46, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Bailey, J.M.; Alsina, J.; Rasheed, Z.A.; McAllister, F.M.; Fu, Y.; Plentz, R.; Zhang, H.; Pasricha, P.J.; Bardeesy, N.; Matsui, W.; et al. DCLK1 marks a morphologically distinct subpopulation of cells with stem cell properties in preinvasive pancreatic cancer. Gastroenterology 2014, 146, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Middelhoff, M.; Westphalen, C.B.; Hayakawa, Y.; Yan, K.S.; Gershon, M.D.; Wang, T.C.; Quante, M. DCLK1-expressing tuft cells: Critical modulators of the intestinal niche? Am. J. Physiol. Gastrointest. Liver Physiol. 2017, 313, G285–G299. [Google Scholar] [CrossRef]
- O’Leary, C.E.; Schneider, C.; Locksley, R.M. Tuft cells-systemically dispersed sensory epithelia integrating immune and neural circuitry. Annu. Rev. Immunol. 2019, 37, 47–72. [Google Scholar] [CrossRef]


| N = 29 (%) | ||
|---|---|---|
| Age, years, median (range) | 74 (51–84) | |
| Sex | ||
| Men | 13 (44.8) | |
| Women | 16 (55.2) | |
| Tumour location | ||
| Head | 11 (37.9) | |
| Body and tail | 18 (62.1) | |
| Tumour histopathology | ||
| PDAC | 7 (24.1) | |
| IPMN with low-grade dysplasia | 10 (34.5) | |
| IPMN with high-grade dysplasia or associated invasive carcinoma | 10 (34.5) | |
| SPN | 1 (3.4) | |
| NET and IPMN with low-grade dysplasia | 1 (3.4) |
| POU2F3-Positive Tuft Cells per PanIN Area (Median) | Percentage of POU2F3-Positive Tuft Cells per Total Cells (Median) | |
|---|---|---|
| Low-grade PanIN | 2 cells (range, 0–20) | 1.1% (range, 0–2.5%) |
| High-grade PanIN | 0 cell (range, 0–4) | 0% (range, 0–1.1%) |
| p = 0.0050 | p = 0.0044 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakanishi, K.; Ishida, M.; Taniguchi, K.; Hosomi, K.; Arima, J.; Tomioka, A.; Asakuma, M.; Miyamoto, Y.; Fujimori, K.; Hirose, Y.; et al. Immunohistochemical Demonstration of Tuft Cells in Human Acinar-to-Ductal Metaplasia and Pancreatic Intraepithelial Neoplasia. Biomedicines 2025, 13, 1944. https://doi.org/10.3390/biomedicines13081944
Nakanishi K, Ishida M, Taniguchi K, Hosomi K, Arima J, Tomioka A, Asakuma M, Miyamoto Y, Fujimori K, Hirose Y, et al. Immunohistochemical Demonstration of Tuft Cells in Human Acinar-to-Ductal Metaplasia and Pancreatic Intraepithelial Neoplasia. Biomedicines. 2025; 13(8):1944. https://doi.org/10.3390/biomedicines13081944
Chicago/Turabian StyleNakanishi, Kensuke, Mitsuaki Ishida, Kohei Taniguchi, Kenta Hosomi, Jun Arima, Atsushi Tomioka, Mitsuhiro Asakuma, Yoshiharu Miyamoto, Ko Fujimori, Yoshinobu Hirose, and et al. 2025. "Immunohistochemical Demonstration of Tuft Cells in Human Acinar-to-Ductal Metaplasia and Pancreatic Intraepithelial Neoplasia" Biomedicines 13, no. 8: 1944. https://doi.org/10.3390/biomedicines13081944
APA StyleNakanishi, K., Ishida, M., Taniguchi, K., Hosomi, K., Arima, J., Tomioka, A., Asakuma, M., Miyamoto, Y., Fujimori, K., Hirose, Y., & Lee, S.-W. (2025). Immunohistochemical Demonstration of Tuft Cells in Human Acinar-to-Ductal Metaplasia and Pancreatic Intraepithelial Neoplasia. Biomedicines, 13(8), 1944. https://doi.org/10.3390/biomedicines13081944







