Nanofiber Graft Therapy to Prevent Shoulder Stiffness and Adhesions after Rotator Cuff Tendon Repair: A Comprehensive Review
Abstract
1. Introduction
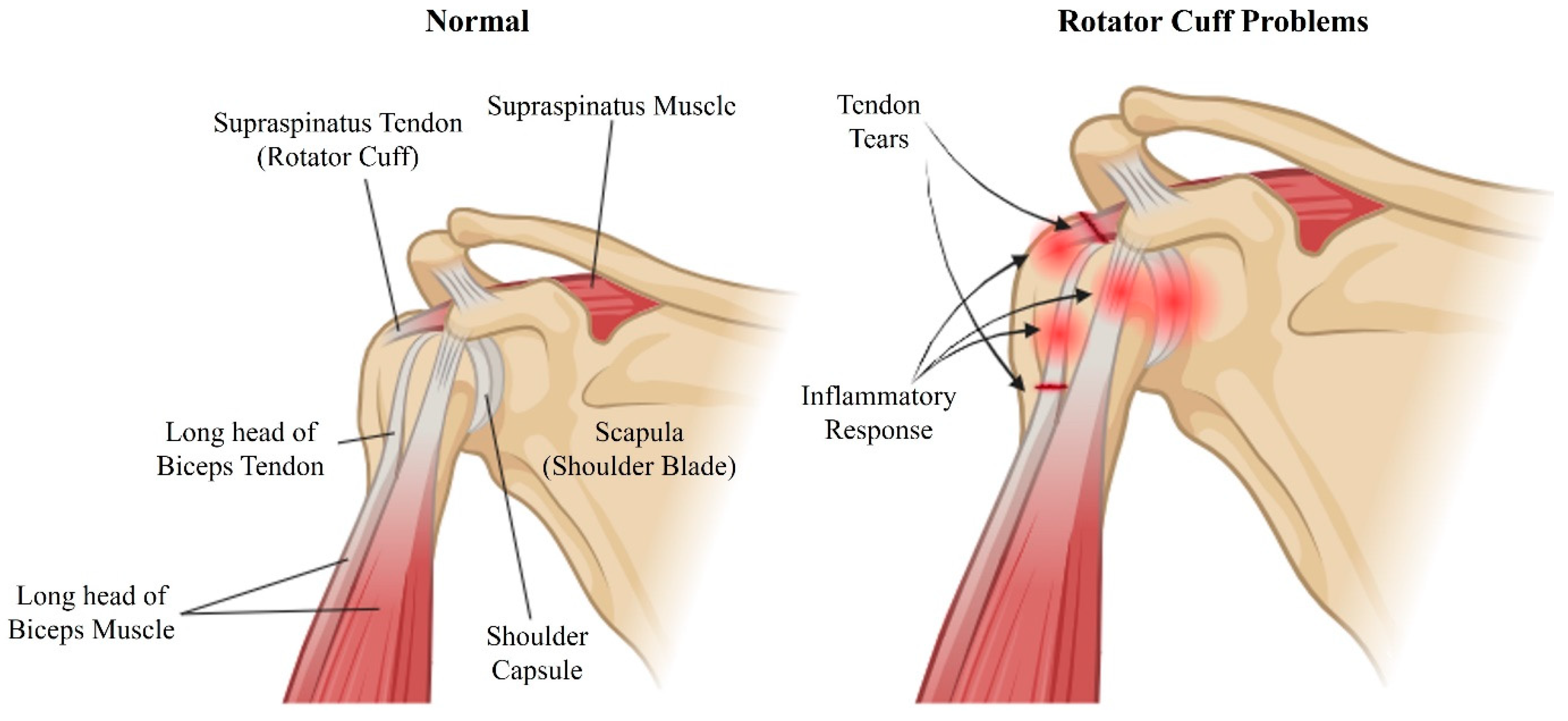
2. Mechanisms and Challenges in Managing Stiffness and Adhesions after Rotator Cuff Repair
2.1. Mechanism of Stiffness and Adhesion Formation
2.2. Current Treatment Techniques for Stiffness and Adhesions
3. Characteristics and Mechanisms of Nanofiber-Based Approaches in Tissue Engineering
3.1. Characteristics of Nanofiber Scaffolds
3.1.1. Structural and Morphological Properties
3.1.2. Compositional Properties
3.1.3. Physico-Chemical Behavior
3.1.4. Fabrication Techniques
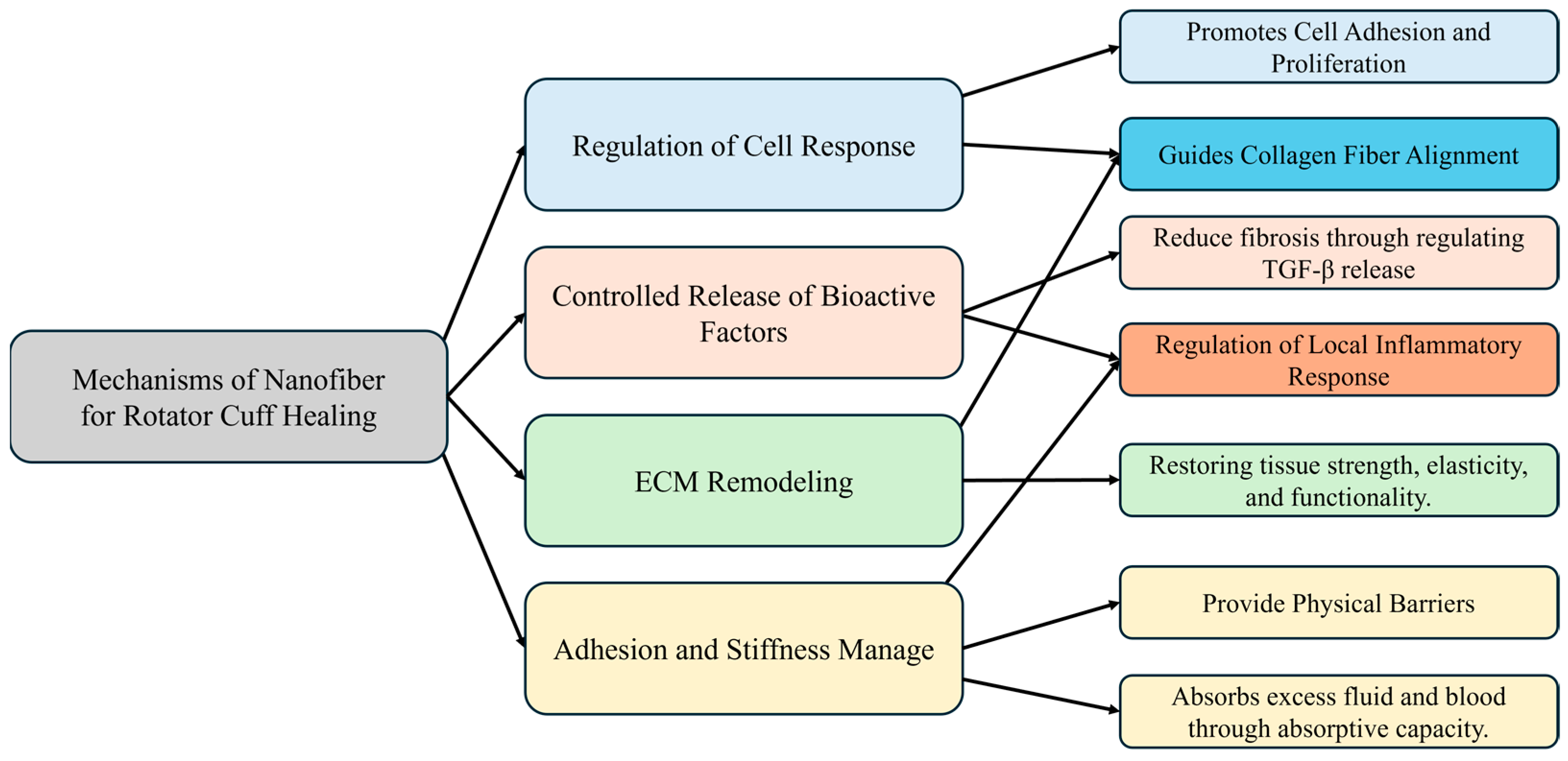
3.2. Mechanism of Nanofiber Scaffolds for Rotator Cuff Healing
3.2.1. Regulation of Cell Response
3.2.2. Controlled Release of Bioactive Factors and Regulation of Local Inflammatory Response
3.2.3. ECM Remodeling
3.2.4. Adhesion and Stiffness Management
3.3. Improvement of Rotator Cuff Adhesion through Preclinical Cases
4. Nanofiber-Based Strategies for Managing Stiffness and Adhesions in Rotator Cuff Healing
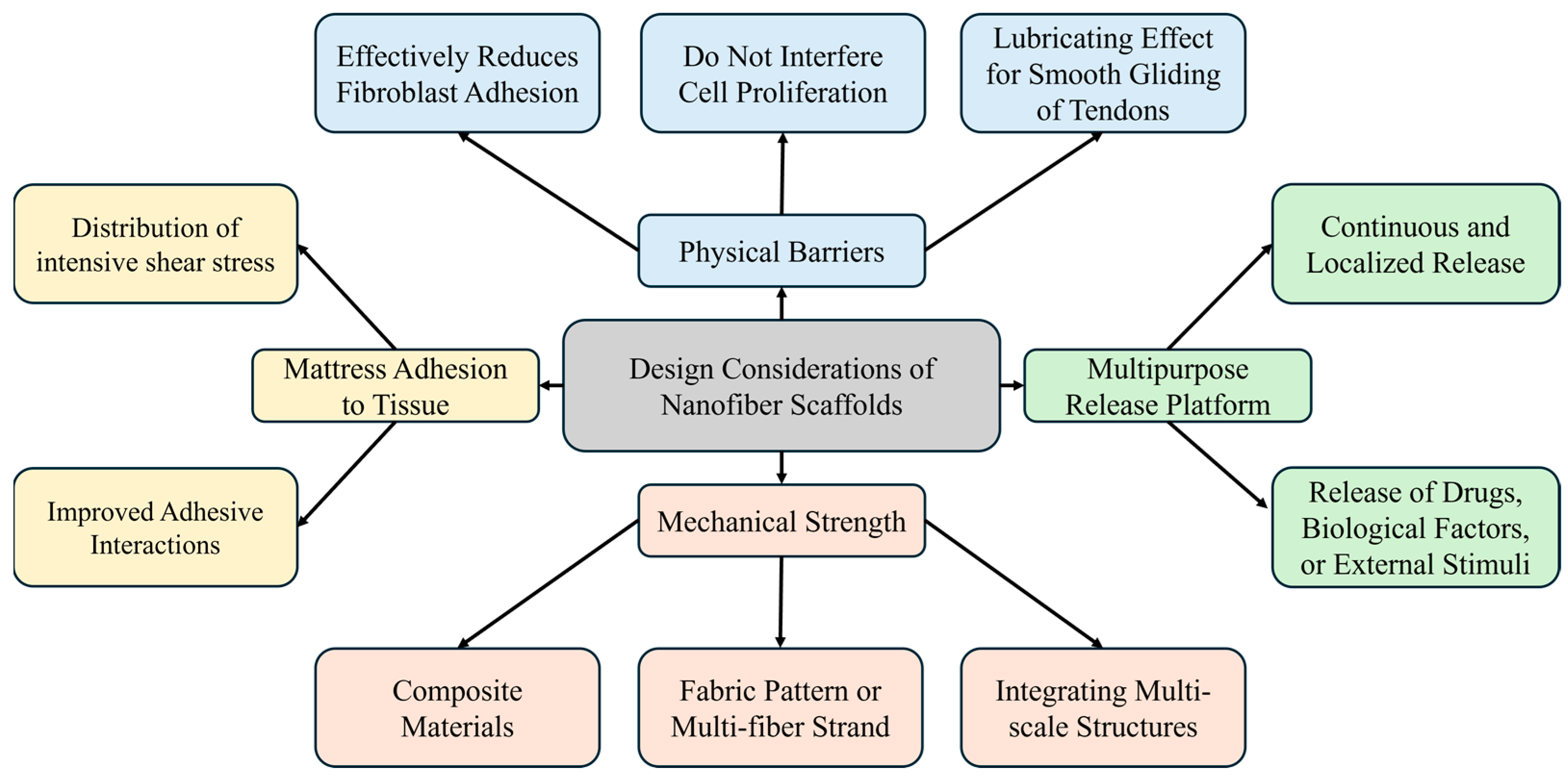
4.1. Design Considerations for Nanofiber Scaffolds
4.1.1. Overview of Design Considerations
4.1.2. Anti-Adhesion Properties
4.1.3. Piezoelectric Materials
4.1.4. Based on Polymer Types
4.1.5. Enhancing Mechanical Properties
4.1.6. Alternative Fixation Methods
4.2. Clinical Trials for Rotator Cuff Healing and Adhesion Management
4.3. Commercialization Cases for Tendon Reconstruction and Adhesion Prevention
5. Challenges and Future Directions
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ghodadra, N.S.; Provencher, M.T.; Verma, N.N.; Wilk, K.E.; Romeo, A.A. Open, mini-open, and all-arthroscopic rotator cuff repair surgery: Indications and implications for rehabilitation. J. Orthop. Sports Phys. Ther. 2009, 39, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Yang, J., Jr.; Robbins, M.; Reilly, J.; Maerz, T.; Anderson, K. The Clinical Effect of a Rotator Cuff Retear: A Meta-analysis of Arthroscopic Single-Row and Double-Row Repairs. Am. J. Sports Med. 2017, 45, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Akdemir, M.; Kılıç, A.; Kurt, C.; Çapkın, S. Better short-term outcomes of mini-open rotator cuff repair compared to full arthroscopic repair. Clin. Shoulder. Elb. 2024, 27, 212–218. [Google Scholar] [CrossRef]
- Cofield, R.H.; Parvizi, J.; Hoffmeyer, P.J.; Lanzer, W.L.; Ilstrup, D.M.; Rowland, C.M. Surgical repair of chronic rotator cuff tears. A prospective long-term study. J. Bone Jt. Surg. Am. 2001, 83, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Vollans, S.; Ali, A. Rotator cuff tears. Surgery 2016, 34, 129–133. [Google Scholar] [CrossRef]
- Denard, P.J.; Lädermann, A.; Burkhart, S.S. Prevention and management of stiffness after arthroscopic rotator cuff repair: Systematic review and implications for rotator cuff healing. Arthroscopy 2011, 27, 842–848. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Chung, S.W.; Kim, J.Y.; Ok, J.H.; Park, I.; Oh, J.H. Is early passive motion exercise necessary after arthroscopic rotator cuff repair? Am. J. Sports Med. 2012, 40, 815–821. [Google Scholar] [CrossRef]
- Koo, S.S.; Parsley, B.K.; Burkhart, S.S.; Schoolfield, J.D. Reduction of postoperative stiffness after arthroscopic rotator cuff repair: Results of a customized physical therapy regimen based on risk factors for stiffness. Arthroscopy 2011, 27, 155–160. [Google Scholar] [CrossRef]
- Tan, Z.; Hendy, B.A.; Zmistowski, B.; Camp, R.S.; Getz, C.L.; Abboud, J.A.; Namdari, S. Glenohumeral synovitis score predicts early shoulder stiffness following arthroscopic rotator cuff repair. J. Orthop. 2020, 22, 17–21. [Google Scholar] [CrossRef]
- Kholinne, E.; Jeon, I.H.; Kwak, J.M. Basic to advanced elbow arthroscopy: The history, basic set up, and indications. Clin. Shoulder. Elb. 2024, epub ahead of print. [Google Scholar] [CrossRef]
- Akbar, M.; McLean, M.; Garcia-Melchor, E.; Crowe, L.A.; McMillan, P.; Fazzi, U.G.; Martin, D.; Arthur, A.; Reilly, J.H.; McInnes, I.B.; et al. Fibroblast activation and inflammation in frozen shoulder. PLoS ONE 2019, 14, e0215301. [Google Scholar] [CrossRef] [PubMed]
- Van Eecke, E.; Struelens, B.; Muermans, S. Long-term clinical and radiographic outcomes of arthroscopic acromioclavicular stabilization for acute acromioclavicular joint dislocation. Clin. Shoulder. Elb. 2024, 27, 219–228. [Google Scholar] [CrossRef]
- Radhakrishnan, R.; Goh, J.; Tan, A.H.C. Partial-thickness rotator cuff tears: A review of current literature on evaluation and management. Clin. Shoulder. Elb. 2024, 27, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.B.; Jung, D.W. An Intra-articular Steroid Injection at 6 Weeks Postoperatively for Shoulder Stiffness After Arthroscopic Rotator Cuff Repair Does Not Affect Repair Integrity. Am. J. Sports Med. 2018, 46, 2192–2202. [Google Scholar] [CrossRef]
- Cho, C.H.; Bae, K.C.; Kim, D.H. Incidence and risk factors for early postoperative stiffness after arthroscopic rotator cuff repair in patients without preoperative stiffness. Sci. Rep. 2022, 12, 3132. [Google Scholar] [CrossRef] [PubMed]
- Millican, C.R.; Lam, P.H.; Murrell, G.A.C. Shoulder stiffness after rotator cuff repair: The fate of stiff shoulders up to 9 years after rotator cuff repair. J. Shoulder. Elb. Surg. 2020, 29, 1323–1331. [Google Scholar] [CrossRef] [PubMed]
- Morgan, A.M.; Li, Z.I.; Garra, S.; Bi, A.S.; Gonzalez-Lomas, G.; Jazrawi, L.M.; Campbell, K.A. Patient-reported allergies are associated with increased rate of postoperative stiffness after arthroscopic rotator cuff repair. J. Shoulder. Elb. Surg. 2024, 33, 1050–1057. [Google Scholar] [CrossRef]
- Huberty, D.P.; Schoolfield, J.D.; Brady, P.C.; Vadala, A.P.; Arrigoni, P.; Burkhart, S.S. Incidence and treatment of postoperative stiffness following arthroscopic rotator cuff repair. Arthroscopy 2009, 25, 880–890. [Google Scholar] [CrossRef]
- Papalia, R.; Franceschi, F.; Vasta, S.; Gallo, A.; Maffulli, N.; Denaro, V. Shoulder stiffness and rotator cuff repair. Br. Med. Bull. 2012, 104, 163–174. [Google Scholar] [CrossRef]
- Gumina, S.; Kim, H.; Jung, Y.; Song, H.S. Rotator cuff degeneration and healing after rotator cuff repair. Clin. Shoulder. Elb. 2023, 26, 323–329. [Google Scholar] [CrossRef]
- Seo, S.S.; Choi, J.S.; An, K.C.; Kim, J.H.; Kim, S.B. The factors affecting stiffness occurring with rotator cuff tear. J. Shoulder. Elb. Surg. 2012, 21, 304–309. [Google Scholar] [CrossRef] [PubMed]
- Eshima, K.; Ohzono, H.; Gotoh, M.; Shimokobe, H.; Tanaka, K.; Nakamura, H.; Kanazawa, T.; Okawa, T.; Shiba, N. Effect of suprascapular nerve injury on muscle and regenerated enthesis in a rat rotator cuff tear model. Clin. Shoulder. Elb. 2023, 26, 131–139. [Google Scholar] [CrossRef]
- Rawson, S.; Cartmell, S.; Wong, J. Suture techniques for tendon repair; a comparative review. Muscles Ligaments Tendons. J. 2013, 3, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Verstift, D.E.; Heisen, J.; Somford, M.P.; van den Bekerom, M.P.J. Eponymous terms in acromioclavicular joint surgery. Clin. Shoulder. Elb. 2024, 27, 229–236. [Google Scholar] [CrossRef]
- Zhao, S.; Su, W.; Shah, V.; Hobson, D.; Yildirimer, L.; Yeung, K.W.K.; Zhao, J.; Cui, W.; Zhao, X. Biomaterials based strategies for rotator cuff repair. Colloids Surf. B Biointerfaces 2017, 157, 407–416. [Google Scholar] [CrossRef]
- Lee, H.J. Long head biceps tendon as a graft material. Clin. Shoulder. Elb. 2024, 27, 138–140. [Google Scholar] [CrossRef] [PubMed]
- Saveh-Shemshaki, N.; Nair, L.S.; Laurencin, C.T. Nanofiber-based matrices for rotator cuff regenerative engineering. Acta Biomater. 2019, 94, 64–81. [Google Scholar] [CrossRef]
- Giordano, J.R.; Klein, B.; Hershfeld, B.; Gruber, J.; Trasolini, R.; Cohn, R.M. A review of chronic pectoralis major tears: What options are available? Clin. Shoulder. Elb. 2023, 26, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Qinglin, K.; Rui, Z.; Yanhao, L.; Rong, B. Tendon Adhesion and Novel Solutions. In Tendons; Nahum, R., Ed.; IntechOpen: Rijeka, Croatia, 2022; Chapter 3. [Google Scholar]
- Fares, M.Y.; Daher, M.; Boufadel, P.; Haikal, E.R.; Koa, J.; Singh, J.; Abboud, J.A. Arthroscopic remplissage: History, indications, and clinical outcomes. Clin. Shoulder. Elb. 2024, 27, 254–262. [Google Scholar] [CrossRef]
- Titan, A.L.; Foster, D.S.; Chang, J.; Longaker, M.T. Flexor Tendon: Development, Healing, Adhesion Formation, and Contributing Growth Factors. Plast. Reconstr. Surg. 2019, 144, 639e–647e. [Google Scholar] [CrossRef]
- Coccolini, F.; Ansaloni, L.; Manfredi, R.; Campanati, L.; Poiasina, E.; Bertoli, P.; Capponi, M.G.; Sartelli, M.; Di Saverio, S.; Cucchi, M.; et al. Peritoneal adhesion index (PAI): Proposal of a score for the “ignored iceberg” of medicine and surgery. World J. Emerg. Surg. 2013, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Fatehi Hassanabad, A.; Zarzycki, A.N.; Jeon, K.; Deniset, J.F.; Fedak, P.W.M. Post-Operative Adhesions: A Comprehensive Review of Mechanisms. Biomedicines 2021, 9, 867. [Google Scholar] [CrossRef] [PubMed]
- Nikolaidou, O.; Migkou, S.; Karampalis, C. Rehabilitation after Rotator Cuff Repair. Open Orthop. J. 2017, 11, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Redler, L.H.; Dennis, E.R. Treatment of Adhesive Capsulitis of the Shoulder. J. Am. Acad. Orthop. Surg. 2019, 27, e544–e554. [Google Scholar] [CrossRef] [PubMed]
- Hong, W.K.; Kim, S.; Gong, H.S. Fracture Management in Chronic Kidney Disease: Challenges and Considerations for Orthopedic Surgeons. Clin. Orthop. Surg. 2024, 16, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Bianco, S.T.; Moser, H.L.; Galatz, L.M.; Huang, A.H. Biologics and stem cell-based therapies for rotator cuff repair. Ann. N. Y. Acad. Sci. 2019, 1442, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Quigley, R.; Kaiser, J.T.; Cole, B.J. Editorial Commentary: Rotator Cuff Repair: Graft Augmentation Provides Promising Clinical Outcomes. Arthroscopy 2022, 38, 2348–2349. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.K.; Dorthé, E.; Williams, A.; D’Lima, D.D. Nanofiber Scaffolds by Electrospinning for Rotator Cuff Tissue Engineering. Chonnam. Med. J. 2021, 57, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ding, B.; Li, B. Biomimetic electrospun nanofibrous structures for tissue engineering. Mater. Today 2013, 16, 229–241. [Google Scholar] [CrossRef]
- Hiwrale, A.; Bharati, S.; Pingale, P.; Rajput, A. Nanofibers: A current era in drug delivery system. Heliyon 2023, 9, e18917. [Google Scholar] [CrossRef]
- Li, H.; Zhu, C.; Xue, J.; Ke, Q.; Xia, Y. Enhancing the Mechanical Properties of Electrospun Nanofiber Mats through Controllable Welding at the Cross Points. Macromol. Rapid. Commun. 2017, 38, 1600723. [Google Scholar] [CrossRef]
- Tang, H.; Yi, B.; Wang, X.; Shen, Y.; Zhang, Y. Understanding the cellular responses based on low-density electrospun fiber networks. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 119, 111470. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Gong, M.; Song, J.; Chen, S.; Meng, Q.; Shi, R.; Zhang, L.; Xue, J. Integrating Inflammation-Responsive Prodrug with Electrospun Nanofibers for Anti-Inflammation Application. Pharmaceutics 2022, 14, 1273. [Google Scholar] [CrossRef] [PubMed]
- Vasita, R.; Katti, D.S. Nanofibers and their applications in tissue engineering. Int. J. Nanomed. 2006, 1, 15–30. [Google Scholar] [CrossRef]
- Kumbar, S.G.; Nair, L.S.; Bhattacharyya, S.; Laurencin, C.T. Polymeric nanofibers as novel carriers for the delivery of therapeutic molecules. J. Nanosci. Nanotechnol. 2006, 6, 2591–2607. [Google Scholar] [CrossRef]
- Ma, P.X.; Zhang, R. Synthetic nano-scale fibrous extracellular matrix. J. Biomed. Mater. Res. 1999, 46, 60–72. [Google Scholar] [CrossRef]
- Matthews, J.A.; Wnek, G.E.; Simpson, D.G.; Bowlin, G.L. Electrospinning of collagen nanofibers. Biomacromolecules 2002, 3, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S. Fabrication of novel biomaterials through molecular self-assembly. Nat. Biotechnol. 2003, 21, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Kwon, I.K.; Kidoaki, S.; Matsuda, T. Electrospun nano- to microfiber fabrics made of biodegradable copolyesters: Structural characteristics, mechanical properties and cell adhesion potential. Biomaterials 2005, 26, 3929–3939. [Google Scholar] [CrossRef]
- Yoo, H.S.; Kim, T.G.; Park, T.G. Surface-functionalized electrospun nanofibers for tissue engineering and drug delivery. Adv. Drug. Deliv. Rev. 2009, 61, 1033–1042. [Google Scholar] [CrossRef]
- Dahlin, R.L.; Kasper, F.K.; Mikos, A.G. Polymeric nanofibers in tissue engineering. Tissue Eng. Part B Rev. 2011, 17, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.K.; Kim, S.I.; Kim, S.J.; Kim, S.-K.; Lee, H.; Spinks, G.M. Size-dependent elastic modulus of single electroactive polymer nanofibers. Appl. Phys. Lett. 2006, 89, 231929. [Google Scholar] [CrossRef]
- Tan, E.P.S.; Lim, C.T. Physical properties of a single polymeric nanofiber. Appl. Phys. Lett. 2004, 84, 1603–1605. [Google Scholar] [CrossRef]
- Chew, S.Y.; Hufnagel, T.C.; Lim, C.T.; Leong, K.W. Mechanical properties of single electrospun drug-encapsulated nanofibres. Nanotechnology 2006, 17, 3880–3891. [Google Scholar] [CrossRef]
- Yang, L.; Fitié, C.F.; van der Werf, K.O.; Bennink, M.L.; Dijkstra, P.J.; Feijen, J. Mechanical properties of single electrospun collagen type I fibers. Biomaterials 2008, 29, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Tan, E.P.; Lim, C.T. Effects of annealing on the structural and mechanical properties of electrospun polymeric nanofibres. Nanotechnology 2006, 17, 2649–2654. [Google Scholar] [CrossRef]
- Santoro, M.; Shah, S.R.; Walker, J.L.; Mikos, A.G. Poly(lactic acid) nanofibrous scaffolds for tissue engineering. Adv. Drug. Deliv. Rev. 2016, 107, 206–212. [Google Scholar] [CrossRef]
- Salaris, V.; López, D.; Kenny, J.M.; Peponi, L. Hydrolytic Degradation and Bioactivity of Electrospun PCL-Mg-NPs Fibrous Mats. Molecules 2023, 28, 1001. [Google Scholar] [CrossRef]
- Guzmán-Soria, A.; Moreno-Serna, V.; Canales, D.A.; García-Herrera, C.; Zapata, P.A.; Orihuela, P.A. Effect of Electrospun PLGA/Collagen Scaffolds on Cell Adhesion, Viability, and Collagen Release: Potential Applications in Tissue Engineering. Polymers 2023, 15, 1079. [Google Scholar] [CrossRef]
- Yang, D.; Xiao, J.; Wang, B.; Li, L.; Kong, X.; Liao, J. The immune reaction and degradation fate of scaffold in cartilage/bone tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 104, 109927. [Google Scholar] [CrossRef]
- Mohammadalipour, M.; Asadolahi, M.; Mohammadalipour, Z.; Behzad, T.; Karbasi, S. Plasma surface modification of electrospun polyhydroxybutyrate (PHB) nanofibers to investigate their performance in bone tissue engineering. Int. J. Biol. Macromol. 2023, 230, 123167. [Google Scholar] [CrossRef]
- Kalvand, E.; Bakhshandeh, H.; Nadri, S.; Habibizadeh, M.; Rostamizadeh, K. Poly-ε-caprolactone (PCL)/poly-l-lactic acid (PLLA) nanofibers loaded by nanoparticles-containing TGF-β1 with linearly arranged transforming structure as a scaffold in cartilage tissue engineering. J. Biomed. Mater. Res. A 2023, 111, 1838–1849. [Google Scholar] [CrossRef]
- Lee, J.-H.; Kim, Y.-J. Hydroxyapatite nanofibers fabricated through electrospinning and sol–gel process. Ceram. Int. 2014, 40, 3361–3369. [Google Scholar] [CrossRef]
- Diaz, F.; Zimmermann, L.; Dale, T.P.; Forsyth, N.R.; Boccaccini, A.R. Tuning the properties of all natural polymeric scaffolds for tendon repair with cellulose microfibers. Carbohydr. Polym. Technol. Appl. 2024, 7, 100447. [Google Scholar] [CrossRef]
- Elnaggar, M.A.; El-Fawal, H.A.N.; Allam, N.K. Biocompatible PCL-nanofibers scaffold with immobilized fibronectin and laminin for neuronal tissue regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 119, 111550. [Google Scholar] [CrossRef]
- Lin, Y.; Zhang, L.; Liu, N.Q.; Yao, Q.; Van Handel, B.; Xu, Y.; Wang, C.; Evseenko, D.; Wang, L. In vitro behavior of tendon stem/progenitor cells on bioactive electrospun nanofiber membranes for tendon-bone tissue engineering applications. Int. J. Nanomed. 2019, 14, 5831–5848. [Google Scholar] [CrossRef] [PubMed]
- Yadav, P.; Beniwal, G.; Saxena, K.K. A review on pore and porosity in tissue engineering. Mater. Today Proc. 2021, 44, 2623–2628. [Google Scholar] [CrossRef]
- Laccourreye, O.; Maisonneuve, H. French scientific medical journals confronted by developments in medical writing and the transformation of the medical press. Eur. Ann. Otorhinolaryngol. Head. Neck. Dis. 2019, 136, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; He, Z.; Ji, L.; Zhang, W.; Tong, Y.; Luo, J.; Zhang, Y.; Li, Y.; Meng, X.; Bi, Q. Advanced Nanofiber-Based Scaffolds for Achilles Tendon Regenerative Engineering. Front. Bioeng. Biotechnol. 2022, 10, 897010. [Google Scholar] [CrossRef]
- Lowery, J.L.; Datta, N.; Rutledge, G.C. Effect of fiber diameter, pore size and seeding method on growth of human dermal fibroblasts in electrospun poly(epsilon-caprolactone) fibrous mats. Biomaterials 2010, 31, 491–504. [Google Scholar] [CrossRef]
- Arima, Y.; Iwata, H. Effect of wettability and surface functional groups on protein adsorption and cell adhesion using well-defined mixed self-assembled monolayers. Biomaterials 2007, 28, 3074–3082. [Google Scholar] [CrossRef] [PubMed]
- Faucheux, N.; Schweiss, R.; Lützow, K.; Werner, C.; Groth, T. Self-assembled monolayers with different terminating groups as model substrates for cell adhesion studies. Biomaterials 2004, 25, 2721–2730. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Deng, G.; Chen, W.; Ye, X.; Mo, X. A novel electrospun-aligned nanoyarn-reinforced nanofibrous scaffold for tendon tissue engineering. Colloids Surf. B Biointerfaces 2014, 122, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Svensson, R.B.; Hansen, P.; Hassenkam, T.; Haraldsson, B.T.; Aagaard, P.; Kovanen, V.; Krogsgaard, M.; Kjaer, M.; Magnusson, S.P. Mechanical properties of human patellar tendon at the hierarchical levels of tendon and fibril. J. Appl. Physiol. 2012, 112, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Wren, T.A.; Yerby, S.A.; Beaupré, G.S.; Carter, D.R. Mechanical properties of the human achilles tendon. Clin. Biomech. 2001, 16, 245–251. [Google Scholar] [CrossRef]
- Wu, S.; Wang, Y.; Streubel, P.N.; Duan, B. Living nanofiber yarn-based woven biotextiles for tendon tissue engineering using cell tri-culture and mechanical stimulation. Acta Biomater. 2017, 62, 102–115. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yu, D.G.; Liu, Y.; Liu, Y.N. Progress of Electrospun Nanofibrous Carriers for Modifications to Drug Release Profiles. J. Funct. Biomater. 2022, 13, 289. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Hota, G. Chapter 21—Electrospun polymer composites and ceramics nanofibers: Synthesis and environmental remediation applications. In Design, Fabrication, and Characterization of Multifunctional Nanomaterials; Thomas, S., Kalarikkal, N., Abraham, A.R., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 503–525. [Google Scholar]
- Buzgo, M.; Mickova, A.; Rampichova, M.; Doupnik, M. 11—Blend electrospinning, coaxial electrospinning, and emulsion electrospinning techniques. In Core-Shell Nanostructures for Drug Delivery and Theranostics; Focarete, M.L., Tampieri, A., Eds.; Woodhead Publishing: Sawston, UK, 2018; pp. 325–347. [Google Scholar]
- Cui, J.; Yu, X.; Shen, Y.; Sun, B.; Guo, W.; Liu, M.; Chen, Y.; Wang, L.; Zhou, X.; Shafiq, M.; et al. Electrospinning Inorganic Nanomaterials to Fabricate Bionanocomposites for Soft and Hard Tissue Repair. Nanomaterials 2023, 13, 204. [Google Scholar] [CrossRef] [PubMed]
- Khajavi, R.; Abbasipour, M. Electrospinning as a versatile method for fabricating coreshell, hollow and porous nanofibers. Sci. Iran. 2012, 19, 2029–2034. [Google Scholar] [CrossRef]
- Li, Y.; Liu, X.; Liu, X.; Peng, Y.; Zhu, B.; Guo, S.; Wang, C.; Wang, D.; Li, S. Transforming growth factor-β signalling pathway in tendon healing. Growth Factors 2022, 40, 98–107. [Google Scholar] [CrossRef]
- Anjum, F.; Agabalyan, N.A.; Sparks, H.D.; Rosin, N.L.; Kallos, M.S.; Biernaskie, J. Biocomposite nanofiber matrices to support ECM remodeling by human dermal progenitors and enhanced wound closure. Sci. Rep. 2017, 7, 10291. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Song, W.; Xu, L.; Yu, D.G.; Annie Bligh, S.W. A Review on Electrospun Poly(amino acid) Nanofibers and Their Applications of Hemostasis and Wound Healing. Biomolecules 2022, 12, 794. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.; Park, H.; Park, Y.; Kang, H.; Lee, D. Development of a Lidocaine-Loaded Alginate/CMC/PEO Electrospun Nanofiber Film and Application as an Anti-Adhesion Barrier. Polymers 2020, 12, 618. [Google Scholar] [CrossRef] [PubMed]
- Romeo, A.; Easley, J.; Regan, D.; Hackett, E.; Johnson, J.; Johnson, J.; Puttlitz, C.; McGilvray, K. Rotator cuff repair using a bioresorbable nanofiber interposition scaffold: A biomechanical and histologic analysis in sheep. J. Shoulder. Elb. Surg. 2022, 31, 402–412. [Google Scholar] [CrossRef]
- Han, J.; Rhee, S.M.; Kim, Y.W.; Park, S.H.; Oh, J.H. Three-dimensionally printed recombinant human parathyroid hormone-soaked nanofiber sheet accelerates tendon-to-bone healing in a rabbit model of chronic rotator cuff tear. J. Shoulder. Elb. Surg. 2022, 31, 1628–1639. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.T.; Chen, C.H.; Sheu, C.; Chen, J.P. Ibuprofen-Loaded Hyaluronic Acid Nanofibrous Membranes for Prevention of Postoperative Tendon Adhesion through Reduction of Inflammation. Int. J. Mol. Sci. 2019, 20, 5038. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Chen, F.; Geng, Z.; Cui, R.; Zhao, Y.; Liu, C. Self-assembling RATEA16 peptide nanofiber designed for rapid hemostasis. J. Mater. Chem. B 2020, 8, 1897–1905. [Google Scholar] [CrossRef]
- Alimohammadi, M.; Aghli, Y.; Fakhraei, O.; Moradi, A.; Passandideh-Fard, M.; Ebrahimzadeh, M.H.; Khademhosseini, A.; Tamayol, A.; Mousavi Shaegh, S.A. Electrospun Nanofibrous Membranes for Preventing Tendon Adhesion. ACS Biomater. Sci. Eng. 2020, 6, 4356–4376. [Google Scholar] [CrossRef]
- Chen, C.H.; Cheng, Y.H.; Chen, S.H.; Chuang, A.D.; Chen, J.P. Functional Hyaluronic Acid-Polylactic Acid/Silver Nanoparticles Core-Sheath Nanofiber Membranes for Prevention of Post-Operative Tendon Adhesion. Int. J. Mol. Sci. 2021, 22, 8781. [Google Scholar] [CrossRef]
- Chen, S.H.; Chen, C.H.; Shalumon, K.T.; Chen, J.P. Preparation and characterization of antiadhesion barrier film from hyaluronic acid-grafted electrospun poly(caprolactone) nanofibrous membranes for prevention of flexor tendon postoperative peritendinous adhesion. Int. J. Nanomed. 2014, 9, 4079–4092. [Google Scholar] [CrossRef]
- Jiang, S.; Yan, H.; Fan, D.; Song, J.; Fan, C. Multi-layer electrospun membrane mimicking tendon sheath for prevention of tendon adhesions. Int. J. Mol. Sci. 2015, 16, 6932–6944. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Ma, K.; Lam, C.-H.; Bei, H.-P.; Liu, Y.; Yang, X.; Zhao, X. Micro- and nano-environment dual-modulated anti-tendon adhesion barrier membranes. Mater. Des. 2022, 219, 110737. [Google Scholar] [CrossRef]
- Chen, C.H.; Chen, S.H.; Chen, S.H.; Chuang, A.D.; Darshan, T.G.; Chen, J.P. Hyaluronic acid/platelet rich plasma-infused core-shell nanofiber membrane to prevent postoperative tendon adhesion and promote tendon healing. Int. J. Biol. Macromol. 2023, 231, 123312. [Google Scholar] [CrossRef]
- Chen, J.; Sheng, D.; Ying, T.; Zhao, H.; Zhang, J.; Li, Y.; Xu, H.; Chen, S. MOFs-Based Nitric Oxide Therapy for Tendon Regeneration. Nanomicro Lett. 2020, 13, 23. [Google Scholar] [CrossRef]
- Fernandez-Yague, M.A.; Trotier, A.; Demir, S.; Abbah, S.A.; Larrañaga, A.; Thirumaran, A.; Stapleton, A.; Tofail, S.A.M.; Palma, M.; Kilcoyne, M.; et al. A Self-Powered Piezo-Bioelectric Device Regulates Tendon Repair-Associated Signaling Pathways through Modulation of Mechanosensitive Ion Channels. Adv. Mater. 2021, 33, e2008788. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Lu, L.; Yang, Q.; Zupanic, A.; Xu, Q.; Jiang, L. Using MoS2 Nanomaterials to Generate or Remove Reactive Oxygen Species: A Review. ACS Appl. Nano Mater. 2021, 4, 7523–7537. [Google Scholar] [CrossRef]
- Kim, T.I.; Kwon, B.; Yoon, J.; Park, I.J.; Bang, G.S.; Park, Y.; Seo, Y.S.; Choi, S.Y. Antibacterial Activities of Graphene Oxide-Molybdenum Disulfide Nanocomposite Films. ACS Appl. Mater. Interfaces 2017, 9, 7908–7917. [Google Scholar] [CrossRef] [PubMed]
- Jose, G.; Shalumon, K.T.; Chen, J.P. Natural Polymers Based Hydrogels for Cell Culture Applications. Curr. Med. Chem. 2020, 27, 2734–2776. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Mo, Q.; Sheng, R.; Zhu, A.; Ling, C.; Luo, Y.; Zhang, A.; Chen, Z.; Yao, Q.; Cai, Z.; et al. The application of human periodontal ligament stem cells and biomimetic silk scaffold for in situ tendon regeneration. Stem. Cell Res. Ther. 2021, 12, 596. [Google Scholar] [CrossRef]
- Yang, S.; Shi, X.; Li, X.; Wang, J.; Wang, Y.; Luo, Y. Oriented collagen fiber membranes formed through counter-rotating extrusion and their application in tendon regeneration. Biomaterials 2019, 207, 61–75. [Google Scholar] [CrossRef]
- Nikolova, M.P.; Chavali, M.S. Recent advances in biomaterials for 3D scaffolds: A review. Bioact. Mater. 2019, 4, 271–292. [Google Scholar] [CrossRef]
- Chen, S.; Wang, J.; Chen, Y.; Mo, X.; Fan, C. Tenogenic adipose-derived stem cell sheets with nanoyarn scaffolds for tendon regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 119, 111506. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Yang, C.; Ma, Z.; Wei, X.; Younis, M.R.; Wang, H.; Li, W.; Wang, Z.; Wang, W.; Luo, Y.; et al. Multiscale Hierarchical Architecture-Based Bioactive Scaffolds for Versatile Tissue Engineering. Adv. Heal. Mater. 2022, 11, e2102837. [Google Scholar] [CrossRef]
- Jiang, C.; Wang, K.; Liu, Y.; Zhang, C.; Wang, B. Application of textile technology in tissue engineering: A review. Acta Biomater. 2021, 128, 60–76. [Google Scholar] [CrossRef] [PubMed]
- Linderman, S.W.; Golman, M.; Gardner, T.R.; Birman, V.; Levine, W.N.; Genin, G.M.; Thomopoulos, S. Enhanced tendon-to-bone repair through adhesive films. Acta Biomater. 2018, 70, 165–176. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, Y.; Suo, D.; Zhao, S.; Cheung, J.C.; Leung, P.H.; Zhao, X. A Biomimetic Adhesive and Robust Janus Patch with Anti-Oxidative, Anti-Inflammatory, and Anti-Bacterial Activities for Tendon Repair. ACS Nano 2023, 17, 16798–16816. [Google Scholar] [CrossRef] [PubMed]
- Balkenende, D.W.R.; Winkler, S.M.; Messersmith, P.B. Marine-Inspired Polymers in Medical Adhesion. Eur. Polym. J. 2019, 116, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Chou, P.Y.; Chen, S.H.; Chen, C.H.; Chen, S.H.; Fong, Y.T.; Chen, J.P. Thermo-responsive in-situ forming hydrogels as barriers to prevent post-operative peritendinous adhesion. Acta Biomater. 2017, 63, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Barbash, S.; Denny, C.; Collin, P.; Reish, T.; Hart, J.; Brockmeier, S. Clinical Outcomes and Structural Healing After Arthroscopic Rotator Cuff Repair Reinforced With A Novel Absorbable Biologic Scaffold: A Prospective, Multicenter Trial. Orthop. J. Sports Med. 2015, 3 (Suppl. S2), 2325967115S00071. [Google Scholar] [CrossRef]
- Ferreira De Barros, A.A. Bioinductive Scafold Augmentation in Complete and Massive Rotator Cuff Tears—Prospective Randomized Trial. J. Shoulder Elb. Surg. 2023, 32, e251. [Google Scholar] [CrossRef]
- Beleckas, C.M.; Bishai, S.K.; Badman, B.L. Rotator Cuff Repair Augmented With Interpositional Nanofiber Scaffold. Arthrosc. Tech. 2023, 12, e77–e81. [Google Scholar] [CrossRef]
- Seetharam, A.; Abad, J.; Baessler, A.; Badman, B.L. Use of a Nanofiber Resorbable Scaffold During Rotator Cuff Repair: Surgical Technique and Results After Repair of Small- to Medium-Sized Tears. Orthop. J. Sports Med. 2022, 10, 23259671221094848. [Google Scholar] [CrossRef]
- Beleckas, C.M.; Minetos, P.D.; Badman, B.L. Short-term radiographic and clinical outcomes of arthroscopic rotator cuff repair with and without augmentation with an interpositional nanofiber scaffold. J. Orthop. Exp. Innov. 2023, 4. [Google Scholar] [CrossRef]
- Cai, Y.Z.; Zhang, C.; Jin, R.L.; Shen, T.; Gu, P.C.; Lin, X.J.; Chen, J.D. Arthroscopic Rotator Cuff Repair With Graft Augmentation of 3-Dimensional Biological Collagen for Moderate to Large Tears: A Randomized Controlled Study. Am. J. Sports Med. 2018, 46, 1424–1431. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xue, Y.; Ren, Y.; Li, X.; Liu, Y. Biodegradable Polymer Electrospinning for Tendon Repairment. Polymers 2023, 15, 1566. [Google Scholar] [CrossRef]
- TAPESTRY, Embody, Inc. Available online: https://embody-inc.com/ (accessed on 8 May 2024).
- GTR® Medical Collagen Repair Membrane. Available online: https://www.gtrbio.cn/ (accessed on 8 May 2024).
- TenoMend® Collagen Tendon Wrap. Available online: https://www.exac.com/ (accessed on 8 May 2024).
- GYNECARE INTERCEED™ Absorbable Adhesion Barrier. Available online: https://www.jnjmedtech.com/en-US/product/gynecare-interceed-absorbable-adhesion-barrier (accessed on 8 May 2024).
- Seprafilm®, Baxter. Available online: https://advancedsurgery.baxter.co.kr/resource-library/seprafilm (accessed on 8 May 2024).
- Wang, Y.; Xu, Y.; Zhai, W.; Zhang, Z.; Liu, Y.; Cheng, S.; Zhang, H. In-situ growth of robust superlubricated nano-skin on electrospun nanofibers for post-operative adhesion prevention. Nat. Commun. 2022, 13, 5056. [Google Scholar] [CrossRef] [PubMed]
- Maghdouri-White, Y.; Sori, N.; Petrova, S.; Wriggers, H.; Kemper, N.; Dasgupta, A.; Coughenour, K.; Polk, S.; Thayer, N.; Mario, R.D.; et al. Biomanufacturing organized collagen-based microfibers as a Tissue ENgineered Device (TEND) for tendon regeneration. Biomed. Mater. 2021, 16, 025025. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.H.; Arcidiacono, J.A.; Bilek, A.M.; Wille, J.J.; Hamill, C.A.; Wonnacott, K.M.; Wells, M.A.; Oh, S.S. Considerations for tissue-engineered and regenerative medicine product development prior to clinical trials in the United States. Tissue Eng. Part B Rev. 2010, 16, 41–54. [Google Scholar] [CrossRef]
- Saed, G.M.; Munkarah, A.R.; Diamond, M.P. Cyclooxygenase-2 is expressed in human fibroblasts isolated from intraperitoneal adhesions but not from normal peritoneal tissues. Fertil. Steril. 2003, 79, 1404–1408. [Google Scholar] [CrossRef]
- Belsky, K.; Smiell, J. Navigating the Regulatory Pathways and Requirements for Tissue-Engineered Products in the Treatment of Burns in the United States. J. Burn. Care Res. 2021, 42, 774–784. [Google Scholar] [CrossRef]

| Physicochemical Parameters | Optimal Range | Importance |
|---|---|---|
| Fiber Diameter | 10–500 nm [70] | Influences surface area-to-volume ratio, cell attachment, and mechanical properties. |
| Porosity | 80–90 % [67,68] | Affects nutrient diffusion, waste removal, and cell infiltration. |
| Pore Size | 6–20 μm [68,71] | Essential for tissue integration and cell migration. |
| Surface Hydrophilicity | Water contact angle 35°–60° [65,72,73] | Enhances cell attachment and proliferation. |
| Mechanical Properties | Tensile Strength: 4.4–660 MPa [74,75] Elastic Modulus: 200–1500 MPa [74,75,76] Strain: ~35% [77] | Ensures scaffold integrity and mimics native tissue mechanics. |
| Biocompatibility | Nontoxic, nonimmunogenic [70] | Ensures safe integration with host tissue. |
| Author | Study | Scaffold Material | Study Type | Main Findings |
|---|---|---|---|---|
| Barbash et al. [112] | Clinical Outcomes and Structural Healing After Arthroscopic Rotator Cuff Repair Reinforced With A Novel Absorbable Biologic Scaffold | BioFiber, bi-layer absorbable scaffold | Prospective, multicenter clinical trial | Improved repair integrity and functional outcomes, 96% repair success rate at 6 months |
| Ferreira De Barros [113] | Bioinductive Scaffold Augmentation in Complete and Massive Rotator Cuff Tears | Bioinductive porcine collagen scaffold | Randomized trial | Significant improvements in functional scores, reduced retear rate |
| Beleckas et al. [114] | Rotator Cuff Repair Augmented With Interpositional Nanofiber Scaffold | Rotium wick, interpositional nanofiber scaffold | Study with technical note | Enhanced cellular organization and tendon strength, 91% tendon healing rate |
| Seetharam et al. [115] | Use of a Nanofiber Resorbable Scaffold During Rotator Cuff Repair | Rotium wick, interpositional nanofiber scaffold | Case series | Significant improvements in functional outcomes, high rate of tendon healing in small to medium RC tears |
| Beleckas et al. [116] | Short-Term Radiographic and Clinical Outcomes of Arthroscopic Rotator Cuff Repair with and without Augmentation with an Interpositional Nanofiber Scaffold | Interpositional nanofiber scaffold | Case series | Improved radiographic and clinical outcomes |
| Cai et al. [117] | Arthroscopic Rotator Cuff Repair With Graft Augmentation of Three-Dimensional Biological Collagen for Moderate to Large Tears | 3D biological collagen | Randomized controlled study | Significant improvements in functional scores, reduced retear rate to 13.7%, better tendon-bone healing |
| Product | Company | Compositions | Applications |
|---|---|---|---|
| TAPESTRY® [119] | Embody, Inc. (Norfolk, VA, USA) | Collagen and PDLA | Tendon and ligament healing |
| GTR® [120] | GTR BioTech. Co., Ltd. (Fuzhou, China) | Collagen separated from bovine tendon tissue. | Tendon healing |
| TenoMed® [121] | Exactech, Inc. (Gainesville, FL, USA) | Absorbable type I collagen matrix | tendon healing and provide a sliding surface |
| Interceed® [122] | Johnson & Johnson MedTech Co. (New Brunswick, NJ, USA) | Oxidized regenerated cellulose | Tendon and abdominal adhesion prevention and protective coating |
| Seprafilm® [123] | Baxter International Inc. (Deerfield, IL, USA) | HA and carboxymethylcellulose (CMC) based | Abdominal and pelvic adhesion prevention |
| DK-film® [124] | Chengdu Dickon Pharmaceutical Co. (Chengdu, China) | PLA based | Tendon and abdominal adhesion prevention |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoon, J.P.; Kim, H.; Park, S.-J.; Kim, D.-H.; Kim, J.-Y.; Kim, D.H.; Chung, S.W. Nanofiber Graft Therapy to Prevent Shoulder Stiffness and Adhesions after Rotator Cuff Tendon Repair: A Comprehensive Review. Biomedicines 2024, 12, 1613. https://doi.org/10.3390/biomedicines12071613
Yoon JP, Kim H, Park S-J, Kim D-H, Kim J-Y, Kim DH, Chung SW. Nanofiber Graft Therapy to Prevent Shoulder Stiffness and Adhesions after Rotator Cuff Tendon Repair: A Comprehensive Review. Biomedicines. 2024; 12(7):1613. https://doi.org/10.3390/biomedicines12071613
Chicago/Turabian StyleYoon, Jong Pil, Hyunjin Kim, Sung-Jin Park, Dong-Hyun Kim, Jun-Young Kim, Du Han Kim, and Seok Won Chung. 2024. "Nanofiber Graft Therapy to Prevent Shoulder Stiffness and Adhesions after Rotator Cuff Tendon Repair: A Comprehensive Review" Biomedicines 12, no. 7: 1613. https://doi.org/10.3390/biomedicines12071613
APA StyleYoon, J. P., Kim, H., Park, S.-J., Kim, D.-H., Kim, J.-Y., Kim, D. H., & Chung, S. W. (2024). Nanofiber Graft Therapy to Prevent Shoulder Stiffness and Adhesions after Rotator Cuff Tendon Repair: A Comprehensive Review. Biomedicines, 12(7), 1613. https://doi.org/10.3390/biomedicines12071613