Exploring Motor Network Connectivity in State-Dependent Transcranial Magnetic Stimulation: A Proof-of-Concept Study
Abstract
1. Introduction
2. Material and Methods
2.1. Participants and Experiment
2.2. Data Processing
2.3. Source Estimation
2.4. Connectivity Analysis
2.5. Relation between Functional Connectivity and Motor-Evoked Potential
2.6. Coupling Directionality
2.7. Phase Estimation
2.8. Linear Regression Analysis
3. Results
3.1. Functional Connectivity at the μ-Rhythm Frequency Highlights Coupling within the Motor Network
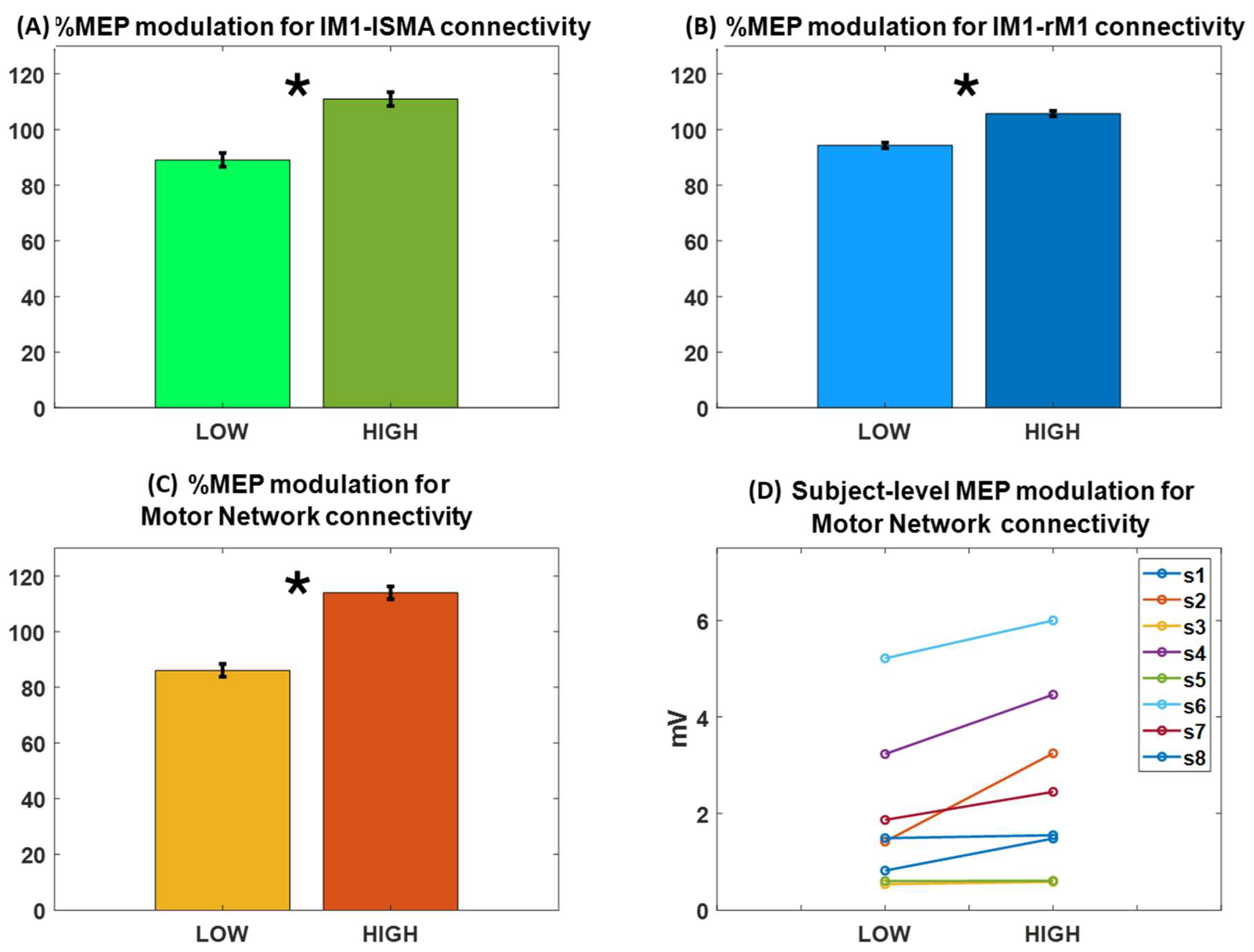
3.2. MEP Amplitude Modulates with Functional Connectivity of the Motor Network
3.3. Coupling Directionality Reveals the Top-Down Control of SMA on Bilateral M1
3.4. A Linear Regression Model That Relies on Network Connectivity and the lM1 Phase Best Predicts MEP
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hallett, M. Transcranial Magnetic Stimulation: A Primer. Neuron 2007, 55, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Hallett, M. Transcranial magnetic stimulation and the human brain. Nature 2000, 406, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Rossini, P.M.; Burke, D.; Chen, R.; Cohen, L.G.; Daskalakis, Z.; Di Iorio, R.; Di Lazzaro, V.; Ferreri, F.; Fitzgerald, P.B.; George, M.S.; et al. Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: Basic principles and procedures for routine clinical and research application. An updated report from an I.F.C.N. Committee. Clin. Neurophysiol. 2015, 126, 1071–1107. [Google Scholar] [CrossRef] [PubMed]
- Corp, D.T.; Bereznicki, H.G.K.; Clark, G.M.; Youssef, G.J.; Fried, P.J.; Jannati, A.; Davies, C.B.; Gomes-Osman, J.; Kirkovski, M.; Albein-Urios, N.; et al. Large-scale analysis of interindividual variability in single and paired-pulse TMS data. Clin. Neurophysiol. 2021, 132, 2639–2653. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, T.O. Brain State-Dependent Brain Stimulation. Front. Psychol. 2018, 9, 2108. [Google Scholar] [CrossRef] [PubMed]
- Silvanto, J.; Muggleton, N.; Walsh, V. State-dependency in brain stimulation studies of perception and cognition. Trends Cogn. Sci. 2008, 12, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Silvanto, J.; Pascual-Leone, A. State-dependency of transcranial magnetic stimulation. Brain Topogr. 2008, 21, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Hartwigsen, G.; Silvanto, J. Noninvasive Brain Stimulation: Multiple Effects on Cognition. Neuroscientist 2023, 29, 639–653. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, T.O.; Karabanov, A.; Hartwigsen, G.; Thielscher, A.; Siebner, H.R. Combining non-invasive transcranial brain stimulation with neuroimaging and electrophysiology: Current approaches and future perspectives. NeuroImage 2016, 140, 4–19. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.W.; Rogasch, N.C.; Hoy, K.E.; Fitzgerald, P.B. Measuring Brain Stimulation Induced Changes in Cortical Properties Using TMS-EEG. Brain Stimul. 2015, 8, 1010–1020. [Google Scholar] [CrossRef] [PubMed]
- Zrenner, C.; Desideri, D.; Belardinelli, P.; Ziemann, U. Real-time EEG-defined excitability states determine efficacy of TMS-induced plasticity in human motor cortex. Brain Stimul. 2018, 11, 374–389. [Google Scholar] [CrossRef] [PubMed]
- Mäki, H.; Ilmoniemi, R.J. EEG oscillations and magnetically evoked motor potentials reflect motor system excitability in overlapping neuronal populations. Clin. Neurophysiol. 2010, 121, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Schilberg, L.; Ten Oever, S.; Schuhmann, T.; Sack, A.T. Phase and power modulations on the amplitude of TMS-induced motor evoked potentials. PLoS ONE 2021, 16, e0255815. [Google Scholar] [CrossRef] [PubMed]
- Wischnewski, M.; Haigh, Z.J.; Shirinpour, S.; Alekseichuk, I.; Opitz, A. The phase of sensorimotor mu and beta oscillations has the opposite effect on corticospinal excitability. Brain Stimul. 2022, 15, 1093–1100. [Google Scholar] [CrossRef] [PubMed]
- Karabanov, A.N.; Madsen, K.H.; Krohne, L.G.; Siebner, H.R. Does pericentral mu-rhythm “power” corticomotor excitability?—A matter of EEG perspective. Brain Stimul. 2021, 14, 713–722. [Google Scholar] [CrossRef]
- Madsen, K.H.; Karabanov, A.N.; Krohne, L.G.; Safeldt, M.G.; Tomasevic, L.; Siebner, H.R. No trace of phase: Corticomotor excitability is not tuned by phase of pericentral mu-rhythm. Brain Stimul. 2019, 12, 1261–1270. [Google Scholar] [CrossRef] [PubMed]
- Magnuson, J.; Ozdemir, M.A.; Mathieson, E.; Kirkman, S.; Passera, B.; Rampersad, S.; Dufour, A.B.; Brooks, D.; Pascual-Leone, A.; Fried, P.J.; et al. Neuromodulatory effects and reproducibility of the most widely used repetitive transcranial magnetic stimulation protocols. PLoS ONE 2023, 18, e0286465. [Google Scholar] [CrossRef] [PubMed]
- Bressler, S.L.; Menon, V. Large-scale brain networks in cognition: Emerging methods and principles. Trends Cogn. Sci. 2010, 14, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Fox, M.D.; Raichle, M.E. Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nat. Rev. Neurosci. 2007, 8, 700–711. [Google Scholar] [CrossRef]
- Brookes, M.J.; Hale, J.R.; Zumer, J.M.; Stevenson, C.M.; Francis, S.T.; Barnes, G.R.; Owen, J.P.; Morris, P.G.; Nagarajan, S.S. Measuring functional connectivity using MEG: Methodology and comparison with fcMRI. Neuroimage 2011, 56, 1082–1104. [Google Scholar] [CrossRef] [PubMed]
- Brookes, M.J.; Woolrich, M.; Luckhoo, H.; Price, D.; Hale, J.R.; Stephenson, M.C.; Barnes, G.R.; Smith, S.M.; Morris, P.G. Investigating the electrophysiological basis of resting state networks using magnetoencephalography. Proc. Natl. Acad. Sci. USA 2011, 108, 16783–16788. [Google Scholar] [CrossRef] [PubMed]
- Brookes, M.J.; O’Neill, G.C.; Hall, E.L.; Woolrich, M.W.; Baker, A.; Palazzo Corner, S.; Robson, S.E.; Morris, P.G.; Barnes, G.R. Measuring temporal, spectral and spatial changes in electrophysiological brain network connectivity. Neuroimage 2014, 91, 282–299. [Google Scholar] [CrossRef] [PubMed]
- Arzate-Mena, J.D.; Abela, E.; Olguín-Rodríguez, P.V.; Ríos-Herrera, W.; Alcauter, S.; Schindler, K.; Wiest, R.; Müller, M.F.; Rummel, C. Stationary EEG pattern relates to large-scale resting state networks—An EEG-fMRI study connecting brain networks across time-scales. Neuroimage 2022, 246, 118763. [Google Scholar] [CrossRef] [PubMed]
- Engel, A.K.; Gerloff, C.; Hilgetag, C.C.; Nolte, G. Intrinsic Coupling Modes: Multiscale Interactions in Ongoing Brain Activity. Neuron 2013, 80, 867–886. [Google Scholar] [CrossRef] [PubMed]
- Ganzetti, M.; Mantini, D. Functional connectivity and oscillatory neuronal activity in the resting human brain. Neuroscience 2013, 240, 297–309. [Google Scholar] [CrossRef] [PubMed]
- Marzetti, L.; Basti, A.; Chella, F.; D’Andrea, A.; Syrjälä, J.; Pizzella, V. Brain Functional Connectivity through Phase Coupling of Neuronal Oscillations: A Perspective from Magnetoencephalography. Front. Neurosci. 2019, 13, 964. [Google Scholar] [CrossRef] [PubMed]
- Marzetti, L.; Della Penna, S.; Snyder, A.Z.; Pizzella, V.; Nolte, G.; de Pasquale, F.; Romani, G.L.; Corbetta, M. Frequency specific interactions of MEG resting state activity within and across brain networks as revealed by the multivariate interaction measure. NeuroImage 2013, 79, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Bastos, A.M.; Vezoli, J.; Fries, P. Communication through coherence with inter-areal delays. Curr. Opin. Neurobiol. 2015, 31, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Fries, P. Rhythms For Cognition: Communication through Coherence. Neuron 2015, 88, 220–235. [Google Scholar] [CrossRef] [PubMed]
- Fries, P. A mechanism for cognitive dynamics: Neuronal communication through neuronal coherence. Trends Cogn. Sci. 2005, 9, 474–480. [Google Scholar] [CrossRef]
- Kringelbach, M.L.; Deco, G. Brain States and Transitions: Insights from Computational Neuroscience. Cell Rep. 2020, 32, 108128. [Google Scholar] [CrossRef] [PubMed]
- Deco, G.; Corbetta, M. The Dynamical Balance of the Brain at Rest. Neuroscientist 2011, 17, 107–123. [Google Scholar] [CrossRef] [PubMed]
- Beynel, L.; Powers, J.P.; Appelbaum, L.G. Effects of repetitive transcranial magnetic stimulation on resting-state connectivity: A systematic review. NeuroImage 2020, 211, 116596. [Google Scholar] [CrossRef] [PubMed]
- Boutet, A.; Jain, M.; Elias, G.J.B.; Gramer, R.; Germann, J.; Davidson, B.; Coblentz, A.; Giacobbe, P.; Kucharczyk, W.; Wennberg, R.A.; et al. Network Basis of Seizures Induced by Deep Brain Stimulation: Literature Review and Connectivity Analysis. World Neurosurg. 2019, 132, 314–320. [Google Scholar] [CrossRef]
- Pieramico, G.; Guidotti, R.; Nieminen, A.E.; D’Andrea, A.; Basti, A.; Souza, V.H.; Nieminen, J.O.; Lioumis, P.; Ilmoniemi, R.J.; Romani, G.L.; et al. TMS-Induced Modulation of EEG Functional Connectivity Is Affected by the E-Field Orientation. Brain Sci. 2023, 13, 418. [Google Scholar] [CrossRef] [PubMed]
- Cheyuo, C.; Germann, J.; Yamamoto, K.; Vetkas, A.; Loh, A.; Sarica, C.; Milano, V.; Zemmar, A.; Flouty, O.; Harmsen, I.E.; et al. Connectomic neuromodulation for Alzheimer’s disease: A systematic review and meta-analysis of invasive and non-invasive techniques. Transl. Psychiatry 2022, 12, 490. [Google Scholar] [CrossRef] [PubMed]
- Horn, A.; Fox, M.D. Opportunities of connectomic neuromodulation. NeuroImage 2020, 221, 117180. [Google Scholar] [CrossRef] [PubMed]
- Ozdemir, R.A.; Tadayon, E.; Boucher, P.; Momi, D.; Karakhanyan, K.A.; Fox, M.D.; Halko, M.A.; Pascual-Leone, A.; Shafi, M.M.; Santarnecchi, E. Individualized perturbation of the human connectome reveals reproducible biomarkers of network dynamics relevant to cognition. Proc. Natl. Acad. Sci. USA 2020, 117, 8115–8125. [Google Scholar] [CrossRef]
- Ozdemir, R.A.; Kirkman, S.; Magnuson, J.R.; Fried, P.J.; Pascual-Leone, A.; Shafi, M.M. Phase matters when there is power: Phasic modulation of corticospinal excitability occurs at high amplitude sensorimotor mu-oscillations. Neuroimage Rep. 2022, 2, 100132. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, S.H.; Taylor, S.F.; Cooke, D.; Pascual-Leone, A.; George, M.S.; Fox, M.D. Distinct Symptom-Specific Treatment Targets for Circuit-Based Neuromodulation. Am. J. Psychiatry 2020, 177, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, S.H.; Kording, K.P.; Parvizi, J.; Fox, M.D. Causal mapping of human brain function. Nat. Rev. Neurosci. 2022, 23, 361–375. [Google Scholar] [CrossRef] [PubMed]
- Ferreri, F.; Vecchio, F.; Ponzo, D.; Pasqualetti, P.; Rossini, P.M. Time-varying coupling of EEG oscillations predicts excitability fluctuations in the primary motor cortex as reflected by motor evoked potentials amplitude: An EEG-TMS study. Hum. Brain Mapp. 2014, 35, 1969–1980. [Google Scholar] [CrossRef] [PubMed]
- Vetter, D.E.; Zrenner, C.; Belardinelli, P.; Mutanen, T.P.; Kozák, G.; Marzetti, L.; Ziemann, U. Targeting motor cortex high-excitability states defined by functional connectivity with real-time EEG–TMS. NeuroImage 2023, 284, 120427. [Google Scholar] [CrossRef] [PubMed]
- Marzetti, L.; Makkinayeri, S.; Pieramico, G.; Guidotti, R.; D’Andrea, A.; Roine, T.; Mutanen, T.P.; Souza, V.H.; Kičić, D.; Baldassarre, A.; et al. Towards real-time identification of large-scale brain states for improved brain state-dependent stimulation. Clin. Neurophysiol. 2024, 158, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Sinisalo, H.; Rissanen, I.; Kahilakoski, O.P.; Souza, V.H.; Tommila, T.; Laine, M.; Nyrhinen, M.; Ukharova, E.; Granö, I.; Soto, A.M.; et al. Modulating brain networks in space and time: Multi-locus transcranial magnetic stimulation. Clin. Neurophysiol. 2024, 158, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Metsomaa, J.; Belardinelli, P.; Ermolova, M.; Ziemann, U.; Zrenner, C. Causal decoding of individual cortical excitability states. NeuroImage 2021, 245, 118652. [Google Scholar] [CrossRef] [PubMed]
- Hari, R. Action–perception connection and the cortical mu rhythm. In Progress in Brain Research; Elsevier: Amsterdam, The Netherlands, 2006; pp. 253–260. [Google Scholar] [CrossRef]
- Hyvarinen, A. Fast and robust fixed-point algorithms for independent component analysis. IEEE Trans. Neural Netw. 1999, 10, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Pascual-Marqui, R.D.; Lehmann, D.; Koukkou, M.; Kochi, K.; Anderer, P.; Saletu, B.; Tanaka, H.; Hirata, K.; John, E.R.; Prichep, L.; et al. Assessing interactions in the brain with exact low-resolution electromagnetic tomography. Philos. Trans. R. Soc. Math. Phys. Eng. Sci. 2011, 369, 3768–3784. [Google Scholar] [CrossRef] [PubMed]
- Oostenveld, R.; Fries, P.; Maris, E.; Schoffelen, J.-M. FieldTrip: Open Source Software for Advanced Analysis of MEG, EEG, and Invasive Electrophysiological Data. Comput. Intell. Neurosci. 2011, 2011, 156869. [Google Scholar] [CrossRef] [PubMed]
- Dykstra, A.R.; Chan, A.M.; Quinn, B.T.; Zepeda, R.; Keller, C.J.; Cormier, J.; Madsen, J.R.; Eskandar, E.N.; Cash, S.S. Individualized localization and cortical surface-based registration of intracranial electrodes. NeuroImage 2012, 59, 3563–3570. [Google Scholar] [CrossRef] [PubMed]
- Palva, S.; Palva, J.M. Discovering oscillatory interaction networks with M/EEG: Challenges and breakthroughs. Trends Cogn. Sci. 2012, 16, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Basti, A.; Pizzella, V.; Chella, F.; Romani, G.L.; Nolte, G.; Marzetti, L. Disclosing large-scale directed functional connections in MEG with the multivariate phase slope index. NeuroImage 2018, 175, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Nolte, G.; Ziehe, A.; Nikulin, V.V.; Schlögl, A.; Krämer, N.; Brismar, T.; Müller, K.R. Robustly estimating the flow direction of information in complex physical systems. Phys. Rev. Lett. 2008, 100, 234101. [Google Scholar] [CrossRef] [PubMed]
- Ewald, A.; Avarvand, F.S.; Nolte, G. Identifying causal networks of neuronal sources from EEG/MEG data with the phase slope index: A simulation study. Biomed. Tech. 2013, 58, 165–178. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, D.P. Robust regression computation using iteratively reweighted least squares. SIAM J. Matrix Anal. Appl. 1990, 11, 466–480. [Google Scholar] [CrossRef]
- Akaike, H. Information Theory and an Extension of the Maximum Likelihood Principle. In Selected Papers of Hirotugu Akaike; Parzen, E., Tanabe, K., Kitagawa, G., Eds.; Springer Series in Statistics; Springer: New York, NY, USA, 1998; pp. 199–213. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. (Eds.) Model Selection and Multimodel Inference; Springer: New York, NY, USA, 2004. [Google Scholar] [CrossRef]
- Marino, M.; Liu, Q.; Samogin, J.; Tecchio, F.; Cottone, C.; Mantini, D.; Porcaro, C. Neuronal dynamics enable the functional differentiation of resting state networks in the human brain. Hum. Brain Mapp. 2019, 40, 1445–1457. [Google Scholar] [CrossRef] [PubMed]
- Samogin, J.; Marino, M.; Porcaro, C.; Wenderoth, N.; Dupont, P.; Swinnen, S.P.; Mantini, D. Frequency-dependent functional connectivity in resting state networks. Hum. Brain Mapp. 2020, 41, 5187–5198. [Google Scholar] [CrossRef] [PubMed]
- Arai, N.; Müller-Dahlhaus, F.; Murakami, T.; Bliem, B.; Lu, M.-K.; Ugawa, Y.; Ziemann, U. State-Dependent and Timing-Dependent Bidirectional Associative Plasticity in the Human SMA-M1 Network. J. Neurosci. 2011, 31, 15376–15383. [Google Scholar] [CrossRef] [PubMed]
- Cárdenas-Morales, L.; Volz, L.J.; Michely, J.; Rehme, A.K.; Pool, E.-M.; Nettekoven, C.; Eickhoff, S.B.; Fink, G.R.; Grefkes, C. Network Connectivity and Individual Responses to Brain Stimulation in the Human Motor System. Cereb. Cortex 2014, 24, 1697–1707. [Google Scholar] [CrossRef] [PubMed]
- Pool, E.-M.; Rehme, A.K.; Fink, G.R.; Eickhoff, S.B.; Grefkes, C. Network dynamics engaged in the modulation of motor behavior in healthy subjects. NeuroImage 2013, 82, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Grefkes, C.; Nowak, D.A.; Wang, L.E.; Dafotakis, M.; Eickhoff, S.B.; Fink, G.R. Modulating cortical connectivity in stroke patients by rTMS assessed with fMRI and dynamic causal modeling. NeuroImage 2010, 50, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Chen, J.; Hu, X.; Hu, G.; Liu, Y.; Yang, K.; Xiao, C.; Zou, Y.; Liu, H. Contralesional homotopic functional plasticity in patients with temporal glioma. J. Neurosurg. 2021, 134, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Carter, A.R.; Astafiev, S.V.; Lang, C.E.; Connor, L.T.; Rengachary, J.; Strube, M.J.; Pope, D.L.W.; Shulman, G.L.; Corbetta, M. Resting Inter-hemispheric fMRI Connectivity Predicts Performance after Stroke. Ann. Neurol. 2010, 67, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Diekhoff-Krebs, S.; Pool, E.-M.; Sarfeld, A.-S.; Rehme, A.K.; Eickhoff, S.B.; Fink, G.R.; Grefkes, C. Interindividual differences in motor network connectivity and behavioral response to iTBS in stroke patients. NeuroImage Clin. 2017, 15, 559–571. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, A.; Kim, H.; Shin, M.; Yun, S.M.; Jung, Y.; Chang, W.H.; Kim, Y.H. Different Brain Connectivity between Responders and Nonresponders to Dual-Mode Noninvasive Brain Stimulation over Bilateral Primary Motor Cortices in Stroke Patients. Neural Plast. 2019, 2019, 3826495. [Google Scholar] [CrossRef] [PubMed]
- Anwar, A.R.; Muthalib, M.; Perrey, S.; Galka, A.; Granert, O.; Wolff, S.; Deuschl, G.; Raethjen, J.; Heute, U.; Muthuraman, M. Directionality analysis on functional magnetic resonance imaging during motor task using Granger causality. In Proceedings of the 2012 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Diego, CA, USA, 28 August–1 September 2012; pp. 2287–2290. [Google Scholar] [CrossRef]
- Grefkes, C.; Eickhoff, S.B.; Nowak, D.A.; Dafotakis, M.; Fink, G.R. Dynamic intra- and interhemispheric interactions during unilateral and bilateral hand movements assessed with fMRI and DCM. NeuroImage 2008, 41, 1382–1394. [Google Scholar] [CrossRef] [PubMed]
- Stefanou, M.-I.; Desideri, D.; Belardinelli, P.; Zrenner, C.; Ziemann, U. Phase Synchronicity of μ-Rhythm Determines Efficacy of Interhemispheric Communication Between Human Motor Cortices. J. Neurosci. 2018, 38, 10525–10534. [Google Scholar] [CrossRef] [PubMed]
- Sommariva, S.; Sorrentino, A.; Piana, M.; Pizzella, V.; Marzetti, L. A Comparative Study of the Robustness of Frequency-Domain Connectivity Measures to Finite Data Length. Brain Topogr. 2019, 32, 675–695. [Google Scholar] [CrossRef]
- Basti, A.; Chella, F.; Guidotti, R.; Ermolova, M.; D’Andrea, A.; Stenroos, M.; Romani, G.L.; Pizzella, V.; Marzetti, L. Looking through the windows: A study about the dependency of phase-coupling estimates on the data length. J. Neural Eng. 2022, 19, 016039. [Google Scholar] [CrossRef]
- Vidaurre, C.; Haufe, S.; Jorajuría, T.; Müller, K.R.; Nikulin, V.V. Sensorimotor Functional Connectivity: A Neurophysiological Factor Related to BCI Performance. Front. Neurosci. 2020, 14, 575081. [Google Scholar] [CrossRef] [PubMed]
- Momi, D.; Ozdemir, R.A.; Tadayon, E.; Boucher, P.; Shafi, M.M.; Pascual-Leone, A.; Santarnecchi, E. Network-level macroscale structural connectivity predicts propagation of transcranial magnetic stimulation. NeuroImage 2021, 229, 117698. [Google Scholar] [CrossRef] [PubMed]
- Vlachos, I.; Kugiumtzis, D.; Tsalikakis, D.G.; Kimiskidis, V.K. TMS-induced brain connectivity modulation in Genetic Generalized Epilepsy. Clin. Neurophysiol. 2022, 133, 83–93. [Google Scholar] [CrossRef] [PubMed]

| Subject # | AIC Constant Model | AIC lM1 Phase | AIC Motor Network | AIC Motor Network and lM1 Phase | Preferred Model |
|---|---|---|---|---|---|
| 1 | 2106, 5 | 2004, 8 | 2071, 6 | 1971, 0 | Motor Network and lM1 phase |
| 2 | 1416, 8 | 1414, 6 | 1299, 5 | 1298, 6 | Motor Network and lM1 phase |
| 3 | 1724, 4 | 1716, 9 | 1726, 9 | 1719, 1 | lM1 Phase |
| 4 | 1867, 0 | 1863, 6 | 1858, 4 | 1855, 5 | Motor Network and lM1 phase |
| 5 | 1931, 0 | 1933, 8 | 1932, 9 | 1935, 7 | Constant |
| 6 | 1823, 4 | 1830, 1 | 1822, 4 | 1824, 8 | Motor Network |
| 7 | 2178, 2 | 2180, 3 | 2173, 6 | 2176, 2 | Motor Network |
| 8 | 1626, 0 | 1622, 9 | 1602, 1 | 1597, 7 | Motor Network and lM1 phase |
| Subject # | Constant Model | lM1 Phase | Motor Network | Motor Network and lM1 Phase |
|---|---|---|---|---|
| 1 | Not plausible | Not plausible | Not plausible | Preferred |
| 2 | Not Plausible | Not Plausible | Plausible | Preferred |
| 3 | Not plausible | (Mildly) Preferred | Not plausible | Plausible |
| 4 | Not Plausible | Not Plausible | Plausible | Preferred |
| 5 | Preferred | Plausible | Plausible | Plausible |
| 6 | Plausible | Not Plausible | Preferred | Plausible |
| 7 | Not Plausible | Not Plausible | Preferred | Plausible |
| 8 | Not plausible | Not plausible | Not plausible | Preferred |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marzetti, L.; Basti, A.; Guidotti, R.; Baldassarre, A.; Metsomaa, J.; Zrenner, C.; D’Andrea, A.; Makkinayeri, S.; Pieramico, G.; Ilmoniemi, R.J.; et al. Exploring Motor Network Connectivity in State-Dependent Transcranial Magnetic Stimulation: A Proof-of-Concept Study. Biomedicines 2024, 12, 955. https://doi.org/10.3390/biomedicines12050955
Marzetti L, Basti A, Guidotti R, Baldassarre A, Metsomaa J, Zrenner C, D’Andrea A, Makkinayeri S, Pieramico G, Ilmoniemi RJ, et al. Exploring Motor Network Connectivity in State-Dependent Transcranial Magnetic Stimulation: A Proof-of-Concept Study. Biomedicines. 2024; 12(5):955. https://doi.org/10.3390/biomedicines12050955
Chicago/Turabian StyleMarzetti, Laura, Alessio Basti, Roberto Guidotti, Antonello Baldassarre, Johanna Metsomaa, Christoph Zrenner, Antea D’Andrea, Saeed Makkinayeri, Giulia Pieramico, Risto J. Ilmoniemi, and et al. 2024. "Exploring Motor Network Connectivity in State-Dependent Transcranial Magnetic Stimulation: A Proof-of-Concept Study" Biomedicines 12, no. 5: 955. https://doi.org/10.3390/biomedicines12050955
APA StyleMarzetti, L., Basti, A., Guidotti, R., Baldassarre, A., Metsomaa, J., Zrenner, C., D’Andrea, A., Makkinayeri, S., Pieramico, G., Ilmoniemi, R. J., Ziemann, U., Romani, G. L., & Pizzella, V. (2024). Exploring Motor Network Connectivity in State-Dependent Transcranial Magnetic Stimulation: A Proof-of-Concept Study. Biomedicines, 12(5), 955. https://doi.org/10.3390/biomedicines12050955







