NOS1AP Gene Variants and Their Role in Metabolic Syndrome: A Study of Patients with Schizophrenia
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects
- Either concentration of TGs above 1.7 mmol/L or use of lipid-lowering therapy.
- Concentration of high-density lipoprotein cholesterol (HDL-C) less than 1.03 mmol/L in males or 1.29 mmol/L in females.
- Either BP ≥ 130/85 mm Hg or use of antihypertensive therapy.
- Either a blood serum glucose concentration of ≥5.6 mmol/L or previously diagnosed type 2 diabetes mellitus.
2.2. Laboratory Methods
- Minor allele frequency (MAF) of at least 5%.
- Availability of information from previous studies on a given SNP.
- Marker localization.
2.3. Statistics
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nolan, P.B.; Carrick-Ranson, G.; Stinear, J.W.; Reading, S.A.; Dalleck, L.C. Prevalence of Metabolic Syndrome and Metabolic Syndrome Components in Young Adults: A Pooled Analysis. Prev. Med. Rep. 2017, 7, 211–215. [Google Scholar] [CrossRef]
- Hx, G.; Ee, R.; Kl, C. International Diabetes Federation World Diabetes Congress 2015. J. Diabetes 2016, 8, 300–302. [Google Scholar] [CrossRef]
- Wilson, P.W.F.; D’Agostino, R.B.; Parise, H.; Sullivan, L.; Meigs, J.B. Metabolic Syndrome as a Precursor of Cardiovascular Disease and Type 2 Diabetes Mellitus. Circulation 2005, 112, 3066–3072. [Google Scholar] [CrossRef] [PubMed]
- Barnes, A.S. The Epidemic of Obesity and Diabetes. Tex. Heart Inst. J. 2011, 38, 142–144. [Google Scholar] [PubMed]
- Penninx, B.W.J.H.; Lange, S.M.M. Metabolic Syndrome in Psychiatric Patients: Overview, Mechanisms, and Implications. Dialogues Clin. Neurosci. 2018, 20, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Nousen, E.K.; Franco, J.G.; Sullivan, E.L. Unraveling the Mechanisms Responsible for the Comorbidity between Metabolic Syndrome and Mental Health Disorders. Neuroendocrinology 2013, 98, 254–266. [Google Scholar] [CrossRef] [PubMed]
- Ohaeri, J.U.; Akanji, A.O. Metabolic Syndrome in Severe Mental Disorders. Metab. Syndr. Relat. Disord. 2011, 9, 91–98. [Google Scholar] [CrossRef] [PubMed]
- de Hert, M.; Schreurs, V.; Vancampfort, D.; van Winkel, R. Metabolic Syndrome in People with Schizophrenia: A Review. World Psychiatry 2009, 8, 15–22. [Google Scholar] [CrossRef]
- Yoca, G.; Anıl Yağcıoğlu, A.E.; Eni, N.; Karahan, S.; Türkoğlu, İ.; Akal Yıldız, E.; Mercanlıgil, S.M.; Yazıcı, M.K. A Follow-up Study of Metabolic Syndrome in Schizophrenia. Eur. Arch. Psychiatry Clin. Neurosci. 2020, 270, 611–618. [Google Scholar] [CrossRef]
- Yazıcı, M.K.; Anıl Yağcıoğlu, A.E.; Ertuğrul, A.; Eni, N.; Karahan, S.; Karaağaoğlu, E.; Tokgözoğlu, S.L. The Prevalence and Clinical Correlates of Metabolic Syndrome in Patients with Schizophrenia: Findings from a Cohort in Turkey. Eur. Arch. Psychiatry Clin. Neurosci. 2011, 261, 69–78. [Google Scholar] [CrossRef]
- Laursen, T.M.; Munk-Olsen, T.; Vestergaard, M. Life Expectancy and Cardiovascular Mortality in Persons with Schizophrenia. Curr. Opin. Psychiatry 2012, 25, 83. [Google Scholar] [CrossRef]
- Medeiros-Ferreira, L.; Navarro-Pastor, J.B.; Zúñiga-Lagares, A.; Romaní, R.; Muray, E.; Obiols, J.E. Perceived Needs and Health-Related Quality of Life in People with Schizophrenia and Metabolic Syndrome: A “Real-World” Study. BMC Psychiatry 2016, 16, 414. [Google Scholar] [CrossRef]
- Hert, M.D.; Vancampfort, D.; Correll, C.U.; Mercken, V.; Peuskens, J.; Sweers, K.; van Winkel, R.; Mitchell, A.J. Guidelines for Screening and Monitoring of Cardiometabolic Risk in Schizophrenia: Systematic Evaluation. Br. J. Psychiatry 2011, 199, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Gurusamy, J.; Gandhi, S.; Damodharan, D.; Ganesan, V.; Palaniappan, M. Exercise, Diet and Educational Interventions for Metabolic Syndrome in Persons with Schizophrenia: A Systematic Review. Asian J. Psychiatry 2018, 36, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Heald, A.; Pendlebury, J.; Anderson, S.; Narayan, V.; Guy, M.; Gibson, M.; Haddad, P.; Livingston, M. Lifestyle Factors and the Metabolic Syndrome in Schizophrenia: A Cross-Sectional Study. Ann. Gen. Psychiatry 2017, 16, 12. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-P.; Gallego, J.A.; Robinson, D.G.; Malhotra, A.K.; Kane, J.M.; Correll, C.U. Efficacy and Safety of Individual Second-Generation vs. First-Generation Antipsychotics in First-Episode Psychosis: A Systematic Review and Meta-Analysis. Int. J. Neuropsychopharmacol. 2013, 16, 1205–1218. [Google Scholar] [CrossRef] [PubMed]
- Cason, N.-M.; Babes, P.A.; Béres, E.; Babes, K. Assessment of Long Term Metabolic Effects of Atypical Antipsychotics in Schizophrenia Patients. Rom. J. Diabetes Nutr. Metab. Dis. 2019, 26, 267–277. [Google Scholar] [CrossRef][Green Version]
- Kelly, D.L.; McMahon, R.P.; Liu, F.; Love, R.C.; Wehring, H.J.; Shim, J.-C.; Warren, K.R.; Conley, R.R. Cardiovascular Disease Mortality in Patients with Chronic Schizophrenia Treated with Clozapine: A Retrospective Cohort Study. J. Clin. Psychiatry 2010, 71, 304–311. [Google Scholar] [CrossRef]
- Papanastasiou, E. Interventions for the Metabolic Syndrome in Schizophrenia: A Review. Ther. Adv. Endocrinol. Metab. 2012, 3, 141–162. [Google Scholar] [CrossRef]
- Fernandez-Egea, E.; Bernardo, M.; Donner, T.; Conget, I.; Parellada, E.; Justicia, A.; Esmatjes, E.; Garcia-Rizo, C.; Kirkpatrick, B. Metabolic Profile of Antipsychotic-Naive Individuals with Non-Affective Psychosis. Br. J. Psychiatry 2009, 194, 434–438. [Google Scholar] [CrossRef]
- Malan-Müller, S.; Kilian, S.; van den Heuvel, L.L.; Bardien, S.; Asmal, L.; Warnich, L.; Emsley, R.A.; Hemmings, S.M.J.; Seedat, S. A Systematic Review of Genetic Variants Associated with Metabolic Syndrome in Patients with Schizophrenia. Schizophr. Res. 2016, 170, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Lv, H.; Li, J.; Gao, K.; Zeng, L.; Xue, R.; Liu, X.; Zhou, C.; Yue, W.; Yu, H. Identification of Genetic Loci That Overlap between Schizophrenia and Metabolic Syndrome. Psychiatry Res. 2022, 318, 114947. [Google Scholar] [CrossRef] [PubMed]
- Guevara-Ramírez, P.; Cadena-Ullauri, S.; Ruiz-Pozo, V.A.; Tamayo-Trujillo, R.; Paz-Cruz, E.; Simancas-Racines, D.; Zambrano, A.K. Genetics, Genomics, and Diet Interactions in Obesity in the Latin American Environment. Front. Nutr. 2022, 9, 1063286. [Google Scholar] [CrossRef] [PubMed]
- Kochetova, O.V.; Akhmadishina, L.Z.; Korytina, G.F.; Karpov, A.A.; Mustafina, O.E. Association of Obesity Susceptibility Gene Variants with Metabolic Syndrome in Women. Obes. Metab. 2017, 14, 33–40. [Google Scholar] [CrossRef][Green Version]
- Paderina, D.Z.; Boiko, A.S.; Pozhidaev, I.V.; Bocharova, A.V.; Mednova, I.A.; Fedorenko, O.Y.; Kornetova, E.G.; Loonen, A.J.M.; Semke, A.V.; Bokhan, N.A.; et al. Genetic Polymorphisms of 5-HT Receptors and Antipsychotic-Induced Metabolic Dysfunction in Patients with Schizophrenia. J. Pers. Med. 2021, 11, 181. [Google Scholar] [CrossRef] [PubMed]
- Boiko, A.S.; Pozhidaev, I.V.; Paderina, D.Z.; Mednova, I.A.; Goncharova, A.A.; Fedorenko, O.Y.; Kornetova, E.G.; Semke, A.V.; Bokhan, N.A.; Loonen, A.J.M.; et al. Gene Polymorphisms of Hormonal Regulators of Metabolism in Patients with Schizophrenia with Metabolic Syndrome. Genes 2022, 13, 844. [Google Scholar] [CrossRef]
- Boiko, A.S.; Pozhidaev, I.V.; Paderina, D.Z.; Bocharova, A.V.; Mednova, I.A.; Fedorenko, O.Y.; Kornetova, E.G.; Loonen, A.J.M.; Semke, A.V.; Bokhan, N.A.; et al. Search for Possible Associations of FTO Gene Polymorphic Variants with Metabolic Syndrome, Obesity and Body Mass Index in Schizophrenia Patients. Pharmgenomics Pers. Med. 2021, 14, 1123–1131. [Google Scholar] [CrossRef]
- Paderina, D.Z.; Boiko, A.S.; Pozhidaev, I.V.; Mednova, I.A.; Goncharova, A.A.; Bocharova, A.V.; Fedorenko, O.Y.; Kornetova, E.G.; Semke, A.V.; Bokhan, N.A.; et al. The Gender-Specific Association of DRD2 Polymorphism with Metabolic Syndrome in Patients with Schizophrenia. Genes 2022, 13, 1312. [Google Scholar] [CrossRef]
- Edwards, K.L.; Wan, J.Y.; Hutter, C.M.; Fong, P.Y.; Santorico, S.A. Multivariate Linkage Scan for Metabolic Syndrome Traits in Families with Type 2 Diabetes. Obesity 2011, 19, 1235–1243. [Google Scholar] [CrossRef]
- Meigs, J.B.; Panhuysen, C.I.; Myers, R.H.; Wilson, P.W.; Cupples, L.A. A Genome-Wide Scan for Loci Linked to Plasma Levels of Glucose and HbA1c in a Community-Based Sample of Caucasian Pedigrees: The Framingham Offspring Study. Diabetes 2002, 51, 833–840. [Google Scholar] [CrossRef]
- Wiltshire, S.; Hattersley, A.T.; Hitman, G.A.; Walker, M.; Levy, J.C.; Sampson, M.; O’Rahilly, S.; Frayling, T.M.; Bell, J.I.; Lathrop, G.M. A Genomewide Scan for Loci Predisposing to Type 2 Diabetes in a UK Population (the Diabetes UK Warren 2 Repository): Analysis of 573 Pedigrees Provides Independent Replication of a Susceptibility Locus on Chromosome 1q. Am. J. Hum. Genet. 2001, 69, 553–569. [Google Scholar] [CrossRef]
- Ng, M.C.Y.; So, W.-Y.; Lam, V.K.L.; Cockram, C.S.; Bell, G.I.; Cox, N.J.; Chan, J.C.N. Genome-Wide Scan for Metabolic Syndrome and Related Quantitative Traits in Hong Kong Chinese and Confirmation of a Susceptibility Locus on Chromosome 1q21-Q25. Diabetes 2004, 53, 2676–2683. [Google Scholar] [CrossRef][Green Version]
- Freudenberg, F.; Alttoa, A.; Reif, A. Neuronal Nitric Oxide Synthase (NOS1) and Its Adaptor, NOS1AP, as a Genetic Risk Factors for Psychiatric Disorders. Genes Brain Behav. 2015, 14, 46–63. [Google Scholar] [CrossRef]
- Brzustowicz, L.M.; Hodgkinson, K.A.; Chow, E.W.C.; Honer, W.G.; Bassett, A.S. Location of a Major Susceptibility Locus for Familial Schizophrenia on Chromosome 1q21-Q22. Science 2000, 288, 678–682. [Google Scholar] [CrossRef]
- Gough, S.C.L.; O’Donovan, M.C. Clustering of Metabolic Comorbidity in Schizophrenia: A Genetic Contribution? J. Psychopharmacol. 2005, 19, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Jaffrey, S.R.; Snowman, A.M.; Eliasson, M.J.; Cohen, N.A.; Snyder, S.H. CAPON: A Protein Associated with Neuronal Nitric Oxide Synthase That Regulates Its Interactions with PSD95. Neuron 1998, 20, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Matiiv, A.B.; Moskalenko, S.E.; Sergeeva, O.S.; Zhouravleva, G.A.; Bondarev, S.A. NOS1AP Interacts with α-Synuclein and Aggregates in Yeast and Mammalian Cells. Int. J. Mol. Sci. 2022, 23, 9102. [Google Scholar] [CrossRef]
- Nasyrova, R.F.; Moskaleva, P.V.; Vaiman, E.E.; Shnayder, N.A.; Blatt, N.L.; Rizvanov, A.A. Genetic Factors of Nitric Oxide’s System in Psychoneurologic Disorders. Int. J. Mol. Sci. 2020, 21, 1604. [Google Scholar] [CrossRef] [PubMed]
- Tziomalos, K.; Athyros, V.G.; Karagiannis, A.; Mikhailidis, D.P. Endothelial Dysfunction in Metabolic Syndrome: Prevalence, Pathogenesis and Management. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Francisqueti, F.V.; Chiaverini, L.C.T.; dos Santos, K.C.; Minatel, I.O.; Ronchi, C.B.; Ferron, A.J.T.; Ferreira, A.L.A.; Corrêa, C.R. The Role of Oxidative Stress on the Pathophysiology of Metabolic Syndrome. Rev. Assoc. Médica Bras. 2017, 63, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Nasykhova, Y.A.; Tonyan, Z.N.; Mikhailova, A.A.; Danilova, M.M.; Glotov, A.S. Pharmacogenetics of Type 2 Diabetes—Progress and Prospects. Int. J. Mol. Sci. 2020, 21, 6842. [Google Scholar] [CrossRef] [PubMed]
- Marjamaa, A.; Newton-Cheh, C.; Porthan, K.; Reunanen, A.; Lahermo, P.; Väänänen, H.; Jula, A.; Karanko, H.; Swan, H.; Toivonen, L.; et al. Common Candidate Gene Variants Are Associated with QT Interval Duration in the General Population. J. Intern. Med. 2009, 265, 448–458. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.A.; Herrington, D.M.; Howard, T.D.; Divers, J.; Arnett, D.K.; Burke, G.L.; Hong Kao, W.; Guo, X.; Siscovick, D.S.; Chakravarti, A.; et al. Associations between NOS1AP Single Nucleotide Polymorphisms (SNPs) and QT Interval Duration in Four Racial/Ethnic Groups in the Multi-Ethnic Study of Atherosclerosis (MESA). Ann. Noninvasive Electrocardiol. 2013, 18, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Aarnoudse, A.-J.L.H.J.; Newton-Cheh, C.; de Bakker, P.I.W.; Straus, S.M.J.M.; Kors, J.A.; Hofman, A.; Uitterlinden, A.G.; Witteman, J.C.M.; Stricker, B.H.C. Common NOS1AP Variants Are Associated with a Prolonged QTc Interval in the Rotterdam Study. Circulation 2007, 116, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Arking, D.E.; Khera, A.; Xing, C.; Kao, W.H.L.; Post, W.; Boerwinkle, E.; Chakravarti, A. Multiple Independent Genetic Factors at NOS1AP Modulate the QT Interval in a Multi-Ethnic Population. PLoS ONE 2009, 4, e4333. [Google Scholar] [CrossRef] [PubMed]
- Eijgelsheim, M.; Aarnoudse, A.L.H.J.; Rivadeneira, F.; Kors, J.A.; Witteman, J.C.M.; Hofman, A.; van Duijn, C.M.; Uitterlinden, A.G.; Stricker, B.H.C. Identification of a Common Variant at the NOS1AP Locus Strongly Associated to QT-Interval Duration. Hum. Mol. Genet. 2009, 18, 347–357. [Google Scholar] [CrossRef]
- Tomás, M.; Napolitano, C.; De, G.L.; Bloise, R.; Subirana, I.; Malovini, A.; Bellazzi, R.; Arking, D.E.; Marban, E.; Chakravarti, A.; et al. Polymorphisms in the NOS1APGene Modulate QT Interval Duration and Risk of Arrhythmias in the Long QT Syndrome. J. Am. Coll. Cardiol. 2010, 55, 2745–2752. [Google Scholar] [CrossRef]
- Chu, A.Y.; Coresh, J.; Arking, D.E.; Pankow, J.S.; Tomaselli, G.F.; Chakravarti, A.; Post, W.S.; Spooner, P.H.; Boerwinkle, E.; Kao, W.H.L. NOS1AP Variant Associated with Incidence of Type 2 Diabetes in Calcium Channel Blocker Users in the Atherosclerosis Risk in Communities (ARIC) Study. Diabetologia 2010, 53, 510–516. [Google Scholar] [CrossRef]
- Hu, C.; Wang, C.; Zhang, R.; Ng, M.C.; Bao, Y.; Wang, C.; So, W.Y.; Ma, R.C.; Ma, X.; Chan, J.C.; et al. Association of Genetic Variants of NOS1AP with Type 2 Diabetes in a Chinese Population. Diabetologia 2010, 53, 290–298. [Google Scholar] [CrossRef]
- Becker, M.L.; Visser, L.E.; Newton-Cheh, C.; Witteman, J.C.M.; Hofman, A.; Uitterlinden, A.G.; Stricker, B.H.C. Genetic Variation in the NOS1AP Gene Is Associated with the Incidence of Diabetes Mellitus in Users of Calcium Channel Blockers. Diabetologia 2008, 51, 2138–2140. [Google Scholar] [CrossRef][Green Version]
- Alberti, K.G.M.M.; Zimmet, P.; Shaw, J. Metabolic Syndrome—A New World-wide Definition. A Consensus Statement from the International Diabetes Federation. Diabet. Med. 2006, 23, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Lewontin, R.C. The Interaction of Selection and Linkage. I. General Considerations; Heterotic Models. Genetics 1964, 49, 49. [Google Scholar] [CrossRef]
- Chang, K.-C.; Barth, A.S.; Sasano, T.; Kizana, E.; Kashiwakura, Y.; Zhang, Y.; Foster, D.B.; Marbán, E. CAPON Modulates Cardiac Repolarization via Neuronal Nitric Oxide Synthase Signaling in the Heart. Proc. Natl. Acad. Sci. USA 2008, 105, 4477–4482. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Hu, C.; Hu, W.; Zhang, R.; Wang, C.; Qin, W.; Yu, W.; Xiang, K.; International Type 2 Diabetes 1q Consortium; Jia, W. A Common Variant of NOS1AP Is Associated with QT Interval Duration in a Chinese Population with Type 2 Diabetes. Diabet. Med. 2010, 27, 1074–1079. [Google Scholar] [CrossRef] [PubMed]
- Zang, X.; Li, S.; Zhao, Y.; Chen, K.; Wang, X.; Song, W.; Ma, J.; Tu, X.; Xia, Y.; Zhang, S. Systematic Meta-Analysis of the Association between a Common NOS1AP Genetic Polymorphism, the QTc Interval, and Sudden Death. Int. Heart J. 2019, 60, 1083–1090. [Google Scholar] [CrossRef] [PubMed]
- The GTEx Consortium. The GTEx Consortium Atlas of Genetic Regulatory Effects across Human Tissues. Science 2020, 369, 1318–1330. [Google Scholar] [CrossRef]
- Ronchi, C.; Bernardi, J.; Mura, M.; Stefanello, M.; Badone, B.; Rocchetti, M.; Crotti, L.; Brink, P.; Schwartz, P.J.; Gnecchi, M. NOS1AP Polymorphisms Reduce NOS1 Activity and Interact with Prolonged Repolarization in Arrhythmogenesis. Cardiovasc. Res. 2021, 117, 472–483. [Google Scholar] [CrossRef]
- Jaensch, M.; Trum, M.; Williams, T.; Schmitt, J.; Schuh, K.; Qadri, F.; Maier, L.; Bader, M.; Ritter, O. Investigations Regarding the Role of NOS1AP in the Heart Using a Conditional Overexpression Mouse Model. Eur. Heart J. 2021, 42, ehab724-3217. [Google Scholar] [CrossRef]
- Andreasen, C.H.; Mogensen, M.S.; Borch-Johnsen, K.; Sandbæk, A.; Lauritzen, T.; Almind, K.; Hansen, L.; Jørgensen, T.; Pedersen, O.; Hansen, T. Lack of Association between PKLR Rs3020781 and NOS1AP Rs7538490 and Type 2 Diabetes, Overweight, Obesity and Related Metabolic Phenotypes in a Danish Large-Scale Study: Case-Control Studies and Analyses of Quantitative Traits. BMC Med. Genet. 2008, 9, 118. [Google Scholar] [CrossRef]
- Lee, M.-K.; Han, K.; Kim, M.K.; Koh, E.S.; Kim, E.S.; Nam, G.E.; Kwon, H.-S. Changes in Metabolic Syndrome and Its Components and the Risk of Type 2 Diabetes: A Nationwide Cohort Study. Sci. Rep. 2020, 10, 2313. [Google Scholar] [CrossRef] [PubMed]
- Lehtinen, A.B.; Newton-Cheh, C.; Ziegler, J.T.; Langefeld, C.D.; Freedman, B.I.; Daniel, K.R.; Herrington, D.M.; Bowden, D.W. Abstract 2924: Association of NOS1AP Genetic Variants with QT Interval Duration in Families from the Diabetes Heart Study. Circulation 2007, 116, 1108–1114. [Google Scholar] [CrossRef]
- Lehtinen, A.B.; Newton-Cheh, C.; Ziegler, J.T.; Langefeld, C.D.; Freedman, B.I.; Daniel, K.R.; Herrington, D.M.; Bowden, D.W. Association of NOS1AP Genetic Variants with QT Interval Duration in Families from the Diabetes Heart Study. Diabetes 2008, 57, 1108–1114. [Google Scholar] [CrossRef] [PubMed]
- Park, B.; Lee, Y.-J. Metabolic Syndrome and Its Components as Risk Factors for Prolonged Corrected QT Interval in Apparently Healthy Korean Men and Women. J. Clin. Lipidol. 2018, 12, 1298–1304. [Google Scholar] [CrossRef]
- Faramawi, M.F.; Wildman, R.P.; Gustat, J.; Rice, J.; Abdul Kareem, M.Y. The Association of the Metabolic Syndrome with QTc Interval in NHANES III. Eur. J. Epidemiol. 2008, 23, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Mottillo, S.; Filion, K.B.; Genest, J.; Joseph, L.; Pilote, L.; Poirier, P.; Rinfret, S.; Schiffrin, E.L.; Eisenberg, M.J. The Metabolic Syndrome and Cardiovascular Risk. J. Am. Coll. Cardiol. 2010, 56, 1113–1132. [Google Scholar] [CrossRef] [PubMed]
- Orlov, P.S.; Ivanoshchuk, D.Е.; Ivanova, А.А.; Malyutina, S.К.; Novosyolov, V.P.; Voevoda, М.I.; Maximov, V.N. Association of Polymorphisms KCNN2 and NOS1AP with Sudden Cardiac Death. Russ. J. Cardiol. 2018, 10, 59–63. [Google Scholar] [CrossRef]
- Nielsen, J.; Graff, C.; Kanters, J.K.; Toft, E.; Taylor, D.; Meyer, J.M. Assessing QT Interval Prolongation and Its Associated Risks with Antipsychotics. CNS Drugs 2011, 25, 473–490. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association; American Psychiatric Association; American Association of Clinical Endocrinologists; North American Association for the Study of Obesity. Consensus Development Conference on Antipsychotic Drugs and Obesity and Diabetes. Diabetes Care 2004, 27, 596–601. [Google Scholar] [CrossRef] [PubMed]
- Zhai, D.; Lang, Y.; Dong, G.; Liu, Y.; Wang, X.; Zhou, D.; Cui, T.; Yang, Y.; Zhang, W.; Zhao, Y. QTc Interval Lengthening in First-Episode Schizophrenia (FES) Patients in the Earliest Stages of Antipsychotic Treatment. Schizophr. Res. 2017, 179, 70–74. [Google Scholar] [CrossRef]
- Åberg, K.; Adkins, D.E.; Liu, Y.; McClay, J.L.; Bukszár, J.; Jia, P.; Zhao, Z.; Perkins, D.; Stroup, T.S.; Lieberman, J.A.; et al. Genome-Wide Association Study of Antipsychotic Induced QTc Interval Prolongation. Pharmacogenomics J. 2012, 12, 165–172. [Google Scholar] [CrossRef]
- Esen-Sehir, D.; Kopf, J.; Hägele, S.; Plichta, M.M.; Reif, A.; Freudenberg, F. Influence of NOS1AP Risk Variants on the Corrected QT (QTc) Interval in the Pharmacotherapy of Schizophrenia. Pharmacopsychiatry 2022, 55, 266–273. [Google Scholar] [CrossRef]
- Fattakhov, N.; Smirnova, L.; Atochin, D.; Parshukova, D.; Skuratovskaia, D.; Painter, Q.; Zatolokin, P.; Semke, A.; Litvinova, L.; Ivanova, S. Haplotype Analysis of Endothelial Nitric Oxide Synthase (NOS3) Genetic Variants and Metabolic Syndrome in Healthy Subjects and Schizophrenia Patients. Int. J. Obes. 2018, 42, 2036–2046. [Google Scholar] [CrossRef] [PubMed]
- de Caluwe, L.; van Buitenen, N.; Gelan, P.J.; Crunelle, C.L.; Thomas, R.; Casseres, S.; Matthys, F.; van Harten, P.; Cahn, W. Prevalence of Metabolic Syndrome and Its Associated Risk Factors in an African–Caribbean Population with Severe Mental Illness. Psychiatry Res. 2019, 281, 112558. [Google Scholar] [CrossRef] [PubMed]
- Hildrum, B.; Mykletun, A.; Hole, T.; Midthjell, K.; Dahl, A.A. Age-Specific Prevalence of the Metabolic Syndrome Defined by the International Diabetes Federation and the National Cholesterol Education Program: The Norwegian HUNT 2 Study. BMC Public Health 2007, 7, 220. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, N.; Yasui-Furukori, N.; Sato, Y.; Umeda, T.; Kishida, I.; Yamashita, H.; Saito, M.; Furukori, H.; Nakagami, T.; Hatakeyama, M. Prevalence of Metabolic Syndrome among Patients with Schizophrenia in Japan. Schizophr. Res. 2010, 123, 244–250. [Google Scholar] [CrossRef]
- Bermudes, R.A.; Keck, P.E., Jr.; Welge, J.A. The Prevalence of the Metabolic Syndrome in Psychiatric Inpatients with Primary Psychotic and Mood Disorders. Psychosomatics 2006, 47, 491–497. [Google Scholar] [CrossRef]
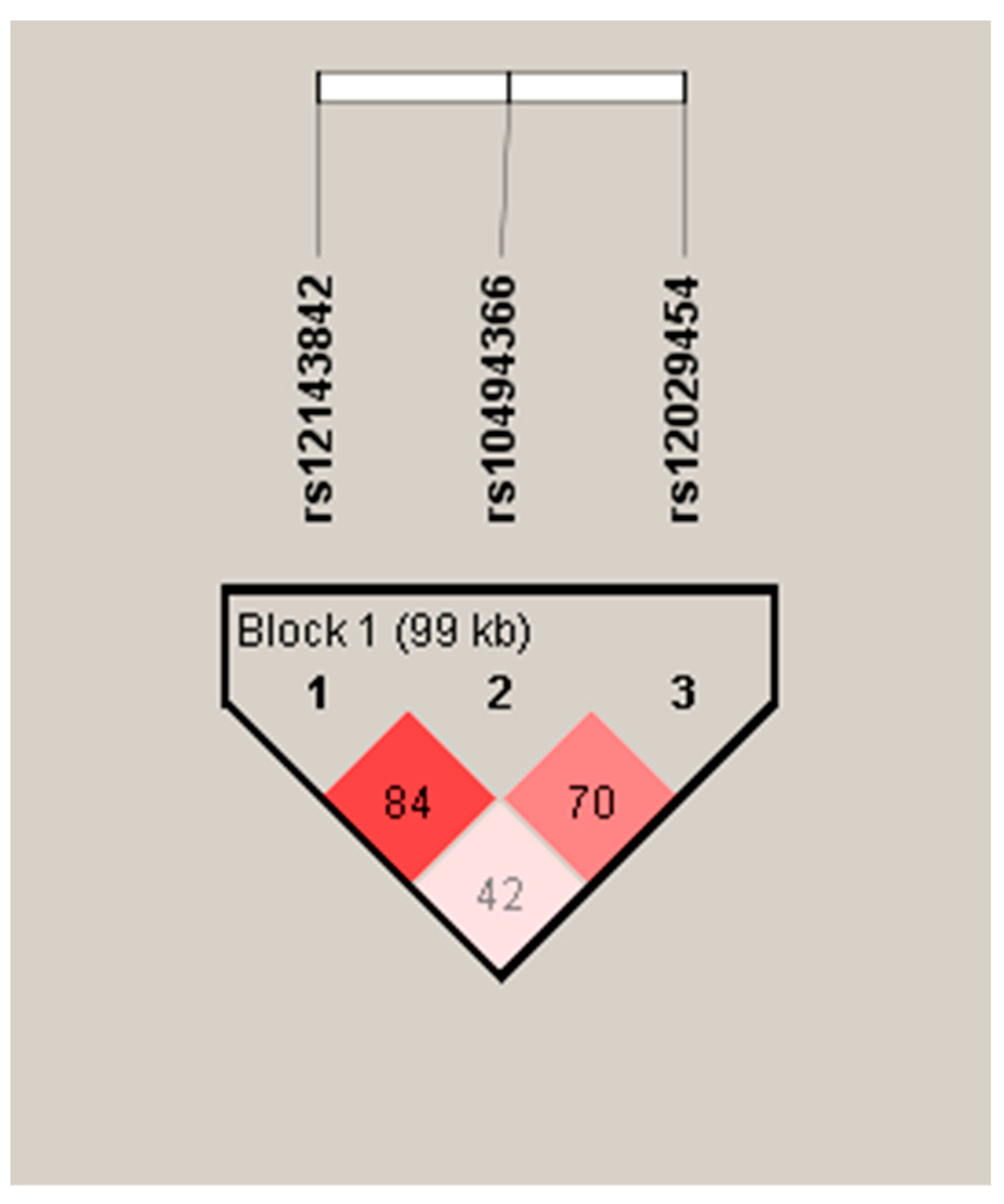
| Parameter | Total, n = 489 | Patients without MetS, n = 358 (73.2%) | Patients with MetS, n = 131 (26.8%) | p Value | |
|---|---|---|---|---|---|
| Gender, n (%) | Females | 257 (52.6%) | 158 (44.1%) | 74 (56.5%) | 0.021 |
| Males | 232 (47.4%) | 200 (55.9%) | 57 (43.5%) | ||
| Age, years, Me (Q1; Q3) | 38 (31;49) | 36 (30;47) | 44 (34;54) | 0.000 | |
| Age of schizophrenia onset, years, Me (Q1; Q3) | 23 (19;30) | 23 (19;29) | 26 (21;31) | 0.003 | |
| Duration of schizophrenia, years, Me (Q1; Q3) | 13 (7;21) | 12 (7;20) | 17 (10;22) | 0.000 | |
| Parameter | Overall, n = 489 | Patients without MetS, n = 358 (73.2%) | Patients with MetS, n = 131 (26.8%) | p Value | |
|---|---|---|---|---|---|
| Monotherapy or combined therapy, n (%) | Monotherapy | 263 (53.8%) | 197 (55.0%) | 66 (50.4%) | 0.649 |
| Two antipsychotics | 186 (38.0%) | 132 (36.9%) | 54 (41.2%) | ||
| Three antipsychotics | 40 (8.2%) | 29 (8.1%) | 11 (8.4%) | ||
| Antipsychotic generation for basic therapy, n (%) | First generation | 288 (58.9%) | 222 (62.0%) | 66 (50.4%) | 0.023 |
| Second generation | 201 (41.2%) | 136 (38.0%) | 65 (49.6%) | ||
| CPZeq, dose, mg, Me (Q1; Q3) | 450 (225;775) | 429.1 (244.3;775) | 450 (225;713.1) | 0.600 | |
| Duration of basic therapy, years, Me (Q1; Q3) | 9 (3;17) | 9 (3;16) | 9 (3; 17) | 0.550 | |
| Gene | NOS1AP | NOS1AP | NOS1AP |
|---|---|---|---|
| SNP | rs12029454 | rs10494366 | rs12143842 |
| Chromosome | chr1 | chr1 | chr1 |
| Position | 162163327 | 162115895 | 162064100 |
| Alleles | G/A | G > T | C > T |
| Type | intron variant | intron variant | intergenic variant |
| MAF (Ensembl; EUR) | 0.179 | 0.361 | 0.257 |
| MAF (study) | 0.182 | 0.375 | 0.245 |
| p HWE | 0.446 | 0.847 | 0.903 |
| Gene | SNP | OR | 95% CI Lower Bound | 95% CI Upper Bound | p Value |
|---|---|---|---|---|---|
| NOS1AP | rs12029454 | 1.026 | 0.955 | 1.103 | 0.4858 |
| NOS1AP | rs10494366 | 1.084 | 1.026 | 1.146 | 0.0046 |
| NOS1AP | rs12143842 | 0.906 | 0.851 | 0.964 | 0.0021 |
| SNP | Alleles | Patients without MetS | Patients with MetS | OR (95% CI) | χ2 | p |
|---|---|---|---|---|---|---|
| rs10494366 | T | 59.6% | 70.2% | 1.60 (1.18–2.16) | 9.179 | 0.002 |
| G | 40.4% | 29.8% | 0.63 (0.46–0.85) | |||
| rs12143842 | C | 72.9% | 82.4% | 1.75 (1.22–2.50) | 9.423 | 0.002 |
| T | 27.1% | 17.6% | 0.57 (0.40–0.82) |
| SNP.Gene | Genotypes | Patients without MetS, n (%) | Patients with MetS, n (%) | OR (95% CI) | χ2 | p |
|---|---|---|---|---|---|---|
| rs12029454.NOS1AP (dominant) | G/G | 235 (65.6%) | 89 (67.9%) | 0.90 (0.59–1.38) | 0.228 | 0.633 |
| A/G + A/A | 123 (34.4%) | 42 (32.1%) | ||||
| rs12029454.NOS1AP (recessive) | G/G + A/G | 346 (96.6%) | 130 (99.2%) | 0.22 (0.03–1.72) | 3.127 | 0.077 |
| A/A | 12 (3.4%) | 1 (0.8%) | ||||
| rs10494366.NOS1AP (dominant) | T/T | 128 (35.8%) | 64 (48.9%) | 0.58 (0.39–0.87) | 6.823 | 0.009 |
| G/T + G/G | 230 (64.2%) | 67 (51.1%) | ||||
| rs10494366.NOS1AP (recessive) | T/T + G/T | 299 (83.5%) | 120 (91.6%) | 0.46 (0.24–1.00) | 5.596 | 0.018 |
| G/G | 59 (16.5%) | 11 (8.4%) | ||||
| rs12143842.NOS1AP (dominant) | C/C | 191 (53.4%) | 88 (67.2%) | 0.56 (0.37–0.58) | 7.550 | 0.006 |
| C/T + T/T | 167 (46.6%) | 43 (32.8%) | ||||
| rs12143842.NOS1AP (recessive) | C/C + C/T | 331 (92.5%) | 128 (97.7%) | 0.29 (0.09–1.01) | 5.502 | 0.019 |
| T/T | 27 (7.5%) | 3 (2.3%) |
| Haplotype | Frequency | OR | 95% CI | p Value |
|---|---|---|---|---|
| GTC | 0.5692 | 1 (Ref) | ||
| AGT | 0.1014 | 0.48 | 0.27–0.86 | 0.0139 |
| GGC | 0.1070 | 0.60 | 0.35–1.01 | 0.0557 |
| GGT | 0.1197 | 0.57 | 0.34–0.94 | 0.0294 |
| Rare | 0.1027 | 0.98 | 0.58–1.64 | 0.9306 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mednova, I.A.; Pozhidaev, I.V.; Tiguntsev, V.V.; Bocharova, A.V.; Paderina, D.Z.; Boiko, A.S.; Fedorenko, O.Y.; Kornetova, E.G.; Bokhan, N.A.; Stepanov, V.A.; et al. NOS1AP Gene Variants and Their Role in Metabolic Syndrome: A Study of Patients with Schizophrenia. Biomedicines 2024, 12, 627. https://doi.org/10.3390/biomedicines12030627
Mednova IA, Pozhidaev IV, Tiguntsev VV, Bocharova AV, Paderina DZ, Boiko AS, Fedorenko OY, Kornetova EG, Bokhan NA, Stepanov VA, et al. NOS1AP Gene Variants and Their Role in Metabolic Syndrome: A Study of Patients with Schizophrenia. Biomedicines. 2024; 12(3):627. https://doi.org/10.3390/biomedicines12030627
Chicago/Turabian StyleMednova, Irina A., Ivan V. Pozhidaev, Vladimir V. Tiguntsev, Anna V. Bocharova, Diana Z. Paderina, Anastasiia S. Boiko, Olga Y. Fedorenko, Elena G. Kornetova, Nikolay A. Bokhan, Vadim A. Stepanov, and et al. 2024. "NOS1AP Gene Variants and Their Role in Metabolic Syndrome: A Study of Patients with Schizophrenia" Biomedicines 12, no. 3: 627. https://doi.org/10.3390/biomedicines12030627
APA StyleMednova, I. A., Pozhidaev, I. V., Tiguntsev, V. V., Bocharova, A. V., Paderina, D. Z., Boiko, A. S., Fedorenko, O. Y., Kornetova, E. G., Bokhan, N. A., Stepanov, V. A., & Ivanova, S. A. (2024). NOS1AP Gene Variants and Their Role in Metabolic Syndrome: A Study of Patients with Schizophrenia. Biomedicines, 12(3), 627. https://doi.org/10.3390/biomedicines12030627





