A Comprehensive Review of Muscle–Tendon Junction: Structure, Function, Injury and Repair
Abstract
1. Introduction
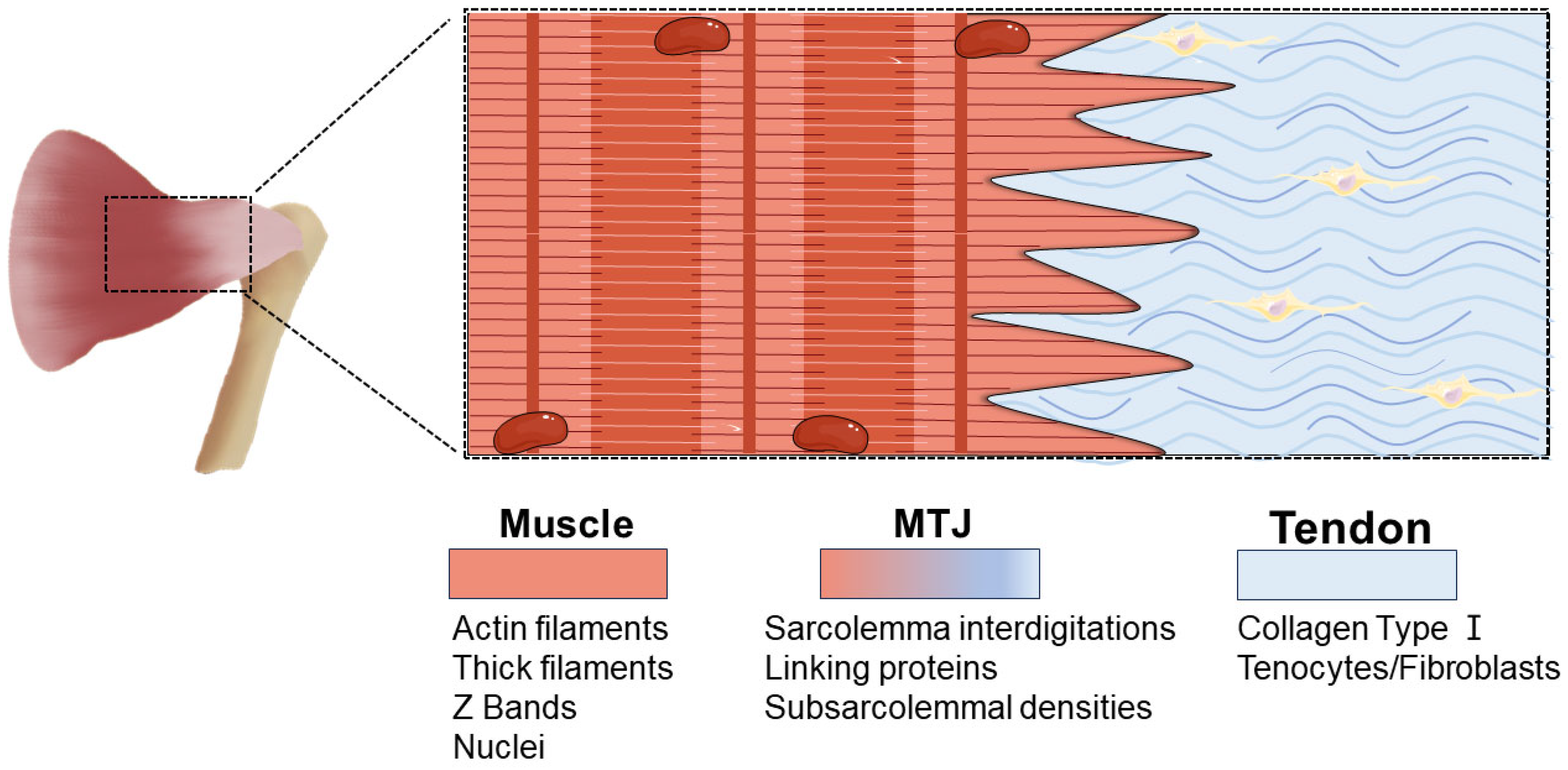
2. The Structure and Definition of MTJ
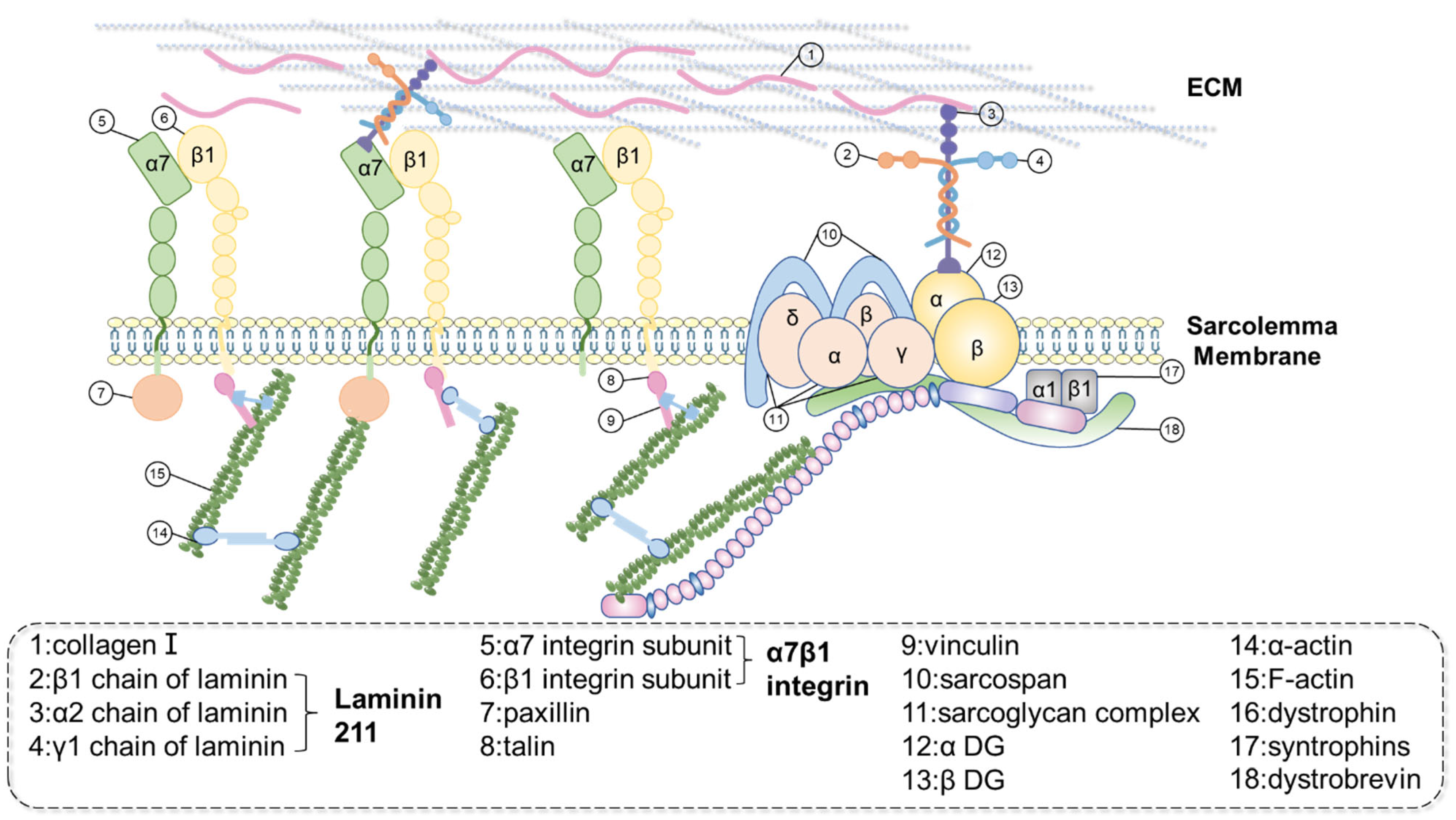
3. Subtle Structure and Characteristic Proteins of MTJ
3.1. Dystrophin
3.2. α7β1 Integrin
3.3. Col XXII
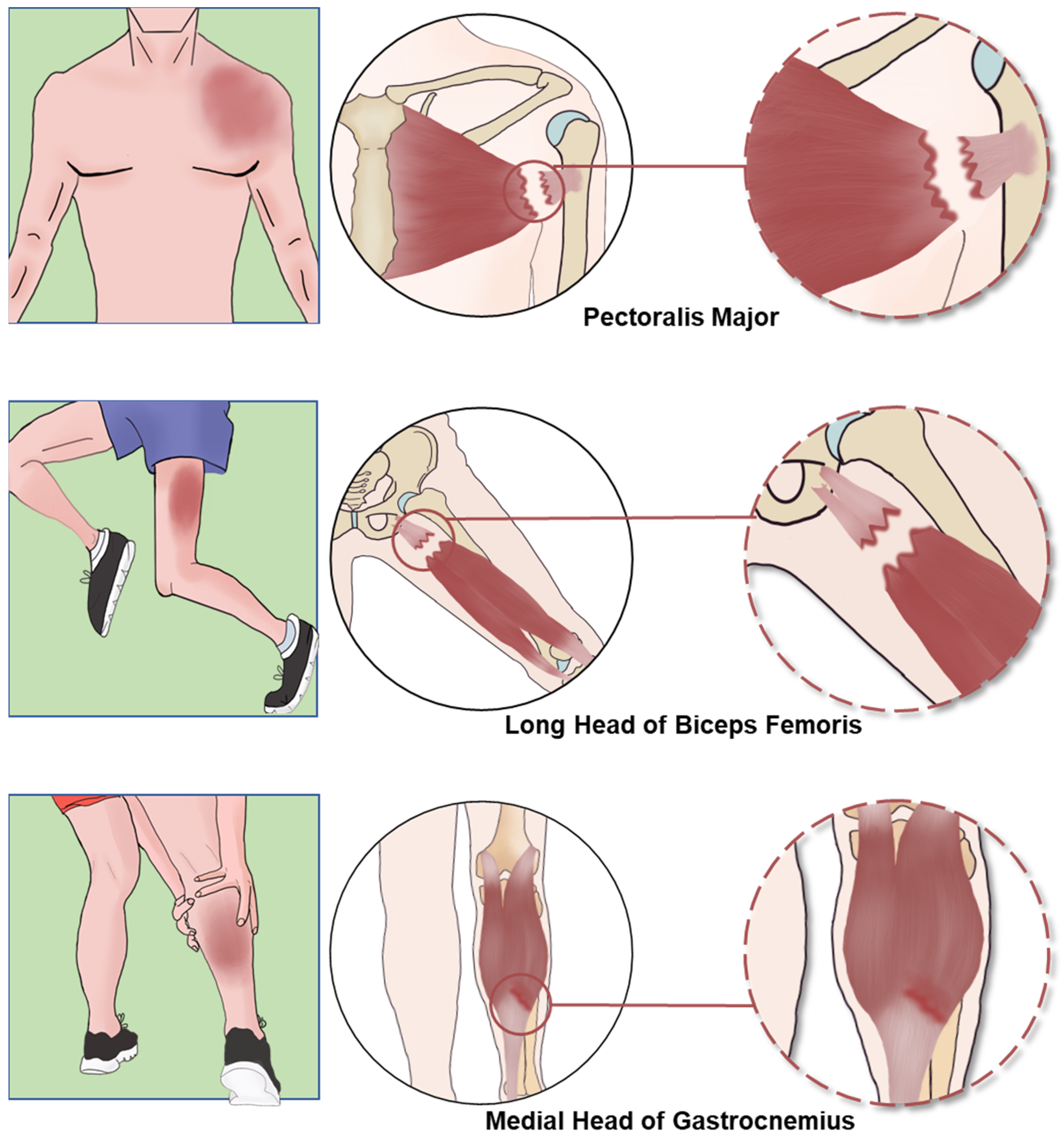
4. MTJ Injuries
Traditional Therapies for MTJ Injuries
5. Tissue Engineering Strategies for MTJ Injuries
5.1. Biological Scaffold
5.2. Synthetic Scaffold
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kääriäinen, M.; Jarvinen, T.; Jarvinen, M.; Rantanen, J.; Kalimo, H. Relation between myofibers and connective tissue during muscle injury repair. Scand. J. Med. Sci. Sports 2000, 10, 332–337. [Google Scholar] [CrossRef]
- Bloch, R.J.; Gonzalez-Serratos, H. Lateral force transmission across costameres in skeletal muscle. Exerc. Sport Sci. Rev. 2003, 31, 73–78. [Google Scholar] [CrossRef]
- Passerieux, E.; Rossignol, R.; Letellier, T.; Delage, J. Physical continuity of the perimysium from myofibers to tendons: Involvement in lateral force transmission in skeletal muscle. J. Struct. Biol. 2007, 159, 19–28. [Google Scholar] [CrossRef]
- Kage, T.; Sanada, T.; Iwaso, H.; Goto, H.; Fukai, A.; Yamagami, R.; Honda, E.; Uchiyama, E. Morphology of Acute Achilles Tendon Rupture by Intraoperative Evaluation. J. Foot Ankle Surg. 2021, 60, 1198–1203. [Google Scholar] [CrossRef]
- Yan, R.; Zhang, H.; Ma, Y.; Lin, R.; Zhou, B.; Zhang, T.; Fan, C.; Zhang, Y.; Wang, Z.; Fang, T.; et al. Discovery of muscle-tendon progenitor subpopulation in human myotendinous junction at single-cell resolution. Research 2022, 2022, 9760390. [Google Scholar] [CrossRef] [PubMed]
- Charvet, B.; Ruggiero, F.; Le Guellec, D. The development of the myotendinous junction. A review. Muscles Ligaments Tendons J. 2012, 2, 53–63. [Google Scholar] [PubMed]
- Yu, J.; Zhang, C.; Horner, N.; Ayeni, O.R.; Leroux, T.; Alolabi, B.; Khan, M. Outcomes and return to sport after pectoralis major tendon repair: A systematic review. Sports Health 2019, 11, 134–141. [Google Scholar] [CrossRef]
- Heidari, B.S.; Ruan, R.; De-Juan-Pardo, E.M.; Zheng, M.; Doyle, B. Biofabrication and signaling strategies for tendon/ligament interfacial tissue engineering. ACS Biomater. Sci. Eng. 2021, 7, 383–399. [Google Scholar] [CrossRef]
- Baldino, L.; Cardea, S.; Maffulli, N.; Reverchon, E. Regeneration techniques for bone-to-tendon and muscle-to-tendon interfaces reconstruction. Br. Med. Bull. 2016, 117, 25–37. [Google Scholar] [CrossRef] [PubMed]
- VanDusen, K.; Larkin, L. Muscle–tendon interface. In Regenerative Engineering of Musculoskeletal Tissues and Interfaces; Elsevier: Amsterdam, The Netherlands, 2015; pp. 409–429. [Google Scholar]
- Yamamoto, M.; Sakiyama, K.; Kitamura, K.; Yamamoto, Y.; Takagi, T.; Sekiya, S.; Watanabe, G.; Taniguchi, S.; Ogawa, Y.; Ishizuka, S.; et al. Development and regeneration of muscle, tendon, and myotendinous junctions in striated skeletal muscle. Int. J. Mol. Sci. 2022, 23, 3006. [Google Scholar] [CrossRef]
- Charvet, B.; Malbouyres, M.; Pagnon-Minot, A.; Ruggiero, F.; Le Guellec, D. Development of the zebrafish myoseptum with emphasis on the myotendinous junction. Cell Tissue Res. 2011, 346, 439–449. [Google Scholar] [CrossRef]
- Knudsen, A.; Larsen, M.; Mackey, A.L.; Hjort, M.; Hansen, K.K.; Qvortrup, K.; Kjær, M.; Krogsgaard, M.R. The human myotendinous junction: An ultrastructural and 3 D analysis study. Scand. J. Med. Sci. Sports 2015, 25, e116–e123. [Google Scholar] [CrossRef]
- Trotter, J.A. Structure–function considerations of muscle–tendon junctions. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2002, 133, 1127–1133. [Google Scholar] [CrossRef]
- Trotter, J.A.; Eberhard, S.; Samora, A. Structural connections of the muscle-tendon junction. Cell Motil. 1983, 3, 431–438. [Google Scholar] [CrossRef]
- Ovalie, W.K. The human muscle-tendon junction. Anat. Embryol. 1987, 176, 281–294. [Google Scholar] [CrossRef]
- Ciena, A.P.; Luques, I.U.; Dias, F.J.; de Almeida, S.R.Y.; Iyomasa, M.M.; Watanabe, I.-S. Ultrastructure of the myotendinous junction of the medial pterygoid muscle of adult and aged Wistar rats. Micron 2010, 41, 1011–1014. [Google Scholar] [CrossRef]
- Heyde, C.-E.; Kayser, R.; Jungmichel, D.; Melzer, C. Die Grenzen der Sonographie in der Diagnostik der Achillessehnenteilruptur im Muskelsehnenübergang anhand einer Kasuistik. Sportverletz.·Sportschaden 2003, 17, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Clark, B.B.; Jaffe, D.; Henn, F.R.; Lovering, R.M. Evaluation and imaging of an untreated grade III hamstring tear: A case report. Clin. Orthop. Relat. Res. 2011, 469, 3248–3252. [Google Scholar] [CrossRef] [PubMed]
- Morin, A.; Stantzou, A.; Petrova, O.N.; Hildyard, J.; Tensorer, T.; Matouk, M.; Petkova, M.V.; Richard, I.; Manoliu, T.; Goyenvalle, A.; et al. Dystrophin myonuclear domain restoration governs treatment efficacy in dystrophic muscle. Proc. Natl. Acad. Sci. USA 2023, 120, e2206324120. [Google Scholar] [CrossRef] [PubMed]
- Blake, D.J.; Weir, A.; Newey, S.E.; Davies, K.E.; Spaulding, H.R.; Quindry, T.; Hammer, K.; Quindry, J.C.; Selsby, J.T.; Amirouche, A.; et al. Function and Genetics of Dystrophin and Dystrophin-Related Proteins in Muscle. Physiol. Rev. 2002, 82, 291–329. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; McNally, E.M. The Dystrophin Complex: Structure, function and implications for therapy. Compr. Physiol. 2015, 5, 1223. [Google Scholar]
- Prins, K.W.; Humston, J.L.; Mehta, A.; Tate, V.; Ralston, E.; Ervasti, J.M. Dystrophin is a microtubule-associated protein. J. Cell Biol. 2009, 186, 363–369. [Google Scholar] [CrossRef]
- Muntoni, F.; Torelli, S.; Ferlini, A. Dystrophin and mutations: One gene, several proteins, multiple phenotypes. Lancet Neurol. 2003, 2, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Bhat, H.F.; Mir, S.S.; Dar, K.B.; Bhat, Z.F.; Shah, R.A.; Ganai, N.A. ABC of multifaceted dystrophin glycoprotein complex (DGC). J. Cell. Physiol. 2018, 233, 5142–5159. [Google Scholar] [CrossRef] [PubMed]
- Bassett, D.I.; Bryson-Richardson, R.J.; Daggett, D.F.; Gautier, P.; Keenan, D.G.; Currie, P.D. Dystrophin is required for the formation of stable muscle attachments in the zebrafish embryo. Development 2003, 130, 5851–5860. [Google Scholar] [CrossRef] [PubMed]
- Campbell, I.D.; Humphries, M.J. Integrin structure, activation, and interactions. Cold Spring Harb. Perspect. Biol. 2011, 3, a004994. [Google Scholar] [CrossRef] [PubMed]
- Song, W.K.; Wang, W.; Foster, R.F.; Bielser, D.A.; Kaufman, S.J. H36-alpha 7 is a novel integrin alpha chain that is developmentally regulated during skeletal myogenesis. J. Cell Biol. 1992, 117, 643–657. [Google Scholar] [CrossRef] [PubMed]
- LaFlamme, S.E.; Akiyama, S.K.; Yamada, K.M. Regulation of fibronectin receptor distribution. J. Cell Biol. 1992, 117, 437–447. [Google Scholar] [CrossRef]
- Bao, Z.; Lakonishok, M.; Kaufman, S.; Horwitz, A.F. α7β1 integrin is a component of the myotendinous junction on skeletal muscle. J. Cell Sci. 1993, 106, 579–589. [Google Scholar] [CrossRef]
- Welser, J.V.; Rooney, J.E.; Cohen, N.C.; Gurpur, P.B.; Singer, C.A.; Evans, R.A.; Haines, B.A.; Burkin, D.J. Myotendinous junction defects and reduced force transmission in mice that lack α7 integrin and utrophin. Am. J. Pathol. 2009, 175, 1545–1554. [Google Scholar] [CrossRef]
- Jacobson, K.R.; Lipp, S.; Acuna, A.; Leng, Y.; Bu, Y.; Calve, S. Comparative analysis of the extracellular matrix proteome across the myotendinous junction. J. Proteome Res. 2020, 19, 3955–3967. [Google Scholar] [CrossRef]
- Koch, M.; Schulze, J.; Hansen, U.; Ashwodt, T.; Keene, D.R.; Brunken, W.J.; Burgeson, R.E.; Bruckner, P.; Bruckner-Tuderman, L. A novel marker of tissue junctions, collagen XXII. J. Biol. Chem. 2004, 279, 22514–22521. [Google Scholar] [CrossRef] [PubMed]
- Zwolanek, D.; Veit, G.; Eble, J.A.; Gullberg, D.; Ruggiero, F.; Heino, J.; Meier, M.; Stetefeld, J.; Koch, M. Collagen XXII binds to collagen-binding integrins via the novel motifs GLQGER and GFKGER. Biochem. J. 2014, 459, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Charvet, B.; Guiraud, A.; Malbouyres, M.; Zwolanek, D.; Guillon, E.; Bretaud, S.; Monnot, C.; Schulze, J.; Bader, H.L.; Allard, B.; et al. Knockdown of col22a1 gene in zebrafish induces a muscular dystrophy by disruption of the myotendinous junction. Development 2013, 140, 4602–4613. [Google Scholar] [CrossRef] [PubMed]
- Malbouyres, M.; Guiraud, A.; Lefrançois, C.; Salamito, M.; Nauroy, P.; Bernard, L.; Sohm, F.; Allard, B.; Ruggiero, F. The myotendinous junction marker collagen XXII enables zebrafish postural control learning and optimal swimming performance through its force transmission activity. bioRxiv 2021. [Google Scholar] [CrossRef]
- Kim, M.; Franke, V.; Brandt, B.; Lowenstein, E.D.; Schöwel, V.; Spuler, S.; Akalin, A.; Birchmeier, C. Single-nucleus transcriptomics reveals functional compartmentalization in syncytial skeletal muscle cells. Nat. Commun. 2020, 11, 6375. [Google Scholar] [CrossRef]
- Petrany, M.J.; Swoboda, C.O.; Sun, C.; Chetal, K.; Chen, X.; Weirauch, M.T.; Salomonis, N.; Millay, D.P. Single-nucleus RNA-seq identifies transcriptional heterogeneity in multinucleated skeletal myofibers. Nat. Commun. 2020, 11, 6374. [Google Scholar] [CrossRef]
- Karlsen, A.; Gonzalez-Franquesa, A.; Jakobsen, J.R.; Krogsgaard, M.R.; Koch, M.; Kjaer, M.; Schiaffino, S.; Mackey, A.L.; Deshmukh, A.S. The proteomic profile of the human myotendinous junction. Iscience 2022, 25, 103836. [Google Scholar] [CrossRef]
- Maffulli, N.; Renström, P.; Leadbetter, W.B. Tendon Injuries; Springer: Berlin/Heidelberg, Germany, 2005. [Google Scholar]
- Vila Pouca, M.C.; Parente, M.P.L.; Jorge, R.M.N.; Ashton-Miller, J.A. Injuries in muscle-tendon-bone units: A systematic review considering the role of passive tissue fatigue. Orthop. J. Sports Med. 2021, 9, 23259671211020731. [Google Scholar] [CrossRef]
- Huijing, P.A. Muscle as a collagen fiber reinforced composite: A review of force transmission in muscle and whole limb. J. Biomech. 1999, 32, 329–345. [Google Scholar] [CrossRef]
- Danielsson, A.; Horvath, A.; Senorski, C.; Alentorn-Geli, E.; Garrett, W.E.; Cugat, R.; Samuelsson, K.; Senorski, E.H. The mechanism of hamstring injuries—A systematic review. BMC Musculoskelet. Disord. 2020, 21, 641. [Google Scholar] [CrossRef] [PubMed]
- Grassi, A.; Quaglia, A.; Canata, G.L.; Zaffagnini, S. An update on the grading of muscle injuries: A narrative review from clinical to comprehensive systems. Joints 2016, 4, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Kakwani, R.; Matthews, J.J.; Kumar, K.M.; Pimpalnerkar, A.; Mohtadi, N. Rupture of the pectoralis major muscle: Surgical treatment in athletes. Int. Orthop. 2007, 31, 159–163. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Orchard, J.; Seward, H. Epidemiology of injuries in the Australian Football League, seasons 1997–2000. Br. J. Sports Med. 2002, 36, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Woods, C.; Hawkins, R.D.; Maltby, S.; Hulse, M.; Thomas, A.; Hodson, A. The Football Association Medical Research Programme: An audit of injuries in professional football—Analysis of hamstring injuries. Br. J. Sports Med. 2004, 38, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Green, B.; Lin, M.; Schache, A.G.; McClelland, J.A.; Semciw, A.I.; Rotstein, A.; Cook, J.; Pizzari, T. Calf muscle strain injuries in elite Australian Football players: A descriptive epidemiological evaluation. Scand. J. Med. Sci. Sports 2020, 30, 174–184. [Google Scholar] [CrossRef] [PubMed]
- Tidball, J.G. Force transmission across muscle cell membranes. J. Biomech. 1991, 24, 43–52. [Google Scholar] [CrossRef]
- Brotto, M.; Bonewald, L. Bone and muscle: Interactions beyond mechanical. Bone 2015, 80, 109–114. [Google Scholar] [CrossRef]
- Curzi, D.; Sartini, S.; Guescini, M.; Lattanzi, D.; Di Palma, M.; Ambrogini, P.; Savelli, D.; Stocchi, V.; Cuppini, R.; Falcieri, E. Effect of different exercise intensities on the myotendinous junction plasticity. PLoS ONE 2016, 11, e0158059. [Google Scholar] [CrossRef]
- Clanton, T.O.; Coupe, K.J. Hamstring Strains in Athletes: Diagnosis and Treatment. J. Am. Acad. Orthop. Surg. 1998, 6, 237–248. [Google Scholar] [CrossRef]
- Järvinen, M.J.; Lehto, M.U. The effects of early mobilisation and immobilisation on the healing process following muscle injuries. Sports Med. 1993, 15, 78–89. [Google Scholar] [CrossRef]
- Noonan, T.J.; Garrett, W.E., Jr. Muscle strain injury: Diagnosis and treatment. J. Am. Acad. Orthop. Surg. 1999, 7, 262–269. [Google Scholar] [CrossRef]
- Travis, R.D.; Doane, R.; Burkhead, W.Z. Tendon ruptures about the shoulder. Orthop. Clin. 2000, 31, 313–330. [Google Scholar] [CrossRef]
- Butt, U.; Mehta, S.; Funk, L.; Monga, P. Pectoralis major ruptures: A review of current management. J. Shoulder Elb. Surg. 2015, 24, 655–662. [Google Scholar] [CrossRef]
- de Castro Pochini, A.; Ejnisman, B.; Andreoli, C.V.; Monteiro, G.C.; Silva, A.C.; Cohen, M.; Albertoni, W.M. Pectoralis major muscle rupture in athletes: A prospective study. Am. J. Sports Med. 2010, 38, 92–98. [Google Scholar] [CrossRef]
- Petilon, J.; Carr, D.R.; Sekiya, J.K.; Unger, D.V. Pectoralis major muscle injuries: Evaluation and management. J. Am. Acad. Orthop. Surg. 2005, 13, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, B.; McAleer, S.; Kelly, S.; Chakraverty, R.; Johnston, M.; Pollock, N. Hamstring rehabilitation in elite track and field athletes: Applying the British Athletics Muscle Injury Classification in clinical practice. Br. J. Sports Med. 2019, 53, 1464–1473. [Google Scholar] [CrossRef] [PubMed]
- Brockett, C.L.; Morgan, D.L.; Proske, U. Human hamstring muscles adapt to eccentric exercise by changing optimum length. Med. Sci. Sports Exerc. 2001, 33, 783–790. [Google Scholar] [CrossRef] [PubMed]
- Croisier, J.-L. Factors associated with recurrent hamstring injuries. Sports Med. 2004, 34, 681–695. [Google Scholar] [CrossRef] [PubMed]
- Dürig, M.; Schuppisser, J.; Gauer, E.; Müller, W. Spontaneous rupture of the gastrocnemius muscle. Injury 1977, 9, 143–145. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Yang, H.-L.; Sun, Z.-Y.; Ni, L.; Zhang, H.-T. Surgical treatment of gastrocnemius muscle ruptures. Orthop. Surg. 2012, 4, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Bak, K.; Cameron, E.; Henderson, I. Rupture of the pectoralis major: A meta-analysis of 112 cases. Knee Surg. Sports Traumatol. Arthrosc. 2000, 8, 113–119. [Google Scholar] [CrossRef] [PubMed]
- de Castro Pochini, A.; Andreoli, C.V.; Belangero, P.S.; Figueiredo, E.A.; Terra, B.B.; Cohen, C.; Andrade, M.D.S.; Cohen, M.; Ejnisman, B. Clinical considerations for the surgical treatment of pectoralis major muscle ruptures based on 60 cases: A prospective study and literature review. Am. J. Sports Med. 2014, 42, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Järvinen, T.A.; Järvinen, T.L.N.; Kääriäinen, M.; Kalimo, H.; Järvinen, M. Muscle injuries: Biology and treatment. Am. J. Sports Med. 2005, 33, 745–764. [Google Scholar] [CrossRef] [PubMed]
- Wood, D.G.; Packham, I.; Trikha, S.P.; Linklater, J. Avulsion of the proximal hamstring origin. J. Bone Jt. Surg. Am. 2008, 90, 2365–2374. [Google Scholar] [CrossRef] [PubMed]
- Sarimo, J.; Lempainen, L.; Mattila, K.; Orava, S. Complete proximal hamstring avulsions: A series of 41 patients with operative treatment. Am. J. Sports Med. 2008, 36, 1110–1115. [Google Scholar] [CrossRef]
- Cohen, S.; Bradley, J. Acute proximal hamstring rupture. J. Am. Acad. Orthop. Surg. 2007, 15, 350–355. [Google Scholar] [CrossRef]
- Ikada, Y. Challenges in tissue engineering. J. R. Soc. Interface 2006, 3, 589–601. [Google Scholar] [CrossRef]
- Scott, R.W.; Arostegui, M.; Schweitzer, R.; Rossi, F.M.; Underhill, T.M. Hic1 defines quiescent mesenchymal progenitor subpopulations with distinct functions and fates in skeletal muscle regeneration. Cell Stem Cell 2019, 25, 797–813.e9. [Google Scholar] [CrossRef]
- Yaseen, W.; Kraft-Sheleg, O.; Zaffryar-Eilot, S.; Melamed, S.; Sun, C.; Millay, D.P.; Hasson, P. Fibroblast fusion to the muscle fiber regulates myotendinous junction formation. Nat. Commun. 2021, 12, 3852. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Yu, J.-M.; Gan, Y.-C.; Qiu, X.-Z.; Gao, Z.-C.; Wang, H.; Chen, S.-X.; Xiong, Y.; Liu, G.-H.; Lin, S.-E.; et al. Biomimetic natural biomaterials for tissue engineering and regenerative medicine: New biosynthesis methods, recent advances, and emerging applications. Mil. Med. Res. 2023, 10, 16. [Google Scholar] [CrossRef]
- Turner, N.J.; Badylak, S.F. Biologic scaffolds for musculotendinous tissue repair. Eur. Cell Mater. 2013, 25, 43. [Google Scholar] [CrossRef]
- Turner, N.J.; Yates, A.J.; Weber, D.J.; Qureshi, I.R.; Stolz, D.B.; Gilbert, T.W.; Badylak, S.F. Xenogeneic extracellular matrix as an inductive scaffold for regeneration of a functioning musculotendinous junction. Tissue Eng. Part A 2010, 16, 3309–3317. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, X.; Hong, H.; Hu, R.; Liu, J.; Liu, C. Decellularized extracellular matrix scaffolds: Recent trends and emerging strategies in tissue engineering. Bioact. Mater. 2022, 10, 15–31. [Google Scholar] [CrossRef]
- Klatte-Schulz, F.; Schmidt, T.; Uckert, M.; Scheffler, S.; Kalus, U.; Rojewski, M.; Schrezenmeier, H.; Pruss, A.; Wildemann, B. Comparative analysis of different platelet lysates and platelet rich preparations to stimulate tendon cell biology: An in vitro study. Int. J. Mol. Sci. 2018, 19, 212. [Google Scholar] [CrossRef]
- Kim, B.-S.; Mooney, D.J. Development of biocompatible synthetic extracellular matrices for tissue engineering. Trends Biotechnol. 1998, 16, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Li, X.; Xia, Y. Putting electrospun nanofibers to work for biomedical research. Macromol. Rapid Commun. 2008, 29, 1775–1792. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Ouyang, G.; McCann, J.T.; Xia, Y. Collecting electrospun nanofibers with patterned electrodes. Nano Lett. 2005, 5, 913–916. [Google Scholar] [CrossRef]
- Merceron, T.K.; Burt, M.; Seol, Y.-J.; Kang, H.-W.; Lee, S.J.; Yoo, J.J.; Atala, A. A 3D bioprinted complex structure for engineering the muscle–tendon unit. Biofabrication 2015, 7, 035003. [Google Scholar] [CrossRef]
- Gaffney, L.S.; Davis, Z.G.; Mora-Navarro, C.; Fisher, M.B.; Freytes, D.O. Extracellular matrix hydrogels promote expression of muscle-tendon junction proteins. Tissue Eng. Part A 2022, 28, 270–282. [Google Scholar] [CrossRef]
- Chen, J.; Xu, J.; Wang, A.; Zheng, M. Scaffolds for tendon and ligament repair: Review of the efficacy of commercial products. Expert Rev. Med. Devices 2009, 6, 61–73. [Google Scholar] [CrossRef]
- Keane, T.J.; Londono, R.; Turner, N.J.; Badylak, S.F. Consequences of ineffective decellularization of biologic scaffolds on the host response. Biomaterials 2012, 33, 1771–1781. [Google Scholar] [CrossRef] [PubMed]
- Bn, B. Macrophage phenotype and remodeling out-comes in response to biologic scaffolds with and without a cellular component. Biomaterials 2009, 30, 1482–1489. [Google Scholar]
- Valentin, J.E.; Stewart-Akers, A.M.; Gilbert, T.W.; Badylak, S.F. Macrophage participation in the degradation and remodeling of extracellular matrix scaffolds. Tissue Eng. Part A 2009, 15, 1687–1694. [Google Scholar] [CrossRef]
- Longo, U.G.; Lamberti, A.; Petrillo, S.; Maffulli, N.; Denaro, V. Scaffolds in tendon tissue engineering. Stem Cells Int. 2012, 2012, 517165. [Google Scholar] [CrossRef]
- Wolf, M.T.; Dearth, C.L.; Sonnenberg, S.B.; Loboa, E.G.; Badylak, S.F. Naturally derived and synthetic scaffolds for skeletal muscle reconstruction. Adv. Drug Deliv. Rev. 2015, 84, 208–221. [Google Scholar] [CrossRef]
- Reddy, M.S.B.; Ponnamma, D.; Choudhary, R.; Sadasivuni, K.K. A comparative review of natural and synthetic biopolymer composite scaffolds. Polymers 2021, 13, 1105. [Google Scholar] [CrossRef] [PubMed]
- Spalazzi, J.P.; Dagher, E.; Doty, S.B.; Guo, X.E.; Rodeo, S.A.; Lu, H.H. In vivo evaluation of a multiphased scaffold designed for orthopaedic interface tissue engineering and soft tissue-to-bone integration. J. Biomed. Mater. Res. Part A 2008, 86, 1–12. [Google Scholar] [CrossRef]
- Ladd, M.R. Electrospun Dual Scaffolding System with Potential for the Tissue Engineering of Muscle-Tendon Junctions; Wake Forest University: Winston-Salem, NC, USA, 2011. [Google Scholar]
- Kuo, C.-C.; Qin, H.; Cheng, Y.; Jiang, X.; Shi, X. An integrated manufacturing strategy to fabricate delivery system using gelatin/alginate hybrid hydrogels: 3D printing and freeze-drying. Food Hydrocoll. 2021, 111, 106262. [Google Scholar] [CrossRef]
- Wu, X.; Liu, Y.; Li, X.; Wen, P.; Zhang, Y.; Long, Y.; Wang, X.; Guo, Y.; Xing, F.; Gao, J. Preparation of aligned porous gelatin scaffolds by unidirectional freeze-drying method. Acta Biomater. 2010, 6, 1167–1177. [Google Scholar] [CrossRef]
- Zeiger, E.; Gollapudi, B.; Spencer, P. Genetic toxicity and carcinogenicity studies of glutaraldehyde—A review. Mutat. Res./Rev. Mutat. Res. 2005, 589, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Baldino, L.; Concilio, S.; Cardea, S.; De Marco, I.; Reverchon, E.J. Complete glutaraldehyde elimination during chitosan hydrogel drying by SC-CO2 processing. Supercrit. Fluids 2015, 103, 70–76. [Google Scholar] [CrossRef]
- Xu, H.-Q.; Liu, J.-C.; Zhang, Z.-Y.; Xu, C.-X. A review on cell damage, viability, and functionality during 3D bioprinting. Mil. Med. Res. 2022, 9, 70. [Google Scholar] [CrossRef] [PubMed]
| Techniques | Advantages | References |
|---|---|---|
| Decellularized ECM scaffolds | Mimicking a non-immune environment with native three-dimensional structures and various bioactive components | [74,75,76] |
| Hydrogel scaffolds | Nontoxicity, low production costs, tunable and replicable mechanical and chemical properties | [77,78] |
| Electrospinning | High porosity and a larger surface area, promoting cell adhesion and nutrient transport | [79,80] |
| 3D printing | Easy to create, simulating muscle and tendon interfaces | [81] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tong, S.; Sun, Y.; Kuang, B.; Wang, M.; Chen, Z.; Zhang, W.; Chen, J. A Comprehensive Review of Muscle–Tendon Junction: Structure, Function, Injury and Repair. Biomedicines 2024, 12, 423. https://doi.org/10.3390/biomedicines12020423
Tong S, Sun Y, Kuang B, Wang M, Chen Z, Zhang W, Chen J. A Comprehensive Review of Muscle–Tendon Junction: Structure, Function, Injury and Repair. Biomedicines. 2024; 12(2):423. https://doi.org/10.3390/biomedicines12020423
Chicago/Turabian StyleTong, Siqi, Yuzhi Sun, Baian Kuang, Mingyue Wang, Zhixuan Chen, Wei Zhang, and Jialin Chen. 2024. "A Comprehensive Review of Muscle–Tendon Junction: Structure, Function, Injury and Repair" Biomedicines 12, no. 2: 423. https://doi.org/10.3390/biomedicines12020423
APA StyleTong, S., Sun, Y., Kuang, B., Wang, M., Chen, Z., Zhang, W., & Chen, J. (2024). A Comprehensive Review of Muscle–Tendon Junction: Structure, Function, Injury and Repair. Biomedicines, 12(2), 423. https://doi.org/10.3390/biomedicines12020423







