Do Vedolizumab trough Levels Predict the Outcome of Subsequent Therapy in Inflammatory Bowel Disease?
Abstract
1. Introduction
2. Materials and Methods
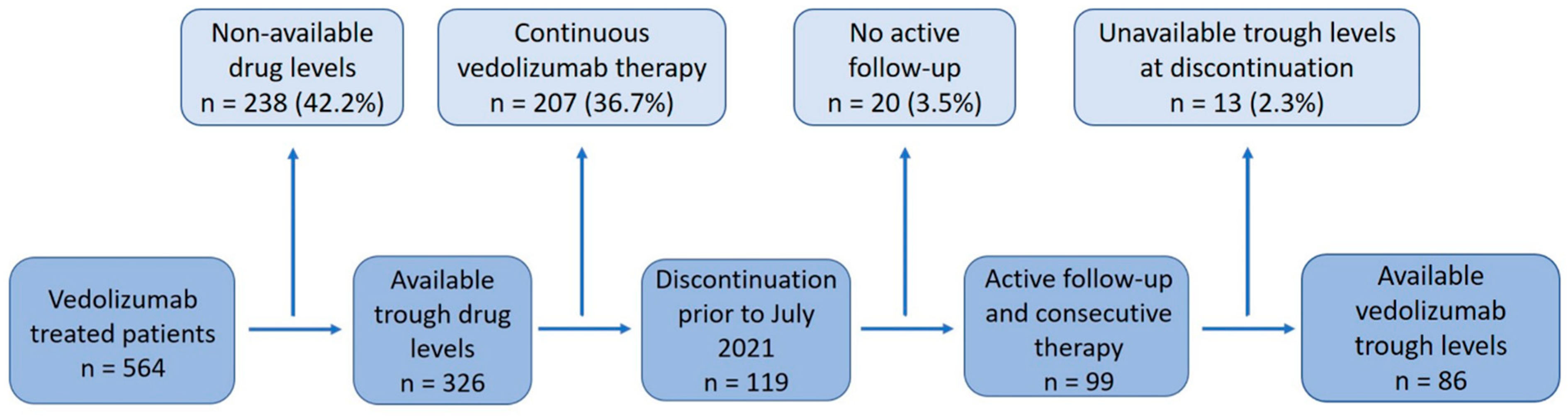
2.1. Patient Population
2.2. Measurement of Vedolizumab Concentrations
2.3. Determination of Anti-Vedolizumab Antibody Concentration
2.4. Clinical and Endoscopic Scores
2.5. Statistical Analysis
3. Results
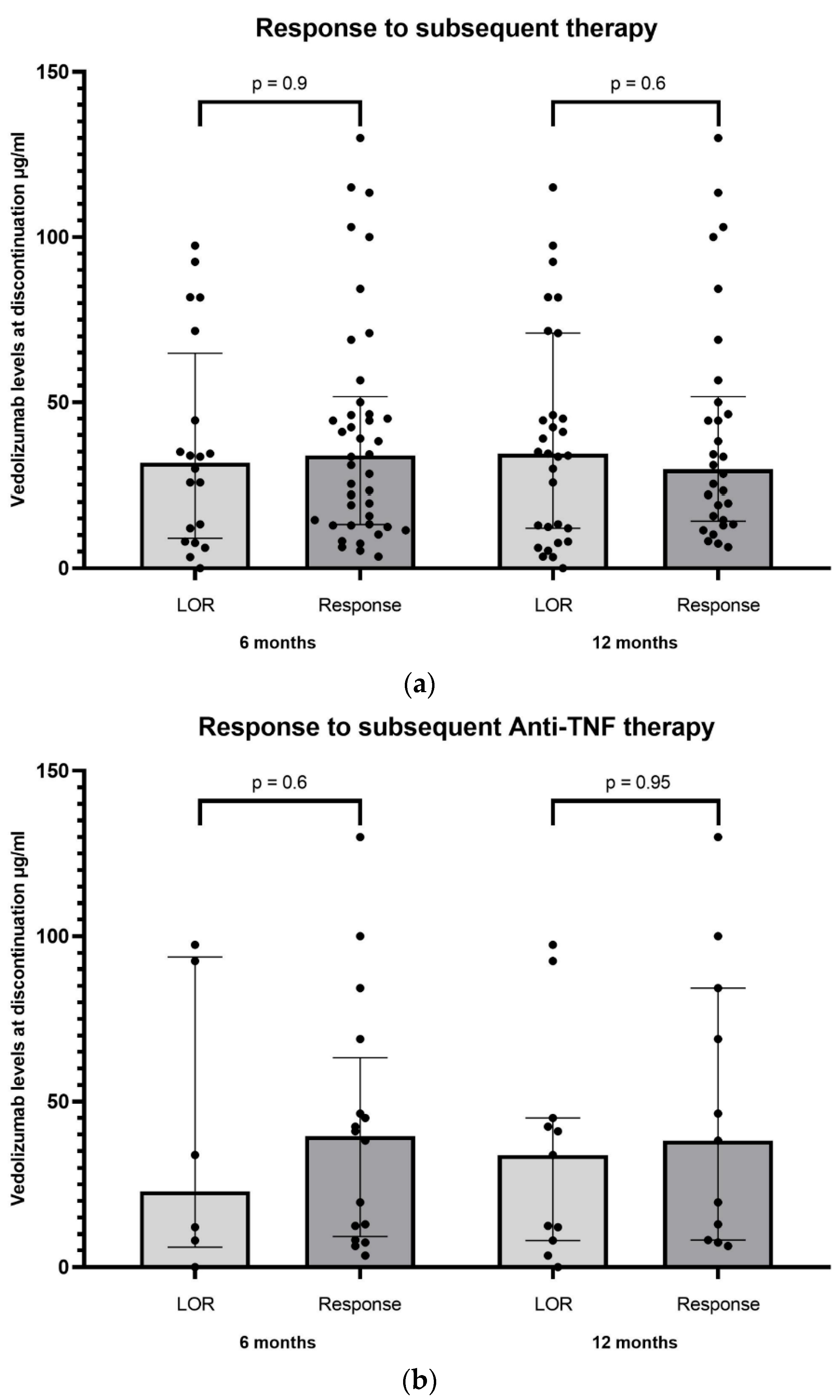
3.1. Vedolizumab and Subsequent Therapy
3.2. Outcome of Subsequent Medical (Non-Surgical) Therapy
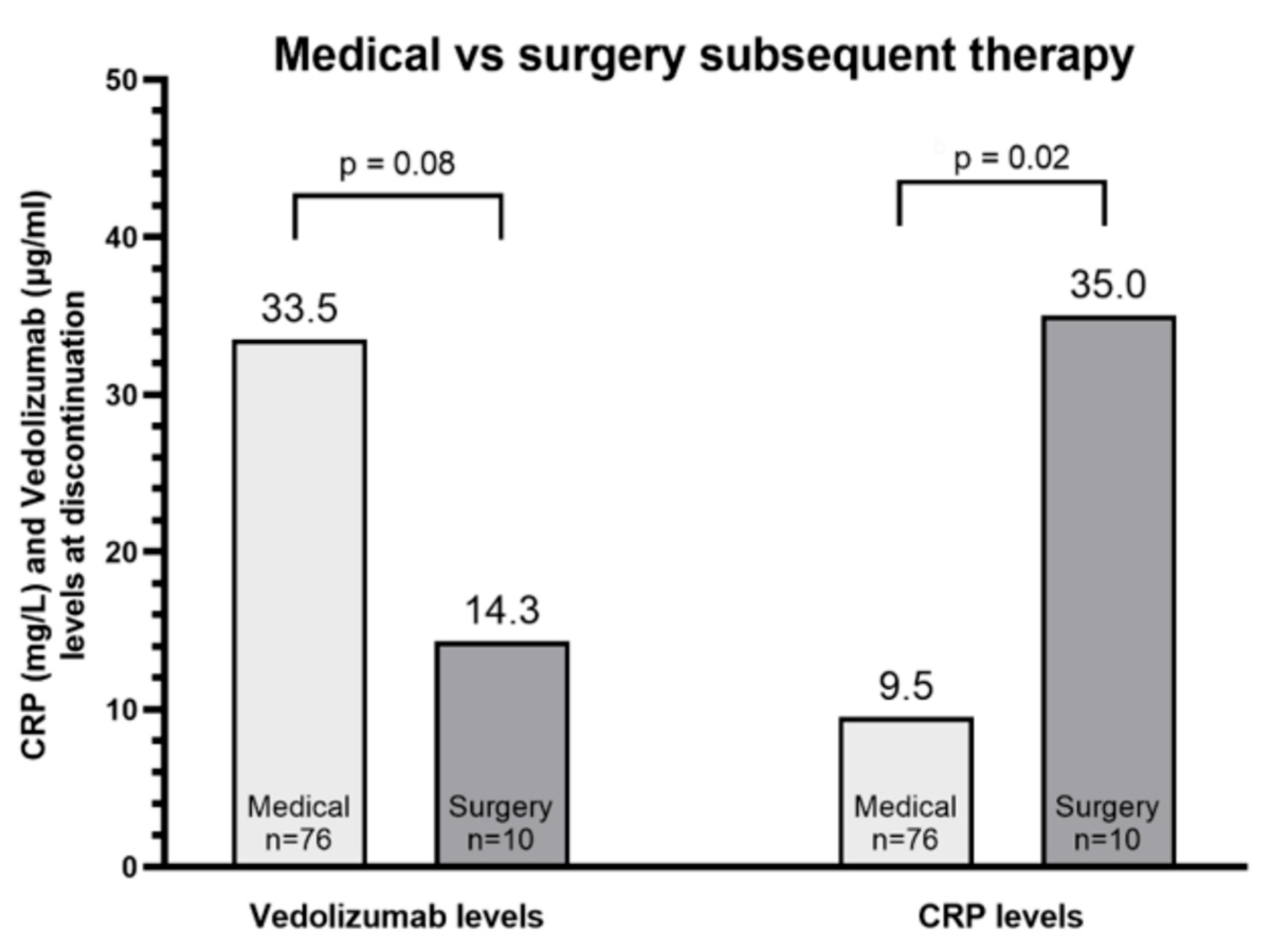
3.3. Subsequent Surgical Therapy
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Feagan, B.G.; Rutgeerts, P.; Sands, B.E.; Hanauer, S.; Colombel, J.-F.; Sandborn, W.J.; Van Assche, G.; Axler, J.; Kim, H.-J.; Danese, S.; et al. Vedolizumab as Induction and Maintenance Therapy for Ulcerative Colitis. N. Engl. J. Med. 2013, 369, 699–710. [Google Scholar] [CrossRef] [PubMed]
- Sandborn, W.J.; Feagan, B.G.; Rutgeerts, P.; Hanauer, S.; Colombel, J.F.; Sands, B.E.; Lukas, M.; Fedorak, R.N.; Lee, S.; Bressler, B.; et al. Vedolizumab as induction and maintenance therapy for Crohn’s disease: Commentary. Inflamm. Bowel Dis. Monit. 2014, 14, 55–56. [Google Scholar] [CrossRef] [PubMed]
- Rosario, M.; French, J.L.; Dirks, N.L.; Sankoh, S.; Parikh, A.; Yang, H.; Danese, S.; Colombel, J.-F.; Smyth, M.; Sandborn, W.J.; et al. Exposure–efficacy Relationships for Vedolizumab Induction Therapy in Patients with Ulcerative Colitis or Crohn’s Disease. J. Crohn’s Colitis 2017, 11, 921–929. [Google Scholar] [CrossRef]
- Vermeire, S.; Loftus, E.V., Jr.; Colombel, J.-F.; Feagan, B.G.; Sandborn, W.J.; Sands, B.E.; Danese, S.; D’Haens, G.R.; Kaser, A.; Panaccione, R.; et al. Long-term Efficacy of Vedolizumab for Crohn’s Disease. J. Crohn’s Colitis 2017, 11, 412–424. [Google Scholar] [CrossRef]
- Loftus, E.V., Jr.; Colombel, J.-F.; Feagan, B.G.; Vermeire, S.; Sandborn, W.J.; Sands, B.E.; Danese, S.; D’Haens, G.R.; Kaser, A.; Panaccione, R.; et al. Long-term Efficacy of Vedolizumab for Ulcerative Colitis. J. Crohn’s Colitis 2017, 11, 400–411. [Google Scholar] [CrossRef] [PubMed]
- Ungar, B.; Levy, I.; Yavne, Y.; Yavzori, M.; Picard, O.; Fudim, E.; Loebstein, R.; Chowers, Y.; Eliakim, R.; Kopylov, U.; et al. Optimizing Anti-TNF-α Therapy: Serum Levels of Infliximab and Adalimumab Are Associated with Mucosal Healing in Patients with Inflammatory Bowel Diseases. Clin. Gastroenterol. Hepatol. 2016, 14, 550–557.e2. [Google Scholar] [CrossRef]
- Davidov, Y.; Ungar, B.; Bar-Yoseph, H.; Carter, D.; Haj-Natour, O.; Yavzori, M.; Chowers, Y.; Eliakim, R.; Ben-Horin, S.; Kopylov, U. Association of Induction Infliximab Levels with Clinical Response in Perianal Crohn’s Disease. J. Crohn’s Colitis 2016, 11, 549–555. [Google Scholar] [CrossRef]
- Bortlik, M.; Duricova, D.; Malickova, K.; Machkova, N.; Bouzkova, E.; Hrdlicka, L.; Komárek, A.; Lukas, M. Infliximab trough levels may predict sustained response to infliximab in patients with Crohn’s disease. J. Crohn’s Colitis 2013, 7, 736–743. [Google Scholar] [CrossRef]
- Wilson, A.; Choi, B.; Sey, M.; Ponich, T.; Beaton, M.; Kim, R.B. High infliximab trough concentrations are associated with sustained histologic remission in inflammatory bowel disease: A prospective cohort study. BMC Gastroenterol. 2021, 21, 77. [Google Scholar] [CrossRef]
- Dreesen, E.; Verstockt, B.; Bian, S.; de Bruyn, M.; Compernolle, G.; Tops, S.; Noman, M.; Van Assche, G.; Ferrante, M.; Gils, A.; et al. Evidence to Support Monitoring of Vedolizumab Trough Concentrations in Patients with Inflammatory Bowel Diseases. Clin. Gastroenterol. Hepatol. 2018, 16, 1937–1946.e8. [Google Scholar] [CrossRef]
- Al-Bawardy, B.; Ramos, G.P.; Willrich, M.A.V.; Jenkins, S.M.; Park, S.H.; Aniwan, S.; Schoenoff, S.A.; Bruining, D.H.; Papadakis, K.A.; Raffals, L.; et al. Vedolizumab Drug Level Correlation with Clinical Remission, Biomarker Normalization, and Mucosal Healing in Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2019, 25, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Yacoub, W.; Williet, N.; Pouillon, L.; Di-Bernado, T.; Bittencourt, M.D.C.; Nancey, S.; Lopez, A.; Paul, S.; Zallot, C.; Roblin, X.; et al. Early vedolizumab trough levels predict mucosal healing in inflammatory bowel disease: A multicentre prospective observational study. Aliment. Pharmacol. Ther. 2018, 47, 906–912. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Dulai, P.S.; Casteele, N.V.; Battat, R.; Fumery, M.; Boland, B.S.; Sandborn, W.J. Systematic review with meta-analysis: Association between vedolizumab trough concentration and clinical outcomes in patients with inflammatory bowel diseases. Aliment. Pharmacol. Ther. 2019, 50, 848–857. [Google Scholar] [CrossRef]
- Peyrin-Biroulet, L.; Danese, S.; Argollo, M.; Pouillon, L.; Peppas, S.; Gonzalez-Lorenzo, M.; Lytras, T.; Bonovas, S. Loss of Response to Vedolizumab and Ability of Dose Intensification to Restore Response in Patients with Crohn’s Disease or Ulcerative Colitis: A Systematic Review and Meta-analysis. Clin. Gastroenterol. Hepatol. 2019, 17, 838–846.e2. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, B.P.; Yarur, A.J.; Graziano, E.; Campbell, J.P.; Bhattacharya, A.; Lee, J.Y.; Gheysens, K.; Papamichael, K.; Osterman, M.T.; Cheifetz, A.S.; et al. Vedolizumab Serum Trough Concentrations and Response to Dose Escalation in Inflammatory Bowel Disease. J. Clin. Med. 2020, 9, 3142. [Google Scholar] [CrossRef]
- Ungar, B.; Malickova, K.; Hanžel, J.; Abu Arisha, M.; Paul, S.; Rocha, C.; Ben Shatach, Z.; Abitbol, C.M.; Natour, O.H.; Selinger, L.; et al. Dose optimisation for Loss of Response to Vedolizumab— Pharmacokinetics and Immune Mechanisms. J. Crohn’s Colitis 2021, 15, 1707–1719. [Google Scholar] [CrossRef]
- Wu, Y.; Lin, B.; Thilakanathan, C.; Lehmann, P.; Xuan, W.; Mohsen, W.; Toong, C.; Williams, A.; Ng, W.; Connor, S. Therapeutic drug monitoring in inflammatory bowel disease reduces unnecessary use of infliximab with substantial associated cost-savings. Intern. Med. J. 2021, 51, 739–745. [Google Scholar] [CrossRef]
- Cheifetz, A.S.; Abreu, M.T.; Afif, W.; Cross, R.K.; Dubinsky, M.C.; Loftus, E.V.; Osterman, M.T.; Saroufim, A.; Siegel, C.A.; Yarur, A.J.; et al. A Comprehensive Literature Review and Expert Consensus Statement on Therapeutic Drug Monitoring of Biologics in Inflammatory Bowel Disease. Am. J. Gastroenterol. 2021, 116, 2014–2025. [Google Scholar] [CrossRef]
- Higgins, P.D.R.; Schwartz, M.; Mapili, J.; Krokos, I.; Leung, J.; Zimmermann, E.M. Patient defined dichotomous end points for remission and clinical improvement in ulcerative colitis. Gut 2005, 54, 782–788. [Google Scholar] [CrossRef]
- Brooke, B.N. Index of Crohn’S Disease Activity. Lancet 1980, 315, 711. [Google Scholar] [CrossRef]
- Daperno, M.; Haens, G.D.; Assche, G.; Van Baert, F. Development and validation of a new, simplified endoscopic activity score for Crohn’s disease: The SES-CD. Gastrointest. Endosc. 2004, 60, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Osterman, M.T.; Rosario, M.; Lasch, K.; Barocas, M.; Wilbur, J.D.; Dirks, N.L.; Gastonguay, M.R. Vedolizumab exposure levels and clinical outcomes in ulcerative colitis: Determining the potential for dose optimisation. Aliment. Pharmacol. Ther. 2019, 49, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Casteele, N.V.; Sandborn, W.J.; Feagan, B.G.; Vermeire, S.; Dulai, P.S.; Yarur, A.; Roblin, X.; Ben-Horin, S.; Dotan, I.; Osterman, M.T.; et al. Real-world multicentre observational study including population pharmacokinetic modelling to evaluate the exposure–response relationship of vedolizumab in inflammatory bowel disease: ERELATE Study. Aliment. Pharmacol. Ther. 2022, 56, 463–476. [Google Scholar] [CrossRef] [PubMed]
- Guidi, L.; Pugliese, D.; Tonucci, T.P.; Bertani, L.; Costa, F.; Privitera, G.; Tolusso, B.; Di Mario, C.; Albano, E.; Tapete, G.; et al. Early vedolizumab trough levels predict treatment persistence over the first year in inflammatory bowel disease. United Eur. Gastroenterol. J. 2019, 7, 1189–1197. [Google Scholar] [CrossRef]
- Hanžel, J.; Sever, N.; Ferkolj, I.; Štabuc, B.; Smrekar, N.; Kurent, T.; Koželj, M.; Novak, G.; Compernolle, G.; Tops, S.; et al. Early vedolizumab trough levels predict combined endoscopic and clinical remission in inflammatory bowel disease. United Eur. Gastroenterol. J. 2019, 7, 741–749. [Google Scholar] [CrossRef]
- Liefferinckx, C.; Minsart, C.; Cremer, A.; Amininejad, L.; Tafciu, V.; Quertinmont, E.; Tops, S.; Devière, J.; Gils, A.; van Gossum, A.; et al. Early vedolizumab trough levels at induction in inflammatory bowel disease patients with treatment failure during maintenance. Eur. J. Gastroenterol. Hepatol. 2019, 31, 478–485. [Google Scholar] [CrossRef]
- Yarur, A.J.; Bruss, A.; Naik, S.; Beniwal-Patel, P.; Fox, C.; Jain, A.; Berens, B.; Patel, A.; Ungaro, R.; Bahur, B.; et al. Vedolizumab Concentrations Are Associated with Long-Term Endoscopic Remission in Patients with Inflammatory Bowel Diseases. Dig. Dis. Sci. 2019, 64, 1651–1659. [Google Scholar] [CrossRef]
- Outtier, A.; Wauters, L.; Rahier, J.F.; Bossuyt, P.; Colard, A.; Franchimont, D.; Lambrecht, G.; Macken, E.; Van Moerkercke, W.; Baert, F.; et al. Effect of vedolizumab dose intensification on serum drug concentrations and regain of response in inflammatory bowel disease patients with secondary loss of response. GastroHep 2021, 3, 63–71. [Google Scholar] [CrossRef]
- Vermeire, S.; Lukáš, M.; Magro, F.; Adsul, S.; Lindner, D.; Rosario, M.; Roth, J.; Danese, S. Vedolizumab efficacy, safety, and pharmacokinetics with reduced frequency of dosing from every 4 weeks to every 8 weeks in patients with Crohn’s disease or ulcerative colitis. J. Crohn’s Colitis 2020, 14, 1066–1073. [Google Scholar] [CrossRef]
- Osterman, M.T.; Jairath, V.; Rana-Khan, Q.; James, A.; Balma, D.; Mehrotra, S.; Yang, L.; Lasch, K.; Yarur, A.J. A randomized trial of vedolizumab dose optimization in patients with moderate to severe ulcerative colitis who have early nonresponse and high drug clearance: The enterpret trial. Gastroenterology 2022, 162, 7. [Google Scholar] [CrossRef]
- Ungar, B.; Kopylov, U.; Yavzori, M.; Fudim, E.; Picard, O.; Lahat, A.; Coscas, D.; Waterman, M.; Haj-Natour, O.; Orbach-Zingboim, N.; et al. Association of Vedolizumab Level, Anti-Drug Antibodies, and α4β7 Occupancy with Response in Patients with Inflammatory Bowel Diseases. Clin. Gastroenterol. Hepatol. 2018, 16, 697–705.e7. [Google Scholar] [CrossRef] [PubMed]
- Schulze, H.; Esters, P.; Hartmann, F.; Stein, J.; Christ, C.; Zorn, M.; Dignass, A. A prospective cohort study to assess the relevance of vedolizumab drug level monitoring in IBD patients. Scand. J. Gastroenterol. 2018, 53, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Dulai, P.S.; Boland, B.S.; Singh, S.; Chaudrey, K.; Koliani-Pace, J.L.; Kochhar, G.; Parikh, M.P.; Shmidt, E.; Hartke, J.; Chilukuri, P.; et al. Development and Validation of a Scoring System to Predict Outcomes of Vedolizumab Treatment in Patients with Crohn’s Disease. Gastroenterology 2018, 155, 687–695.e10. [Google Scholar] [CrossRef] [PubMed]
- Parrish, A.B.; Lopez, N.E.; Truong, A.; Zaghiyan, K.; Melmed, G.Y.; McGovern, D.P.B.; Ha, C.; Syal, G.; Bonthala, N.; Jain, A.; et al. Preoperative Serum Vedolizumab Levels Do Not Impact Postoperative Outcomes in Inflammatory Bowel Disease. Dis. Colon Rectum 2021, 10, 1259–1266. [Google Scholar] [CrossRef]


| Study Population n = 86 | |
| Age of diagnosis, n (median, IQR) | 26 (18, 39.5) |
| Male, n (%) Female, n (%) | 48 (55.8) 38 (44.2) |
| CD, n (%) UC, n (%) | 51 (59.3) 35 (40.7) |
| CD extent, n (% of CD patients) L1 (ileal) L2 (colonic) L3 (ileo-colonic) Perianal disease | 19 (37.2) 4 (7.8) 28 (55) 16 (31.4) |
| CD behavior, n (% of CD patients) B1 (non-stricturing and non-penetrating) B2 (stricturing) B3 (penetrating) B2 + B3 | 13 (25.5) 16 (31.4) 18 (35.3) 4 (7.8) |
| UC extent, n (% of UC patients) E1 (proctitis) E2 (left sided colitis) E3 (right sided colitis) | 0 (0) 13 (37.1) 22 (62.9) |
| Extra-intestinal manifestations, n (%) | 39 (45.3) |
| Smoking (current or past), n (%) | 19 (22.1) |
| Past IBD Treatment | |
| Immunomodulator, n (%) | 56 (65.1) |
| 5-ASA derivatives, n (%) | 49 (57) |
| Infliximab and reason for cessation, n (%) LOR Immunogenicity Adverse event Compliance Other | 53 (61.6) 26 (49.1) 13 (24.5) 11 (20.7) 0 (0) 3 (5.7) |
| Adalimumab and reason for cessation, n (%) LOR Immunogenicity Adverse event Compliance Other | 37 (43) 23 (62.1) 6 (16.2) 5 (13.5) 1 (2.7) 2 (5.4) |
| Ustekinumab and reason for cessation, n (%) LOR | 5 (5.8) 5 (100) |
| Vedolizumab Treatment | |
| Duration of treatment, median weeks, n (IQR) | 46.4 (29.6, 83.6) |
| Reason for therapy cessation, n (%) LOR Adverse event Compliance | 72 (83.7) 13 (15.1) 1 (1.2) |
| Adverse events leading to therapy cessation, n (% of adverse events) Erythema Nodosum Arthralgia Paraesthesia Rash Acute otitis media Headache Recurrent pharyngitis Elevated liver enzymes | 1 (7.7) 4 (30.7) 1 (7.7) 3 (23.1) 1 (7.7) 1 (7.7) 1 (7.7) 1 (7.7) |
| Additional therapy on stop, n (%) Steroids Immunomodulators 5-ASA derivatives | 28 (32.5) 9 (10.4) 24 (27.9) |
| Last drug levels, μg/mL (IQR) | 30 (12.5, 53) |
| Vedolizumab dose interval on discontinuation, n (%) 4 weeks 8 weeks | 60 (69.8) 26 (30.2) |
| CRP on stop, mg/L (IQR) | 11 (4.6, 34.25) |
| Sequential Treatment, n (%) | |
| Infliximab Adalimumab Ustekinumab Certolizumab Golimumab Tofacitinib Tacrolimus Research Surgery | 19 (22.1) 7 (8.1) 24 (27.9) 6 (7) 1 (1.2) 7 (8.1) 1 (1.2) 11 (12.8) 10 (11.6) |
| Duration between vedolizumab cessation and initiation of subsequent therapy, median weeks, n (IQR) | 8.8 (5.32, 21.1) |
| Reason for cessation of subsequent therapy, n (%) LOR Immunogenicity Adverse event Compliance Other Therapy ongoing | 39 (51.3) 2 (2.6) 9 (11.8) 1 (1.3) 2 (2.6) 24 (31.6) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Levartovsky, A.; Cohen, I.; Abitbol, C.M.; Yavzori, M.; Fudim, E.; Picard, O.; Kopylov, U.; Ben-Horin, S.; Ungar, B. Do Vedolizumab trough Levels Predict the Outcome of Subsequent Therapy in Inflammatory Bowel Disease? Biomedicines 2023, 11, 1553. https://doi.org/10.3390/biomedicines11061553
Levartovsky A, Cohen I, Abitbol CM, Yavzori M, Fudim E, Picard O, Kopylov U, Ben-Horin S, Ungar B. Do Vedolizumab trough Levels Predict the Outcome of Subsequent Therapy in Inflammatory Bowel Disease? Biomedicines. 2023; 11(6):1553. https://doi.org/10.3390/biomedicines11061553
Chicago/Turabian StyleLevartovsky, Asaf, Ido Cohen, Chaya Mushka Abitbol, Miri Yavzori, Ella Fudim, Orit Picard, Uri Kopylov, Shomron Ben-Horin, and Bella Ungar. 2023. "Do Vedolizumab trough Levels Predict the Outcome of Subsequent Therapy in Inflammatory Bowel Disease?" Biomedicines 11, no. 6: 1553. https://doi.org/10.3390/biomedicines11061553
APA StyleLevartovsky, A., Cohen, I., Abitbol, C. M., Yavzori, M., Fudim, E., Picard, O., Kopylov, U., Ben-Horin, S., & Ungar, B. (2023). Do Vedolizumab trough Levels Predict the Outcome of Subsequent Therapy in Inflammatory Bowel Disease? Biomedicines, 11(6), 1553. https://doi.org/10.3390/biomedicines11061553






