Lung Transplantation for Pleuroparenchymal Fibroelastosis: A Single-Center Experience with Revision of Literature
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Methods
2.2. Review of Current Literature
3. Results
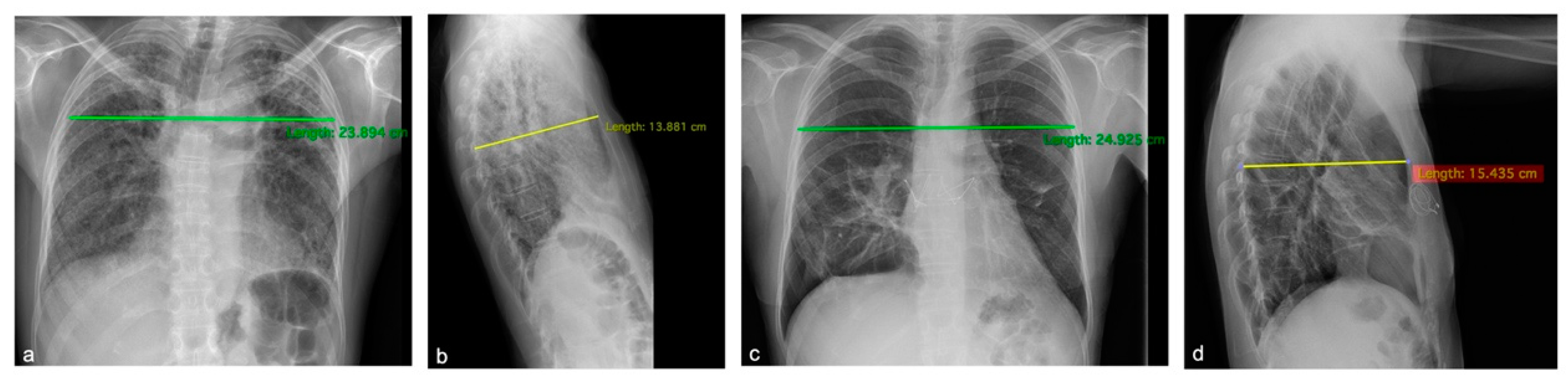
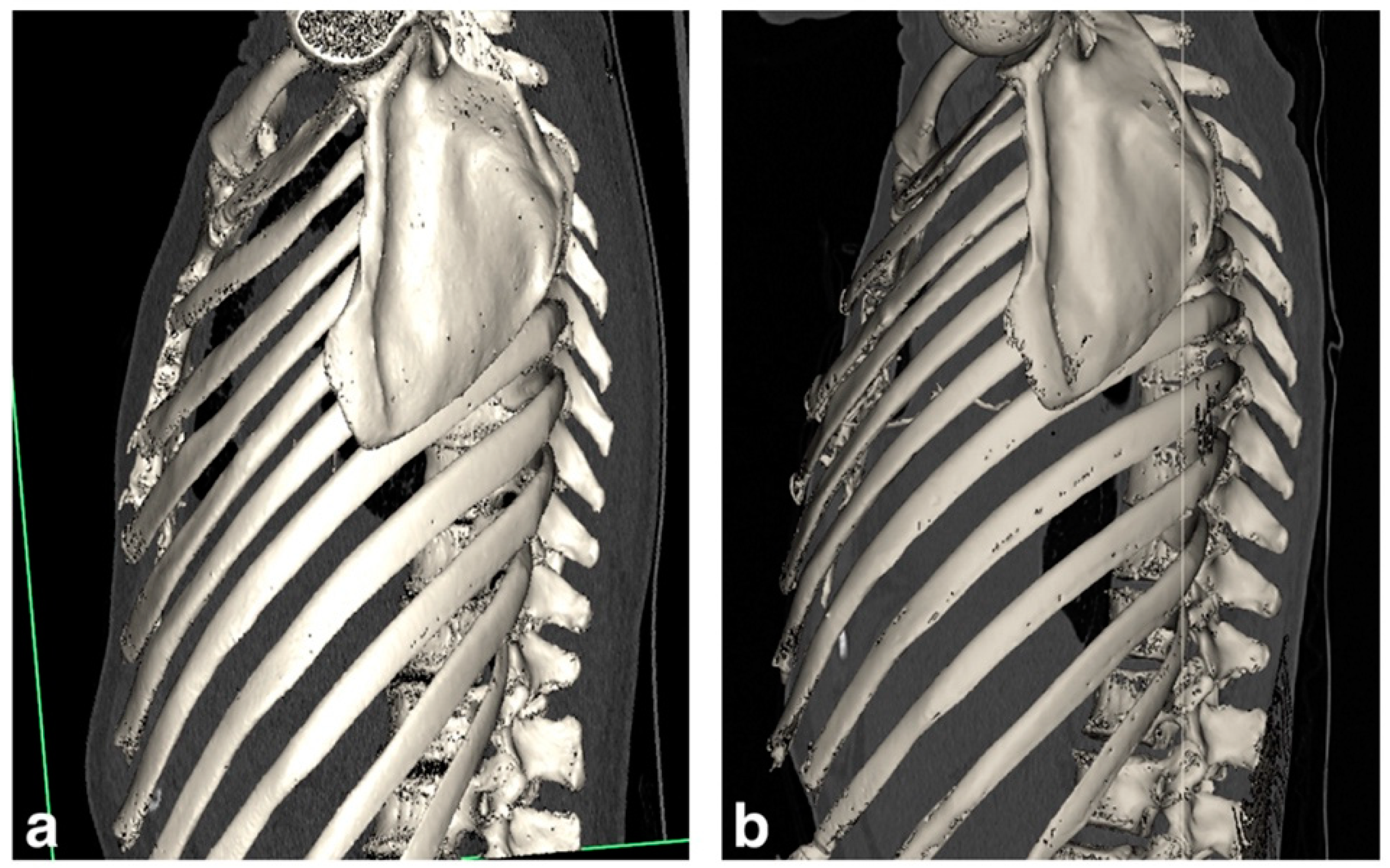
3.1. Case Presentations
3.1.1. Patient 1
3.1.2. Patient 2
3.1.3. Patient 3
3.1.4. Patient 4
3.1.5. Patient 5
3.1.6. Patient 6
3.2. Revision of the Available Literature
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Travis, W.D.; Costabel, U.; Hansell, D.M.; King, T.E., Jr.; Lynch, D.A.; Nicholson, A.G.; Ryerson, C.J.; Ryu, J.H.; Selman, M.; Wells, A.U.; et al. An official American Thoracic Society/European Respiratory Society statement: Update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. Am. J. Respir. Crit. Care Med. 2013, 188, 733–748. [Google Scholar] [CrossRef] [PubMed]
- Harada, T.; Yoshida, Y.; Kitasato, Y.; Tsuruta, N.; Wakamatsu, K.; Hirota, T.; Tanaka, M.; Tashiro, N.; Ishii, H.; Shiraishi, M.; et al. The thoracic cage becomes flattened in the progression of pleuroparenchymal fibroelastosis. Eur. Respir. Rev. 2014, 23, 263–266. [Google Scholar] [CrossRef] [PubMed]
- Yanagiya, M.; Sato, M.; Kawashima, S.; Kuwano, H.; Nagayama, K.; Nitadori, J.-I.; Anraku, M.; Nakajima, J. Flat Chest of Pleuroparenchymal Fibroelastosis Reversed by Lung Transplantation. Ann. Thorac. Surg. 2016, 102, e347–e349. [Google Scholar] [CrossRef] [PubMed]
- Mariani, F.; Gatti, B.; Rocca, A.; Bonifazi, F.; Cavazza, A.; Fanti, S.; Tomassetti, S.; Piciucchi, S.; Poletti, V.; Zompatori, M. Pleuroparenchymal fibroelastosis: The prevalence of secondary forms in hematopoietic stem cell and lung transplantation recipients. Diagn. Interv. Radiol. 2016, 22, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Beynat-Mouterde, C.; Beltramo, G.; Lezmi, G.; Pernet, D.; Camus, C.; Fanton, A.; Foucher, P.; Cottin, V.; Bonniaud, P. Pleuroparenchymal fibroelastosis as a late complication of chemotherapy agents. Eur. Respir. J. 2014, 44, 523–527. [Google Scholar] [CrossRef]
- Baroke, E.; Heussel, C.P.; Warth, A.; Eichinger, M.; Oltmanns, U.; Palmowski, K.; Herth, F.J.; Kreuter, M. Pleuroparenchymal fibroelastosis in association with carcinomas. Respirology 2015, 21, 191–194. [Google Scholar] [CrossRef]
- Huang, Z.; Li, S.; Zhu, Y.; Zhu, H.; Yi, X. Pleuroparenchymal fibroelastosis associated with aluminosilicate dust: A case report. Int. J. Clin. Exp. Pathol. 2015, 8, 8676–8679. [Google Scholar]
- Reddy, T.L.; Tominaga, M.; Hansell, D.M.; von der Thusen, J.; Rassl, D.; Parfrey, H.; Guy, S.; Twentyman, O.; Rice, A.; Maher, T.M.; et al. Pleuroparenchymal fibroelastosis: A spectrum of histopathological and imaging phenotypes. Eur. Respir. J. 2012, 40, 377–385. [Google Scholar] [CrossRef]
- Frankel, S.K.; Cool, C.D.; Lynch, D.A.; Brown, K.K. Idiopathic Pleuroparenchymal Fibroelastosis. Chest 2004, 126, 2007–2013. [Google Scholar] [CrossRef]
- Ali, M.S.; Ramalingam, V.S.; Haasler, G.; Presberg, K. Pleuroparenchymal fibroelastosis (PPFE) treated with lung transplantation and review of the literature. BMJ Case Rep. 2019, 12, e229402. [Google Scholar] [CrossRef]
- Aljefri, N.A.; Abothenain, F.F.; Hussein, A.M.; Saleh, W.; Alkattan, K.; Mohammed, S.F.; Alhajji, M. Idiopathic pleuroparenchymal fibroelastosis: The first case to be managed with a successful lung transplant at King Faisal Specialist Hospital and Research Center, Riyadh. Ann. Thorac. Med. 2019, 14, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Sekine, A.; Seo, K.; Matsukura, S.; Sato, M.; Shinozaki-Ushiku, A.; Ogura, T.; Kitami, A.; Kadokura, M.; Dohi, S.; Ichizuka, K.; et al. A case report of idiopathic pleuroparenchymal fibroelastosis with severe respiratory failure in pregnancy. BMC Pulm. Med. 2020, 20, 264. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Matsubara, K.; Miyagawa-Hayashino, A.; Tada, K.; Handa, T.; Yamada, T.; Sato, M.; Aoyama, A.; Date, H. Lung Transplantation for Pleuroparenchymal Fibroelastosis After Chemotherapy. Ann. Thorac. Surg. 2014, 98, e115–e117. [Google Scholar] [CrossRef]
- Portillo, K.; Guasch, I.; Becker, C.; Andreo, F.; Fernández-Figueras, M.T.; Ramirez Ruz, J.; Martinez-Barenys, C.; García-Reina, S.; Lopez de Castro, P.; Sansano, I.; et al. Pleuroparenchymal Fibroelastosis: A New Entity within the Spectrum of Rare Idiopathic Interstitial Pneumonias. Case Rep. Pulmonol. 2015, 2015, 810515. [Google Scholar] [CrossRef]
- Hata, A.; Nakajima, T.; Yoshida, S.; Kinoshita, T.; Terada, J.; Tatsumi, K.; Matsumiya, G.; Date, H.; Yoshino, I. Living Donor Lung Transplantation for Pleuroparenchymal Fibroelastosis. Ann. Thorac. Surg. 2016, 101, 1970–1972. [Google Scholar] [CrossRef]
- Huang, H.; Feng, R.; Li, S.; Wu, B.; Xu, K.; Xu, Z.; Chen, J. A CARE-compliant case report. Lung transplantation for a Chinese young man with idiopathic pleuroparenchymal fibroelastosis. Medicine 2017, 96, e6900. [Google Scholar] [CrossRef]
- Righi, I.; Morlacchi, L.C.; Rossetti, V.; Mendogni, P.; Palleschi, A.; Tosi, D.; Pieropan, S.; Del Gobbo, A.; Nosotti, M. Lung Transplantation as Successful Treatment of End-stage Idiopathic Pleuroparenchymal Fibroelastosis: A Case Report. Transplant. Proc. 2019, 51, 235–238. [Google Scholar] [CrossRef]
- Shimada, A.; Terada, J.; Tsushima, K.; Tateishi, Y.; Abe, R.; Oda, S.; Kobayashi, M.; Yamane, M.; Oto, T.; Tatsumi, K. Veno-venous extracorporeal membrane oxygenation bridged living-donor lung transplantation for rapid progressive respiratory failure with pleuroparenchymal fibroelastosis after allogeneic hematopoietic stem cell transplantation. Respir. Investig. 2018, 56, 258–262. [Google Scholar] [CrossRef]
- Tsubosaka, A.; Matsushima, J.; Ota, M.; Suzuki, M.; Yonemori, Y.; Ota, S.; Yoshino, I.; Tsushima, K.; Tatsumi, K.; Nakatani, Y. Whole-lung pathology of pleuroparenchymal fibroelastosis (PPFE) in an explanted lung: Significance of elastic fiber-rich, non-specific interstitial pneumonia-like change in chemotherapy-related PPFE. Pathol. Int. 2019, 69, 547–555. [Google Scholar] [CrossRef]
- Rasciti, E.; Cancellieri, A.; Romagnoli, M.; Dell’Amore, A.; Zompatori, M. Suspected pleuroparenchymal fibroelastosis relapse after lung transplantation: A case report and literature review. BJR Case Rep. 2019, 5, 20190040. [Google Scholar] [CrossRef] [PubMed]
- Shiiya, H.; Nakajima, J.; Date, H.; Chen-Yoshikawa, T.F.; Tanizawa, K.; Handa, T.; Oto, T.; Otani, S.; Shiotani, T.; Okada, Y.; et al. Outcomes of lung transplantation for idiopathic pleuroparenchymal fibroelastosis. Surg. Today 2021, 51, 1276–1284. [Google Scholar] [CrossRef] [PubMed]
- Shiiya, H.; Sato, M.; Shinozaki-Ushiku, A.; Konoeda, C.; Kitano, K.; Nakajima, J. Exacerbation of Secondary Pulmonary Hypertension by Flat Chest after Lung Transplantation. Ann. Thorac. Cardiovasc. Surg. 2022, 28, 298–301. [Google Scholar] [CrossRef] [PubMed]
- Ruaro, B.; Tavano, S.; Confalonieri, P.; Pozzan, R.; Hughes, M.; Braga, L.; Volpe, M.C.; Ligresti, G.; Andrisano, A.G.; Lerda, S.; et al. Transbronchial lung cryobiopsy and pulmonary fibrosis: A never-ending story? Heliyon 2023, 9, e14768. [Google Scholar] [CrossRef]
- Ando, H.; Suzuki, K.; Yanagihara, T. Insights into Potential Pathogenesis and Treatment Options for Immune-Checkpoint Inhibitor-Related Pneumonitis. Biomedicines 2021, 9, 1484. [Google Scholar] [CrossRef]
- Watanabe, K. Pleuroparenchymal Fibroelastosis: Its Clinical Characteristics. Curr. Respir. Med. Rev. 2014, 9, 229–237. [Google Scholar] [CrossRef]
- Balestro, E.; Cocconcelli, E.; Tinè, M.; Biondini, D.; Faccioli, E.; Saetta, M.; Rea, F. Idiopathic Pulmonary Fibrosis and Lung Transplantation: When it is Feasible. Medicina 2019, 55, 702. [Google Scholar] [CrossRef]
- Leard, L.E.; Holm, A.M.; Valapour, M.; Glanville, A.R.; Attawar, S.; Aversa, M.; Campos, S.V.; Christon, L.M.; Cypel, M.; Dellgren, G.; et al. Consensus document for the selection of lung transplant candidates: An update from the International Society for Heart and Lung Transplantation. J. Heart Lung Transplant. 2021, 40, 1349–1379. [Google Scholar] [CrossRef]
- Zhang, S.; Xie, W.; Wang, Z.; Tian, Y.; Da, J.; Zhai, Z. Pleuroparenchymal fibroelastosis secondary to autologous hematopoietic stem cell transplantation: A case report. Exp. Ther. Med. 2019, 17, 2557–2560. [Google Scholar] [CrossRef]
- Oda, T.; Sekine, A.; Tabata, E.; Iwasawa, T.; Takemura, T.; Ogura, T. Comparison of Clinical Characteristics and Outcomes between Idiopathic and Secondary Pleuroparenchymal Fibroelastosis. J. Clin. Med. 2021, 10, 846. [Google Scholar] [CrossRef]
| Patient | Age | Sex | BMI | LAS | Waiting List Time (Days) | Diagnosis | Time from Diagnosis to LT (Months) | FVC Pre (%) | DLCO Pre (%) | mPAP (mmHg) |
|---|---|---|---|---|---|---|---|---|---|---|
| # 1 | 62 | M | 19.8 | 32.9 | 469 | IPPFE | 72 | 47 | 38 | 14 |
| # 2 | 61 | F | 18.2 | 33.9 | 144 | IPPFE | 84 | 58 | 23 | 13.5 |
| # 3 | 51 | F | 19.8 | 38.5 | 456 | IPPFE | 60 | 31 | 20 | 20 |
| # 4 | 53 | M | 21.2 | 36.9 | 29 | UIP + IPPFE | 12 | 56 | 24 | 10 |
| # 5 | 51 | M | 18.5 | 41.1 | 90 | BOS in IPPFE | 12 * | 41 | 46 | 17 |
| # 6 | 39 | F | 19.5 | 50.8 | 136 | Secondary PPFE | 10 | 13 | 30 | 32 |
| LT Type | LT Time (min) | ECMO | MV (h) | ICU Stay (Days) | In H Stay (Days) | * PGD | CLAD | ** FVC Post (%) | ** DLCO Post (%) | OS (Months) | Status | Death Cause | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pre | Intra | Prolonged | ||||||||||||
| BLT | 580 | No | Yes | No | 280 | 12 | 32 | 0 | No | 73 | 35 | 7 | Alive | - |
| BLT | 320 | No | Yes | No | 16 | 4 | 28 | 0 | No | 87 | 39 | 13 | Death | MOF |
| BLT | 350 | No | Yes | No | 40 | 5 | 30 | 0 | No | 86 | 44 | 29 | Alive | - |
| BLT | 345 | No | Yes | No | 18 | 4 | 31 | 0 | No | 86 | 44 | 6 | Death | COVID-19 Pneumonia |
| BLT | 560 | No | Yes | No | 560 | 38 | 38 | 3 | - | - | - | 0 | Death | Aspergillus Pneumonia |
| BLT | 410 | Yes | Yes | Yes | 480 | 18 | 18 | 3 | - | - | - | 0 | Death | MOF |
| Author, Year | Nº of Reported Cases | Age | Sex | Diagnosis | Time from Diagnosis to LT (Months) | Type of LT | In H Stay (Days) | In H Mortality (Days) | * Status | OS (Months) |
|---|---|---|---|---|---|---|---|---|---|---|
| Chen, 2014 [14] | 1 | 14 | M | Secondary PPFE | After LT | Left SLT | NR | No | Alive | 4 |
| Portillo, 2015 [15] | 1 | 25 | M | Secondary PPFE | 14 | BLT | NR | No | Alive | 24 |
| Yanagiya, 2016 [3] | 1 | 27 | F | IPPFE | 3 | LDLLT | NR | No | Alive | 6 |
| Hata, 2016 [16] | 1 | 19 | M | Secondary PPFE | 60 | LDLLT | 25 | No | Alive | 1 |
| Huang, 2017 [17] | 1 | 36 | M | IPPFE | 36 | BLT | NR | No | Alive | NR |
| Aljefri, 2018 [11] | 1 | 27 | M | IPPFE | After LT | BLT | 45 | No | NR | NR |
| Righi, 2018 [18] | 1 | 42 | M | IPPFE | 36 | BLT | 30 | No | Alive | 24 |
| Shimada, 2018 [19] | 1 | 21 | F | Secondary PPFE | 24 | LDLLT | 120 | No | Alive | 12 |
| Tsubosaka, 2018 [20] | 1 | 19 | M | Secondary PPFE | 24 | LDLLT | NR | NR | NR | NR |
| Ali, 2019 [10] | 1 | 26 | F | IPPFE | 24 | BLT | NR | No | Alive | 44 |
| Rasciti, 2019 [21] | 1 | 48 | M | IPPFE | 12 | First BLT Re-BLT | 20 28 | No No | Alive | 12 (from Re.LT) |
| Sekine, 2020 [12] | 1 | 29 | F | IPPFE | 36 | LDLLT | 365 | No | Alive | 24 |
| Shiiya, 2021 [22] | 31 | 51 ** | F 58% M 42% | IPPFE | NR | 65% BLT 35% SLT | 99 ** | 3% | 61% Alive 39% Dead | ~100 |
| Shiiya, 2022 [23] | 1 | 40 | F | IPPFE | NR | Left SLT | 120 | No | Dead | 18 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faccioli, E.; Verzeletti, V.; Giraudo, C.; Schiavon, M.; Calabrese, F.; Loy, M.; Rea, F.; Dell’Amore, A. Lung Transplantation for Pleuroparenchymal Fibroelastosis: A Single-Center Experience with Revision of Literature. Biomedicines 2023, 11, 1505. https://doi.org/10.3390/biomedicines11061505
Faccioli E, Verzeletti V, Giraudo C, Schiavon M, Calabrese F, Loy M, Rea F, Dell’Amore A. Lung Transplantation for Pleuroparenchymal Fibroelastosis: A Single-Center Experience with Revision of Literature. Biomedicines. 2023; 11(6):1505. https://doi.org/10.3390/biomedicines11061505
Chicago/Turabian StyleFaccioli, Eleonora, Vincenzo Verzeletti, Chiara Giraudo, Marco Schiavon, Fiorella Calabrese, Monica Loy, Federico Rea, and Andrea Dell’Amore. 2023. "Lung Transplantation for Pleuroparenchymal Fibroelastosis: A Single-Center Experience with Revision of Literature" Biomedicines 11, no. 6: 1505. https://doi.org/10.3390/biomedicines11061505
APA StyleFaccioli, E., Verzeletti, V., Giraudo, C., Schiavon, M., Calabrese, F., Loy, M., Rea, F., & Dell’Amore, A. (2023). Lung Transplantation for Pleuroparenchymal Fibroelastosis: A Single-Center Experience with Revision of Literature. Biomedicines, 11(6), 1505. https://doi.org/10.3390/biomedicines11061505







