Checkpoint Inhibitor-Induced Colitis: An Update
Abstract
1. Introduction
1.1. General Principles of Immunotherapy and Immune Checkpoint Inhibitors (ICIs)
1.2. Immuno-Related Adverse Events (irAEs) and ICI Colitis
2. Epidemiology
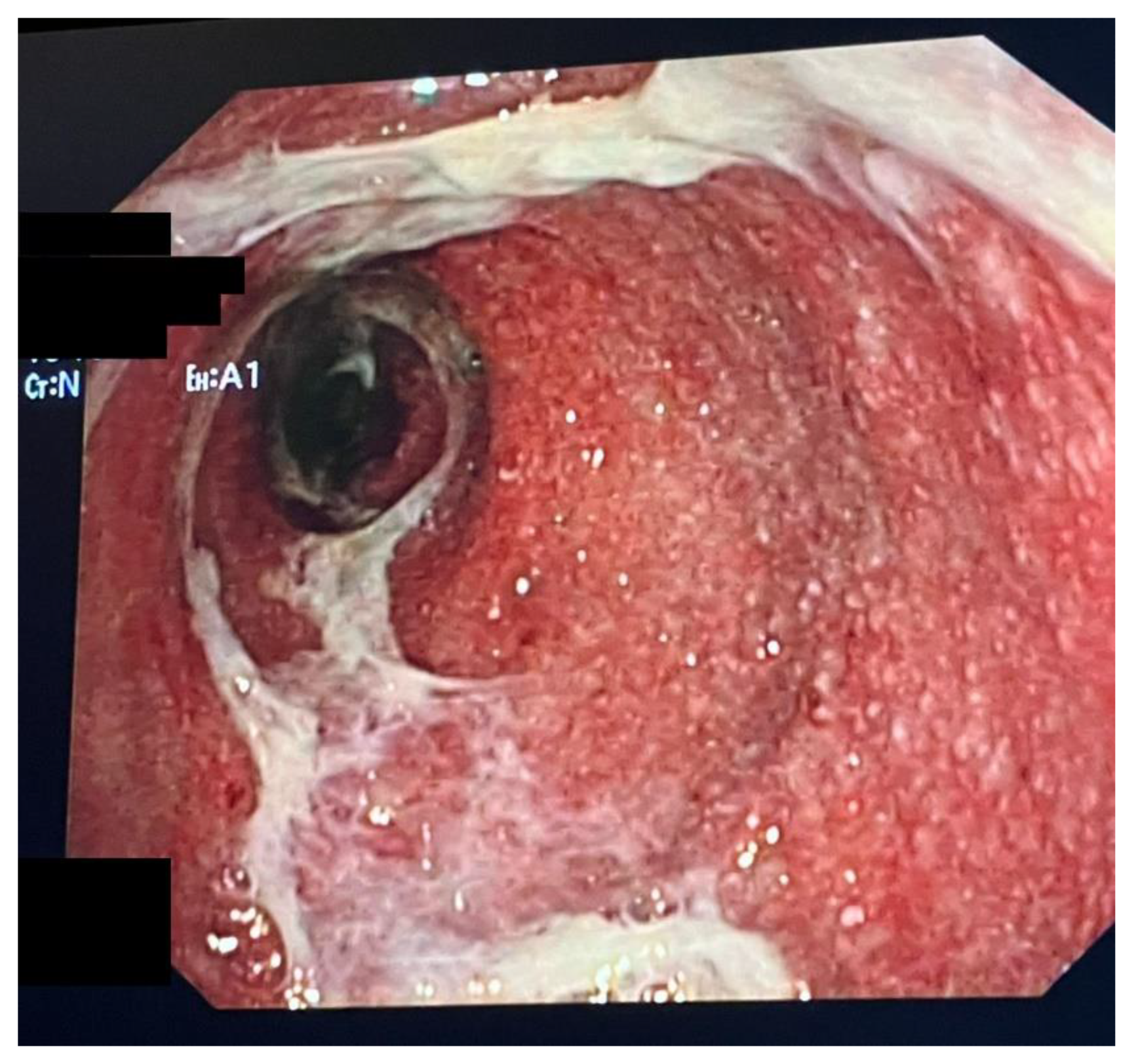
3. Clinical Presentation, Diagnosis, Endoscopic and Histopathological Findings
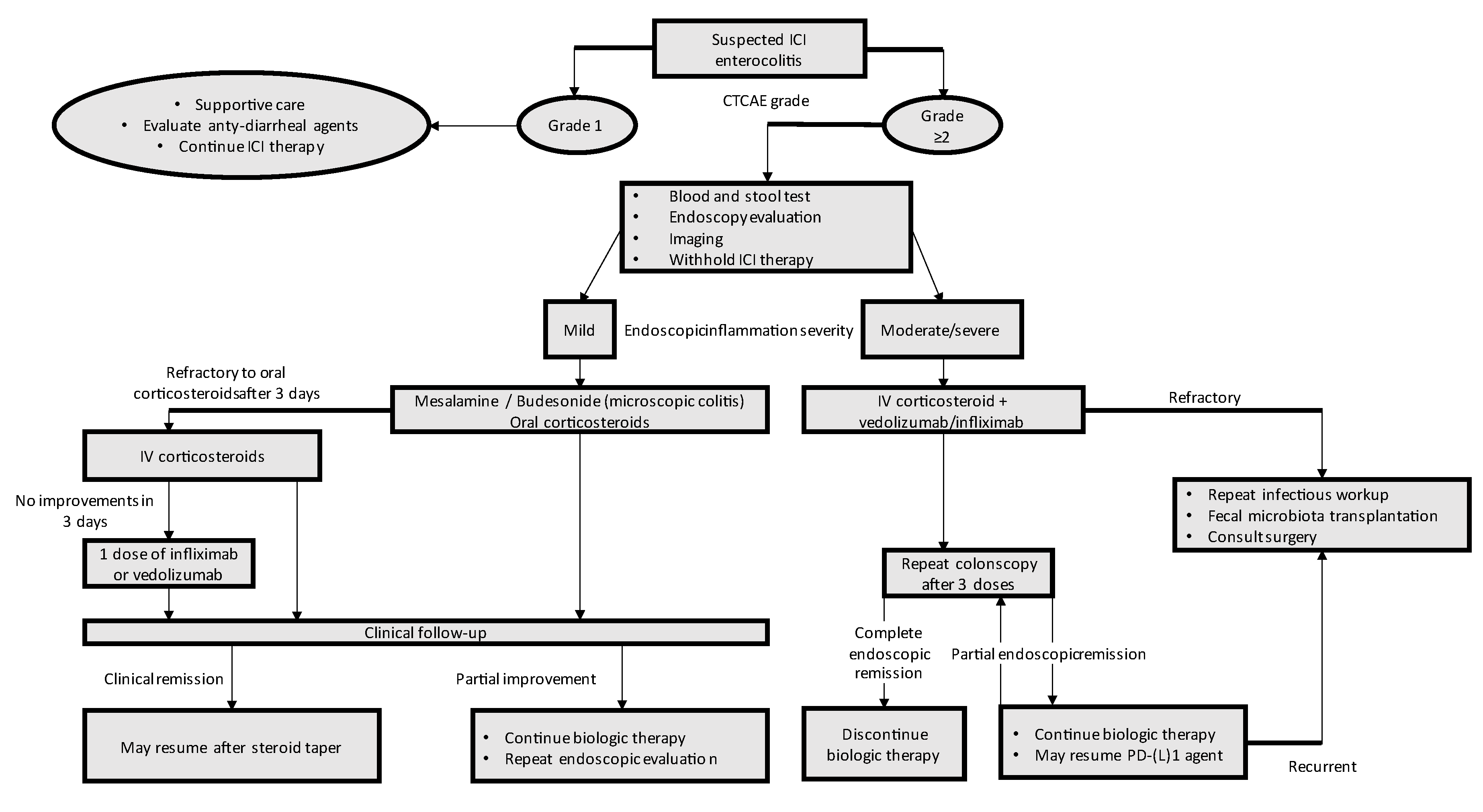
4. Therapy and Management
4.1. First-Line Management
4.2. Refractory Colitis
4.3. Resuming ICI Treatment
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Olszanski, A.J. Principles of immunotherapy. J. Natl. Compr. Canc. Netw. 2015, 13 (Suppl. S5), 670–672. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, Z. The history and advances in cancer immunotherapy: Understanding the characteristics of tumor-infiltrating immune cells and their therapeutic implications. Cell. Mol. Immunol. 2020, 17, 807–821. [Google Scholar] [CrossRef]
- Onitilo, A.A.; Wittig, J.A. Principles of Immunotherapy in Melanoma. Surg. Clin. N. Am. 2020, 100, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Bagchi, S.; Yuan, R.; Engleman, E.G. Immune Checkpoint Inhibitors for the Treatment of Cancer: Clinical Impact and Mechanisms of Response and Resistance. Annu. Rev. Pathol. 2021, 16, 223–249. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Ciuleanu, T.-E.; Cobo, M.; Schenker, M.; Zurawski, B.; Menezes, J.; Richardet, E.; Bennouna, J.; Felip, E.; Juan-Vidal, O.; et al. First-line nivolumab plus ipilimumab combined with two cycles of chemotherapy in patients with non-small-cell lung cancer (CheckMate 9LA): An international, randomised, open-label, phase 3 trial. Lancet Oncol. 2021, 22, 198–211, Correction in Lancet Oncol. 2021, 22, e92. [Google Scholar] [CrossRef]
- Albiges, L.; Tannir, N.M.; Burotto, M.; McDermott, D.; Plimack, E.R.; Barthélémy, P.; Porta, C.; Powles, T.; Donskov, F.; George, S.; et al. Nivolumab plus ipilimumab versus sunitinib for first-line treatment of advanced renal cell carcinoma: Extended 4-year follow-up of the phase III CheckMate 214 trial. ESMO Open 2020, 5, e001079. [Google Scholar] [CrossRef] [PubMed]
- Hodi, F.S.; Chiarion-Sileni, V.; Gonzalez, R.; Grob, J.-J.; Rutkowski, P.; Cowey, C.L.; Lao, C.D.; Schadendorf, D.; Wagstaff, J.; Dummer, R.; et al. Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (CheckMate 067): 4-year outcomes of a multicentre, randomised, phase 3 trial. Lancet Oncol. 2018, 19, 1480–1492, Correction in Lancet Oncol. 2018, 19, e668; Correction in Lancet Oncol. 2018, 19, e581.. [Google Scholar] [CrossRef] [PubMed]
- Abou-Alfa, G.K.; Chan, S.L.; Kudo, M.; Lau, G.; Kelley, R.K.; Furuse, J.; Sukeepaisarnjaroen, W.; Kang, Y.-K.; Dao, T.V.; De Toni, E.N.; et al. Phase 3 randomized, open-label, multicenter study of tremelimumab (T) and durvalumab (D) as first-line therapy in patients (pts) with unresectable hepatocellular carcinoma (uHCC): HIMALAYA. J. Clin. Oncol. 2022, 40, 379. [Google Scholar] [CrossRef]
- Qin, S.; Xu, L.; Yi, M.; Yu, S.; Wu, K.; Luo, S. Novel immune checkpoint targets: Moving beyond PD-1 and CTLA-4. Mol. Cancer 2019, 18, 155. [Google Scholar] [CrossRef]
- Conroy, M.; Naidoo, J. Immune-Related Adverse Events and the Balancing Act of Immunotherapy. Nat. Commun. 2022, 13, 392. [Google Scholar] [CrossRef]
- Raschi, E.; Gatti, M.; Gelsomino, F.; Ardizzoni, A.; Poluzzi, E.; De Ponti, F. Lessons to Be Learnt from Real-World Studies on Immune-Related Adverse Events with Checkpoint Inhibitors: A Clinical Perspective from Pharmacovigilance. Target Oncol. 2020, 15, 449–466. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.-F.; Chen, Y.; Song, S.-Y.; Wang, T.-J.; Ji, W.-J.; Li, S.-W.; Liu, N.; Yan, C.-X. Immune-Related Adverse Events Associated with Anti-PD-1/PD-L1 Treatment for Malignancies: A Meta-Analysis. Front. Pharmacol. 2017, 8, 730. [Google Scholar] [CrossRef]
- Schneider, B.J.; Naidoo, J.; Santomasso, B.D.; Lacchetti, C.; Adkins, S.; Anadkat, M.; Atkins, M.B.; Brassil, K.J.; Caterino, J.M.; Chau, I.; et al. Management of Immune-Related Adverse Events in Patients Treated with Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J. Clin. Oncol. 2021, 39, 4073–4126. [Google Scholar] [CrossRef] [PubMed]
- Sasson, S.C.; Slevin, S.M.; Cheung, V.T.F.; Nassiri, I.; Olsson-Brown, A.; Fryer, E.; Ferreira, R.C.; Trzupek, D.; Gupta, T.; Al-Hillawi, L.; et al. Interferon-Gamma-Producing CD8+ Tissue Resident Memory T Cells Are a Targetable Hallmark of Immune Checkpoint Inhibitor-Colitis. Gastroenterology 2021, 161, 1229–1244.e9. [Google Scholar] [CrossRef]
- Luoma, A.M.; Suo, S.; Williams, H.L.; Sharova, T.; Sullivan, K.; Manos, M.; Bowling, P.; Hodi, F.S.; Rahma, O.; Sullivan, R.J.; et al. Molecular Pathways of Colon Inflammation Induced by Cancer Immunotherapy. Cell 2020, 182, 655–671.e22. [Google Scholar] [CrossRef] [PubMed]
- Westdorp, H.; Sweep, M.W.D.; Gorris, M.A.J.; Hoentjen, F.; Boers-Sonderen, M.J.; Post, R.S.V.D.; Heuvel, M.M.V.D.; Piet, B.; Boleij, A.; Bloemendal, H.J.; et al. Mechanisms of Immune Checkpoint Inhibitor-Mediated Colitis. Front. Immunol. 2021, 12, 768957. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, T.; De Velasco, M.A.; Sakai, K.; Nagai, T.; Nishiyama, H.; Hashimoto, K.; Uemura, H.; Kawakami, H.; Nakagawa, K.; Ogata, H.; et al. Integrative Analysis of Gut Microbiome and Host Transcriptomes Reveals Associations between Treatment Outcomes and Immunotherapy-Induced Colitis. Mol. Oncol. 2022, 16, 1493–1507. [Google Scholar] [CrossRef]
- Dubin, K.; Callahan, M.K.; Ren, B.; Khanin, R.; Viale, A.; Ling, L.; No, D.; Gobourne, A.; Littmann, E.; Huttenhower, C.; et al. Intestinal Microbiome Analyses Identify Melanoma Patients at Risk for Checkpoint-Blockade-Induced Colitis. Nat. Commun. 2016, 7, 10391. [Google Scholar] [CrossRef]
- Gupta, A.; De Felice, K.M.; Loftus, E.V.; Khanna, S. Systematic Review: Colitis Associated with Anti-CTLA-4 Therapy. Aliment. Pharmacol. Ther. 2015, 42, 406–417. [Google Scholar] [CrossRef]
- Haanen, J.; Obeid, M.; Spain, L.; Carbonnel, F.; Wang, Y.; Robert, C.; Lyon, A.R.; Wick, W.; Kostine, M.; Peters, S.; et al. Management of Toxicities from Immunotherapy: ESMO Clinical Practice Guideline for Diagnosis, Treatment and Follow-Up. Ann. Oncol. 2022, 33, 1217–1238. [Google Scholar] [CrossRef]
- Tran, T.; Tran, N.G.T.; Ho, V. Checkpoint Inhibitors and the Gut. J. Clin. Med. 2022, 11, 824. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, D.L.; Juhl, C.B.; Chen, I.M.; Kellermann, L.; Nielsen, O.H. Immune checkpoint inhibitor induced diarrhea and colitis: Incidence and management. A systematic review and metanalysis. Cancer Treat. Rev. 2022, 109, 102440. [Google Scholar] [CrossRef] [PubMed]
- OPDIVO. INN-Nivolumab-European Medicines Agency. 2021. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/opdivo (accessed on 20 May 2023).
- Yau, T.; Kang, Y.; Kim, T.; El-Khoueiry, A.B.; Santoro, A.; Sangro, B.; Melero, I.; Kudo, M.; Hou, M.-M.; Matilla, A.; et al. Efficacy and Safety of Nivolumab Plus Ipilimumab in Patients with Advanced Hepatocellular Carcinoma Previously Treated with Sorafenib: The CheckMate 040 Randomized Clinical Trial. JAMA Oncol. 2020, 6, e204564. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.R.; Eyada, M.; Kono, M.; Varatharajalu, K.; Lu, Y.; Xu, G.; Panneerselvam, K.; Shatila, M.; Altan, M.; Wang, J.; et al. Characteristics, treatment, and outcome of diverticulitis after immune checkpoint inhibitor treatment in patients with malignancies. J. Cancer Res. Clin. Oncol. 2022; Online ahead of print. [Google Scholar]
- Yamada, T.; Alpers, D.H.; Kalloo, A.N.; Kaplowitz, N.; Owyang, C.; Powell, D.W. Yamada Textbook of Gastroenterology, 7th ed.; John Wiley & Sons: New York, NY, USA, 2022. [Google Scholar]
- Haanen, J.; Ernstoff, M.S.; Wang, Y.; Menzies, A.M.; Puzanov, I.; Grivas, P.; Larkin, J.; Peters, S.; Thompson, J.A.; Obeid, M. Autoimmune diseases and immune-checkpoint inhibitors for cancer therapy: Review of the literature and personalized risk-based prevention strategy. Ann. Oncol. 2020, 31, 724–744. [Google Scholar] [CrossRef]
- Steiger, S.; Marcucci, V.; Desai, V.; Zheng, M.; Parker, G. Refractory Fulminant Colitis Requiring Surgical Intervention in a Patient with Ulcerative Colitis on Atezolizumab Therapy for Small Cell Lung Cancer: An Atypical Case. Cureus 2022, 14, e25437. [Google Scholar] [CrossRef] [PubMed]
- Rubin, S.J.S.; Balabanis, T.; Gubatan, J.; Habtezion, A. Disease exacerbation is common in inflammatory bowel disease patients treated with immune checkpoint inhibitors for malignancy. World J. Clin. Cases 2022, 10, 1787–1794. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Elias, R.; Peng, L.; Levonyak, N.; Asokan, A.; Christie, A.; Kubiliun, N.; Brugarolas, J.; Hammers, H.J. Chronic Use of Proton Pump Inhibitors Is Associated with an Increased Risk of Immune Checkpoint Inhibitor Colitis in Renal Cell Carcinoma. Clin. Genitourin. Cancer 2022, 20, 260–269. [Google Scholar] [CrossRef] [PubMed]
- US Department of Health and Human Services. Common Terminology Criteria for Adverse Events; Version 5; National Institutes of Health, National Cancer Institute: Bethesda, MD, USA, 2017.
- Abu-Sbeih, H.; Ali, F.S.; Luo, W.; Qiao, W.; Raju, G.S.; Wang, Y. Importance of endoscopic and histological evaluation in the management of immune checkpoint inhibitor-induced colitis. J. Immunother. Cancer 2018, 6, 95. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.S.; Molina, G.E.; Chen, S.T.; Zheng, H.; Deshpande, V.; Fadden, R.; Sullivan, R.J.; Dougan, M. Budesonide treatment for microscopic colitis from immune checkpoint inhibitors. J. Immunother. Cancer 2019, 7, 292. [Google Scholar] [CrossRef]
- Wang, Y.; Abu-Sbeih, H.; Mao, E.; Ali, N.; Qiao, W.; Trinh, V.A.; Zobniw, C.; Johnson, D.H.; Samdani, R.; Lum, P.; et al. Endoscopic and histologic features of immune checkpoint inhibitor related colitis. Inflamm. Bowel Dis. 2018, 24, 1695–1705. [Google Scholar] [CrossRef]
- Mooradian, M.J.; Wang, D.Y.; Coromilas, A.; Lumish, M.; Chen, T.; Giobbie-Hurder, A.; Johnson, D.B.; Sullivan, R.J.; Dougan, M. Mucosal inflammation predicts response to systemic steroids in immune checkpoint inhibitor colitis. J. Immunother. Cancer 2020, 8, e000451. [Google Scholar] [CrossRef] [PubMed]
- Marthey, L.; Mateus, C.; Mussini, C.; Nachury, M.; Nancey, S.; Grange, F.; Zallot, C.; Peyrin-Biroulet, L.; Rahier, J.F.; DE Beauregard, M.B.; et al. Cancer Immunotherapy with anti-CTLA-4 monoclonal antibodies induces an inflammatory bowel disease. J. Crohn’s Colitis 2016, 10, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Marchal-Bressenot, A.; Salleron, J.; Boulagnon-Rombi, C.; Bastien, C.; Cahn, V.; Cadiot, G.; Diebold, M.-D.; Danese, S.; Reinisch, W.; Schreiber, S.; et al. Development and validation of the Nancy histological index for UC. Gut 2017, 66, 43–49. [Google Scholar] [CrossRef]
- Mosli, M.; Feagan, B.G.; Zou, G.; Sandborn, W.J.; Dhaens, G.R.; Khanna, R.; Shackelton, L.M.; Walker, C.W.; Nelson, S.; Vandervoort, M.K.; et al. Development and validation of a histological index for UC. Gut 2017, 66, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Pai, R.K.; Schaeffer, D.F.; Krell, J.; Guizzetti, L.; McFarlane, S.C.; MacDonald, J.K.; Choi, W.-T.; Feakins, R.M.; Kirsch, R.; et al. Recommendations for standardizing biopsy acquisition and histological assessment of immune checkpoint inhibitor associated colitis. J. Immunother. Cancer 2022, 10, e004560. [Google Scholar] [CrossRef]
- Gonzalez, R.S.; Salaria, S.N.; Bohannon, C.D.; Huber, A.R.; Feely, M.M.; Shi, C. PD-1 inhibitor gastroenterocolitis: Case series and appraisal of ‘immunomodulatory gastroenterocolitis’. Histopathology 2017, 70, 558–567. [Google Scholar] [CrossRef] [PubMed]
- Isidro, R.A.; Ruan, A.B.; Gannarapu, S.; Raj, D.; Rahma, O.; Grover, S.; Srivastava, A. Medication-specific variations in morphological patterns of injury in immune check-point inhibitor-associated colitis. Histopathology 2021, 78, 532–541. [Google Scholar] [CrossRef]
- Patil, P.A.; Zhang, X. Pathologic manifestations of gastrointestinal and hepatobiliary injury in immune checkpoint inhibitor therapy. Arch. Pathol. Lab. Med. 2021, 145, 571–582. [Google Scholar] [CrossRef]
- Chen, J.H.; Pezhouh, M.K.; Lauwers, G.Y.; Masia, R. Histopathologic features of colitis due to immunotherapy with anti-PD-1 antibodies. Am. J. Surg. Pathol. 2017, 41, 643–654. [Google Scholar] [CrossRef]
- Adler, B.L.; Pezhouh, M.K.; Kim, A.; Luan, L.; Zhu, Q.; Gani, F.; Yarchoan, M.; Chen, J.; Voltaggio, L.; Parian, A.; et al. Histopathological and immunophenotypic features of ipilimumab-associated colitis compared to ulcerative colitis. J. Intern. Med. 2018, 283, 568–577. [Google Scholar] [CrossRef]
- Dougan, M.; Wang, Y.; Rubio-Tapia, A.; Lim, J.K. AGA clinical practice update on diagnosis and management of immune checkpoint inhibitor colitis and hepatitis: Expert review. Gastroenterology 2021, 160, 1384–1393. [Google Scholar] [CrossRef] [PubMed]
- Brahmer, J.R.; Lacchetti, C.; Schneider, B.J.; Atkins, M.B.; Brassil, K.J.; Caterino, J.M.; Chau, I.; Ernstoff, M.S.; Gardner, J.M.; Ginex, P.; et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American society of clinical oncology clinical practice guideline. J. Clin. Oncol. J. Am. Soc. Clin. Oncol. 2018, 36, 1714–1768. [Google Scholar] [CrossRef] [PubMed]
- Dougan, M. Checkpoint Blockade Toxicity and Immune Homeostasis in the Gastrointestinal Tract. Front. Immunol. 2017, 8, 1547. [Google Scholar] [CrossRef]
- Saha, A.; Dreyfuss, I.; Sarfraz, H.; Friedman, M.; Markowitz, J. Dietary Considerations for Inflammatory Bowel Disease Are Useful for Treatment of Checkpoint Inhibitor-Induced Colitis. Cancers 2023, 15, 84. [Google Scholar] [CrossRef] [PubMed]
- Amiot, A.; Laharie, D.; Malamut, G.; Serrero, M.; Poullenot, F.; Educational Committee of the GETAID. Management of immune checkpoint inhibitor in patients with cancer and pre-existing inflammatory bowel disease: Recommendations from the GETAID. Dig. Liver Dis. 2022, 54, 1162–1167. [Google Scholar] [CrossRef]
- Johnson, D.H.; Zobniw, C.M.; Trinh, V.A.; Ma, J.; Bassett, R.L., Jr.; Abdel-Wahab, N.A.; Anderson, J.; Davis, J.E.; Joseph, J.; Uemura, M.; et al. Infliximab associated with faster symptom resolution compared with corticosteroids alone for the management of immune-related enterocolitis. J. Immunother. Cancer 2018, 6, 103. [Google Scholar] [CrossRef] [PubMed]
- Abu-Sbeih, H.; Ali, F.S.; Alsaadi, D.; Jennings, J.; Luo, W.; Gong, Z.; Richards, D.M.; Charabaty, A.; Wang, Y. Outcomes of vedolizumab therapy in patients with immune checkpoint inhibitor-induced colitis: A multi-center study. J. Immunother. Cancer 2018, 6, 142. [Google Scholar] [CrossRef] [PubMed]
- Zou, F.; Faleck, D.; Thomas, A.; Harris, J.; Satish, D.; Wang, X.; Charabaty, A.; Ernstoff, M.S.; Glitza Oliva, I.C.; Hanauer, S.; et al. Efficacy and safety of vedolizumab and infiximab treatment for immune-mediated diarrhea and colitis in patients with cancer: A two-center observational study. J. Immunother. Cancer 2021, 9, e003277. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wiesnoski, D.H.; Helmink, B.A.; Gopalakrishnan, V.; Choi, K.; DuPont, H.L.; Jiang, Z.-D.; Abu-Sbeih, H.; Sanchez, C.A.; Chang, C.-C.; et al. Fecal microbiota transplantation for refractory immune checkpoint inhibitor associated colitis. Nat. Med. 2018, 24, 1804–1808. [Google Scholar] [CrossRef]
- Perez Del Nogal, G.; Patel, N. Refractory Checkpoint Inhibitor Colitis Responsive to Ustekinumab. ACG Case Rep. J. 2022, 9, e00946. [Google Scholar] [CrossRef]
- Thomas, A.S.; Ma, W.; Wang, Y. Ustekinumab for Refractory Colitis Associated with Immune Checkpoint Inhibitors. N. Engl. J. Med. 2021, 384, 581–583. [Google Scholar] [CrossRef]
- Esfahani, K.; Hudson, M.; Batist, G. Tofacitinib for Refractory Immune-Related Colitis from PD-1 Therapy. N. Engl. J. Med. 2020, 382, 2374–2375. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Liu, M.; Li, C.; Peng, S.; Li, Y.; Xu, X.; Sun, M.; Sun, X. Fecal Microbiota Transplantation Effectively Cures a Patient with Severe Bleeding Immune Checkpoint Inhibitor-Associated Colitis and a Short Review. Front. Oncol. 2022, 12, 913217. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Yin, Q.; Chen, L.; Davis, M.M. Bifidobacterium Can Mitigate Intestinal Immunopathology in the Context of CTLA-4 Blockade. Proc. Natl. Acad. Sci. USA 2018, 115, 157–161. [Google Scholar] [CrossRef]
- Wang, Y.; Wiesnoski, D.H.; Helmink, B.A.; Gopalakrishnan, V.; Choi, K.; DuPont, H.L.; Jiang, Z.-D.; Abu-Sbeih, H.; Sanchez, C.A.; Chang, C.-C.; et al. Author Correction: Fecal Microbiota Transplantation for Refractory Immune Checkpoint Inhibitor-Associated Colitis. Nat. Med. 2019, 25, 188. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.; Thompson, J.A.; Hamid, O.; Minor, D.; Amin, A.; Ron, I.; Ridolfi, R.; Assi, H.; Maraveyas, A.; Berman, D.; et al. A randomized, double-blind, placebo-controlled, phase II study comparing the tolerability and efficacy of ipilimumab administered with or without prophylactic budesonide in patients with unresectable stage III or IV melanoma. Clin. Cancer Res. 2009, 15, 5591–5598. [Google Scholar] [CrossRef]
- Falade, A.S.; Reynolds, K.L.; Zubiri, L.; Deshpande, V.; Fintelmann, F.J.; Dougan, M.; Mooradian, M.J. Case report: Fulminant celiac disease with combination immune checkpoint therapy. Front. Immunol. 2022, 13, 871452. [Google Scholar] [CrossRef]
- Perdigoto, A.L.; Tran, T.; Patel, N.; Clark, P.; Patell, K.; Stamatouli, A.M.; Reddy, V.; Clune, J.; Herold, K.C.; Robert, M.E.; et al. Elective Colectomy in a Patient with Active Ulcerative Colitis and Metastatic Melanoma Enabling Successful Treatment with Immune Checkpoint Inhibitors. Clin. Oncol. Case Rep. 2020, 3, 142. [Google Scholar]
- Marron, T.U.; Ryan, A.E.; Reddy, S.M.; Kaczanowska, S.; Younis, R.H.; Thakkar, D.; Zhang, J.; Bartkowiak, T.; Howard, R.; Anderson, K.G.; et al. Considerations for Treatment Duration in Responders to Immune Checkpoint Inhibitors. J. Immunother. Cancer 2021, 9, e001901. [Google Scholar] [CrossRef]
- Albandar, H.J.; Fuqua, J.; Albandar, J.M.; Safi, S.; Merrill, S.A.; Ma, P.C. Immune-Related Adverse Events (IrAE) in Cancer Immune Checkpoint Inhibitors (ICI) and Survival Outcomes Correlation: To Rechallenge or Not? Cancers 2021, 13, 989. [Google Scholar] [CrossRef]
- Abu-Sbeih, H.; Ali, F.S.; Naqash, A.R.; Owen, D.H.; Patel, S.; Otterson, G.A.; Kendra, K.; Ricciuti, B.; Chiari, R.; De Giglio, A.; et al. Resumption of Immune Checkpoint Inhibitor Therapy After Immune-Mediated Colitis. J. Clin. Oncol. 2019, 37, 2738–2745. [Google Scholar] [CrossRef] [PubMed]
- Badran, Y.R.; Cohen, J.V.; Brastianos, P.K.; Parikh, A.R.; Hong, T.S.; Dougan, M. Concurrent Therapy with Immune Checkpoint Inhibitors and TNFα Blockade in Patients with Gastrointestinal Immune-Related Adverse Events. J. Immunother. Cancer 2019, 7, 226. [Google Scholar] [CrossRef] [PubMed]
- Jing, Y.; Liu, J.; Ye, Y.; Pan, L.; Deng, H.; Wang, Y.; Yang, Y.; Diao, L.; Lin, S.H.; Mills, G.B.; et al. Multi-Omics Prediction of Immune-Related Adverse Events during Checkpoint Immunotherapy. Nat. Commun. 2020, 11, 4946. [Google Scholar] [CrossRef] [PubMed]
- Valpione, S.; Pasquali, S.; Campana, L.G.; Piccin, L.; Mocellin, S.; Pigozzo, J.; Chiarion-Sileni, V. Sex and Interleukin-6 Are Prognostic Factors for Autoimmune Toxicity Following Treatment with Anti-CTLA4 Blockade. J. Transl. Med. 2018, 16, 94. [Google Scholar] [CrossRef]
- Zhang, H.; Ye, L.; Yu, X.; Jin, K.; Wu, W. Neoadjuvant therapy alters the immune microenvironment in pancreatic cancer. Front. Immunol. 2022, 13, 956984. [Google Scholar] [CrossRef] [PubMed]
- Bell, P.D.; Pai, R.K. Immune response in colorectal carcinoma: A review of its significance as a predictive and prognostic biomarker. Histopathology 2022, 81, 696–714. [Google Scholar] [CrossRef] [PubMed]
| Variable | Grade 1 | Grade 2 | Grade 3 | Grade 4 | Grade 5 |
|---|---|---|---|---|---|
| Diarrhea | Increase in <4 stools/d over baseline; mild increase in ostomy output compared with baseline. | Increase in 4–6 stools/d over baseline; moderate increase in ostomy output compared with baseline; limiting instrumental ADL. | Increase in ≥7 stools/d over baseline; hospitalization indicated; severe increase in ostomy output compared with baseline; limiting self-care ADL. | Life-threatening consequences; urgent intervention indicated. | Death. |
| Enterocolitis | Asymptomatic; clinical or diagnostic observation only; intervention not indicated. | Asymptomatic; clinical or diagnostic observation only; intervention not indicated. | Severe or persistent abdominal pain; fever; ileus; peritoneal signs. | Life-threatening consequences; urgent intervention indicated. | Death. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Losurdo, G.; Angelillo, D.; Favia, N.; Sergi, M.C.; Di Leo, A.; Triggiano, G.; Tucci, M. Checkpoint Inhibitor-Induced Colitis: An Update. Biomedicines 2023, 11, 1496. https://doi.org/10.3390/biomedicines11051496
Losurdo G, Angelillo D, Favia N, Sergi MC, Di Leo A, Triggiano G, Tucci M. Checkpoint Inhibitor-Induced Colitis: An Update. Biomedicines. 2023; 11(5):1496. https://doi.org/10.3390/biomedicines11051496
Chicago/Turabian StyleLosurdo, Giuseppe, Daniele Angelillo, Nicolas Favia, Maria Chiara Sergi, Alfredo Di Leo, Giacomo Triggiano, and Marco Tucci. 2023. "Checkpoint Inhibitor-Induced Colitis: An Update" Biomedicines 11, no. 5: 1496. https://doi.org/10.3390/biomedicines11051496
APA StyleLosurdo, G., Angelillo, D., Favia, N., Sergi, M. C., Di Leo, A., Triggiano, G., & Tucci, M. (2023). Checkpoint Inhibitor-Induced Colitis: An Update. Biomedicines, 11(5), 1496. https://doi.org/10.3390/biomedicines11051496








