Timing Is Important—Management of Metabolic Syndrome According to the Circadian Rhythm
Abstract
1. Introduction
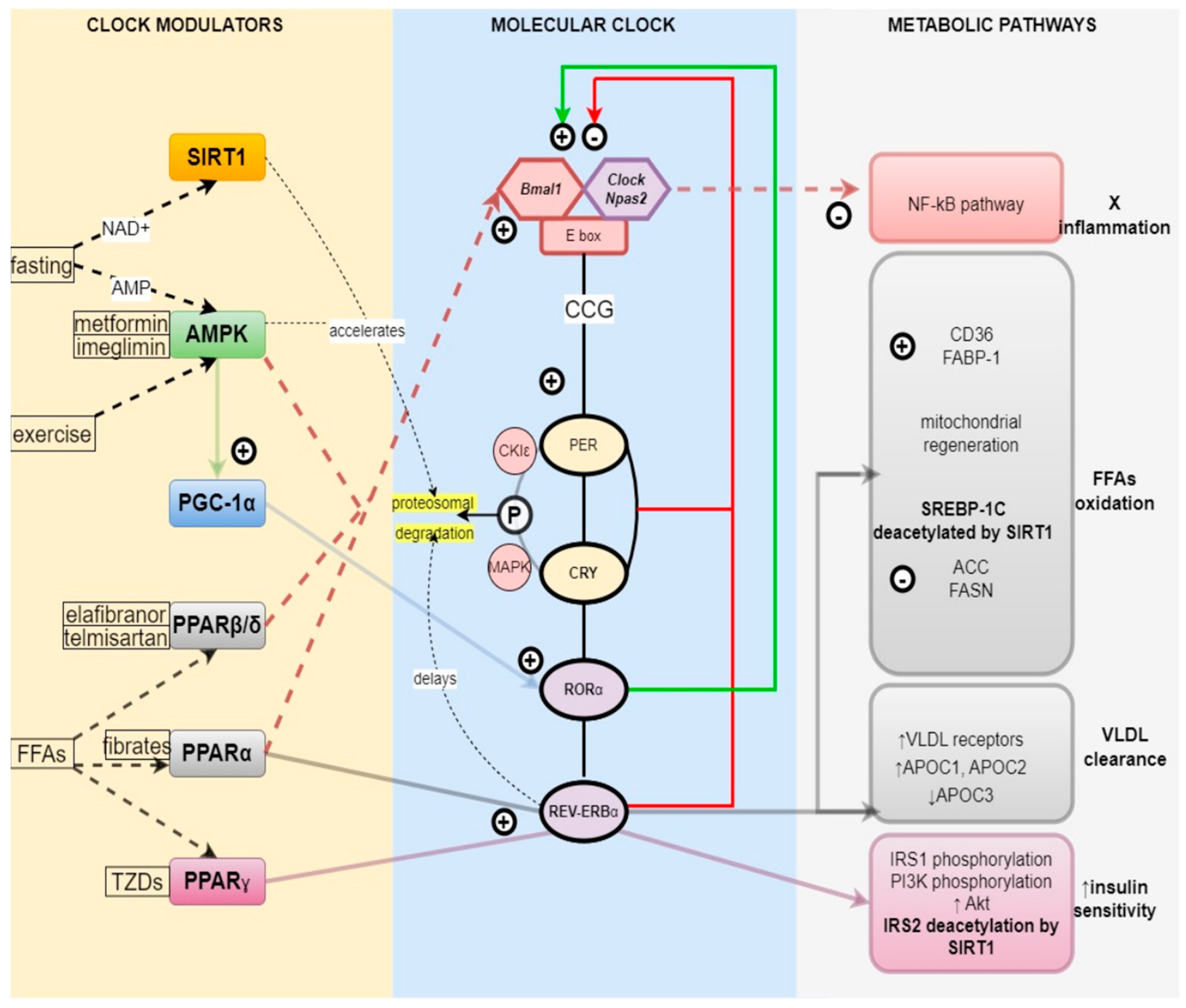
2. The Link between Circadian Clock and Metabolism
2.1. Core Clock Genes
2.2. Reverse-Strand Avian Erythroblastic Leukemia (ERBA) Oncogene Receptors (REV-ERBs) and Retinoic Acid-Related Orphan Receptors (RORs)
2.3. Regulation of the Metabolism by the Circadian Clock
2.3.1. Nutrient Sensors and the Clock—PPARα, β, γ
PPARα
PPARγ
PPARδ/β
2.3.2. Energy Sensors and the Clock—AMPK, SIRT1, PGC1α
SIRT1
AMPK
PGC-1α
2.3.3. Indirect Circadian Control of Metabolism by Hormone Secretion
Insulin
GLP-1
RAAS
3. Circadian Rhythm Disruption
4. Treatment of MetS in Alignment with Circadian Rhythm
4.1. Feeding According to the Biological Clock as a Circadian Rhythm Realignment Strategy
4.2. Exercise around the Clock
4.3. Circadian Medication
4.3.1. Antihypertensives
4.3.2. Antihyperglycemic Medications
Metformin
Imeglimin
PPARγ Agonists
GLP-1 RAs
DPP-4is
SGLT2 Inhibitors
Insulin
4.3.3. Hypolipidemic Agents
Statins
PPARα Agonists
PPARβ/δ Agonists
5. Conclusions
6. Literature Review
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fahed, G.; Aoun, L.; Bou Zerdan, M.; Allam, S.; Bou Zerdan, M.; Bouferraa, Y.; Assi, H.I. Metabolic Syndrome: Updates on Pathophysiology and Management in 2021. Int. J. Mol. Sci. 2022, 23, 786. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.M.M.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.-C.; James, W.P.T.; Loria, C.M.; Smith, S.C.; et al. Harmonizing the Metabolic Syndrome: A Joint Interim Statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar] [CrossRef] [PubMed]
- Engin, A. The Definition and Prevalence of Obesity and Metabolic Syndrome. Adv. Exp. Med. Biol. 2017, 960, 1–17. [Google Scholar] [CrossRef]
- Muzurović, E.; Mikhailidis, D.P.; Mantzoros, C. Non-Alcoholic Fatty Liver Disease, Insulin Resistance, Metabolic Syndrome and Their Association with Vascular Risk. Metabolism 2021, 119, 154770. [Google Scholar] [CrossRef]
- Muzurović, E.; Peng, C.C.-H.; Belanger, M.J.; Sanoudou, D.; Mikhailidis, D.P.; Mantzoros, C.S. Nonalcoholic Fatty Liver Disease and Cardiovascular Disease: A Review of Shared Cardiometabolic Risk Factors. Hypertension 2022, 79, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, L.J.; Hall, J.C.; Rosbash, M. Expression of a Drosophila MRNA Is under Circadian Clock Control during Pupation. Development 1989, 107, 869–880. [Google Scholar] [CrossRef]
- Bargiello, T.A.; Jackson, F.R.; Young, M.W. Restoration of Circadian Behavioural Rhythms by Gene Transfer in Drosophila. Nature 1984, 312, 752–754. [Google Scholar] [CrossRef]
- Froy, O. Metabolism and Circadian Rhythms—Implications for Obesity. Endocr. Rev. 2010, 31, 1–24. [Google Scholar] [CrossRef]
- Acosta-Rodríguez, V.A.; Rijo-Ferreira, F.; Green, C.B.; Takahashi, J.S. Importance of Circadian Timing for Aging and Longevity. Nat. Commun. 2021, 12, 2862. [Google Scholar] [CrossRef]
- Reddy, S.; Reddy, V.; Sharma, S. Physiology, Circadian Rhythm. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Cao, R. Molecular Biology and Physiology of Circadian Clocks. In Oxford Research Encyclopedia of Neuroscience; Oxford University Press: Oxford, UK, 2019; ISBN 978-0-19-026408-6. [Google Scholar]
- Gallego, M.; Kang, H.; Virshup, D.M. Protein Phosphatase 1 Regulates the Stability of the Circadian Protein PER2. Biochem. J. 2006, 399, 169–175. [Google Scholar] [CrossRef]
- Solt, L.A.; Kojetin, D.J.; Burris, T.P. The REV-ERBs and RORs: Molecular Links between Circadian Rhythms and Lipid Homeostasis. Future Med. Chem. 2011, 3, 623–638. [Google Scholar] [CrossRef] [PubMed]
- Gnocchi, D.; Pedrelli, M.; Hurt-Camejo, E.; Parini, P. Lipids around the Clock: Focus on Circadian Rhythms and Lipid Metabolism. Biology 2015, 4, 104–132. [Google Scholar] [CrossRef] [PubMed]
- Adlanmerini, M.; Krusen, B.M.; Nguyen, H.C.B.; Teng, C.W.; Woodie, L.N.; Tackenberg, M.C.; Geisler, C.E.; Gaisinsky, J.; Peed, L.C.; Carpenter, B.J.; et al. REV-ERB Nuclear Receptors in the Suprachiasmatic Nucleus Control Circadian Period and Restrict Diet-Induced Obesity. Sci. Adv. 2021, 7, eabh2007. [Google Scholar] [CrossRef] [PubMed]
- Kojetin, D.J.; Burris, T.P. REV-ERB and ROR Nuclear Receptors as Drug Targets. Nat. Rev. Drug. Discov. 2014, 13, 197–216. [Google Scholar] [CrossRef]
- Duez, H.; Staels, B. Rev-Erb-Alpha: An Integrator of Circadian Rhythms and Metabolism. J. Appl. Physiol. 2009, 107, 1972–1980. [Google Scholar] [CrossRef]
- Yin, L.; Wu, N.; Curtin, J.C.; Qatanani, M.; Szwergold, N.R.; Reid, R.A.; Waitt, G.M.; Parks, D.J.; Pearce, K.H.; Wisely, G.B.; et al. Rev-Erbalpha, a Heme Sensor That Coordinates Metabolic and Circadian Pathways. Science 2007, 318, 1786–1789. [Google Scholar] [CrossRef]
- Raspé, E.; Duez, H.; Gervois, P.; Fiévet, C.; Fruchart, J.C.; Besnard, S.; Mariani, J.; Tedgui, A.; Staels, B. Transcriptional Regulation of Apolipoprotein C-III Gene Expression by the Orphan Nuclear Receptor RORalpha. J. Biol. Chem. 2001, 276, 2865–2871. [Google Scholar] [CrossRef]
- Chen, L.; Yang, G. PPARs Integrate the Mammalian Clock and Energy Metabolism. PPAR Res. 2014, 2014, 653017. [Google Scholar] [CrossRef]
- Canaple, L.; Rambaud, J.; Dkhissi-Benyahya, O.; Rayet, B.; Tan, N.S.; Michalik, L.; Delaunay, F.; Wahli, W.; Laudet, V. Reciprocal Regulation of Brain and Muscle Arnt-Like Protein 1 and Peroxisome Proliferator-Activated Receptor α Defines a Novel Positive Feedback Loop in the Rodent Liver Circadian Clock. Mol. Endocrinol. 2006, 20, 1715–1727. [Google Scholar] [CrossRef]
- Rakhshandehroo, M.; Knoch, B.; Müller, M.; Kersten, S. Peroxisome Proliferator-Activated Receptor Alpha Target Genes. PPAR Res. 2010, 2010, 612089. [Google Scholar] [CrossRef]
- Duez, H.; Staels, B. Rev-Erb Alpha Gives a Time Cue to Metabolism. FEBS Lett. 2008, 582, 19–25. [Google Scholar] [CrossRef]
- Todisco, S.; Santarsiero, A.; Convertini, P.; De Stefano, G.; Gilio, M.; Iacobazzi, V.; Infantino, V. PPAR Alpha as a Metabolic Modulator of the Liver: Role in the Pathogenesis of Nonalcoholic Steatohepatitis (NASH). Biology 2022, 11, 792. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, S.; Gupta, P.; Saini, A.S.; Kaushal, C.; Sharma, S. The Peroxisome Proliferator-Activated Receptor: A Family of Nuclear Receptors Role in Various Diseases. J. Adv. Pharm. Technol. Res. 2011, 2, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Lange, N.F.; Graf, V.; Caussy, C.; Dufour, J.-F. PPAR-Targeted Therapies in the Treatment of Non-Alcoholic Fatty Liver Disease in Diabetic Patients. Int. J. Mol. Sci. 2022, 23, 4305. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Yu, R.; Xiong, Y.; Du, F.; Zhu, S. A Vicious Circle between Insulin Resistance and Inflammation in Nonalcoholic Fatty Liver Disease. Lipids Health Dis. 2017, 16, 203. [Google Scholar] [CrossRef] [PubMed]
- Botta, M.; Audano, M.; Sahebkar, A.; Sirtori, C.R.; Mitro, N.; Ruscica, M. PPAR Agonists and Metabolic Syndrome: An Established Role? Int. J. Mol. Sci. 2018, 19, 1197. [Google Scholar] [CrossRef] [PubMed]
- Lehrke, M.; Lazar, M.A. The Many Faces of PPARγ. Cell 2005, 123, 993–999. [Google Scholar] [CrossRef]
- Lee, Y.K.; Park, J.E.; Lee, M.; Hardwick, J.P. Hepatic Lipid Homeostasis by Peroxisome Proliferator-Activated Receptor Gamma 2. Liver Res. 2018, 2, 209–215. [Google Scholar] [CrossRef]
- Wang, S.; Lin, Y.; Gao, L.; Yang, Z.; Lin, J.; Ren, S.; Li, F.; Chen, J.; Wang, Z.; Dong, Z.; et al. PPAR-γ Integrates Obesity and Adipocyte Clock through Epigenetic Regulation of Bmal1. Theranostics 2022, 12, 1589–1606. [Google Scholar] [CrossRef]
- Leonardini, A.; Laviola, L.; Perrini, S.; Natalicchio, A.; Giorgino, F. Cross-Talk between PPARgamma and Insulin Signaling and Modulation of Insulin Sensitivity. PPAR Res. 2009, 2009, 818945. [Google Scholar] [CrossRef]
- Ivanova, E.A.; Parolari, A.; Myasoedova, V.; Melnichenko, A.A.; Bobryshev, Y.V.; Orekhov, A.N. Peroxisome Proliferator-Activated Receptor (PPAR) Gamma in Cardiovascular Disorders and Cardiovascular Surgery. J. Cardiol. 2015, 66, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Luquet, S.; Lopez-Soriano, J.; Holst, D.; Fredenrich, A.; Melki, J.; Rassoulzadegan, M.; Grimaldi, P.A. Peroxisome Proliferator-Activated Receptor Delta Controls Muscle Development and Oxidative Capability. FASEB J. 2003, 17, 2299–2301. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Dyar, K.A.; Treebak, J.T.; Jepsen, S.L.; Ehrlich, A.M.; Ashcroft, S.P.; Trost, K.; Kunzke, T.; Prade, V.M.; Small, L.; et al. Atlas of Exercise Metabolism Reveals Time-Dependent Signatures of Metabolic Homeostasis. Cell Metab. 2022, 34, 329–345.e8. [Google Scholar] [CrossRef] [PubMed]
- Ruddick-Collins, L.C.; Morgan, P.J.; Johnstone, A.M. Mealtime: A Circadian Disruptor and Determinant of Energy Balance? J. Neuroendocrinol. 2020, 32, e12886. [Google Scholar] [CrossRef]
- Nakahata, Y.; Sahar, S.; Astarita, G.; Kaluzova, M.; Sassone-Corsi, P. Circadian Control of the NAD+ Salvage Pathway by CLOCK-SIRT1. Science 2009, 324, 654–657. [Google Scholar] [CrossRef]
- Liang, F.; Kume, S.; Koya, D. SIRT1 and Insulin Resistance. Nat. Rev. Endocrinol. 2009, 5, 367–373. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, Y.; Wang, Y.; Chao, Y.; Zhang, J.; Jia, Y.; Tie, J.; Hu, D. Regulation of SIRT1 and Its Roles in Inflammation. Front. Immunol. 2022, 13, 831168. [Google Scholar] [CrossRef]
- Kemper, J.K.; Choi, S.-E.; Kim, D.H. Sirtuin 1 Deacetylase: A Key Regulator of Hepatic Lipid Metabolism. Vitam. Horm. 2013, 91, 385–404. [Google Scholar] [CrossRef]
- Henriksson, E.; Huber, A.-L.; Soto, E.K.; Kriebs, A.; Vaughan, M.E.; Duglan, D.; Chan, A.B.; Papp, S.J.; Nguyen, M.; Afetian, M.E.; et al. The Liver Circadian Clock Modulates Biochemical and Physiological Responses to Metformin. J. Biol. Rhythm. 2017, 32, 345–358. [Google Scholar] [CrossRef]
- Bennett, S.; Sato, S. Enhancing the Metabolic Benefits of Exercise: Is Timing the Key? Front. Endocrinol. 2023, 14, 987208. [Google Scholar] [CrossRef]
- Liang, H.; Ward, W.F. PGC-1alpha: A Key Regulator of Energy Metabolism. Adv. Physiol. Educ. 2006, 30, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Hofer, A.; Noe, N.; Tischner, C.; Kladt, N.; Lellek, V.; Schauß, A.; Wenz, T. Defining the Action Spectrum of Potential PGC-1α Activators on a Mitochondrial and Cellular Level in Vivo. Hum. Mol. Genet. 2014, 23, 2400–2415. [Google Scholar] [CrossRef]
- Cipolla-Neto, J.; Amaral, F.G.; Afeche, S.C.; Tan, D.X.; Reiter, R.J. Melatonin, Energy Metabolism, and Obesity: A Review. J. Pineal Res. 2014, 56, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Boden, G.; Ruiz, J.; Urbain, J.L.; Chen, X. Evidence for a Circadian Rhythm of Insulin Secretion. Am. J. Physiol. 1996, 271, E246–E252. [Google Scholar] [CrossRef] [PubMed]
- Schmid, S.M.; Jauch-Chara, K.; Hallschmid, M.; Schultes, B. Mild Sleep Restriction Acutely Reduces Plasma Glucagon Levels in Healthy Men. J. Clin. Endocrinol. Metab. 2009, 94, 5169–5173. [Google Scholar] [CrossRef] [PubMed]
- Oster, H.; Challet, E.; Ott, V.; Arvat, E.; de Kloet, E.R.; Dijk, D.-J.; Lightman, S.; Vgontzas, A.; Van Cauter, E. The Functional and Clinical Significance of the 24-Hour Rhythm of Circulating Glucocorticoids. Endocr. Rev. 2017, 38, 3–45. [Google Scholar] [CrossRef]
- Sun, Q.; Liu, Y.; Wei, W.; Wu, D.; Lin, R.; Wen, D.; Jia, L. Chronic Timed Sleep Restriction Attenuates LepRb-Mediated Signaling Pathways and Circadian Clock Gene Expression in the Rat Hypothalamus. Front. Neurosci. 2020, 14, 909. [Google Scholar] [CrossRef]
- Gómez-Abellán, P.; Gómez-Santos, C.; Madrid, J.A.; Milagro, F.I.; Campion, J.; Martínez, J.A.; Ordovás, J.M.; Garaulet, M. Circadian Expression of Adiponectin and Its Receptors in Human Adipose Tissue. Endocrinology 2010, 151, 115–122. [Google Scholar] [CrossRef]
- Qian, J.; Morris, C.J.; Caputo, R.; Garaulet, M.; Scheer, F.A.J.L. Ghrelin Is Impacted by the Endogenous Circadian System and by Circadian Misalignment in Humans. Int. J. Obes. 2019, 43, 1644–1649. [Google Scholar] [CrossRef]
- Mason, I.C.; Qian, J.; Adler, G.K.; Scheer, F.A.J.L. Impact of Circadian Disruption on Glucose Metabolism: Implications for Type 2 Diabetes. Diabetologia 2020, 63, 462–472. [Google Scholar] [CrossRef]
- Farid, A.; Moussa, P.; Youssef, M.; Haytham, M.; Shamy, A.; Safwat, G. Melatonin Relieves Diabetic Complications and Regenerates Pancreatic Beta Cells by the Reduction in NF-KB Expression in Streptozotocin Induced Diabetic Rats. Saudi J. Biol. Sci. 2022, 29, 103313. [Google Scholar] [CrossRef] [PubMed]
- Boden, G.; Chen, X.; Urbain, J.L. Evidence for a Circadian Rhythm of Insulin Sensitivity in Patients with NIDDM Caused by Cyclic Changes in Hepatic Glucose Production. Diabetes 1996, 45, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, E.T.; Polonsky, K.S.; Copinschi, G.; Bosson, D.; Tillil, H.; Blackman, J.; Lewis, G.; Van Cauter, E. Nocturnal Elevation of Glucose Levels during Fasting in Noninsulin-Dependent Diabetes. J. Clin. Endocrinol. Metab. 1991, 72, 444–454. [Google Scholar] [CrossRef] [PubMed]
- Stamenkovic, J.A.; Olsson, A.H.; Nagorny, C.L.; Malmgren, S.; Dekker-Nitert, M.; Ling, C.; Mulder, H. Regulation of Core Clock Genes in Human Islets. Metabolism 2012, 61, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Kervezee, L.; Kosmadopoulos, A.; Boivin, D.B. Metabolic and Cardiovascular Consequences of Shift Work: The Role of Circadian Disruption and Sleep Disturbances. Eur. J. Neurosci. 2020, 51, 396–412. [Google Scholar] [CrossRef] [PubMed]
- Scheer, F.A.J.L.; Hilton, M.F.; Mantzoros, C.S.; Shea, S.A. Adverse Metabolic and Cardiovascular Consequences of Circadian Misalignment. Proc. Natl. Acad. Sci. USA 2009, 106, 4453–4458. [Google Scholar] [CrossRef]
- Koopman, A.D.M.; Rauh, S.P.; van ’t Riet, E.; Groeneveld, L.; van der Heijden, A.A.; Elders, P.J.; Dekker, J.M.; Nijpels, G.; Beulens, J.W.; Rutters, F. The Association between Social Jetlag, the Metabolic Syndrome, and Type 2 Diabetes Mellitus in the General Population: The New Hoorn Study. J. Biol. Rhythms 2017, 32, 359–368. [Google Scholar] [CrossRef]
- Gozashti, M.H.; Eslami, N.; Radfar, M.H.; Pakmanesh, H. Sleep Pattern, Duration and Quality in Relation with Glycemic Control in People with Type 2 Diabetes Mellitus. Iran. J. Med. Sci. 2016, 41, 531–538. [Google Scholar]
- Wehrens, S.M.T.; Christou, S.; Isherwood, C.; Middleton, B.; Gibbs, M.A.; Archer, S.N.; Skene, D.J.; Johnston, J.D. Meal Timing Regulates the Human Circadian System. Curr. Biol. 2017, 27, 1768–1775.e3. [Google Scholar] [CrossRef]
- Xie, Z.; Sun, Y.; Ye, Y.; Hu, D.; Zhang, H.; He, Z.; Zhao, H.; Yang, H.; Mao, Y. Randomized Controlled Trial for Time-Restricted Eating in Healthy Volunteers without Obesity. Nat. Commun. 2022, 13, 1003. [Google Scholar] [CrossRef]
- Kesztyüs, D.; Cermak, P.; Gulich, M.; Kesztyüs, T. Adherence to Time-Restricted Feeding and Impact on Abdominal Obesity in Primary Care Patients: Results of a Pilot Study in a Pre–Post Design. Nutrients 2019, 11, 2854. [Google Scholar] [CrossRef] [PubMed]
- Tsitsou, S.; Zacharodimos, N.; Poulia, K.-A.; Karatzi, K.; Dimitriadis, G.; Papakonstantinou, E. Effects of Time-Restricted Feeding and Ramadan Fasting on Body Weight, Body Composition, Glucose Responses, and Insulin Resistance: A Systematic Review of Randomized Controlled Trials. Nutrients 2022, 14, 4778. [Google Scholar] [CrossRef]
- Willis, E.A.; Creasy, S.A.; Honas, J.J.; Melanson, E.L.; Donnelly, J.E. The Effects of Exercise Session Timing on Weight Loss and Components of Energy Balance: Midwest Exercise Trial 2. Int. J. Obes. 2020, 44, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Trujillo, J.M.; Nuffer, W.; Ellis, S.L. GLP-1 Receptor Agonists: A Review of Head-to-Head Clinical Studies. Ther. Adv. Endocrinol. Metab. 2015, 6, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Smith, N.K.; Hackett, T.A.; Galli, A.; Flynn, C.R. GLP-1: Molecular Mechanisms and Outcomes of a Complex Signaling System. Neurochem. Int. 2019, 128, 94–105. [Google Scholar] [CrossRef]
- Biancolin, A.D.; Martchenko, A.; Mitova, E.; Gurges, P.; Michalchyshyn, E.; Chalmers, J.A.; Doria, A.; Mychaleckyj, J.C.; Adriaenssens, A.E.; Reimann, F.; et al. The Core Clock Gene, Bmal1, and Its Downstream Target, the SNARE Regulatory Protein Secretagogin, Are Necessary for Circadian Secretion of Glucagon-like Peptide-1. Mol. Metab. 2020, 31, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Liu, Y.; Xin, Y.; Wang, Y. Circadian Secretion Rhythm of GLP-1 and Its Influencing Factors. Front. Endocrinol. 2022, 13, 991397. [Google Scholar] [CrossRef] [PubMed]
- Anini, Y.; Brubaker, P.L. Role of Leptin in the Regulation of Glucagon-Like Peptide-1 Secretion. Diabetes 2003, 52, 252–259. [Google Scholar] [CrossRef]
- Adlanmerini, M.; Nguyen, H.C.B.; Krusen, B.M.; Teng, C.W.; Geisler, C.E.; Peed, L.C.; Carpenter, B.J.; Hayes, M.R.; Lazar, M.A. Hypothalamic REV-ERB Nuclear Receptors Control Diurnal Food Intake and Leptin Sensitivity in Diet-Induced Obese Mice. J. Clin. Investig. 2021, 131, e140424. [Google Scholar] [CrossRef]
- Thornberry, N.A.; Gallwitz, B. Mechanism of Action of Inhibitors of Dipeptidyl-Peptidase-4 (DPP-4). Best. Pract. Res. Clin. Endocrinol. Metab. 2009, 23, 479–486. [Google Scholar] [CrossRef]
- Larochelle, P. Circadian Variation in Blood Pressure: Dipper or Nondipper. J. Clin. Hypertens. 2000, 5, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Douma, L.G.; Gumz, M.L. Circadian Clock-Mediated Regulation of Blood Pressure. Free Radic. Biol. Med. 2018, 119, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Sigmund, C.D. A Clinical Link Between Peroxisome Proliferator-Activated Receptor γ and the Renin–Angiotensin System. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 676–678. [Google Scholar] [CrossRef] [PubMed]
- Lunn, R.M.; Schwingl, P.J.; Atwood, S.T.; Mehta, S.S.; Jahnke, G.D.; Garner, S.C. NTP Cancer Hazard Assessment Report on Night Shift Work and Light at Night, 1st ed.; National Toxicology Program: Research Triangle Park, NC, USA, 2021; pp. 29–61. [Google Scholar] [CrossRef]
- Manfredini, R.; Fabbian, F.; De Giorgi, A.; Zucchi, B.; Cappadona, R.; Signani, F.; Katsiki, N.; Mikhailidis, D.P. Daylight Saving Time and Myocardial Infarction: Should We Be Worried? A Review of the Evidence. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 750–755. [Google Scholar] [CrossRef] [PubMed]
- Manfredini, R.; Fabbian, F.; Cappadona, R.; Modesti, P.A. Daylight Saving Time, Circadian Rhythms, and Cardiovascular Health. Intern. Emerg. Med. 2018, 13, 641–646. [Google Scholar] [CrossRef]
- Frederiksen, L.; Nielsen, T.L.; Wraae, K.; Hagen, C.; Frystyk, J.; Flyvbjerg, A.; Brixen, K.; Andersen, M. Subcutaneous Rather than Visceral Adipose Tissue Is Associated with Adiponectin Levels and Insulin Resistance in Young Men. J. Clin. Endocrinol. Metab. 2009, 94, 4010–4015. [Google Scholar] [CrossRef]
- Cho, S.-A.; Joo, H.J.; Cho, J.-Y.; Lee, S.H.; Park, J.H.; Hong, S.J.; Yu, C.W.; Lim, D.-S. Visceral Fat Area and Serum Adiponectin Level Predict the Development of Metabolic Syndrome in a Community-Based Asymptomatic Population. PLoS ONE 2017, 12, e0169289. [Google Scholar] [CrossRef]
- Awad, K.; Serban, M.-C.; Penson, P.; Mikhailidis, D.P.; Toth, P.P.; Jones, S.R.; Rizzo, M.; Howard, G.; Lip, G.Y.H.; Banach, M.; et al. Effects of Morning vs Evening Statin Administration on Lipid Profile: A Systematic Review and Meta-Analysis. J. Clin. Lipidol. 2017, 11, 972–985.e9. [Google Scholar] [CrossRef]
- Stec, D.E.; Gordon, D.M.; Hipp, J.A.; Hong, S.; Mitchell, Z.L.; Franco, N.R.; Robison, J.W.; Anderson, C.D.; Stec, D.F.; Hinds, T.D. Loss of Hepatic PPARα Promotes Inflammation and Serum Hyperlipidemia in Diet-Induced Obesity. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2019, 317, R733–R745. [Google Scholar] [CrossRef]
- Hermida, R.C.; Calvo, C.; Ayala, D.E.; Fernández, J.R.; Covelo, M.; Mojón, A.; López, J.E. Treatment of Non-Dipper Hypertension with Bedtime Administration of Valsartan. J. Hypertens. 2005, 23, 1913–1922. [Google Scholar] [CrossRef]
- Awazawa, M.; Ueki, K.; Inabe, K.; Yamauchi, T.; Kubota, N.; Kaneko, K.; Kobayashi, M.; Iwane, A.; Sasako, T.; Okazaki, Y.; et al. Adiponectin Enhances Insulin Sensitivity by Increasing Hepatic IRS-2 Expression via a Macrophage-Derived IL-6-Dependent Pathway. Cell Metab. 2011, 13, 401–412. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.M.D. Adiponectin: Role in Physiology and Pathophysiology. Int. J. Prev. Med. 2020, 11, 136. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, M.; Lefebvre, P.; Staels, B. Molecular Mechanism of PPARα Action and Its Impact on Lipid Metabolism, Inflammation and Fibrosis in Non-Alcoholic Fatty Liver Disease. J. Hepatol. 2015, 62, 720–733. [Google Scholar] [CrossRef] [PubMed]
- Jamshed, H.; Beyl, R.A.; Della Manna, D.L.; Yang, E.S.; Ravussin, E.; Peterson, C.M. Early Time-Restricted Feeding Improves 24-Hour Glucose Levels and Affects Markers of the Circadian Clock, Aging, and Autophagy in Humans. Nutrients 2019, 11, 1234. [Google Scholar] [CrossRef]
- Kuroda, T.; Kario, K.; Hoshide, S.; Hashimoto, T.; Nomura, Y.; Saito, Y.; Mito, H.; Shimada, K. Effects of Bedtime vs. Morning Administration of the Long-Acting Lipophilic Angiotensin-Converting Enzyme Inhibitor Trandolapril on Morning Blood Pressure in Hypertensive Patients. Hypertens. Res. 2004, 27, 15–20. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Fontao, M.J.; Mojón, A.; Fernández, J.R. Chronotherapy with Valsartan/Amlodipine Fixed Combination: Improved Blood Pressure Control of Essential Hypertension with Bedtime Dosing. Chronobiol. Int. 2010, 27, 1287–1303. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Fernández, J.R.; Calvo, C. Comparison of the Efficacy of Morning versus Evening Administration of Telmisartan in Essential Hypertension. Hypertension 2007, 50, 715–722. [Google Scholar] [CrossRef]
- Fedchenko, T.; Izmailova, O.; Shynkevych, V.; Shlykova, O.; Kaidashev, I. PPAR-γ Agonist Pioglitazone Restored Mouse Liver MRNA Expression of Clock Genes and Inflammation-Related Genes Disrupted by Reversed Feeding. PPAR Res. 2022, 2022, 7537210. [Google Scholar] [CrossRef]
- Parr, E.B.; Devlin, B.L.; Radford, B.E.; Hawley, J.A. A Delayed Morning and Earlier Evening Time-Restricted Feeding Protocol for Improving Glycemic Control and Dietary Adherence in Men with Overweight/Obesity: A Randomized Controlled Trial. Nutrients 2020, 12, 505. [Google Scholar] [CrossRef]
- Creasy, S.A.; Wayland, L.; Panter, S.L.; Purcell, S.A.; Rosenberg, R.; Willis, E.A.; Shiferaw, B.; Grau, L.; Breit, M.J.; Bessesen, D.H.; et al. Effect of Morning and Evening Exercise on Energy Balance: A Pilot Study. Nutrients 2022, 14, 816. [Google Scholar] [CrossRef]
- Van Someren, E.J.W.; Lijzenga, C.; Mirmiran, M.; Swaab, D.F. Long-Term Fitness Training Improves the Circadian Rest-Activity Rhythm in Healthy Elderly Males. J. Biol. Rhythm. 1997, 12, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Blair, S.N.; Kohl, H.W.; Barlow, C.E.; Paffenbarger, R.S.; Gibbons, L.W.; Macera, C.A. Changes in Physical Fitness and All-Cause Mortality. A Prospective Study of Healthy and Unhealthy Men. JAMA 1995, 273, 1093–1098. [Google Scholar] [CrossRef]
- Blair, S.N.; Kohl, H.W.; Paffenbarger, R.S.; Clark, D.G.; Cooper, K.H.; Gibbons, L.W. Physical Fitness and All-Cause Mortality. A Prospective Study of Healthy Men and Women. JAMA 1989, 262, 2395–2401. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Downes, M.; Yu, R.T.; Bookout, A.L.; He, W.; Straume, M.; Mangelsdorf, D.J.; Evans, R.M. Nuclear Receptor Expression Links the Circadian Clock to Metabolism. Cell 2006, 126, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.; Hitomi, H.; Nishiyama, A. Cardioprotective Effects of SGLT2 Inhibitors Are Possibly Associated with Normalization of the Circadian Rhythm of Blood Pressure. Hypertens. Res. 2017, 40, 535–540. [Google Scholar] [CrossRef]
- ElSayed, N.A.; Aleppo, G.; Aroda, V.R.; Bannuru, R.R.; Brown, F.M.; Bruemmer, D.; Collins, B.S.; Hilliard, M.E.; Isaacs, D.; Johnson, E.L.; et al. 3. Prevention or Delay of Type 2 Diabetes and Associated Comorbidities: Standards of Care in Diabetes—2023. Diabetes Care 2023, 46, S41–S48. [Google Scholar] [CrossRef]
- Schuppelius, B.; Peters, B.; Ottawa, A.; Pivovarova-Ramich, O. Time Restricted Eating: A Dietary Strategy to Prevent and Treat Metabolic Disturbances. Front. Endocrinol. 2021, 12, 683140. [Google Scholar] [CrossRef]
- Gabel, K.; Hoddy, K.K.; Haggerty, N.; Song, J.; Kroeger, C.M.; Trepanowski, J.F.; Panda, S.; Varady, K.A. Effects of 8-Hour Time Restricted Feeding on Body Weight and Metabolic Disease Risk Factors in Obese Adults: A Pilot Study. Nutr. Healthy Aging 2018, 4, 345–353. [Google Scholar] [CrossRef]
- Hutchison, A.T.; Regmi, P.; Manoogian, E.N.C.; Fleischer, J.G.; Wittert, G.A.; Panda, S.; Heilbronn, L.K. Time-Restricted Feeding Improves Glucose Tolerance in Men at Risk for Type 2 Diabetes: A Randomized Crossover Trial. Obesity 2019, 27, 724–732. [Google Scholar] [CrossRef]
- Jakubowicz, D.; Barnea, M.; Wainstein, J.; Froy, O. High Caloric Intake at Breakfast vs. Dinner Differentially Influences Weight Loss of Overweight and Obese Women. Obesity 2013, 21, 2504–2512. [Google Scholar] [CrossRef]
- Timlin, M.T.; Pereira, M.A.; Story, M.; Neumark-Sztainer, D. Breakfast Eating and Weight Change in a 5-Year Prospective Analysis of Adolescents: Project EAT (Eating Among Teens). Pediatrics 2008, 121, e638–e645. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, M.J.; Manoogian, E.N.C.; Zadourian, A.; Lo, H.; Fakhouri, S.; Shoghi, A.; Wang, X.; Fleischer, J.G.; Navlakha, S.; Panda, S.; et al. Ten-Hour Time-Restricted Eating Reduces Weight, Blood Pressure, and Atherogenic Lipids in Patients with Metabolic Syndrome. Cell Metab. 2020, 31, 92–104.e5. [Google Scholar] [CrossRef] [PubMed]
- Patterson, R.E.; Sears, D.D. Metabolic Effects of Intermittent Fasting. Annu. Rev. Nutr. 2017, 37, 371–393. [Google Scholar] [CrossRef]
- Lilja, S.; Stoll, C.; Krammer, U.; Hippe, B.; Duszka, K.; Debebe, T.; Höfinger, I.; König, J.; Pointner, A.; Haslberger, A. Five Days Periodic Fasting Elevates Levels of Longevity Related Christensenella and Sirtuin Expression in Humans. Int. J. Mol. Sci. 2021, 22, 2331. [Google Scholar] [CrossRef] [PubMed]
- Ossoli, A.; Frascarelli, S.; Da Dalt, L.; Norata, G.D.; Calabresi, L.; Gomaraschi, M. Role of Lysosomal Acid Lipase in PPAR-Mediated Reduction of Lipid Accumulation in Hepatocytes. Atherosclerosis 2022, 355, 20. [Google Scholar] [CrossRef]
- Varady, K.A.; Bhutani, S.; Klempel, M.C.; Kroeger, C.M.; Trepanowski, J.F.; Haus, J.M.; Hoddy, K.K.; Calvo, Y. Alternate Day Fasting for Weight Loss in Normal Weight and Overweight Subjects: A Randomized Controlled Trial. Nutr. J. 2013, 12, 146. [Google Scholar] [CrossRef]
- Varady, K.A.; Bhutani, S.; Church, E.C.; Klempel, M.C. Short-Term Modified Alternate-Day Fasting: A Novel Dietary Strategy for Weight Loss and Cardioprotection in Obese Adults. Am. J. Clin. Nutr. 2009, 90, 1138–1143. [Google Scholar] [CrossRef]
- Tripolt, N.J.; Hofer, S.J.; Pferschy, P.N.; Aziz, F.; Durand, S.; Aprahamian, F.; Nirmalathasan, N.; Waltenstorfer, M.; Eisenberg, T.; Obermayer, A.M.A.; et al. Glucose Metabolism and Metabolomic Changes in Response to Prolonged Fasting in Individuals with Obesity, Type 2 Diabetes and Non-Obese People—A Cohort Trial. Nutrients 2023, 15, 511. [Google Scholar] [CrossRef]
- Kang, J.; Shi, X.; Fu, J.; Li, H.; Ma, E.; Chen, W. Effects of an Intermittent Fasting 5:2 Plus Program on Body Weight in Chinese Adults with Overweight or Obesity: A Pilot Study. Nutrients 2022, 14, 4734. [Google Scholar] [CrossRef]
- Sundfør, T.M.; Svendsen, M.; Tonstad, S. Effect of Intermittent versus Continuous Energy Restriction on Weight Loss, Maintenance and Cardiometabolic Risk: A Randomized 1-Year Trial. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 698–706. [Google Scholar] [CrossRef]
- Miyazaki, T.; Hashimoto, S.; Masubuchi, S.; Honma, S.; Honma, K.I. Phase-Advance Shifts of Human Circadian Pacemaker Are Accelerated by Daytime Physical Exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2001, 281, R197–R205. [Google Scholar] [CrossRef]
- Van Reeth, O.; Sturis, J.; Byrne, M.M.; Blackman, J.D.; L’Hermite-Balériaux, M.; Leproult, R.; Oliner, C.; Refetoff, S.; Turek, F.W.; Van Cauter, E. Nocturnal Exercise Phase Delays Circadian Rhythms of Melatonin and Thyrotropin Secretion in Normal Men. Am. J. Physiol. 1994, 266, E964–E974. [Google Scholar] [CrossRef]
- Ezagouri, S.; Zwighaft, Z.; Sobel, J.; Baillieul, S.; Doutreleau, S.; Ladeuix, B.; Golik, M.; Verges, S.; Asher, G. Physiological and Molecular Dissection of Daily Variance in Exercise Capacity. Cell Metab. 2019, 30, 78–91.e4. [Google Scholar] [CrossRef]
- Iwayama, K.; Kurihara, R.; Nabekura, Y.; Kawabuchi, R.; Park, I.; Kobayashi, M.; Ogata, H.; Kayaba, M.; Satoh, M.; Tokuyama, K. Exercise Increases 24-h Fat Oxidation Only When It Is Performed Before Breakfast. EBioMedicine 2015, 2, 2003–2009. [Google Scholar] [CrossRef]
- Cardinali, D.P.; Brown, G.M.; Pandi-Perumal, S.R. Chronotherapy. Handb. Clin. Neurol. 2021, 179, 357–370. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Fontao, M.J.; Mojón, A.; Alonso, I.; Fernández, J.R. Administration-Time-Dependent Effects of Spirapril on Ambulatory Blood Pressure in Uncomplicated Essential Hypertension. Chronobiol. Int. 2010, 27, 560–574. [Google Scholar] [CrossRef]
- Hozumi, K.; Sugawara, K.; Ishihara, T.; Ishihara, N.; Ogawa, W. Effects of Imeglimin on Mitochondrial Function, AMPK Activity, and Gene Expression in Hepatocytes. Sci. Rep. 2023, 13, 746. [Google Scholar] [CrossRef]
- Nowak, M.; Grzeszczak, W. Imeglimin: A New Antidiabetic Drug with Potential Future in the Treatment of Patients with Type 2 Diabetes. Endokrynol. Pol. 2022, 73, 361–370. [Google Scholar] [CrossRef]
- Fonseca-Correa, J.I.; Correa-Rotter, R. Sodium-Glucose Cotransporter 2 Inhibitors Mechanisms of Action: A Review. Front. Med. 2021, 8, 777861. [Google Scholar] [CrossRef] [PubMed]
- ElDash, R.M.; Raslan, M.A.; Shaheen, S.M.; Sabri, N.A. The Effect of Morning versus Evening Administration of Empagliflozin on Its Pharmacokinetics and Pharmacodynamics Characteristics in Healthy Adults: A Two-Way Crossover, Non-Randomised Trial. F1000Research 2021, 10, 321. [Google Scholar] [CrossRef] [PubMed]
- Porcellati, F.; Lucidi, P.; Cioli, P.; Candeloro, P.; Marinelli Andreoli, A.; Marzotti, S.; Ambrogi, M.; Bolli, G.B.; Fanelli, C.G. Pharmacokinetics and Pharmacodynamics of Insulin Glargine Given in the Evening as Compared With in the Morning in Type 2 Diabetes. Diabetes Care 2015, 38, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, Y.; Tanaka, T.; Wakakuri, H.; Kita, Y.; Kanamori, T.; Takamura, T. Metabolic and Sympathovagal Effects of Bolus Insulin Glulisine versus Basal Insulin Glargine Therapy in People with Type 2 Diabetes: A Randomized Controlled Study. J. Diabetes Investig. 2021, 12, 1193–1201. [Google Scholar] [CrossRef] [PubMed]
- Wallace, A.; Chinn, D.; Rubin, G. Taking Simvastatin in the Morning Compared with in the Evening: Randomised Controlled Trial. BMJ 2003, 327, 788. [Google Scholar] [CrossRef] [PubMed]
- Bougarne, N.; Weyers, B.; Desmet, S.J.; Deckers, J.; Ray, D.W.; Staels, B.; De Bosscher, K. Molecular Actions of PPARα in Lipid Metabolism and Inflammation. Endocr. Rev. 2018, 39, 760–802. [Google Scholar] [CrossRef]
- Oishi, K.; Shirai, H.; Ishida, N. CLOCK Is Involved in the Circadian Transactivation of Peroxisome-Proliferator-Activated Receptor Alpha (PPARalpha) in Mice. Biochem. J. 2005, 386, 575–581. [Google Scholar] [CrossRef]
- Post, S.M.; Duez, H.; Gervois, P.P.; Staels, B.; Kuipers, F.; Princen, H.M. Fibrates Suppress Bile Acid Synthesis via Peroxisome Proliferator-Activated Receptor-Alpha-Mediated Downregulation of Cholesterol 7alpha-Hydroxylase and Sterol 27-Hydroxylase Expression. Arterioscler. Thromb. Vasc. Biol. 2001, 21, 1840–1845. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, J. Bile Acid Metabolism and Circadian Rhythms. Am. J. Physiol.-Gastrointest. Liver Physiol. 2020, 319, G549–G563. [Google Scholar] [CrossRef]
- Álvarez-López, H.; Ruiz-Gastélum, E.; Díaz-Aragón, A. Tratamiento Actual de La Hipertrigliceridemia. Cardiovasc. Metab. Sci. 2021, 32, 242–246. [Google Scholar] [CrossRef]
- Knight, B.L.; Hebbachi, A.; Hauton, D.; Brown, A.-M.; Wiggins, D.; Patel, D.D.; Gibbons, G.F. A Role for PPARalpha in the Control of SREBP Activity and Lipid Synthesis in the Liver. Biochem. J. 2005, 389, 413–421. [Google Scholar] [CrossRef]

| IR | Central Obesity | Dyslipidemia | Hypertension |
|---|---|---|---|
Disrupted SIRT1 expression results in:
| Phase delay leads to REV-ERB-dependent leptin resistance in SCN [15,71] and decrease in POMC Low POMC is associated with increased appetite, reduced energy expenditure, and increased fat deposition [49]. | Expression of genes involved in lipid and cholesterol metabolism, such as HMG-CoA reductase and SREBF1 are attenuated or abolished in PPARα-knockout mice [20]. | Obesity correlates with increased activity of ACE in white adipose tissue, which results in higher plasma concentrations of angiotensin II and loss of its diurnal rhythm [74]. |
Dampened PPARγ expression [31]:
| There is a significant difference between circadian rhythm of adiponectin and its receptors in subcutaneous versus visceral fat [50,79,80,81]. Larger amount of visceral white adipose tissue is associated with dampened rhythm of adiponectin, and IR [50,79,80,81]. | The hepatic PPARα-knockout mice show significantly increased levels of LDL subfraction L5 in response to high-fat feeding [82]. | Non-dipping hypertension (i.e., absence of a physiological 10–20% decrease in BP at night) is associated with activation of the RAAS, increased risk of chronic kidney disease, and adverse CV events [74,83]. |
| Adiponectin rhythm delay, associated with both circadian disruption and central obesity [50,84,85]. | Changes in adipocytes’ metabolism, leading to down-regulation of PPARγ [31]. PPARγ repression leads to reduced histone acetylation and methylation, thereby repressing BMAL1 transcription and expression in adipocytes [31]. | Proinflammatory macrophage markers TNF and iNOS are increased in the liver of the PPARα knockout mice compared to controls [86]. |
| Intervention | Recommended Timing | Explanation |
|---|---|---|
| Food consumption (without specific diet plan) | ||
| Time-restricted feeding (TRF) | Early TRF- food intake between 8:00 and 17:00 | Fasting in the late-active and rest phase upregulates SIRT1, AMPK, and PGC-1α, and causes a phase-advance and the alignment of molecular clocks throughout the body [9,92]. |
| Exercise | ||
| Aerobic exercise | Morning | Morning exercise is associated with a 2-fold increase in total body energy expenditure and more extensive FFA oxidation compared to evening exercise [93]. Aerobic exercise restores disrupted circadian rhythm and improves sleep [94,95,96]. |
| Medication | ||
| Antihypertensive medication (e.g., ACEI, ARB) | Bedtime | Better efficacy of ACEI, ARB, and β-blockers at night can be explained by the inhibition of nocturnal RAAS [74,83,88,90]. |
| Extended-release metformin | Bedtime, with dinner | By targeting clock genes, suppresses overnight gluconeogenesis and prevents morning hyperglycemia (i.e., dawn phenomenon) [41]. |
| Thiazolidinediones (PPARγ agonists) | Morning | Boost PPARγ-mediated insulin sensitivity in AM [91,97]. |
| GLP-1 agonists (liraglutide, lixisenatid) | Authors’ suggestion: noon | GLP-1 activity peaks at 14:00 [66,69]. It is possible that GLP-1 analogs could be more effective if administered in sync with endogenous GLP-1 rhythms [66]. |
| DPP4is | Authors’ suggestion: noon | It is possible that DPP4is could be more effective if administered in sync with endogenous GLP-1 rhythms [69,72]. |
| SGLT2is | Authors’ suggestion: morning | No significant difference in effectiveness of SGLT2is in morning vs. evening administration [98]. Evening administration might negatively affect circadian rhythm by increasing diuresis and disrupting night-time sleep [98]. |
| Insulin (long acting) | Bedtime | Evening glargine decreases nocturnal endogenous glucose production, lipolysis, and glucagon concentration. It increases parasympathetic tone during night-time and decreases sympathetic nerve activity at dawn, which can improve treatment of morning hyperglycemia [46,52]. |
| Statins (esp. short-acting) | Bedtime | Statins suppress HMG-CoA reductase-regulated cholesterol biosynthesis, which peaks at night [81]. |
| Fibrates and omega-3-fatty acids (PPARα agonists) | Bedtime, with dinner | At night, they enhance PPARα physiological activity—β-oxidation and ketogenesis. Bedtime administration could reduce side effects—gallstone formation [20,86]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zečević, K.; Popović, N.; Vuksanović Božarić, A.; Vukmirović, M.; Rizzo, M.; Muzurović, E. Timing Is Important—Management of Metabolic Syndrome According to the Circadian Rhythm. Biomedicines 2023, 11, 1171. https://doi.org/10.3390/biomedicines11041171
Zečević K, Popović N, Vuksanović Božarić A, Vukmirović M, Rizzo M, Muzurović E. Timing Is Important—Management of Metabolic Syndrome According to the Circadian Rhythm. Biomedicines. 2023; 11(4):1171. https://doi.org/10.3390/biomedicines11041171
Chicago/Turabian StyleZečević, Ksenija, Nataša Popović, Aleksandra Vuksanović Božarić, Mihailo Vukmirović, Manfredi Rizzo, and Emir Muzurović. 2023. "Timing Is Important—Management of Metabolic Syndrome According to the Circadian Rhythm" Biomedicines 11, no. 4: 1171. https://doi.org/10.3390/biomedicines11041171
APA StyleZečević, K., Popović, N., Vuksanović Božarić, A., Vukmirović, M., Rizzo, M., & Muzurović, E. (2023). Timing Is Important—Management of Metabolic Syndrome According to the Circadian Rhythm. Biomedicines, 11(4), 1171. https://doi.org/10.3390/biomedicines11041171








