An Individualized Low-Pneumoperitoneum-Pressure Strategy May Prevent a Reduction in Liver Perfusion during Colorectal Laparoscopic Surgery
Abstract
1. Introduction
2. Materials and Methods
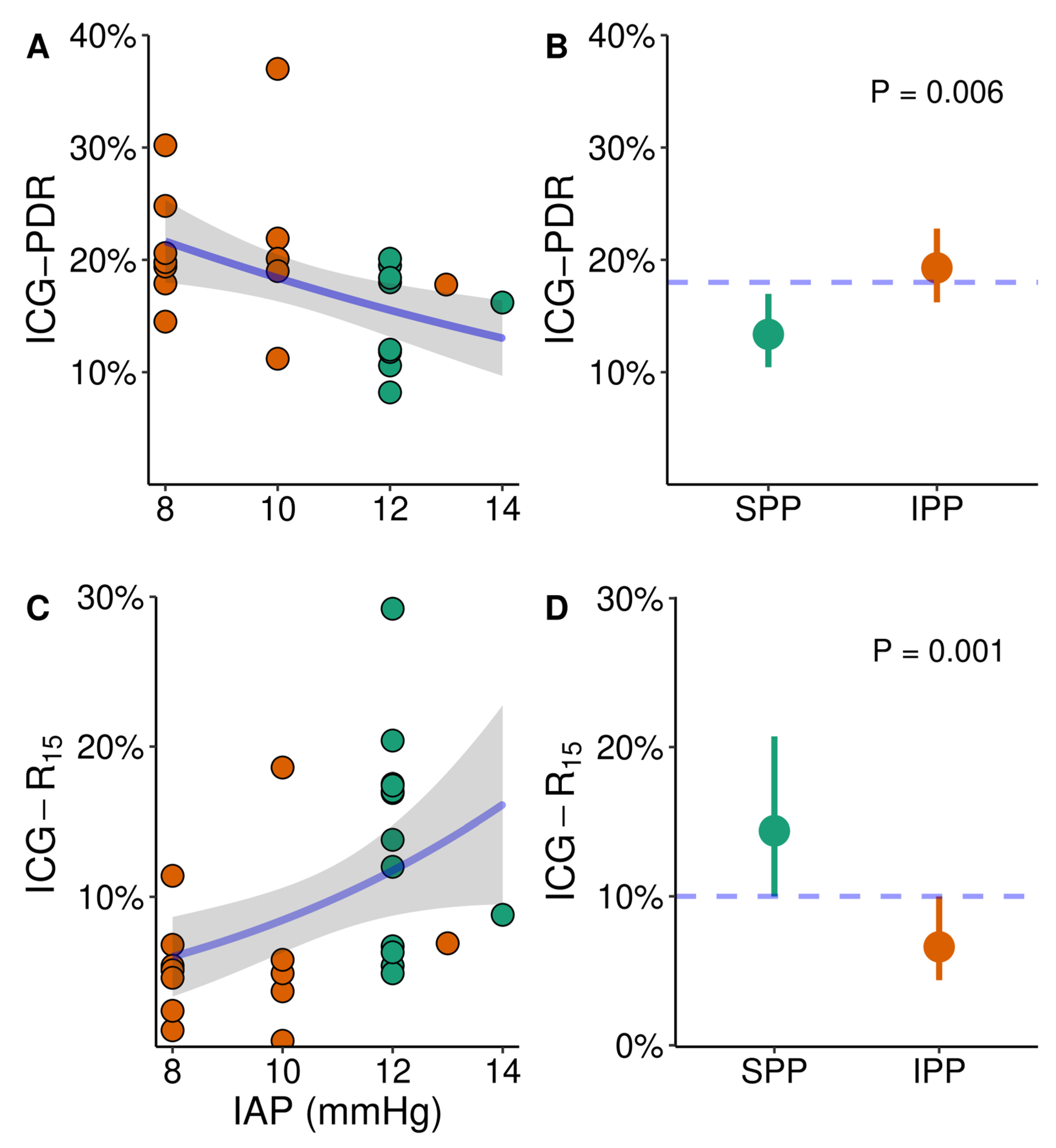
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Blobner, M.; Bogdanski, R.; Kochs, E.; Henke, J.; Findeis, A.; Jelen-Esselborn, J. Effects of intraabdominally insufflated carbon dioxide and elevated intraabdominal pressure on splanchnic circulation: An experimental study in pigs. Anesthesiology 1998, 89, 475–482. [Google Scholar] [CrossRef]
- Eryılmaz, H.B.; Memiş, D.; Sezer, A.; Inal, M.T. The effects of different insufflation pressures on liver functions assessed with LiMON on patients undergoing laparoscopic cholecystectomy. Sci. World J. 2012, 2012, 172575. [Google Scholar] [CrossRef] [PubMed]
- Albers, K.I.; Polat, F.; Loonen, T.; Graat, L.J.; Mulier, J.P.; Snoeck, M.M.J.; Panhuizen, I.F.; Vermulst, A.A.; Scheffer, G.J.; Warlé, M.C. Visualizing improved peritoneal perfusion at lower intra-abdominal pressure by fluorescent imaging during laparoscopic surgery: A randomised controlled study. Int. J. Surg. 2020, 77, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Sakka, S.G. Assessment of liver perfusion and function by indocyanine green in the perioperative setting and in critically ill patients. J. Clin. Monit. Comput. 2018, 32, 787–796. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Cambronero, O.; Mazzinari, G.; Flor Lorente, B.; Garcia Gregorio, N.; Robles-Hernandez, D.; Olmedilla Arnal, L.E.; Martin de Pablos, A.; Schultz, M.J.; Errando, C.L.; Argente Navarro, M.P. Effect of an individualized versus standard pneumoperitoneum pressure strategy on postoperative recovery: A randomized clinical trial in laparoscopic colorectal surgery. Br. J. Surg. 2020, 107, 1605–1614, Erratum in Br. J. Surg. 2020, 107, e630–e631. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Cambronero, O.; Mazzinari, G.; Errando, C.L.; Schultz, M.J.; Flor Lorente, B.; García-Gregorio, N.; Vila Montañés, M.; Robles-Hernández, D.; Olmedilla Arnal, L.E.; Martín-De-Pablos, A.; et al. An individualised versus a conventional pneumoperitoneum pressure strategy during colorectal laparoscopic surgery: Rationale and study protocol for a multicentre randomised clinical study. Trials 2019, 20, 190, Erratum in Trials 2020, 21, 70. [Google Scholar] [CrossRef] [PubMed]
- Meierhenrich, R.; Gauss, A.; Vandenesch, P.; Georgieff, M.; Poch, B.; Schütz, M. The effects of intraabdominally insufflated carbon dioxide on hepatic blood flow during laparoscopic surgery assessed by transesophageal echocardiography. Anesth. Analg. 2005, 100, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Vos, J.J.; Wietasch, J.K.G.; Absalom, A.R.; Hendriks, H.G.D.; Scheeren, T.W.L. Green light for liver function monitoring using indocyanine green? An overview of current clinical applications. Anaesthesia 2014, 69, 1364–1376. [Google Scholar] [CrossRef] [PubMed]
- Neudecker, J.; Sauerland, S.; Neugebauer, E.A.M.; Bergamaschi, R.; Bonjer, H.J.; Cuschieri, A.; Fuchs, K.H.; Jacobi, C.A.; Jansen, F.W.; Koivusalo, A.M.; et al. The EAES Clinical Practice Guidelines on the Pneumoperitoneum for Laparoscopic Surgery (2002). EAES Guidelines for Endoscopic Surgery Twelve Years Evidence-Based Surgery; Springer: Berlin, Germany, 2006; pp. 39–85. [Google Scholar]
- Malbrain, M.L.; Viaene, D.; Kortgen, A.; De Laet, I.; Dits, H.; Van Regenmortel, N.; Schoonheydt, K.; Bauer, M. Relationship between intra-abdominal pressure and indocyanine green plasma disappearance rate: Hepatic perfusion may be impaired in critically ill patients with intra-abdominal hypertension. Ann. Intensive Care 2012, 2 (Suppl. S1), S19. [Google Scholar] [CrossRef] [PubMed]
- Seibel, A.; Sakka, S.G. Indocyanine green plasma disappearance rate: Estimation of abdominal perfusion disturbances. Anaesthsist 2010, 59, 1091–1098. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Cambronero, O.; Flor Lorente, B.; Mazzinari, G.; Vila Montañes, M.; García Gregorio, N.; Robles Hernandez, D.; Olmedilla Arnal, L.; Argente Navarro, M.P.; Schultz, M.J.; Errando, C.L. A multifaceted individualized pneumoperitoneum strategy for laparoscopic colorectal surgery: A multicenter observational feasibility study. Surg. Endosc. 2019, 33, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Özdemir-van Brunschot, D.M.D.; van Laarhoven, K.C.J.H.M.; Scheffer, G.J.; Pouwels, S.; Wever, K.E.; Warlé, M.C. What is the evidence for the use of low-pressure pneumoperitoneum? A systematic review. Surg. Endosc. 2015, 30, 2049–2065. [Google Scholar] [CrossRef] [PubMed]
- Mazzinari, G.; Diaz-Cambronero, O.; Alonso-Iñigo, J.M.; Garcia-Gregorio, N.; Ayas-Montero, B.; Ibañez, J.L.; Serpa Neto, A.; Ball, L.; Gama de Abreu, M.; Pelosi, P.; et al. Intraabdominal Pressure Targeted Positive End-expiratory Pressure during Laparoscopic Surgery: An Open-label, Nonrandomized, Crossover, Clinical Trial. Anesthesiology 2020, 132, 667–677. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.J.; Farrell, S.M. Physiology and Role of Intraocular Pressure in Contemporary Anesthesia. Anest. Analg. 2018, 126, 1551–1562. [Google Scholar] [CrossRef] [PubMed]
| All (n = 29) | SPP Group (n = 14) | IPP Group (n = 15) | SMD | |
|---|---|---|---|---|
| Age (years) | 66 (60–74) | 71 (63–77) | 64 (58–69) | 0.61 |
| Sex (female) | 5 (17.2%) | 5 (35.7%) | 0 (0.0%) | 1.05 |
| ASA risk score | 0.26 | |||
| I | 3 (10.3%) | 1 (7.1%) | 2 (13.3%) | |
| II | 19 (65.5%) | 10 (71.4%) | 9 (60.0%) | |
| III | 7 (24.1%) | 3 (21.4%) | 4 (26.7%) | |
| Intraoperative IAP (mmHg) | 5.32 | |||
| 8 | 8 (27.6%) | 0 (0.0%) | 8 (53.3%) | |
| 10 | 5 (17.2%) | 0 (0.0%) | 5 (33.3%) | |
| 12 | 13 (44.8%) | 12 (85.7%) | 1 (6.7%) | |
| 13 | 1 (3.4%%) | 0 (0.0%) | 1 (6.7%) | |
| 14 | 2 (6.9%) | 2 (14.3%) | 0 (0.0%) | |
| BMI (Kg · m−2) | 26.8 (3.6) | 26.5 (3.6) | 27.1 (3.7) | 0.16 |
| Type of surgery (indication) | 0.80 | |||
| Subtotal colectomy | 1 (3.4%) | 1 (7.1%) | 0 (0.0%) | |
| Total colectomy | 2 (6.9%) | 1 (7.1%) | 1 (6.7%) | |
| Right hemicolectomy | 9 (31.0%) | 6 (42.9%) | 3 (20.0%) | |
| Left hemicolectomy | 1 (3.4%) | 0 (0.0%) | 1 (6.7%) | |
| Sigmoidectomy | 5 (17.2%) | 2 (14.3%) | 3 (20.0%) | |
| Rectal anterior resection | 11 (37.9%) | 4 (28.6%) | 7 (46.7%) | |
| Type of surgery (Rectal) | 11 (37.9%) | 4 (28.6%) | 7 (46.7%) | 0.38 |
| Surgery duration (minutes) | 135 (105–210) | 135 (104–201) | 135 (110–212) | 0.07 |
| ICG–PDR (%) | 18% (6%) | 14% (4%) | 20% (7%) | 1.14 |
| ICG–R15 (%) | 10% (7%) | 13% (7%) | 7% (5%) | 1.04 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olmedilla Arnal, L.E.; Cambronero, O.D.; Mazzinari, G.; Pérez Peña, J.M.; Zorrilla Ortúzar, J.; Rodríguez Martín, M.; Vila Montañes, M.; Schultz, M.J.; Rovira, L.; Argente Navarro, M.P.; et al. An Individualized Low-Pneumoperitoneum-Pressure Strategy May Prevent a Reduction in Liver Perfusion during Colorectal Laparoscopic Surgery. Biomedicines 2023, 11, 891. https://doi.org/10.3390/biomedicines11030891
Olmedilla Arnal LE, Cambronero OD, Mazzinari G, Pérez Peña JM, Zorrilla Ortúzar J, Rodríguez Martín M, Vila Montañes M, Schultz MJ, Rovira L, Argente Navarro MP, et al. An Individualized Low-Pneumoperitoneum-Pressure Strategy May Prevent a Reduction in Liver Perfusion during Colorectal Laparoscopic Surgery. Biomedicines. 2023; 11(3):891. https://doi.org/10.3390/biomedicines11030891
Chicago/Turabian StyleOlmedilla Arnal, Luis Enrique, Oscar Diaz Cambronero, Guido Mazzinari, José María Pérez Peña, Jaime Zorrilla Ortúzar, Marcos Rodríguez Martín, Maria Vila Montañes, Marcus J. Schultz, Lucas Rovira, Maria Pilar Argente Navarro, and et al. 2023. "An Individualized Low-Pneumoperitoneum-Pressure Strategy May Prevent a Reduction in Liver Perfusion during Colorectal Laparoscopic Surgery" Biomedicines 11, no. 3: 891. https://doi.org/10.3390/biomedicines11030891
APA StyleOlmedilla Arnal, L. E., Cambronero, O. D., Mazzinari, G., Pérez Peña, J. M., Zorrilla Ortúzar, J., Rodríguez Martín, M., Vila Montañes, M., Schultz, M. J., Rovira, L., Argente Navarro, M. P., & on behalf of the IPPColLapSe II investigators. (2023). An Individualized Low-Pneumoperitoneum-Pressure Strategy May Prevent a Reduction in Liver Perfusion during Colorectal Laparoscopic Surgery. Biomedicines, 11(3), 891. https://doi.org/10.3390/biomedicines11030891






