Endothelial, Vascular and Sympathetic Alterations as Therapeutic Targets in Chronic Heart Failure
Abstract
1. Introduction
2. Assessment of Vascular–Sympathetic Functions
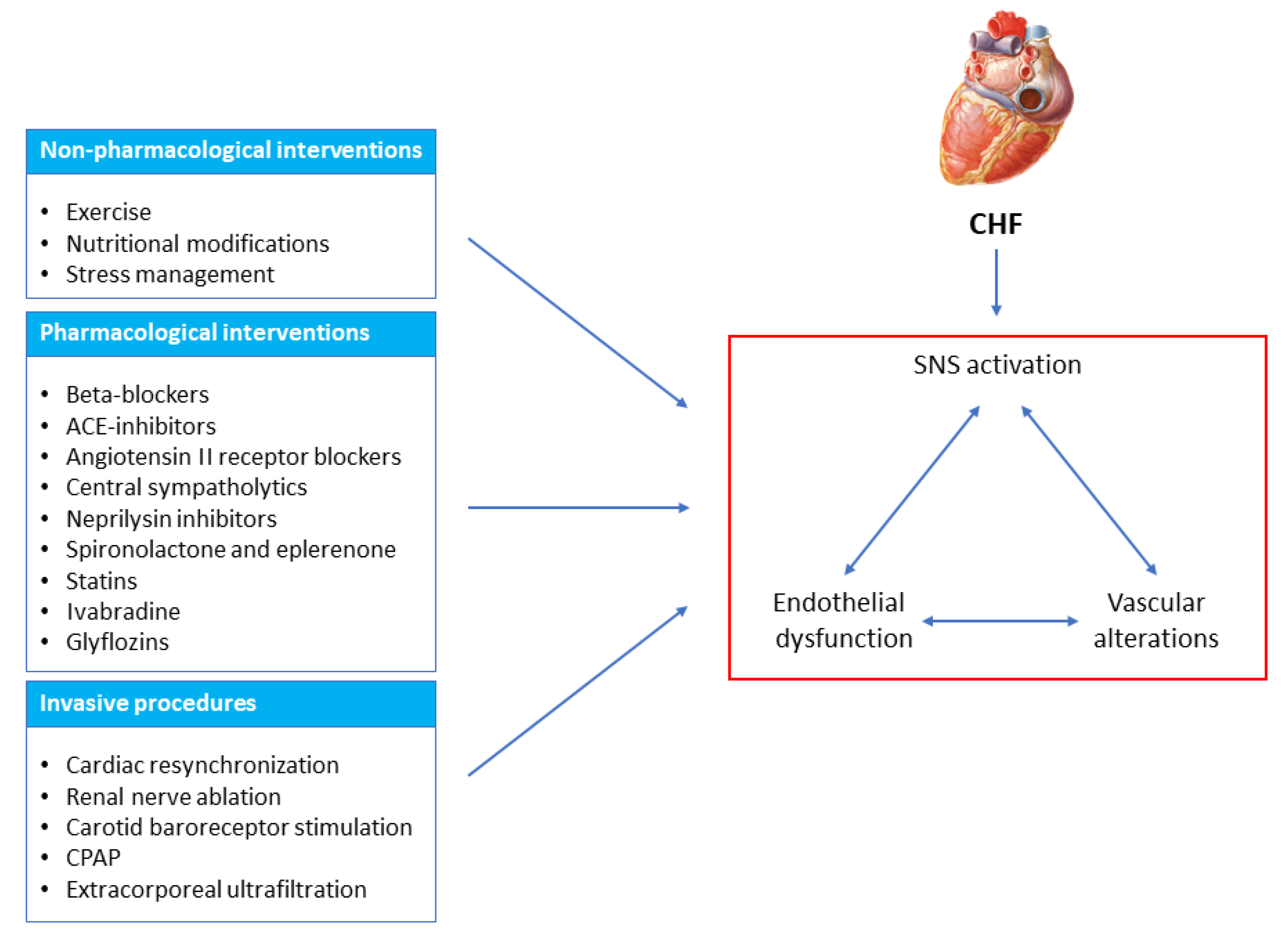
3. Non-Pharmacological Interventions
4. Pharmacological Interventions
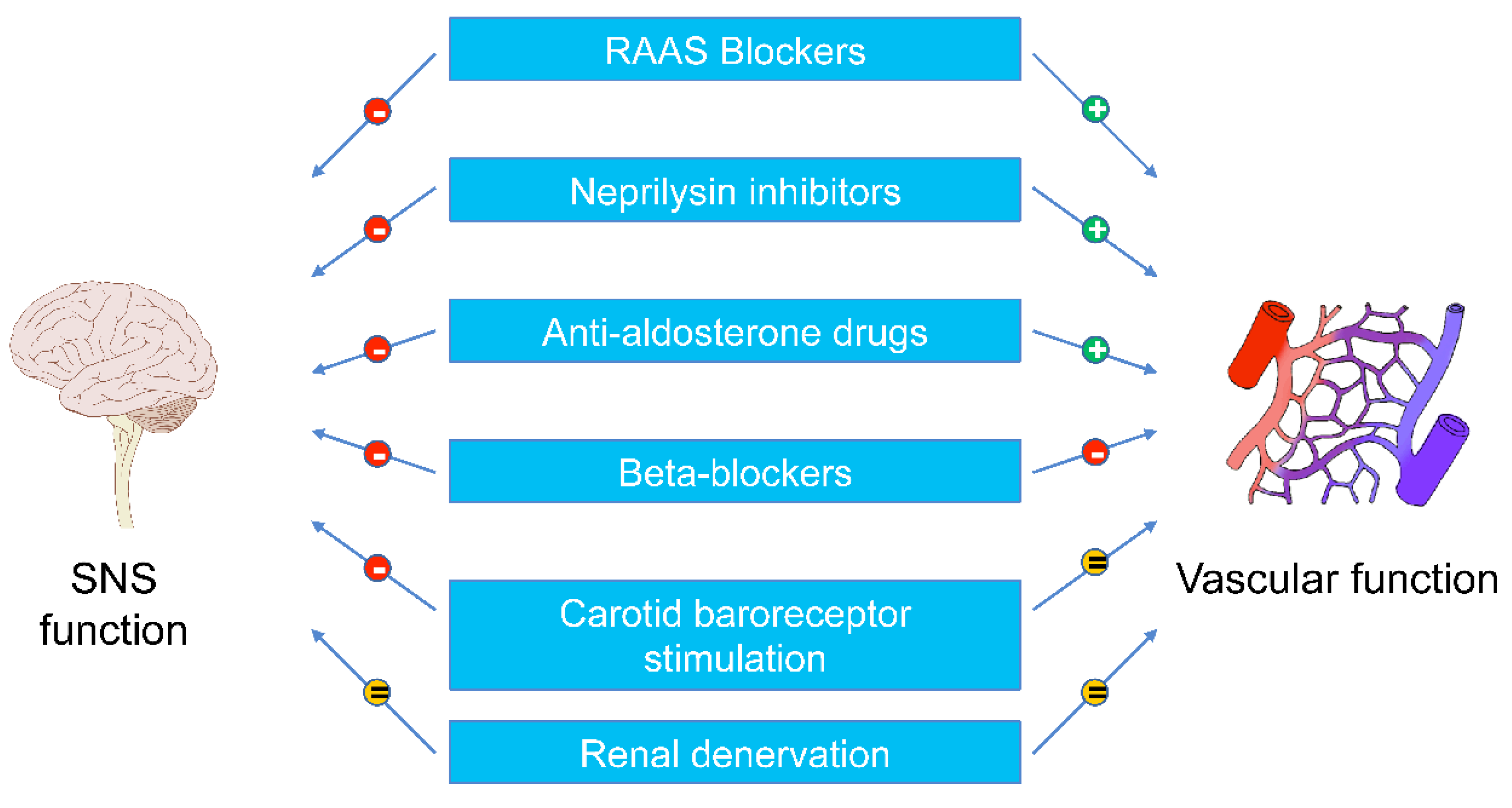
4.1. Beta-Blockers
4.2. Diuretic Drugs
4.3. ACE-Inhibitors and Angiotensin II Receptor Antagonists
4.4. Antialdosterone Drugs
4.5. Old and New Drugs
5. Device-Based Interventions
6. Unmet Goals of Therapeutic Interventions and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Weber, T.; Chirinos, J.A. Pulsatile arterial haemodynamics in heart failure. Eur. Heart J. 2018, 39, 3847–3854. [Google Scholar] [CrossRef] [PubMed]
- Chirinos, J.A.; Segers, P.; Hughes, T.; Townsend, R. Large-artery stiffness in health and disease: JACC state-of-the-art review. J. Am. Coll. Cardiol. 2019, 74, 1237–1263. [Google Scholar] [PubMed]
- Mancia, G.; Luscher, T.F.; Shepherd, J.T.; Noll, G.; Grassi, G. Cardiovascular regulation: Basic considerations. In Cardiovascular Medicine, 3rd ed.; Willerson, J.T., Cohn, J.N., Wellens, H.J.J., Holmes, D., Eds.; Springer: London, UK, 2007; pp. 1525–1540. [Google Scholar]
- Alexander, Y.; Osto, E.; Schmidt-Trucksäss, A.; Shechter, M.; Trifunovic, D.; Duncker, D.J.; Aboyans, V.; Back, M.; Badimon, L.; Cosentino, F.; et al. Endothelial function in cardiovascular medicine: A consensus paper of the European Society of Cardiology working groups on atherosclerosis and vascular biology, aorta and peripheral vascular diseases, coronary pathophysiology and microcirculation, and thrombosis. Cardiovasc. Res. 2021, 117, 29–42. [Google Scholar]
- Little, P.J.; Askew, C.D.; Xu, S.; Kamato, D. Endothelial dysfunction and cardiovascular disease: History and analysis of the clinical utility of the relationship. Biomedicines 2021, 9, 699. [Google Scholar] [CrossRef]
- Ambrosino, P.; D’Anna, S.E.; Grassi, G.; Maniscalco, M. Editorial. It all about endothelial dysfunction? Focusing on the alteration in endothelial integrity as a key determinant of different pathophysiological mechanisms. Biomedicines 2022, 10, 2757. [Google Scholar] [CrossRef] [PubMed]
- Grassi, G.; Mancia, G.; Esler, M. Central and peripheral sympathetic activation in heart failure. Cardiovasc. Res. 2022, 118, 1857–1871. [Google Scholar]
- Hasdai, D.; Lerman, A. The assessment of endothelial function in the cardiac catheterization laboratory in patients with risk factors for atherosclerotic coronary artery disease. Herz 1999, 24, 544–547. [Google Scholar] [CrossRef] [PubMed]
- Corretti, M.C.; Anderson, T.J.; Benjamin, E.J.; Celermajer, D.; Charbonneau, F.; Creager, M.A.; Deanfield, J.; Drexler, H.; Gerhard-Herman, M.; Herrington, D.; et al. Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: A report of the International Brachial Artery Reactivity Task Force. J. Am. Coll. Cardiol. 2002, 39, 257–265. [Google Scholar] [CrossRef]
- Kovacic, S.; Plazonic, Z.; Batinac, T.; Miletic, D.; Ruzic, A. Endothelial dysfunction as assessed with magnetic resonance imaging—A major determinant in chronic heart failure. Med. Hypotheses 2016, 90, 76–78. [Google Scholar]
- Wilkinson, I.B.; Webb, D.J. Venous occlusion plethysmography in cardiovascular research: Methodology and clinical applications. Br. J. Clin. Pharmacol. 2001, 52, 631–646. [Google Scholar]
- Raitakari, O.T.; Celermajer, D.S. Flow-mediated dilatation. Br. J. Clin. Pharmacol. 2000, 50, 397–404. [Google Scholar] [CrossRef]
- Kuvin, J.T.; Patel, A.R.; Sliney, K.A.; Pandian, N.G.; Sheffi, J.; Schnall, R.P.; Karas, R.H.; Udelson, J.E. Assessment of peripheral vascular endothelial function with finger arterial pulse wave amplitude. Am. Heart J. 2003, 146, 168–174. [Google Scholar] [CrossRef]
- Hornig, B.; Maier, V.; Drexler, H. Physical training improves endothelial function in patients with chronic heart failure. Circulation 1996, 93, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Hu, M.; Li, M. Relationship of arterial tonometry and exercise in patients with chronic heart failure: A systematic review with meta-analysis and trial sequential analysis. BMC Cardiovasc. Disord. 2022, 22, 345. [Google Scholar] [CrossRef] [PubMed]
- Van Bilsen, M.; Patel, H.C.; Bauersachs, J.; Bohm, M.; Borggrefe, M.; Brutsaert, D.; Coats, A.J.S.; De Boer, R.A.; De Keulenaer, G.W.; Filippatos, G.S.; et al. The autonomic nervous system as a therapeutic target in heart failure: A scientific position statement from the Translational Research Committee of the European Society of Cardiology. Eur. J. Heart Fail. 2017, 19, 1361–1378. [Google Scholar] [CrossRef]
- Pearson, M.J.; Smart, N.A. Exercise therapy and autonomic function in heart failure patients: A systematic review and meta-analysis. Heart Fail. Rev. 2018, 23, 91–108. [Google Scholar] [CrossRef]
- Carnevale, D. Neural control of immunity in hypertension. Hypertension 2020, 76, 622–628. [Google Scholar]
- Gielen, S.; Adams, V.; Möbius-Winkler, S.; Linke, A.; Erbs, S.; Yu, J.; Kempf, W.; Schubert, A.; Schuler, G.; Hambrecht, R. Anti-inflammatory effects of exercise training in the skeletal muscle of patients with chronic heart failure. J. Am. Coll. Cardiol. 2003, 42, 861–868. [Google Scholar]
- Theofilis, P.; Sagris, M.; Oikonomou, E.; Antonopoulos, A.S.; Siasos, G.; Tsioufis, C.; Tousoulis, D. Inflammatory mechanisms contributing to endothelial dysfunction. Biomedicines 2021, 9, 781. [Google Scholar] [CrossRef] [PubMed]
- Simpson, P.J.L.; Hoyos, C.M.; Celermajer, D.; Liu, P.Y.; Ng, M.K.C. Effects of continuous positive airway pressure on endothelial function and circulating progenitor cells in obstructive sleep apnoea: A randomized sham-controlled study. Int. J. Cardiol. 2013, 168, 2042–2048. [Google Scholar] [CrossRef]
- Kaneko, Y.; Floras, J.S.; Usui, K.; Plante, J.; Tkacova, R.; Kubo, T.; Ando, S.I.; Bradley, T.D. Cardiovascular effects of continuous positive airway pressure in patients with heart failure and obstructive sleep apnea. N. Engl. J. Med. 2003, 348, 1233–1241. [Google Scholar] [CrossRef] [PubMed]
- Kaye, D.M.; Mansfield, D.; Aggarwal, A.; Naughton, M.T.; Esler, M.D. Acute effects of continuous positive airway pressure on cardiac sympathetic tone in congestive heart failure. Circulation 2001, 103, 2336–2338. [Google Scholar] [CrossRef]
- Usui, K.; Bradley, T.D.; Spaak, J.; Ryan, C.M.; Kubo, T.; Kaneko, Y.; Floras, J.S. Inhibition of awake sympathetic nerve activity of heart failure patients with obstructive sleep apnea by nocturnal continuous positive airway pressure. J. Am. Coll. Cardiol. 2005, 45, 2008–2011. [Google Scholar] [CrossRef] [PubMed]
- Kazory, A.; Ross, E.A. Contemporary trends in the pharmacological and extra-corporeal management of heart failure: A nephrologic perspective. Circulation 2008, 117, 975–983. [Google Scholar] [CrossRef] [PubMed]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Bohm, M.; Burri, H.; Butler, J.; Celutkiene, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Do Vale, G.T.; Ceron, C.S.; Gonzaga, N.A.; Simplici, J.A.; Padovan, J.C. Three generation of B-Blockers: History, class difference and clinical applicability. Curr. Hypertens. Rev. 2019, 15, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Felker, G.M.; Ellison, D.H.; Mullens, W.; Cox, Z.L.; Testani, J.M. Diuretic therapy for patients with heart failure. JACC State of the Art Review. J. Am. Coll. Cardiol. 2020, 75, 1178–1195. [Google Scholar] [CrossRef]
- Giannattasio, C.; Achilli, F.; Grappiolo, A.; Failla, M.; Meles, E.; Gentile, G.; Calchera, I.; Capra, A.; Baglivo, J.; Vincenzi, A.; et al. Radial artery flow-mediated dilatation in heart failure patients. Effects of pharmacological and non-pharmacological treatment. Hypertension 2001, 38, 1451–1455. [Google Scholar] [CrossRef]
- Giannattasio, C.; Achilli, F.; Failla, M.; Capra, A.; Vincenzi, A.; Valagussa, F.; Mancia, G. Radial, carotid and aortic distensibility in congestive heart failure. Effects of high-dose angiotensin-converting enzyme inhibitor or low-dose association with angiotensin type 1 receptor blockade. J. Am. Coll. Cardiol. 2002, 39, 1275–1282. [Google Scholar] [CrossRef]
- Fortini, F.; Vieceli dalla Sega, F.; Marracino, L.; Severi, P.; Rapezzi, P.; Rizzo, P.; Ferrari, R. Well-known and novel players in endothelial dysfunction: Updates on a notch(ed) landscape. Biomedicines 2021, 9, 997. [Google Scholar] [CrossRef]
- Kasama, S.; Toyama, T.; Hatori, T.; Sumino, H.; Kumakura, H.; Takayama, Y.; Ichikawa, S.; Suzuki, T.; Kurabayashi, M. Comparative effects of valsartan and enalapril on cardiac sympathetic nerve activity and plasma brain natriuretic peptide in patients with congestive heart failure. Heart 2006, 92, 625–630. [Google Scholar] [CrossRef] [PubMed]
- Blanchet, M.; Sheppard, R.; Racine, N.; Ducharme, A.; Curnier, D.; Tardif, J.C.; Sirois, P.; Lamoureux, M.C.; De Champlain, J.; White, M. Effects of angiotensin-converting enzyme inhibitor plus irbesartan on maximal and submaximal exercise capacity and neurohumoral activation in patients with congestive heart failure. Am. Heart J. 2005, 149, 938. [Google Scholar] [CrossRef]
- Azevedo, E.R.; Mak, S.; Floras, J.S.; Parker, J.D. Acute effects of angiotensin-converting enzyme inhibition versus angiotensin II receptor blockade on cardiac sympathetic activity in patients with heart failure. Am. J. Physiol. 2017, 313, R410–R417. [Google Scholar] [CrossRef] [PubMed]
- Solomon, S.D.; McMurray, J.J.V.; Anand, I.S.; Ge, J.; Lam, C.S.P.; Maggioni, A.P.; Martinez, F.; Packer, M.; Pfeffer, M.; Pieske, B.; et al. Angiotensin–neprilysin inhibition in heart failure with preserved ejection fraction. N. Engl. J. Med. 2019, 381, 1609–1620. [Google Scholar] [CrossRef]
- Bunsawat, K.; Ratchford, S.M.; Alpenglow, J.K.; Stehlik, J.; Smith, A.S.; Richardson, R.S.; Wray, D.W. Sympathoinhibitory effect of sacubitril-valsartan in heart failure with reduced ejection fraction: A pilot study. Auton. Neurosci. 2021, 235, 102834. [Google Scholar] [CrossRef]
- Bunsawat, K.; Ratchford, S.M.; Alpenglow, J.K.; Park, S.H.; Jarrett, C.L.; Stehlik, J.; Smith, A.S.; Richardson, R.S.; Wray, D.W. Sacubitril-valsartan improves conduit vessel function and functional capacity and reduces inflammation in heart failure with reduced ejection fraction. J. Appl. Physiol. 2021, 13, 256–268. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, B.; Nair, A.P.; Misra, A.; Scott, C.Z.; Mahar, J.H.; Fedson, S. Neprilysin inhibitors in heart failure: The science, mechanism of action, clinical studies an unanswered questions. J. Am. Coll. Cardiol. Basic Trans. Sci. 2023, 8, 88–105. [Google Scholar]
- Macdonald, J.E.; Kennedy, N.; Struthers, A.D. Effects of spironolactone on endothelial function, vascular angiotensin converting enyme activity, and other prognostic markers in patients with mild heart failure already taking optimal treatment. Heart 2004, 90, 765–770. [Google Scholar] [CrossRef]
- Jung, C.; Florvaag, A.; Oberle, V.; Fritzenwanger, M.; Kretschmar, D.; Kuethe, F.; Betge, S.; Goebel, B.; Franz, M.; Barz, D.; et al. Positive effect of eplerenone treatment on endothelial progenitor cells in patients with chronic heart failure. J. Renin Ngiotensin Aldosterone Syst. 2012, 13, 401–406. [Google Scholar] [CrossRef]
- Kasama, S.; Toyama, T.; Sumino, H.; Matsumoto, N.; Sato, Y.; Kumakura, H.; Takayama, Y.; Ichikawa, S.; Suzuki, T.; Kurabayashi, M. Additive effects of spironolactone and candesartan on cardiac sympathetic nerve activity and left ventricular remodeling in patients with congestive heart failure. J. Nucl. Med. 2007, 48, 1993–2000. [Google Scholar] [CrossRef]
- Kasama, S.; Toyama, T.; Sumino, H.; Kumakura, H.; Takayama, Y.; Minami, K.; Ichikawa, S.; Matsumoto, N.; Sato, Y.; Kurabayashi, M. Effects of mineral corticoid receptor antagonist spironolactone on cardiac sympathetic nerve activity and prognosis in patients with chronic heart failure. Int. J. Cardiol. 2013, 167, 244–252. [Google Scholar] [CrossRef]
- Ferguson, D.W.; Berg, W.J.; Sanders, J.S.; Roach, P.; Kempf, J.S.; Kienzle, M.G. Sympathoinhibitory responses to digitalis glycosides in heart failure patients. Direct evidence from sympathetic neural recordings. Circulation 1989, 80, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Muhlen, B.V.; Millgard, J.; Lind, L. Effects of digoxin, furosemide, enalaprilat and metoprolol on endothelial function in young normotensive subjects. Clin. Exp. Pharmacol. Physiol. 2001, 28, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Cohn, J.N.; Pfeffer, M.A.; Rouleau, J.R.; Sharpe, N.; Swedberg, K.; Straub, M.; Wiltse, C.; Wright, T.J.; The MOXCON Investigators. Adverse mortality effect of central sympathetic inhibition with sustained-release moxonidine in patients with heart failure (MOXCON). Eur. Heart J. 2003, 5, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Strey, C.H.; Young, J.M.; Lainchbury, J.H.; Frampton, C.M.; Nicholls, M.G.; Richards, A.M.; Scott, R.S. Short-term statin treatment improves endothelial function and neurohumoral imbalance in normocholesterolaemic patients with non-ischaemic heart failure. Heart 2006, 92, 1603–1609. [Google Scholar] [CrossRef] [PubMed]
- Millar, P.J.; Floras, J. Statins and the autonomic nervous system. Clin. Sci. 2014, 126, 401–415. [Google Scholar] [CrossRef]
- Bocchi, E.A.; Salemi, V.M.C. Ivabradine for treatment of heart failure. Expert Opin. Drug Saf. 2019, 18, 393–402. [Google Scholar] [CrossRef]
- Bonadei, I.; Sciatti, E.; Vizzardi, E.; Fabbricatore, D.; Pagnoni, M.; Rossi, L.; Carubelli, V.; Lombardi, C.M.; Metra, M. Effects of ivabradine on endothelial function, aortic properties and ventricular-arterial coupling in chronic systolic heart failure patients. Cardiovasc. Ther. 2018, 36, e12323. [Google Scholar] [CrossRef]
- Bohm, M.; Anker, S.; Mafhoud, F.; Lauder, L.; Filippatos, G.; Ferreira, J.P.; Pocpck, S.J.; Brueckmann, M.; Saloustros, I.; Schuker, E.; et al. Empagliflozin, irrespective of blood pressure, improves outcomes in heart failure with preserved ejection fraction: The EMPEROR-preserved trial. Eur. Heart J. 2023, 44, 396–407. [Google Scholar] [CrossRef]
- Biegus, J.; Voors, A.A.; Collins, S.P.; Kosiborod, M.N.; Teerlink, J.R.; Angermann, C.E.; Tromp, J.; Ferreira, J.P.; Nassif, M.E.; Psotka, M.A.; et al. Impact of empagliflozin on decongestion in acute heart failure: The EMPULSE trial. Eur. Heart J. 2023, 44, 41–50. [Google Scholar] [CrossRef]
- Salvatore, T.; Caturano, A.; Galiero, R.; Di Martino, A.; Albanese, G.; Vetrano, E.; Sardu, C.; Marfella, R.; Rinaldi, L.; Sasso, F.C. Cardiovascular benefits fron gliflozins. Effects on endothelial function. Biomedicines 2021, 9, 1356. [Google Scholar] [CrossRef] [PubMed]
- Azzam, O.; Matthews, V.B.; Schlaich, M.P. Interaction between sodium-glucose co-transporter 2 and the sympathetic nervous sytem. Curr. Opin. Nephrol. Hypertens. 2022, 31, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Coksevim, M.; Akcay, M.; Yuksel, S.; Yenercag, M.; Cerik, B.; Gedikli, O.; Gulele, O.; Sahin, M. The effect of cardiac resynchronization therapy on arterial stiffness and central hemodynamic parameters. J. Arrhythm. 2020, 36, 498–507. [Google Scholar] [CrossRef] [PubMed]
- Zile, M.R.; Lindenfeld, J.; Weaver, F.A.; Zannad, F.; Galle, E.; Rogers, T.; Abraham, W.T. Baroreflex activation therapy in patients with heart failure with reduced ejection fraction. J. Am. Coll. Cardiol. 2020, 76, 1–13. [Google Scholar] [CrossRef]
- Gronda, E.; Seravalle, G.; Brambilla, G.; Costantino, G.; Casini, A.; Alsheraei, A.; Lovett, E.G.; Mancia, G.; Grassi, G. Chronic baroreflex activation effects on sympathetic nerve traffic, baroreflex function and cardiac haemodynamics in heart failure. A proof-of-concept study. Eur. J. Heart Fail. 2014, 16, 977–983. [Google Scholar] [CrossRef]
- Biffi, A.; Dell’Oro, R.; Quarti-Trevano, F.; Cuspidi, C.; Corrao, G.; Mancia, G.; Grassi, G. Effects of renal denervation on sympathetic nerve traffic and correlates in drug-resistant and uncontrolled hypertension: A systematic review and meta-analysis. Hypertension 2023, 80, 659–667. [Google Scholar] [CrossRef]
- Patel, H.C.; Hayward, C.; Keegan, J.; Gatehouse, P.D.; Rajani, R.; Khattar, R.S.; Mohiaddin, R.H.; Rosen, S.D.; Lyon, A.R.; Di Mario, C. Effects of renal denervation on vascular remodelling in patients with heart failure and preserved ejection fraction. A randomized control trial. JRSM Cardiovasc. Dis. 2017, 6, 2048004017690988. [Google Scholar]
- Greene, S.J.; Fonarow, G.C.; Butler, J. Risk profiles in heart failure: Baseline, residual, worsening, and advanced heart failure risk. Circ. Heart Fail. 2020, 13, e007132. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quarti-Trevano, F.; Dell’Oro, R.; Cuspidi, C.; Ambrosino, P.; Grassi, G. Endothelial, Vascular and Sympathetic Alterations as Therapeutic Targets in Chronic Heart Failure. Biomedicines 2023, 11, 803. https://doi.org/10.3390/biomedicines11030803
Quarti-Trevano F, Dell’Oro R, Cuspidi C, Ambrosino P, Grassi G. Endothelial, Vascular and Sympathetic Alterations as Therapeutic Targets in Chronic Heart Failure. Biomedicines. 2023; 11(3):803. https://doi.org/10.3390/biomedicines11030803
Chicago/Turabian StyleQuarti-Trevano, Fosca, Raffaella Dell’Oro, Cesare Cuspidi, Pasquale Ambrosino, and Guido Grassi. 2023. "Endothelial, Vascular and Sympathetic Alterations as Therapeutic Targets in Chronic Heart Failure" Biomedicines 11, no. 3: 803. https://doi.org/10.3390/biomedicines11030803
APA StyleQuarti-Trevano, F., Dell’Oro, R., Cuspidi, C., Ambrosino, P., & Grassi, G. (2023). Endothelial, Vascular and Sympathetic Alterations as Therapeutic Targets in Chronic Heart Failure. Biomedicines, 11(3), 803. https://doi.org/10.3390/biomedicines11030803







