Risk of Severe Alphaherpesvirus Infection after Solid Organ Transplantation: A Nationwide Population-Based Cohort Study
Abstract
1. Introduction
2. Methods
2.1. Data Source
2.2. Study Population
2.3. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Lamb, K.E.; Lodhi, S.; Meier-Kriesche, H.U. Long-term renal allograft survival in the United States: A critical reappraisal. Am. J. Transplant. 2011, 11, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Stegall, M.D.; Gaston, R.S.; Cosio, F.G.; Matas, A. Through a glass darkly: Seeking clarity in preventing late kidney transplant failure. J. Am. Soc. Nephrol. 2015, 26, 20–29. [Google Scholar] [CrossRef]
- Gerada, C.; Campbell, T.M.; Kennedy, J.J.; McSharry, B.P.; Steain, M.; Slobedman, B.; Abendroth, A. Manipulation of the Innate Immune Response by Varicella Zoster Virus. Front. Immunol. 2020, 11, 1. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.I. Herpes zoster. N. Engl. J. Med. 2013, 369, 1766–1767. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.L.; Hall, A.J. What does epidemiology tell us about risk factors for herpes zoster? Lancet Infect. Dis. 2004, 4, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.F. In the clinic. Herpes zoster. Ann. Intern. Med. 2011, 154, ITC31-15. [Google Scholar] [CrossRef]
- Yu, T.-M.; Sun, K.-T.; Kung, S.-C.; Lee, B.K.; Wu, M.-J.; Chen, C.-H.; Sun, C.-S.; Lo, Y.-C.; Lan, T.-H.; Li, C.-Y.; et al. Severe herpes simplex and varicella-zoster viral infection in patients with solid organ transplantation: A nationwide population-based cohort study with propensity score matching analysis. J. Am. Acad. Dermatol. 2021, 87, 472–474. [Google Scholar] [CrossRef]
- Wang, L.; Verschuuren, E.A.M.; van Leer-Buter, C.C.; Bakker, S.J.L.; de Joode, A.A.E.; Westra, J.; Bos, N.A. Herpes Zoster and Immunogenicity and Safety of Zoster Vaccines in Transplant Patients: A Narrative Review of the Literature. Front. Immunol. 2018, 9, 1632. [Google Scholar] [CrossRef]
- Miller, A.E. Selective decline in cellular immune response to varicella-zoster in the elderly. Neurology 1980, 30, 582–587. [Google Scholar] [CrossRef]
- Weinberg, A.; Levin, M.J. VZV T cell-mediated immunity. Curr. Top. Microbiol. Immunol. 2010, 342, 341–357. [Google Scholar]
- Weinberg, A.; Zhang, J.H.; Oxman, M.N.; Johnson, G.R.; Hayward, A.R.; Caulfield, M.J.; Irwin, M.R.; Clair, J.; Smith, J.G.; Stanley, H.; et al. Varicella-zoster virus-specific immune responses to herpes zoster in elderly participants in a trial of a clinically effective zoster vaccine. J. Infect. Dis. 2009, 200, 1068–1077. [Google Scholar] [CrossRef] [PubMed]
- McKay, S.L.; Guo, A.; Pergam, S.A.; Dooling, K. Herpes Zoster Risk in Immunocompromised Adults in the United States: A Systematic Review. Clin. Infect. Dis. 2020, 71, e125–e134. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Quiles, C.; López-Lacort, M.; Díez-Domingo, J.; Orrico-Sánchez, A. Herpes zoster risk and burden of disease in immunocompromised populations: A population-based study using health system integrated databases, 2009–2014. BMC Infect. Dis. 2020, 20, 905. [Google Scholar] [CrossRef] [PubMed]
- Veenstra, J.; Krol, A.; van Praag, R.M.; Frissen, P.H.; Schellekens, P.T.; Lange, J.M.; Coutinho, R.A.; van der Meer, J.T. Herpes zoster, immunological deterioration and disease progression in HIV-1 infection. AIDS 1995, 9, 1153–1158. [Google Scholar] [CrossRef]
- Koo, S.; Gagne, L.S.; Lee, P.; Pratibhu, P.P.; James, L.M.; Givertz, M.M.; Marty, F.M. Incidence and risk factors for herpes zoster following heart transplantation. Transpl. Infect. Dis. 2014, 16, 17–25. [Google Scholar] [CrossRef]
- Pergam, S.A.; Limaye, A.P.; Practice ASTIDCo. Varicella zoster virus in solid organ transplantation. Am. J. Transplant. 2013, 13 (Suppl. S4), 138–146. [Google Scholar] [CrossRef]
- Gourishankar, S.; McDermid, J.C.; Jhangri, G.S.; Preiksaitis, J.K. Herpes zoster infection following solid organ transplantation: Incidence, risk factors and outcomes in the current immunosuppressive era. Am. J. Transplant. 2004, 4, 108–115. [Google Scholar] [CrossRef]
- Pavlopoulou, I.D.; Poulopoulou, S.; Melexopoulou, C.; Papazaharia, I.; Zavos, G.; Boletis, I.N. Incidence and risk factors of herpes zoster among adult renal transplant recipients receiving universal antiviral prophylaxis. BMC Infect. Dis. 2015, 15, 285. [Google Scholar] [CrossRef]
- Harpaz, R.; Leung, J.W. The epidemiology of herpes zoster in the United States during the era of varicella and herpes zoster vaccines: Changing patterns among children. Clin. Infect. Dis. 2019, 69, 345–347. [Google Scholar] [CrossRef]
- Moodley, A.; Swanson, J.; Grose, C.; Bonthius, D.J. Severe herpes zoster following varicella vaccination in immunocompetent young children. J. Child Neurol. 2019, 34, 184–188. [Google Scholar] [CrossRef]
- Cheng, H.Y.; Chang, L.Y.; Lu, C.Y.; Huang, L.M. Epidemiology of Breakthrough Varicella after the Implementation of a Universal Varicella Vaccination Program in Taiwan, 2004–2014. Sci. Rep. 2018, 8, 17192. [Google Scholar] [CrossRef] [PubMed]
| Solid Organ Transplantation | Standardized Mean Differences § | |||
|---|---|---|---|---|
| Variable | Total | No | Yes | |
| (n = 18,064) | (n = 9032) | (n = 9032) | ||
| n | n (%)/Mean ± SD | n (%)/Mean ± SD | ||
| Gender | ||||
| Female | 6754 | 3390 (37.5) | 3364 (37.2) | 0.006 |
| Male | 11,310 | 5642 (62.5) | 5668 (62.8) | 0.006 |
| Age at baseline | ||||
| <50 | 8869 | 4372 (48.4) | 4497 (49.8) | 0.028 |
| 50–64 | 8155 | 4080 (45.2) | 4075 (45.1) | 0.001 |
| ≥65 | 1038 | 578 (6.4) | 460 (5.1) | 0.057 |
| Age, mean (SD) | 49.5 (11.7) | 48.7 (11.1) | 0.069 | |
| Comorbidity | ||||
| Hypertension | 9211 | 4646 (51.4) | 4565 (50.5) | 0.018 |
| Hyperlipidemia | 1892 | 968 (10.7) | 924 (10.2) | 0.016 |
| Diabetes | 3872 | 1972 (21.8) | 1900 (21) | 0.019 |
| Cancer | 2645 | 1360 (15.1) | 1285 (14.2) | 0.023 |
| Chronic kidney disease | 9964 | 4988 (55.2) | 4976 (55.1) | 0.003 |
| COPD | 402 | 203 (2.2) | 199 (2.2) | 0.003 |
| Heart failure | 2286 | 1122 (12.4) | 1164 (12.9) | 0.014 |
| Peripheral vascular disorders | 356 | 158 (1.7) | 198 (2.2) | 0.032 |
| Characteristics | Event | Crude | Adjusted | ||
|---|---|---|---|---|---|
| (n = 676) | HR (95% CI) | p Value | HR (95% CI) | p Value | |
| Solid Organ Transplantation | |||||
| No | 71 | Ref. | Ref. | ||
| Yes | 605 | 9.04 (7.07–11.56) | <0.001 | 9.19 (7.19–11.75) | <0.001 |
| Gender | |||||
| Female | 340 | Ref. | Ref. | ||
| Male | 336 | 0.65 (0.56–0.76) | <0.001 | 0.65 (0.56–0.76) | <0.001 |
| Age at baseline | |||||
| <50 | 290 | Ref. | Ref. | ||
| 50–64 | 352 | 1.60 (1.37–1.87) | <0.001 | 1.73 (1.47–2.03) | <0.001 |
| ≥65 | 34 | 1.33 (0.93–1.89) | 0.121 | 1.47 (1.02–2.12) | 0.038 |
| Baseline comorbidity | |||||
| Hypertension | 411 | 1.47 (1.26–1.71) | <0.001 | 1.18 (0.99–1.40) | 0.060 |
| Hyperlipidemia | 73 | 1.15 (0.90–1.47) | 0.251 | 0.96 (0.74–1.23) | 0.732 |
| Diabetes | 150 | 1.28 (1.07–1.54) | 0.007 | 1.12 (0.92–1.36) | 0.260 |
| Cancer | 54 | 0.67 (0.51–0.89) | 0.006 | 0.81 (0.60–1.08) | 0.153 |
| Chronic kidney disease | 467 | 1.51 (1.28–1.78) | <0.001 | 1.37 (1.13–1.66) | 0.001 |
| COPD | 18 | 1.49 (0.93–2.39) | 0.093 | 1.31 (0.81–2.12) | 0.267 |
| Heart failure | 102 | 1.38 (1.12–1.70) | 0.003 | 1.39 (1.12–1.74) | 0.003 |
| Peripheral vascular disorders | 21 | 1.85 (1.20–2.85) | 0.006 | 1.35 (0.87–2.09) | 0.183 |
| Variables | Non-SOT | SOT | SOT versus Non-SOT | |||||
|---|---|---|---|---|---|---|---|---|
| n = 9032 | n = 9032 | Crude HR | Adjusted HR | |||||
| Event | Person Years | IR | Event | Person Years | IR | (95% CI) | (95% CI) | |
| Kidney type | ||||||||
| <50 years | 29 | 28,545 | 1.02 | 212 | 21,036 | 10.08 | 10.20 (6.92–15.04) *** | 9.28 (6.23–13.83) *** |
| 50–64 | 33 | 20,342 | 1.62 | 203 | 10,563 | 19.22 | 12.56 (8.69–18.15) *** | 10.28 (6.97–15.18) *** |
| ≥65 | 9 | 2396 | 3.76 | 18 | 1066 | 16.89 | 4.66 (2.08–10.42) *** | 5.02 (2.02–12.47) *** |
| Heart type | ||||||||
| <50 years | 29 | 28,545 | 1.02 | 21 | 1710 | 12.28 | 11.91 (6.78–20.93) *** | 19.22 (6.67–55.41) *** |
| 50–64 | 33 | 20,342 | 1.62 | 39 | 1438 | 27.12 | 16.70 (10.50–26.55) *** | 19.08 (7.97–45.67) *** |
| ≥65 | 9 | 2396 | 3.76 | 3 | 203 | 14.81 | 3.92 (1.06–14.53) * | 2.50 (0.45–13.85) |
| Liver type | ||||||||
| <50 years | 29 | 28,545 | 1.02 | 24 | 4866 | 4.93 | 4.66 (2.70–8.04) *** | 9.44 (4.16–21.42) *** |
| 50–64 | 33 | 20,342 | 1.62 | 74 | 6453 | 11.47 | 6.55 (4.33–9.90) *** | 8.45 (5.13–13.90) *** |
| ≥65 | 9 | 2396 | 3.76 | 4 | 690 | 5.80 | 1.58 (0.48–5.17) | 2.37 (0.57–9.81) |
| Variables | Non-SOT | SOT | SOT versus Non-SOT | |||||
|---|---|---|---|---|---|---|---|---|
| n = 9032 | n = 9032 | Crude HR | Adjusted HR | |||||
| Event | Person Years | IR | Event | Person Years | IR | (95% CI) | (95% CI) | |
| Follow-up Year | ||||||||
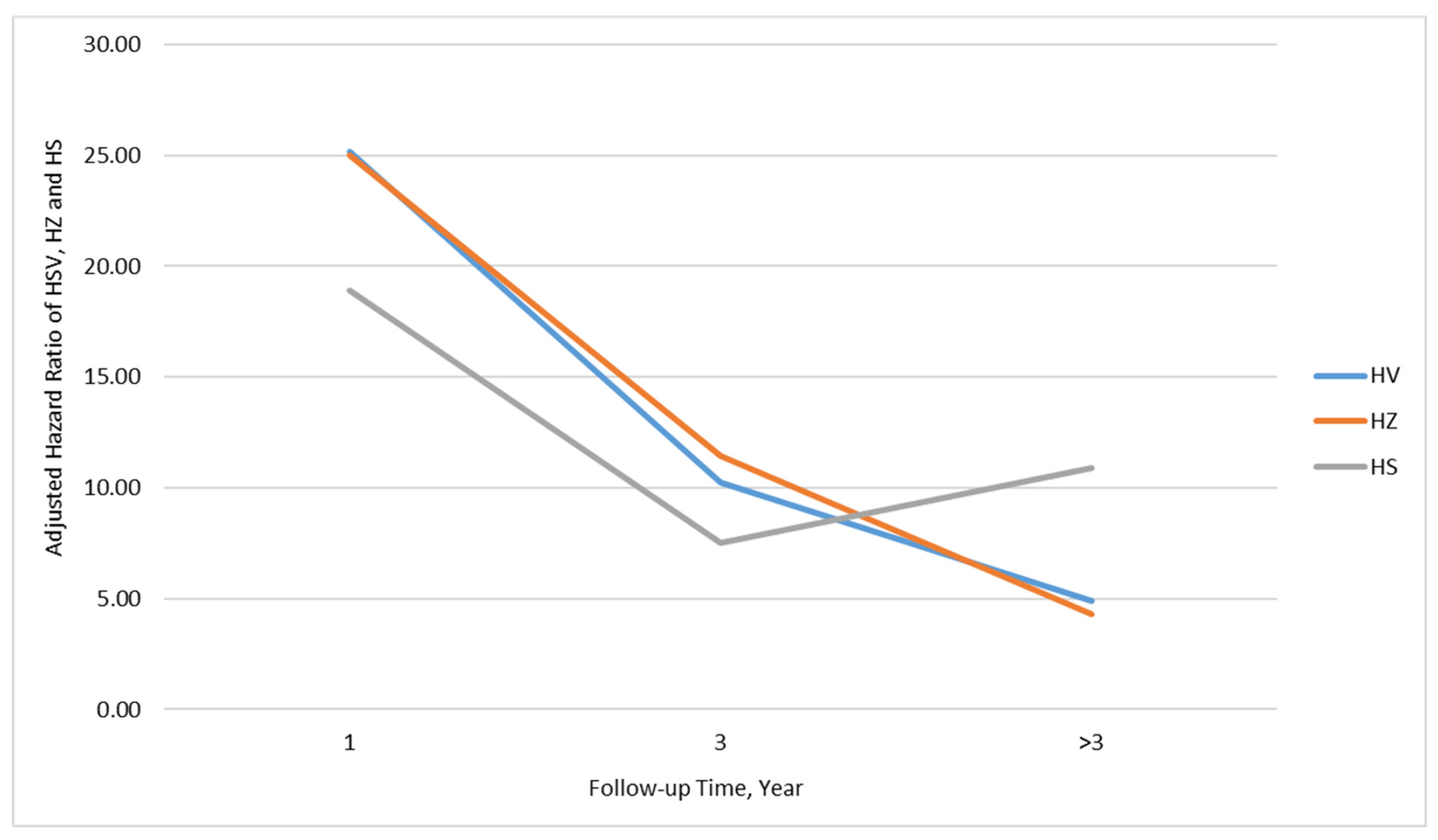
| <1 | 10 | 8825 | 1.13 | 235 | 8403 | 27.97 | 24.53 (13.03–46.20) *** | 25.18 (13.36–47.43) *** |
| 1–3 | 19 | 14,969 | 1.27 | 177 | 13,905 | 12.73 | 10.03 (6.25–16.09) *** | 10.22 (6.36–16.41) *** |
| >3 | 42 | 27,489 | 1.53 | 193 | 25,881 | 7.46 | 4.89 (3.51–6.83) *** | 4.89 (3.50–6.83) *** |
| Variable | Solid Organ Transplantation | p-Value | |
|---|---|---|---|
| No | Yes | ||
| aHV Recurrence (n = 101) | |||
| Crude HR (95% CI) | 1.00 (Ref.) | 1.78 (0.90–3.54) | 0.10 |
| Adjusted HR † (95% CI) | 1.00 (Ref.) | 1.75 (0.87–3.52) | 0.12 |
| 1-year aHV Recurrence (n = 83) | |||
| Crude HR (95% CI) | 1.00 (Ref.) | 0.76 (0.40–1.43) | 0.39 |
| Adjusted HR † (95% CI) | 1.00 (Ref.) | 0.68 (0.35–1.33) | 0.26 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chuang, Y.-W.; Huang, S.-T.; Wang, I.-K.; Lo, Y.-C.; Chang, C.-T.; Lin, C.-L.; Yu, T.-M.; Li, C.-Y. Risk of Severe Alphaherpesvirus Infection after Solid Organ Transplantation: A Nationwide Population-Based Cohort Study. Biomedicines 2023, 11, 637. https://doi.org/10.3390/biomedicines11020637
Chuang Y-W, Huang S-T, Wang I-K, Lo Y-C, Chang C-T, Lin C-L, Yu T-M, Li C-Y. Risk of Severe Alphaherpesvirus Infection after Solid Organ Transplantation: A Nationwide Population-Based Cohort Study. Biomedicines. 2023; 11(2):637. https://doi.org/10.3390/biomedicines11020637
Chicago/Turabian StyleChuang, Ya-Wen, Shih-Ting Huang, I-Kuan Wang, Ying-Chih Lo, Chiz-Tzung Chang, Cheng-Li Lin, Tung-Min Yu, and Chi-Yuan Li. 2023. "Risk of Severe Alphaherpesvirus Infection after Solid Organ Transplantation: A Nationwide Population-Based Cohort Study" Biomedicines 11, no. 2: 637. https://doi.org/10.3390/biomedicines11020637
APA StyleChuang, Y.-W., Huang, S.-T., Wang, I.-K., Lo, Y.-C., Chang, C.-T., Lin, C.-L., Yu, T.-M., & Li, C.-Y. (2023). Risk of Severe Alphaherpesvirus Infection after Solid Organ Transplantation: A Nationwide Population-Based Cohort Study. Biomedicines, 11(2), 637. https://doi.org/10.3390/biomedicines11020637






