The Influence of SARS-CoV-2 Infection on the Thyroid Gland
Abstract
:1. Introduction
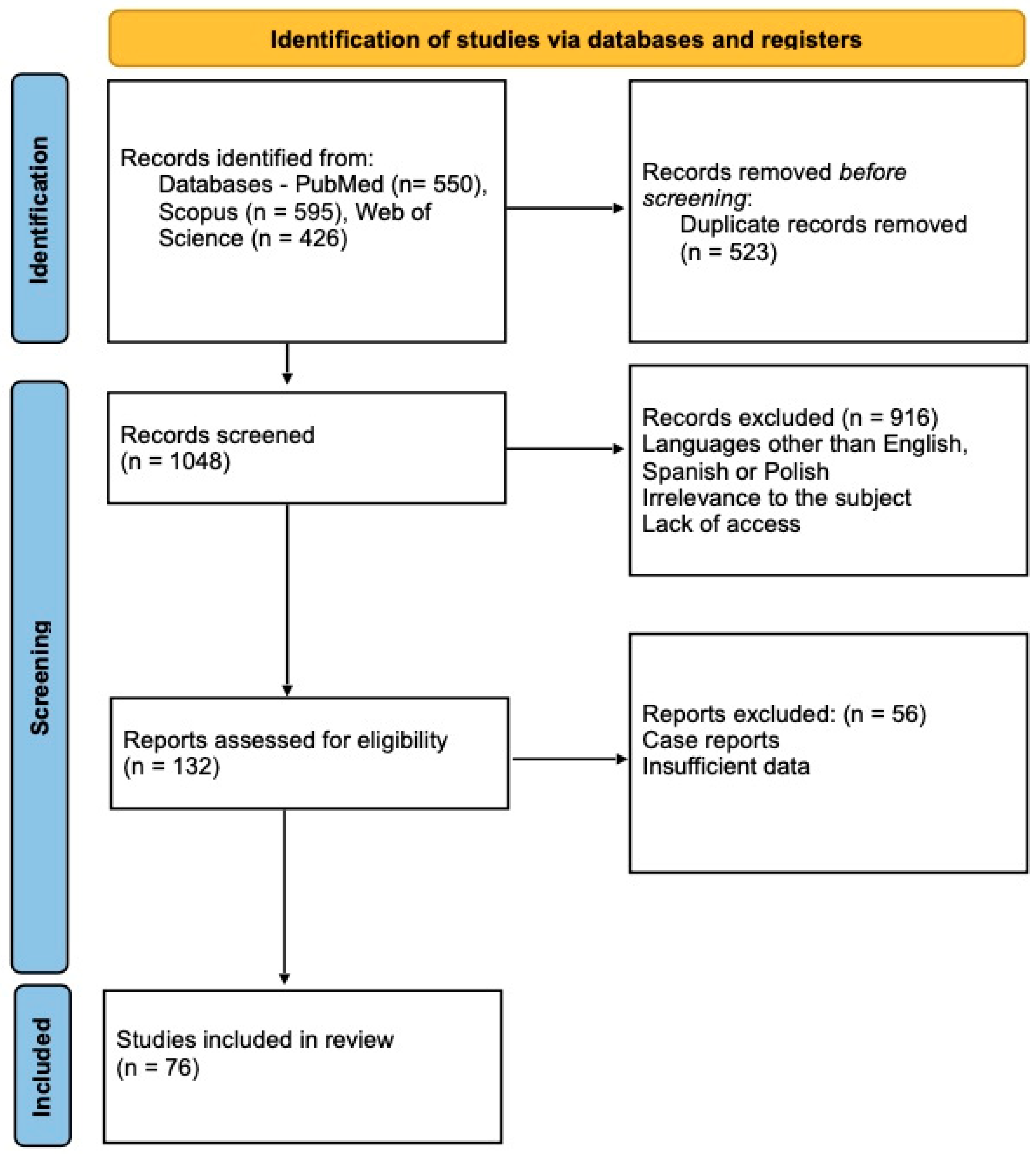
2. Study Selection
3. Pathophysiology
4. ACE-2 and TMPRSS2
5. Cytokine Storm
6. Hypothyroidism
7. Hyperthyroidism
8. Autoimmune Thyroid Diseases
9. Subacute Thyroiditis (SAT)
10. Thyroid Hormonal Function and COVID-19 Severity
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Agarwal, S.; Agarwal, S.K. Endocrine changes in SARS-CoV-2 patients and lessons from SARS-CoV. J. Postgrad. Med. 2020, 96, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Pramono, L.A. COVID-19 and Thyroid Diseases: How the Pandemic Situation Affects Thyroid Disease Patients? J. ASEAN Fed. Endocr. Soc. 2020, 35, 155–157. [Google Scholar] [CrossRef] [PubMed]
- WHO—Coronavirus Disease (COVID-19) Pandemic. Available online: https://covid19.who.int/table (accessed on 20 July 2022).
- Jadali, Z. COVID-19 and thyroid infection: Learning the lessons of the past. Acta Endocrinol. 2020, 16, 375–376. [Google Scholar] [CrossRef]
- Llamas, M.; Garo, M.L.; Giovanella, L. Low free-T3 serum levels and prognosis of COVID-19: Systematic review and meta-analysis. Clin. Chem. Lab. Med. 2021, 59, 1906–1913. [Google Scholar] [CrossRef] [PubMed]
- Șandru, F.; Carsote, M.; Petca, R.C.; Gheorghisan-Galateanu, A.A.; Petca, A.; Valea, A.; Dumitrașcu, M.C. COVID-19-related thyroid conditions (Review). Exp. Ther. Med. 2021, 22, 756. [Google Scholar] [CrossRef]
- Sparano, C.; Zago, E.; Morettini, A.; Nozzoli, C.; Yannas, D.; Adornato, V.; Caldini, E.; Vaudo, M.; Maggi, M.; Petrone, L. Euthyroid sick syndrome as an early surrogate marker of poor outcome in mild SARS-CoV-2 disease. J. Endocrinol. Investig. 2022, 45, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Speer, G.; Somogyi, P. Thyroid complications of SARS and coronavirus disease 2019 (COVID-19). J. Endocrinol. 2021, 68, 129–136. [Google Scholar] [CrossRef]
- Giovanella, L.; Ruggeri, R.M.; Petranović Ovčariček, P.; Campenni, A.; Treglia, G.; Deandreis, D. SARS-CoV-2-related thyroid disorders: A synopsis for nuclear medicine thyroidologists. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 1719–1723. [Google Scholar] [CrossRef]
- Inaba, H.; Aizawa, T. Coronavirus Disease 2019 and the Thyroid—Progress and Perspectives. Front. Endocrinol. 2021, 12, 708333. [Google Scholar] [CrossRef]
- Murugan, A.K.; Alzahrani, A.S. SARS-CoV-2: Emerging Role in the Pathogenesis of Various Thyroid Diseases. J. Inflamm. Res. 2021, 14, 6191–6221. [Google Scholar] [CrossRef]
- Beltrão, F.E.L.; Beltrão, D.C.A.; Carvalhal, G.; Beltrão, F.E.L.; Brito, A.D.S.; Capistrano, K.H.R.D.; Bastos, I.H.A.; Hecht, F.; Daltro, C.H.D.C.; Bianco, A.C.; et al. Thyroid Hormone Levels During Hospital Admission Inform Disease Severity and Mortality in COVID-19 Patients. Thyroid 2021, 31, 1639–1649. [Google Scholar] [CrossRef] [PubMed]
- Caron, P. Thyroiditis and SARS-CoV-2 pandemic: A review. Endocrine 2021, 72, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Garg, M.K.; Gopalakrishnan, M.; Yadav, P.; Misra, S. Endocrine involvement in COVID-19: Mechanisms, clinical features, and implications for care. Indian J. Endocrinol. Metab. 2020, 24, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Lui, D.T.W.; Fung, M.M.H.; Chiu, K.W.H.; Lee, C.H.; Chow, W.S.; Lee, A.C.H.; Tam, A.R.; Pang, P.; Ho, T.Y.; Fong, C.H.Y.; et al. Higher SARS-CoV-2 viral loads correlated with smaller thyroid volumes on ultrasound among male COVID-19 survivors. Endocrine 2021, 74, 205–214. [Google Scholar] [CrossRef]
- Xu, J.; Li, Y.; Xia, Q.; Shi, Q. Association between Thyroid Disease and Severe Coronavirus Disease 2019 (COVID-19) Infection: A Meta-Analysis. Iran. J. Public Health 2021, 50, 1517–1525. [Google Scholar] [CrossRef]
- Kazakou, P.; Paschou, S.A.; Psaltopoulou, T.; Gavriatopoulou, M.; Korompoki, E.; Stefanaki, K.; Kanouta, F.; Kassi, G.N.; Dimopoulos, M.A.; Mitrakou, A. Early and late endocrine complications of COVID-19. Endocr. Connect. 2021, 10, R229–R239. [Google Scholar] [CrossRef]
- Rotondi, M.; Coperchini, F.; Ricci, G.; Denegri, M.; Croce, L.; Ngnitejeu, S.T.; Villani, L.; Magri, F.; Latrofa, F.; Chiovato, L. Detection of SARS-COV-2 receptor ACE-2 mRNA in thyroid cells: A clue for COVID-19-related subacute thyroiditis. J. Endocrinol. Investig. 2021, 44, 1085–1090. [Google Scholar] [CrossRef]
- D’Ardes, D.; Rossi, I.; Bucciarelli, B.; Allegra, M.; Bianco, F.; Sinjari, B.; Marchioni, M.; Di Nicola, M.; Santilli, F.; Guagnano, M.T.; et al. Metabolic Changes in SARS-CoV-2 Infection: Clinical Data and Molecular Hypothesis to Explain Alterations of Lipid Profile and Thyroid Function Observed in COVID-19 Patients. Life 2021, 11, 860. [Google Scholar] [CrossRef]
- Geslot, A.; Chanson, P.; Caron, P. Covid-19, the thyroid and the pituitary—The real state of play. Ann. Endocrinol. 2022, 83, 103–108. [Google Scholar] [CrossRef]
- Piticchio, T.; Le Moli, R.; Tumino, D.; Frasca, F. Relationship between betacoronaviruses and the endocrine system: A new key to understand the COVID-19 pandemic-A comprehensive review. J. Endocrinol. Investig. 2021, 44, 1553–1570. [Google Scholar] [CrossRef]
- Croce, L.; Gangemi, D.; Ancona, G.; Liboà, F.; Bendotti, G.; Minelli, L.; Chiovato, L. The cytokine storm and thyroid hormone changes in COVID-19. J. Endocrinol. Investig. 2021, 44, 891–904. [Google Scholar] [CrossRef] [PubMed]
- Bahçecioğlu, A.B.; Karahan, Z.C.; Aydoğan, B.I.; Kalkan, I.A.; Azap, A.; Erdoğan, M.F. Subacute thyroiditis during the COVID-19 pandemic: A prospective study. J. Endocrinol. Investig. 2022, 45, 865–874. [Google Scholar] [CrossRef]
- Kumari, K.; Chainy, G.B.N.; Subudhi, U. Prospective role of thyroid disorders in monitoring COVID-19 pandemic. Heliyon 2020, 6, e05712. [Google Scholar] [CrossRef] [PubMed]
- Koya, S.F.; Ebrahim, S.H.; Bhat, L.D.; Vijayan, B.; Khan, S.; Jose, S.D.; Pilakkadavath, Z.; Rajeev, P.; Azariah, J.L. COVID-19 and Comorbidities: Audit of 2,000 COVID-19 Deaths in India. J. Epidemiol. Glob. Health 2021, 11, 230–232. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.N.; Silveira, L.F.G.; Guimarães, M.M.M.; Polanczyk, C.A.; Nunes, A.G.S.; Costa, A.S.M.; Farace, B.L.; Cimini, C.C.R.; Carvalho, C.A.; Ponce, D.; et al. Hypothyroidism does not lead to worse prognosis in COVID-19: Findings from the Brazilian COVID-19 registry. Int. J. Infect. Dis. 2022, 116, 319–327. [Google Scholar] [CrossRef] [PubMed]
- van Gerwen, M.; Alsen, M.; Little, C.; Barlow, J.; Naymagon, L.; Tremblay, D.; Sinclair, C.F.; Genden, E. Outcomes of Patients With Hypothyroidism and COVID-19: A Retrospective Cohort Study. Front. Endocrinol. 2020, 11, 565. [Google Scholar] [CrossRef] [PubMed]
- Bogojevic, M.; Bansal, V.; Pattan, V.; Singh, R.; Tekin, A.; Sharma, M.; La Nou, A.T.; LeMahieu, A.M.; Hanson, A.C.; Schulte, P.J.; et al. Association of hypothyroidism with outcomes in hospitalized adults with COVID-19: Results from the International SCCM Discovery Viral Infection and Respiratory Illness Universal Study (VIRUS): COVID-19 Registry. Clin. Endocrinol. 2022. [Google Scholar] [CrossRef]
- Nguyen, C.; Yale, K.; Ghigi, A.; Zheng, K.; Mesinkovska, N.A.; Wambier, C.G.; Cadegiani, F.A.; Goren, A. SARS-CoV-2 infection in patients with thyroid disease: A cross-sectional study. Ann. Thyroid 2021, 6, 7. [Google Scholar] [CrossRef]
- Pizzocaro, A.; Colombo, P.; Vena, W.; Ariano, S.; Magnoni, P.; Reggiani, F.; Favacchio, G.; Mirani, M.; Lavezzi, E.; Voza, A.; et al. Humanitas COVID-19 Task force. Outcome of Sars-COV-2-related thyrotoxicosis in survivors of Covid-19: A prospective study. Endocrine 2021, 73, 255–260. [Google Scholar] [CrossRef]
- Nakamura, S.; Kido, N.; Watanabe, M.; Ohmachi, Y.; Inayama, Y.; Kashitani, Y.; Ishii, K.; Ishida, I.; Goda, N.; Fujita, Y.; et al. Analysis of thyroid function in Japanese patients with coronavirus disease 2019. Endocr. J. 2022, 69, 643–648. [Google Scholar] [CrossRef]
- Murugan, A.K.; Alzahrani, A.S. SARS-CoV-2 plays a pivotal role in inducing hyperthyroidism of Graves’ disease. Endocrine 2021, 73, 243–254. [Google Scholar] [CrossRef]
- Clarke, S.; Abbara, A.; Dhillo, W.S. A Impact of COVID-19 on the Endocrine System: A Mini-review. Endocrinology 2022, 163, bqab203. [Google Scholar] [CrossRef] [PubMed]
- Lui, D.T.W.; Lee, C.H.; Chow, W.S.; Lee, A.C.H.; Tam, A.R.; Fong, C.H.Y.; Law, C.Y.; Leung, E.K.H.; To, K.K.W.; Tan, K.C.B.; et al. Insights from a Prospective Follow-up of Thyroid Function and Autoimmunity among COVID-19 Survivors. Endocrinol. Metab. 2021, 36, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Lui, D.T.W.; Lee, C.H.; Chow, W.S.; Lee, A.C.H.; Tam, A.R.; Fong, C.H.Y.; Law, C.Y.; Leung, E.K.H.; To, K.K.W.; Tan, K.C.B.; et al. Thyroid Dysfunction in Relation to Immune Profile, Disease Status, and Outcome in 191 Patients with COVID-19. J. Clin. Endocrinol. Metab. 2021, 106, e926–e935. [Google Scholar] [CrossRef] [PubMed]
- Santacroce, G.; Lenti, M.V.; Aronico, N.; Miceli, E.; Lovati, E.; Lucotti, P.C.; Coppola, L.; Gentile, A.; Latorre, M.A.; Di Terlizzi, F.; et al. Impact of COVID-19 in immunosuppressive drug-naïve autoimmune disorders: Autoimmune gastritis, celiac disease, type 1 diabetes, and autoimmune thyroid disease. Pediatr. Allergy Immunol. 2022, 33, 105–107. [Google Scholar] [CrossRef]
- Aemaz Ur Rehman, M.; Farooq, H.; Ali, M.M.; Ebaad Ur Rehman, M.; Dar, Q.A.; Hussain, A. The association of subacute thyroiditis with COVID-19: A systematic review. SN Compr. Clin. Med. 2021, 3, 1515–1527. [Google Scholar] [CrossRef]
- Brancatella, A.; Ricci, D.; Viola, N.; Sgrò, D.; Santini, F.; Latrofa, F. Subacute Thyroiditis After Sars-COV-2 Infection. J. Clin. Endocrinol. Metab. 2020, 105, 2367–2370. [Google Scholar] [CrossRef]
- Caron, P. Thyroid disorders and SARS-CoV-2 infection: From pathophysiological mechanism to patient management. Ann. Endocrinol. 2020, 81, 507–510. [Google Scholar] [CrossRef]
- Sariakçali, B.; Duman, G. Subacute thyroid and SARS-CoV-2 relationship. Pak. J. Med. Health Sci. 2021, 15, 2438–2441. [Google Scholar] [CrossRef]
- Abreu, R.; Miguel, R.; Saieg, M. Subacute (De Quervain) thyroiditis during the COVID-19 pandemic. Cancer Cytopathol. 2021, 129, 844–846. [Google Scholar] [CrossRef]
- Christensen, J.; O’Callaghan, K.; Sinclair, H.; Hawke, K.; Love, A.; Hajkowicz, K.; Stewart, A.G. Risk factors, treatment and outcomes of subacute thyroiditis secondary to COVID-19: A systematic review. Intern. Med. J. 2022, 52, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Sohrabpour, S.; Heidari, F.; Karimi, E.; Ansari, R.; Tajdini, A.; Heidari, F. Subacute Thyroiditis in COVID-19 Patients. Eur. Thyroid J. 2021, 9, 321–323. [Google Scholar] [CrossRef] [PubMed]
- Trimboli, P.; Cappelli, C.; Croce, L.; Scappaticcio, L.; Chiovato, L.; Rotondi, M. COVID-19-Associated Subacute Thyroiditis: Evidence-Based Data From a Systematic Review. Front. Endocrinol. 2021, 12, 707726. [Google Scholar] [CrossRef] [PubMed]
- Pirola, I.; Gandossi, E.; Rotondi, M.; Marini, F.; Cristiano, A.; Chiovato, L.; Castellano, M.; Ferlin, A.; Cappelli, C. Incidence of De Quervain’s thyroiditis during the COVID-19 pandemic in an area heavily affected by Sars-CoV-2 infection. Endocrine 2021, 74, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Campi, I.; Bulgarelli, I.; Dubini, A.; Perego, G.B.; Tortorici, E.; Torlasco, C.; Torresani, E.; Rocco, L.; Persani, L.; Fugazzola, L. The spectrum of thyroid function tests during hospitalization for SARS COV-2 infection. Eur. J. Endocrinol. 2021, 184, 699–709. [Google Scholar] [CrossRef]
- Brancatella, A.; Viola, N.; Rutigliano, G.; Sgrò, D.; Santini, F.; Latrofa, F. Subacute Thyroiditis During the SARS-CoV-2 Pandemic. J. Endocr. Soc. 2021, 5, bvab130. [Google Scholar] [CrossRef]
- Seyed Resuli, A.; Bezgal, M. Subacute Thyroiditis in COVID-19 Patients. Ear Nose Throat J. 2022, 101, 501–505. [Google Scholar] [CrossRef]
- Stasiak, M.; Lewiński, A. New aspects in the pathogenesis and management of subacute thyroiditis. Rev. Endocr. Metab. Disord. 2021, 22, 1027–1039. [Google Scholar] [CrossRef]
- Chen, W.; Tian, Y.; Li, Z.; Zhu, J.; Wei, T.; Lei, J. Potential Interaction Between SARS-CoV-2 and Thyroid: A Review. Endocrinology 2021, 162, bqab004. [Google Scholar] [CrossRef]
- Ballesteros Vizoso, M.A.; Castilla, A.F.; Barceló, A.; Raurich, J.M.; Argente del Castillo, P.; Morell-García, D.; Velasco, J.; Pérez-Bárcena, J.; Llompart-Pou, J.A. Thyroid Disfunction in Critically Ill COVID-19 Patients. Relationship with In-Hospital Mortality. J. Clin. Med. 2021, 10, 5057. [Google Scholar] [CrossRef]
- Schwarz, Y.; Percik, R.; Oberman, B.; Yaffe, D.; Zimlichman, E.; Tirosh, A. Sick Euthyroid Syndrome on Presentation of Patients With COVID-19: A Potential Marker for Disease Severity. Endocr. Pract. 2021, 27, 101–109. [Google Scholar] [CrossRef]
- Güven, M.; Gültekin, H. The prognostic impact of thyroid disorders on the clinical severity of COVID-19: Results of single-centre pandemic hospital. Int. J. Clin. Pract. 2021, 75, 14129. [Google Scholar] [CrossRef]
- Dutta, A.; Jevalikar, G.; Sharma, R.; Farooqui, K.J.; Mahendru, S.; Dewan, A.; Bhudiraja, S.; Mithal, A. Low FT3 is an independent marker of disease severity in patients hospitalized for COVID-19. Endocr. Connect. 2021, 10, 1455–1462. [Google Scholar] [CrossRef] [PubMed]
- Ayan, D.; Balcı, T.; Ünal, S.; Ulucan, H.; Türkyürek, C.; Bayram, E. Evaluation of the fT3/fT4 ratio and prevalence of Euthyroid Sick Syndrome among patients with COVID-19: A cross-sectional study. Ann. Clin. Anal. Med. 2021, 12, 1343–1347. [Google Scholar] [CrossRef]
- Ahn, J.; Lee, M.; Lee, J.; Sohn, S. Thyroid Hormone Profile and Its Prognostic Impact on the Coronavirus Disease 2019 in Korean Patients. Endocrinol. Metab. 2021, 36, 769–777. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, X.; Dai, Y.; Zhang, J. The Association between COVID-19 and Thyroxine Levels: A Meta-Analysis. Front. Endocrinol. 2022, 12, 779692. [Google Scholar] [CrossRef] [PubMed]
- Zou, R.; Wu, C.; Zhang, S.; Wang, G.; Zhang, Q.; Yu, B.; Wu, Y.; Dong, H.; Wu, G.; Wu, S.; et al. Euthyroid Sick Syndrome in Patients With COVID-19. Front. Endocrinol. 2020, 11, 566439. [Google Scholar] [CrossRef]
- Lang, S.; Liu, Y.; Qu, X.; Lu, R.; Fu, W.; Zhang, W.; Wang, H.; Hong, T. Association between Thyroid Function and Prognosis of COVID-19: A Retrospective Observational Study. Endocr. Res. 2021, 46, 170–177. [Google Scholar] [CrossRef]
- Gao, W.; Guo, W.; Guo, Y.; Shi, M.; Dong, G.; Wang, G.; Ge, Q.; Zhu, J.; Zhou, X. Thyroid hormone concentrations in severely or critically ill patients with COVID-19. J. Endocrinol. Investig. 2021, 44, 1031–1040. [Google Scholar] [CrossRef]
- Dincer Yazan, C.; Ilgin, C.; Elbasan, O.; Apaydin, T.; Dashdamirova, S.; Yigit, T.; Sili, U.; Karahasan Yagci, A.; Sirikci, O.; Haklar, G.; et al. The Association of Thyroid Hormone Changes with Inflammatory Status and Prognosis in COVID-19. Int. J. Endocrinol. 2021, 2021, 2395212. [Google Scholar] [CrossRef]
- Sciacchitano, S.; Giovagnoli, S.; D’Ascanio, M.; Dominicis, C.; Laghi, A.; Anibaldi, P.; Vitis, C.; Petrucca, A.; Amodeo, R.; Simmaco, M.; et al. Low FT3 Values During the Acute Phase of the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection Correlate to the Severity Indexes of the Disease. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Gong, J.; Wang, D.K.; Dong, H.; Xia, Q.S.; Huang, Z.Y.; Zhao, Y.; Chen, X.; Yuan, F.; Li, J.B.; Lu, F.E. Prognostic significance of low TSH concentration in patients with COVID-19 presenting with non-thyroidal illness syndrome. BMC Endocr. Disord. 2021, 21, 111. [Google Scholar] [CrossRef] [PubMed]
- Baldelli, R.; Nicastri, E.; Petrosillo, N.; Marchioni, L.; Gubbiotti, A.; Sperduti, I.; Di Giacinto, P.; Rizza, L.; Rota, F.; Franco, M.; et al. Thyroid dysfunction in COVID-19 patients. J. Endocrinol. Investig. 2021, 44, 2735–2739. [Google Scholar] [CrossRef]
- Wang, W.; Su, X.; Ding, Y.; Fan, W.; Zhou, W.; Su, J.; Chen, Z.; Zhao, H.; Xu, K.; Ni, Q.; et al. Thyroid Function Abnormalities in COVID-19 Patients. Front. Endocrinol. 2021, 11, 1158. [Google Scholar] [CrossRef] [PubMed]
- Asghar, M.S.; Yasmin, F.; Dapke, K.; Phadke, R.; Shah, S.M.I.; Bin Zafar, M.D. Derangements of biochemical markers and thyroid function analysis among COVID-19-positive patients: A developing country single-center experience. J. Med. Virol. 2021, 93, 5712–5717. [Google Scholar] [CrossRef]
- Lania, A.; Sandri, M.T.; Cellini, M.; Mirani, M.; Lavezzi, E.; Mazziotti, G. Thyrotoxicosis in patients with COVID-19: The THYRCOV study. Eur. J. Endocrinol. 2020, 183, 381–387. [Google Scholar] [CrossRef]
- Clausen, C.L.; Rasmussen, Å.K.; Johannsen, T.H.; Hilsted, L.M.; Skakkebæk, N.E.; Szecsi, P.B.; Pedersen, L.; Benfield, T.; Juul, A. Thyroid function in COVID-19 and the association with cytokine levels and mortality. Endocr. Connect. 2021, 10, 1234–1242. [Google Scholar] [CrossRef]
- Malik, J.; Malik, A.; Javaid, M.; Zahid, T.; Ishaq, U.; Shoaib, M. Thyroid function analysis in COVID-19: A retrospective study from a single center. PLoS ONE 2021, 16, e0249421. [Google Scholar] [CrossRef]
- Das, L.; Dutta, P.; Walia, R.; Mukherjee, S.; Suri, V.; Puri, G.D.; Mahajan, V.; Malhotra, P.; Chaudhary, S.; Gupta, R.; et al. Spectrum of Endocrine Dysfunctionand Association With Disease Severityin Patients With COVID-19: InsightsFrom a Cross-Sectional, Observational Study. Front. Endocrinol. 2021, 12, 645787. [Google Scholar] [CrossRef]
- Sen, K.; Sinha, A.; Sen, S.; Chakraborty, S.; Alam, M.S. Thyroid function test in COVID-19 patients: A cross-sectional study in a tertiary care hospital. Indian J. Endocrinol. Metab. 2020, 24, 532–536. [Google Scholar] [CrossRef]
- Okwor, C.J.; Meka, I.A.; Akinwande, K.S.; Edem, V.F.; Okwor, V.C. Assessment of thyroid function of newly diagnosed SARS-CoV-2 infected patients in Nigeria. Pan Afr. Med. J. 2021, 40, 9. [Google Scholar] [CrossRef]
- Zhang, Y.; Lin, F.; Tu, W.; Zhang, J.; Choudhry, A.A.; Ahmed, O.; Cheng, J.; Cui, Y.; Liu, B.; Dai, M.; et al. Thyroid dysfunction may be associated with poor outcomes in patients with COVID-19. Mol. Cell. Endocrinol. 2021, 521, 111097. [Google Scholar] [CrossRef]
- Czarnywojtek, A.; Ochmańska, A.; Zgorzalewicz-Stachowiak, M.; Sawicka-Gutaj, N.; Matyjaszek-Matuszek, B.; Woźniak, M.; Ruchała, M. Influence of SARS-CoV-2 infection on thyroid gland function: The current knowledge. Adv. Clin. Exp. Med. 2021, 30, 747–755. [Google Scholar] [CrossRef]
- Lisco, G.; De Tullio, A.; Jirillo, E.; Giagulli, V.A.; De Pergola, G.; Guastamacchia, E.; Triggiani, V. Thyroid and COVID-19: A review on pathophysiological, clinical and organizational aspects. J. Endocrinol. Investig. 2021, 44, 1801–1814. [Google Scholar] [CrossRef]
- Dworakowska, D.; Morley, S.; Mulholland, N.; Grossman, A.B. COVID-19-related thyroiditis: A novel disease entity? Clin. Endocrinol. 2021, 95, 369–377. [Google Scholar] [CrossRef]
- Scappaticcio, L.; Pitoia, F.; Esposito, K.; Piccardo, A.; Trimboli, P. Impact of COVID-19 on the thyroid gland: An update. Rev. Endocr. Metab. Disord. 2021, 22, 803–815. [Google Scholar] [CrossRef]
| Reference Number | Author | Levels of Hormones | Hormone Levels Indicating a More Severe Course of COVID-19 | COVID-19 Positive Cases | Country | ||
|---|---|---|---|---|---|---|---|
| TSH | fT3 | fT4 | |||||
| [5] | Llamas et al. | N | ↓ | N | ↓fT3 | 1183 | Switzerland |
| [7] | Sparano et al. | N | ↓ | N | ↓fT3 | 506 | Italy |
| [12] | Beltrao et al. | N | ↓ | N | ↓fT3 | 245 | Brazil |
| [19] | D’Ardes et al. | ↓ | N | N | ↓TSH | 118 | Italy |
| [31] | Nakamura et al. | ↓ | ↓ | N | ↓fT3, TSH | 147 | Japan |
| [33] | Clarke et al. | N | ↓ | N | ↓fT3 | - | United Kingdom |
| [35] | Lui et al. | N | ↓ | N | ↓fT3 | 191 | China |
| [46] | Campi et al. | N | ↓ | N | ↓fT3 | 144 | Italy |
| [51] | Ballesteros Vizoso et al. | ↓ | ↓ | ↓ | fT4 at the fifth day after ICU admission is an important mortality risk factor | 78 | Spain |
| [52] | Schwarz et al. | N | ↓ | N | ↓fT3 | 54 | Israel |
| [53] | Guven et al. | N | ↓/N | ↓/N | ↓fT3 | 250 | Turkey |
| [54] | Dutta et al. | N | ↓/N | N | ↓fT3, TSH | 236 | India |
| [55] | Ayan et al. | N | N | N | ↓fT3, TSH | 114 | Turkey |
| [56] | Ahn et al. | ↓ | ↓ | N | ↓fT3 | 119 | South Korea |
| [57] | Chen et al. | ↓/N | ↓/N | ↓/N | ↓fT3, fT4, TSH | 3609 | China |
| [58] | Zou et al. | N | N | ↓ | ↓fT4 | 149 | China |
| [59] | Lang et al. | ↓ | ↓ | N | ↓fT3 | 127 | China |
| [60] | Gao et al. | ↓ | ↓ | ↓ | ↓fT3 | 100 | China |
| [61] | Dincer et al. | ↓ | ↓ | ↑ | ↓TSH | 205 | Turkey |
| [62] | Sciacchitano et al. | ↓ | ↓ | ↓/N | ↓fT3 | 62 | Italy |
| [63] | Gong et al. | ↓/N | ↓ | ↓/N | ↓TSH | 150 | China |
| [64] | Baldelli et al. | ↓ | ↓ | ↓/N | ↓TSH | 46 | Italy |
| [65] | Wang et al. | ↓ | ↓ | - | ↓TSH | 84 | China |
| [66] | Asghar et al. | ↓ | N | N | ↓TSH | 54 | Pakistan |
| [67] | Lania et al. | ↓/N/↑ | N | N/↑ | ↓TSH | 287 | Italy |
| [68] | Clausen et al. | ↓/N/↑ | - | N/↑ | no correlation between mortality and TSH and fT4 levels | 116 | Denmark |
| [69] | Malik et al. | ↑ | ↓ | N | unrelated | 48 | Pakistan |
| [70] | Das et al. | ↓/N | ↓ | ↓/N/↑ | unrelated | 84 | India |
| [71] | Sen et al. | ↓/N/↑ | ↓/N/↑ | ↓/N/↑ | unrelated | 60 | India |
| [72] | Okwor et al. | ↑ | ↑ | N | no information | 45 | Nigeria |
| [73] | Zhang et al. | ↓/N/↑ | ↓/N/↑ | ↓/N/↑ | pre-existing thyroid disease | 71 | China |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piekarska, A.; Góral, M.; Kozula, M.; Jawiarczyk-Przybyłowska, A.; Zawadzka, K.; Bolanowski, M. The Influence of SARS-CoV-2 Infection on the Thyroid Gland. Biomedicines 2023, 11, 614. https://doi.org/10.3390/biomedicines11020614
Piekarska A, Góral M, Kozula M, Jawiarczyk-Przybyłowska A, Zawadzka K, Bolanowski M. The Influence of SARS-CoV-2 Infection on the Thyroid Gland. Biomedicines. 2023; 11(2):614. https://doi.org/10.3390/biomedicines11020614
Chicago/Turabian StylePiekarska, Aleksandra, Marta Góral, Marta Kozula, Aleksandra Jawiarczyk-Przybyłowska, Katarzyna Zawadzka, and Marek Bolanowski. 2023. "The Influence of SARS-CoV-2 Infection on the Thyroid Gland" Biomedicines 11, no. 2: 614. https://doi.org/10.3390/biomedicines11020614
APA StylePiekarska, A., Góral, M., Kozula, M., Jawiarczyk-Przybyłowska, A., Zawadzka, K., & Bolanowski, M. (2023). The Influence of SARS-CoV-2 Infection on the Thyroid Gland. Biomedicines, 11(2), 614. https://doi.org/10.3390/biomedicines11020614






