Effects of Interleukin-1 Genotype on the Clinical Efficacy of Non-Surgical Periodontal Treatment of Polish Patients with Periodontitis
Abstract
1. Introduction
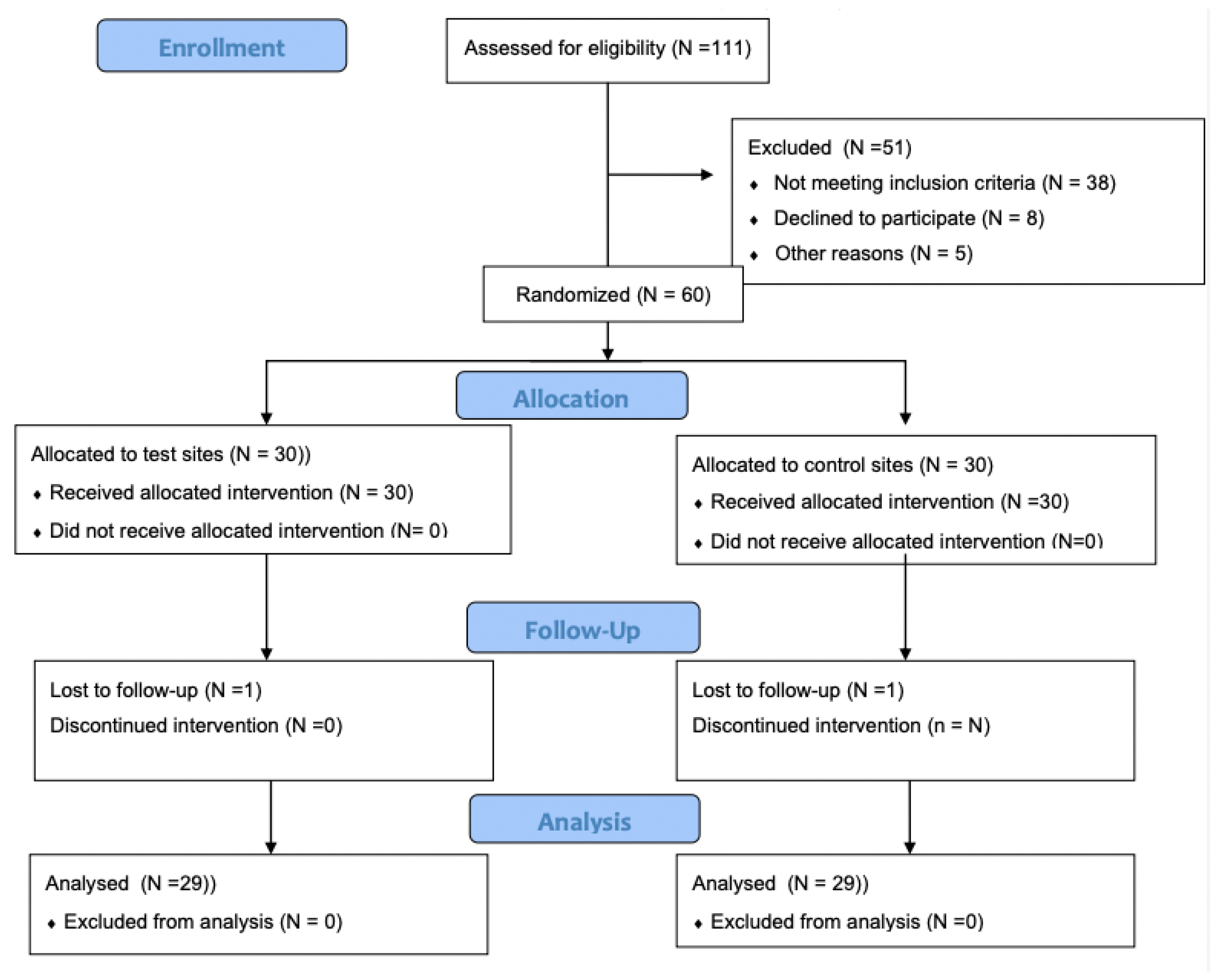
2. Materials and Methods
2.1. Inclusion Criteria
2.2. Exclusion Criteria
2.3. Clinical Examination
2.4. Genetic Examination
2.5. Periodontal Treatment
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liu, X.; Li, H. A systematic review and meta-analysis on multiple cytokine gene polymorphisms in the pathogenesis of periodontitis. Front. Immunol. 2022, 12, 713198. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, F.R.P.; Pessoa, L.D.S.; Shin, J., II; Alves, E.H.P.; Koga, R.S.; Smith, C.V.; Vasconcelos, D.F.P.V.; De Cuhna Pereira, A.C.T. Polymorphisms in the interleukin genes and chronic periodontitis: A field synopsis and revaluation by Bayesian approaches. Cytokine 2021, 138, 155361. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Liu, J. Association between IL1A (-889C/T) polymorphism and susceptibility of chronic periodontitis: A meta-analysis. Gene 2020, 729, 144227. [Google Scholar] [CrossRef] [PubMed]
- Fiorillo, L.; Cervino, G.; Herford, A.S.; Lauritano, F.; D’Amoico, C.; Giudice, R.L.; Laino, L.; Troiano, G.; Crimi, S.; Cicciu, M. Interferon crevicular fluid profile and correlation with periodontal disease and wound healing: A systemioc review of recernt data. Int. J. Mol. Sci. 2018, 19, 1908. [Google Scholar] [CrossRef]
- Kornman, K.S.; Crane, A.; Wang, H.Y.; di Giovine, F.S.; Newman, M.G.; Pirk, F.W.; Wilson, T.G., Jr.; Higginbottom, F.L.; Duff, G.W. The interleukin–1 genotype as a severity gfactor in adult periodontal disease. J. Clin. Periodontol. 1997, 24, 72–77. [Google Scholar] [CrossRef]
- Ebersole, J.L., 3rd; Dawson, D.; Emecen-Huja, P.; Nagarajan, R.; Howard, K.; Grady, M.E.; Thompson, K.; Peyyala, R.; Al-Attar, A.; Lethbridge, K.; et al. The periodontal war: Microbes and immunity. Periodontology 2000 2017, 75, 52–115. [Google Scholar] [CrossRef]
- Shaddox, L.M.; Morford, L.A.; Nibali, L. Periodontal health and disease: The contribution of genetics. Periodontology 2000 2021, 85, 161–181. [Google Scholar] [CrossRef]
- Martu, M.A.; Maftei, G.A.; Sufaru, I.G.; Jelihovscgi, I.; Luchian, I.; Hurjui, L.; Martu, I.; Pasarin, L. COVID-19 and periodontal disease—Ethiopathogenic and clinical implications. Rom. J. Oral Rehabil. 2020, 12, 116–124. [Google Scholar]
- Martu, M.A.; Solomon, S.M.; Toma, V.; Maftei, G.A.; Iovan, A.; Gamen, A.; Hurjui, L.; Rezus, E.; Foia, L.; Forna, N.C. The importance of cytokines in periodontal disease and rheumatoid arthritis. Review. Rom. J. Oral Rehabil. 2019, 11, 230–240. [Google Scholar]
- Citterio, F.; Romano, F.; Ferrarotti, F.; Gualini, G.; Aimetti, M. Quality of methods and reporting in association studies of chronic periodontitis and IL1A -889 and IL1B +3953/4 SNPs: A systematic review. J. Periodontal. Res. 2019, 54, 457–467. [Google Scholar] [CrossRef]
- O’Leary, T.J.; Drake, R.B.; Naylor, J.E. The plaque control record. J. Periodontol. 1972, 43, 38–46. [Google Scholar] [CrossRef]
- Ainamo, J.; Bay, I. Problems and proposal for recording gingivitis and plaque. Int. Dent. J. 1975, 25, 229–235. [Google Scholar]
- Tonetti, M.S.; Greenwell, H.; Kornman, K.S. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J. Clin. Periodontol. 2018, 45 (Suppl. 20), 149–161. [Google Scholar] [CrossRef]
- Cheng, R.; Wu, Z.; Li, M.; Shao, M.; Hu, T. Interleukin-1ß is a potential therapeutic target for periodontitis: A narrative review. Int. J. Oral Sci. 2020, 12, 2. [Google Scholar] [CrossRef]
- Monje, A.; Kramp, A.R.; Criado, E.; Suarez-Lopez Del Amo, F.; Garaicoa-Pazmino, C.; Gargallo-Albiol, J.; Wang, H.L. Effect of periodontal dressing on non-surgical periodontal treatment outcomes: A systemic review. Int. J. Dent. Hyg. 2016, 14, 161–167. [Google Scholar] [CrossRef]
- Cobb, C.M.; Sottosanti, J.S. A re-evaluation of scaling and root planning. J. Periodontol. 2021, 92, 1370–1378. [Google Scholar] [CrossRef]
- Yan, Y.; Zhan, Y.; Wang, X.; Hou, J. Clinical evaluation of ultrasonic subgingival debridement versus ultrasonic subgingival scaling combined with manual root planning in the treatment of periodontitis: Study protocol for randomized controlled trial. Trials 2020, 21, 113. [Google Scholar] [CrossRef]
- Smiley, C.J.; Tracy, S.L.; Abt, E.; Michalowicz, B.S.; John, M.T.; Gunsolley, J.; Cobb, C.M.; Rossmann, J.; Harrel, S.K.; Forrest, J.L.; et al. Systematic review and meta-analysis on the nonsurgical treatment of chronic periodontitis by means of scaling and root planning with or without adjuncts. J. Am. Dent. Assoc. 2015, 146, 508–524. [Google Scholar] [CrossRef]
- Albonni, H.; El Abdelah, A.A.A.D.; Al Hamwi, M.O.M.S.; Al Hamoui, W.B.; Sawaf, H. Clinical effectiveness of a topical subgingival application of injectable platelet-rich fibrin as adjunctive therapy to scaling and root planning: A double-blind, split-mouth, randomized, prospective, comparative controlled trial. Quintessence Int. 2021, 52, 676–685. [Google Scholar]
- Meisel, P.; Siegemund, A.; Grimm, R.; Herrmann, F.H.; John, U.; Schwann, C.; Kocher, T. The interleukin-1 polymorphism, smoking, and the risk of periodontal disease in the population based SHIP study. J. Dent. Res. 2003, 82, 189–193. [Google Scholar] [CrossRef]
- De Sanctis, M.; Zucchelli, G. Interleukin-1 gene polymorphism and long-term stability following guided tissue regeneration therapy. J. Periodontol. 2000, 71, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Darby, I.B.; Hodge, P.J.; Riggio, M.P.; Kinane, D.F. Clinical and microbiological effect of scaling and root planning in smoker and non-smoker chronic and aggressive periodontitis patients. J. Clin. Periodontol. 2005, 32, 200–206. [Google Scholar] [CrossRef]
- Povsic, K.; Cuk, K.; Milavec, S.; Erculj, V.; Seme, K.; Gaspersic, R. Systemic azithromycin as an adjunct to scaling and root planning in patients with stage III/IV periodontitis: 12 month results of a randomized controlled clinical trial. Clin. Oral Investig. 2021, 25, 5997–6006. [Google Scholar] [CrossRef]
- Walther, K.A.; Gonzales, J.R.; Gröger, S.; Ehmke, B.; Kaner, D.; Lorenz, K.; Eickholz, P.; Kocher, T.; Kim, T.S.; Schlagenhauf, U.; et al. The Role of Polymorphisms at the Interleukin-1, Interleukin-4, GATA-3 and Cyclooxygenase-2 Genes in Non-Surgical Periodontal Therapy. Int. J. Mol. Sci. 2022, 23, 7266. [Google Scholar] [CrossRef]
- Pozo, P.; Valenzuela, M.A.; Melej, C.; Zaldivar, M.; Puente, J.; Martinez, B.; Gamonal, J. Longitudinal analysis of metalloproteinases, tissue inhibitors of metalloproteinases and clinical parameters in gingival crevicular fluid from periodontitis-affected patients. J. Periodontal Res. 2005, 40, 199–207. [Google Scholar] [CrossRef]
- Jepsen, S.; Eberhard, J.; Fricke, D.; Hedderich, J.; Siebert, R.; Acil, Y. Interleukin-1 gene polymorphisms and experimental gingivitis. J. Clin. Periodontol. 2003, 30, 102–107. [Google Scholar] [CrossRef]
- Lang, N.P.; Tonnetti, M.S.; Suter, J.; Sorrell, J.; Duff, G.W.; Kornman, K.S. Effect of interleukin-1 gene polymorphisms on gingival inflammation assessed by bleeding on probing in a periodontal maintenance population. J. Periodontal Res. 2000, 35, 102–107. [Google Scholar] [CrossRef]
- Nile, C.J.; Apatizidou, D.A.; Awang, R.A.; Riggo, M.P.; Kinane, D.F.; Lappin, D.F. The effect of periodontal scaling and root polishing on serum IL-17E concentrations and the IL-17A:IL-17E ratio. Clin. Oral Investig. 2016, 20, 2529–2537. [Google Scholar] [CrossRef]
- Cullinam, M.P.; Westerman, B.; Hamlet, S.M.; Palmer, J.E.; Faddy, M.J.; Lang, N.P.; Seymour, G.J. A longitudinal study of Interleukine-1 gene polymorphisms and periodontal disease in a general adult population. J. Clin. Periodontol. 2001, 28, 1137–1144. [Google Scholar] [CrossRef]
- Caffesse, R.G.; De La Rosa, R.M.; De La Rosa, G.M. Effect of interleukin-1 gene polymorphism in a periodontally-healthy Hispanic population treated with mucogingival surgery. J. Clin. Periodontol. 2002, 29, 177–181. [Google Scholar] [CrossRef]
- Caffesse, R.G.; De La Rosa, R.M.; De La Rosa, G.M. Interleukin-1 gene polymorphism in a well-maintained periodontal patient population. Braz. J. Oral Sci. 2002, 1, 1–6. [Google Scholar]
- Christgau, M.; Aslanidis, C.; Felden, A.; Hiller, K.A.; Shmitz, G.; Schmalz, G. Influence of interleukin-1 gene polymorphism on periodontal regeneration in intrabony defects. J. Periodontal Res. 2003, 38, 20–27. [Google Scholar] [CrossRef]
| Variable | IL+ | IL− | p |
|---|---|---|---|
| Age, mean ± SD (years) | 4.3 ± 5.3 | 56.1 ± 4.2 | 0.432 |
| Sex, F/M n (%) | 20/10 (66.6/33/3) | 22/8 (73.3/26.6) | 0.453 |
| Number of teeth, mean ± SD | 21.56 ± 1.62 | 22.53 ± 1.30 | 0.456 |
| FMPI, mean ± SD (%) | 83.71 ± 10.76 | 86.91 ± 10.91 | <0.001 |
| FMBOP, mean ± SD (%) | 52.78 ± 21.6 | 50.95 ± 28.09 | <0.001 |
| PPD, mean ± SD (mm) | 3.55 ± 0.73 | 3.47 ± 1.12 | <0.001 |
| CAL, mean ± SD (mm) | 9.85 ± 1.81 | 10.66 ± 2.13 | <0.001 |
| Stage I, n (%) | 0 (0) | 0 (0) | >0.05 |
| Stage II, n (%) | 10 (33.3) | 9 (30) | >0.05 |
| Stage III, n (%) | 12 (40) | 14 (46.6) | >0.05 |
| Stage IV, n (%) | 8 (26.6) | 7 (23.3) | >0.05 |
| Variable | T1 | T2 | T3 | P T1-T2 | P T1-T3 | P T2-T3 |
|---|---|---|---|---|---|---|
| FMPI IL+ (%) | 83.71 ± 10.76 | 56.61 ± 17.33 | 56.16 ± 17.17 | p < 0.05 | p < 0.05 | p > 0.05 |
| FMPI IL− (%) | 86.91 ± 10.91 | 49.11 ± 21.36 | 45.77 ± 18.79 | p < 0.05 | p < 0.05 | p > 0.05 |
| p | p > 0.05 | p > 0.05 | p > 0.05 | |||
| FMBoP IL+ (%) | 52.78 ± 21.6 | 21.79 ± 13.89 | 22.53 ± 13.06 | p < 0.05 | p < 0.05 | p > 0.05 |
| FMBoP IL− (%) | 50.95 ± 26.09 | 17.1 ± 15.0 | 16.69 ± 13.51 | p < 0.05 | p < 0.05 | p > 0.05 |
| p | p > 0.05 | p > 0.05 | p > 0.05 | |||
| PPD IL+ (mm) | 3.55 ± 0.73 | 2.7 ± 0.69 | 2.6 ± 0.56 | p < 0.05 | p < 0.05 | p > 0.05 |
| PPD IL− (mm) | 3.47 ± 1.12 | 2.52 ± 1.01 | 2.33 ± 0.91 | p < 0.05 | p < 0.05 | p < 0.05 |
| p | p > 0.05 | p > 0.05 | p > 0.05 | |||
| CAL IL+ (mm) | 9.85 ± 1.81 | 9.3 ± 2.01 | 9.06 ± 1.86 | p < 0.05 | p < 0.05 | p > 0.05 |
| CAL IL− (mm) | 10.66 ± 2.13 | 9.89 ± 1.87 | 9.72 ± 1.88 | p < 0.05 | p < 0.05 | p < 0.05 |
| p | p < 0.05 | p < 0.05 | p < 0.05 | |||
| %PPD < 4 mm IL+ | 53.92 ± 19.03 | 77.41 ± 18.07 | 80.18 ± 14.93 | p < 0.05 | p < 0.05 | p > 0.05 |
| %PPD < 4 mm IL− | 58.84 ± 23.95 | 82.85 ± 20.93 | 85.74 ± 19.54 | p < 0.05 | p < 0.05 | p < 0.05 |
| p | p < 0.05 | p < 0.05 | p < 0.05 | |||
| %PPD 4–6 mm IL+ | 40.21+/14.89 | 20.26 ± 15.93 | 18.45 ± 13.84 | p < 0.05 | p < 0.05 | p > 0.05 |
| %PPD 4–6 mm IL− | 33.7 ± 16.41 | 13.9 ± 13.88 | 11.9 ± 13.61 | p < 0.05 | p < 0.05 | p > 0.05 |
| p | p < 0.05 | p < 0.05 | p < 0.05 | |||
| %PPD > 6 mm IL+ | 5.64 ± 5.16 | 2.33 ± 3.44 | 1.37 ± 2.19 | p < 0.05 | p < 0.05 | p < 0.05 |
| %PPD > 6 mm IL− | 7.45 ± 13.39 | 3.24 ± 8.94 | 2.36 ± 6.97 | p < 0.05 | p < 0.05 | p > 0.05 |
| p | p > 0.05 | p > 0.05 | p > 0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brodzikowska, A.; Górski, B.; Bogusławska-Kapała, A. Effects of Interleukin-1 Genotype on the Clinical Efficacy of Non-Surgical Periodontal Treatment of Polish Patients with Periodontitis. Biomedicines 2023, 11, 456. https://doi.org/10.3390/biomedicines11020456
Brodzikowska A, Górski B, Bogusławska-Kapała A. Effects of Interleukin-1 Genotype on the Clinical Efficacy of Non-Surgical Periodontal Treatment of Polish Patients with Periodontitis. Biomedicines. 2023; 11(2):456. https://doi.org/10.3390/biomedicines11020456
Chicago/Turabian StyleBrodzikowska, Aniela, Bartłomiej Górski, and Agnieszka Bogusławska-Kapała. 2023. "Effects of Interleukin-1 Genotype on the Clinical Efficacy of Non-Surgical Periodontal Treatment of Polish Patients with Periodontitis" Biomedicines 11, no. 2: 456. https://doi.org/10.3390/biomedicines11020456
APA StyleBrodzikowska, A., Górski, B., & Bogusławska-Kapała, A. (2023). Effects of Interleukin-1 Genotype on the Clinical Efficacy of Non-Surgical Periodontal Treatment of Polish Patients with Periodontitis. Biomedicines, 11(2), 456. https://doi.org/10.3390/biomedicines11020456






