In Vitro Molecular Study of Titanium-Niobium Alloy Biocompatibility
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Grade 4 and Niobium Titanium Discs
2.2. Cell Culture
2.3. Cell Viability Assay
2.4. Scanning Electron Microscopy
2.5. Total RNA Extraction and Purification
2.6. Microarray Hybridization
2.7. Statistical and Functional Analyses of Microarray Data
3. Results
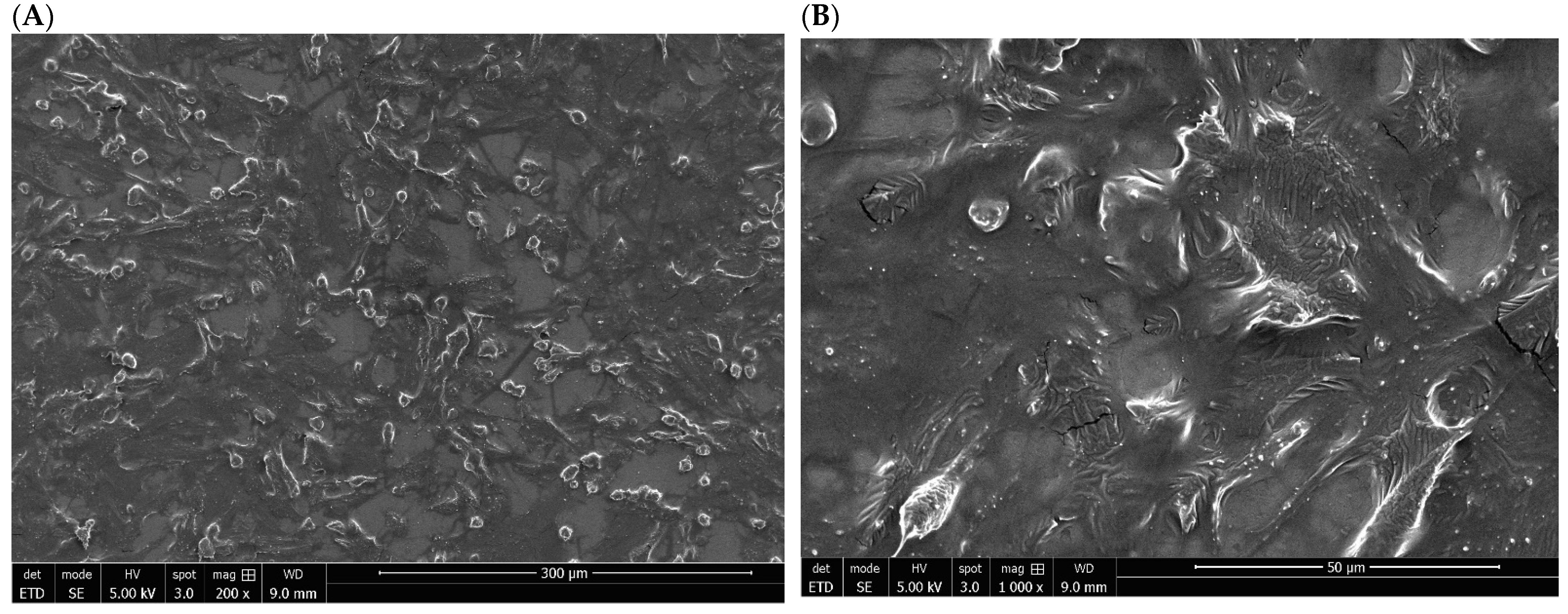
3.1. Structural and Morphological Properties of Titanium Discs
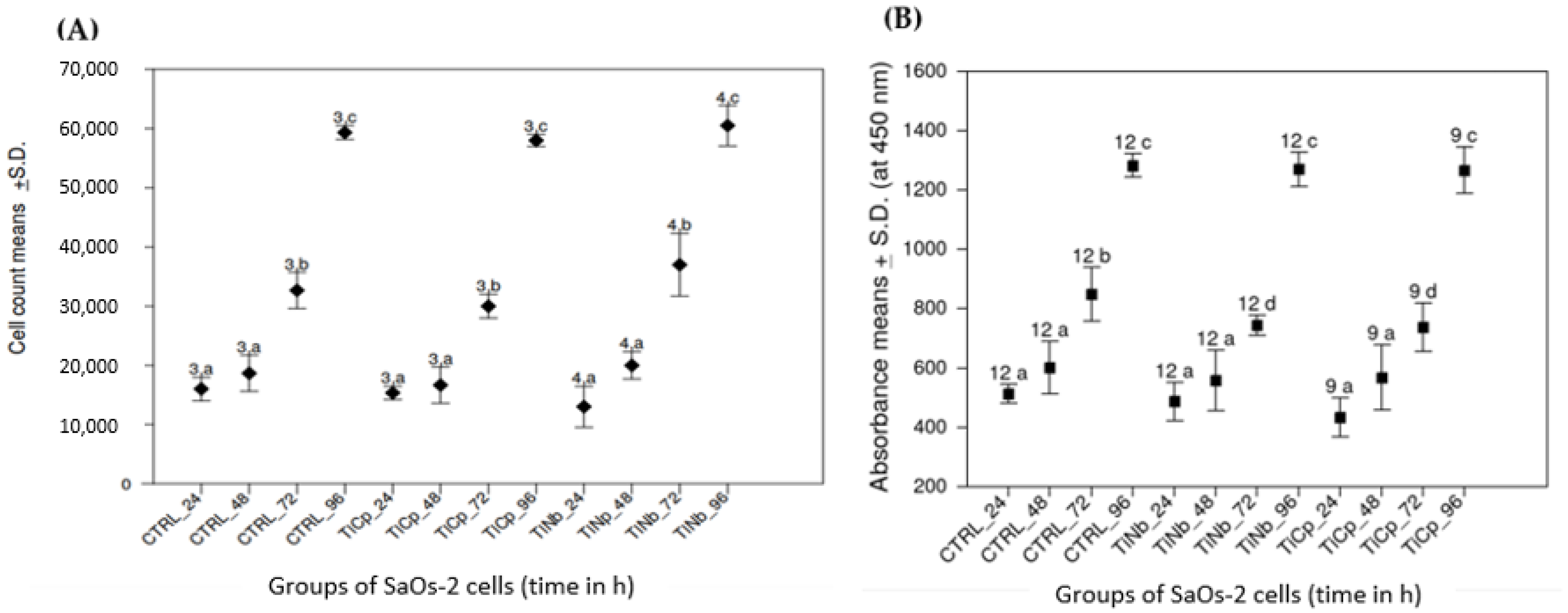
3.2. Proliferation and Metabolic Activity of SaOs-2 Cells
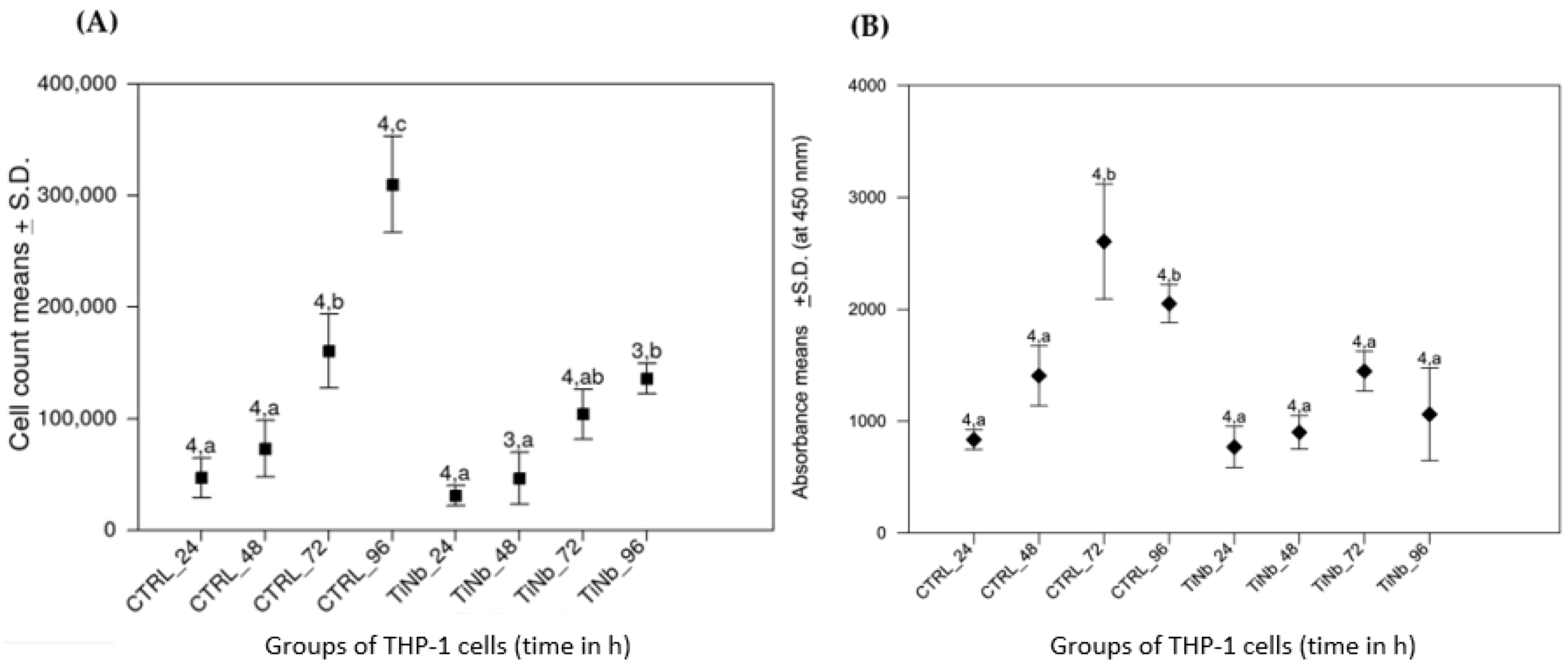
3.3. Viability and Metabolic Activity of THP-1 Cells
3.4. Morphology of SaOs-2 Cells Exposed to S_TiCp and S_TiNb Discs
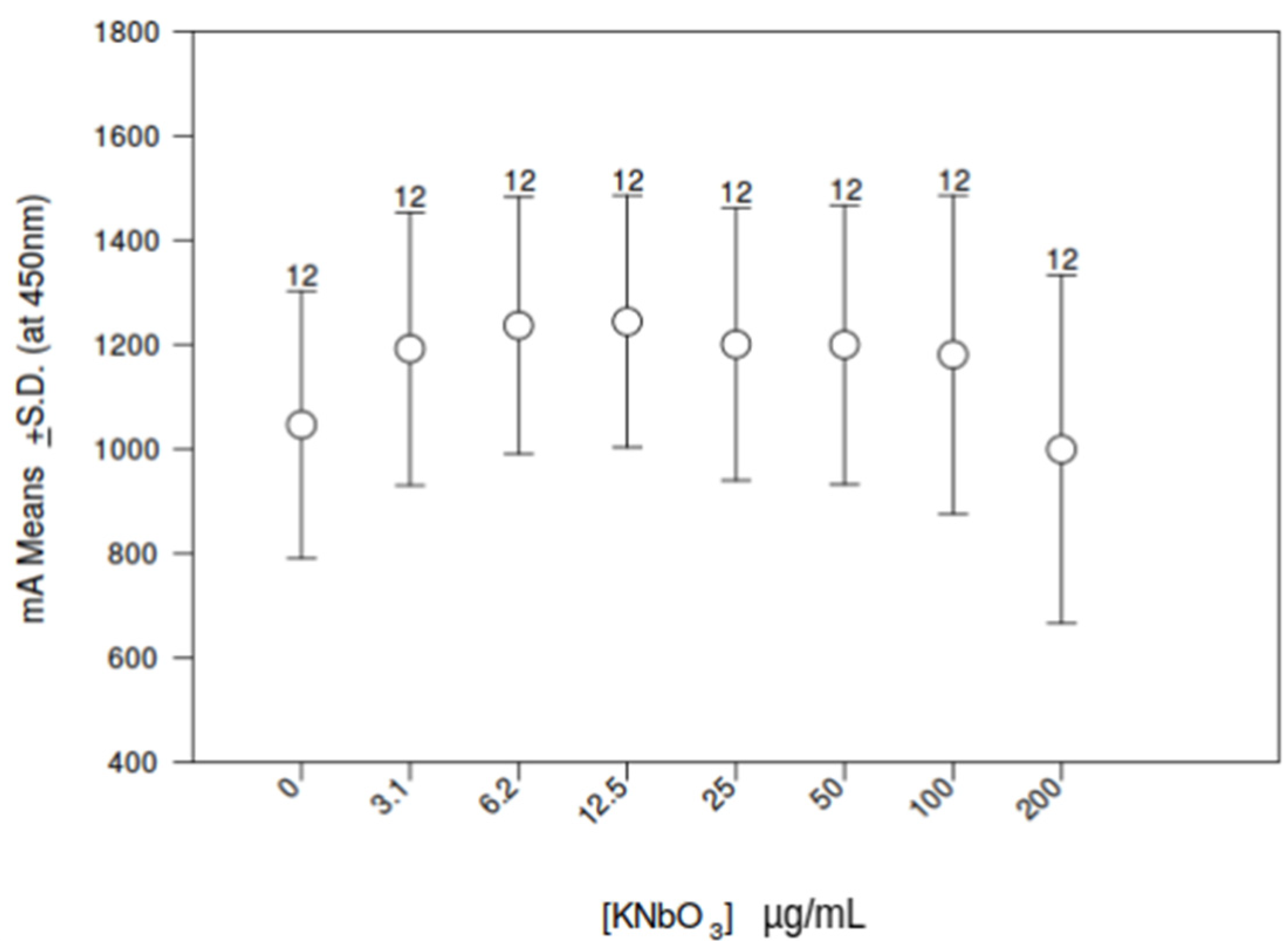
3.5. Influence of Increasing Doses of KNbO3 on THP-1 Cell Metabolic Activity
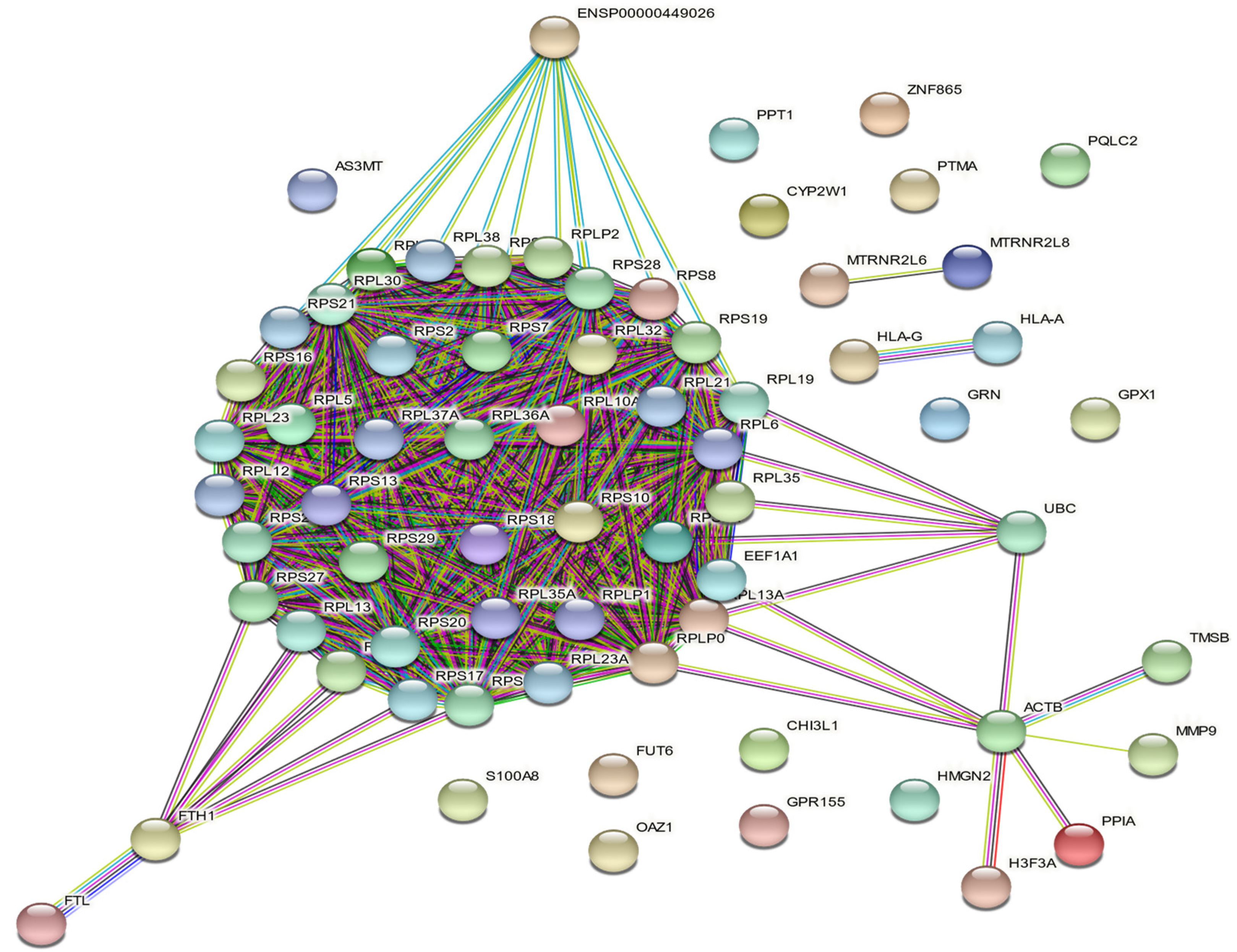
3.6. THP-1 Transcriptome Analysis
- -
- (i) a first cluster of 46 transcripts that encode cytosolic proteins figured as red nodes: AS3MT, ENSP00000449026, FTH1, FTL, FUT6, RPL10A, RPL12, RPL13, RPL13A, RPL19, RPL21, RPL23, RPL23A, RPL30, RPL32, RPL35, RPL35A, RPL36A, RPL37A, RPL38, RPL5, RPL6, RPL7A, RPLP0, RPLP1, RPLP2, RPS10, RPS11, RPS13, RPS16, RPS17, RPS18, RPS19, RPS2, RPS20, RPS21, RPS25, RPS27, RPS28, RPS29, RPS3A, RPS5, RPS6, RPS7, RPS8, S100A8 transcripts that encode proteins mainly involved in biological process ‘GO:0000184—nuclear-transcribed mRNA catabolic process’ (41/46 counts),
- -
- (ii) a second cluster of 11 transcripts figured by green nodes: ACTB, CHI3L1, EEF1A1, GPR155, H3F3A, HMGN2, MMP9, OAZ1, PPIA, TMSB4X, UBC of which a functional enrichment was the cellular component ‘GO:1904813—ficolin-1-rich granule lumen’ (3/11 counts),
- -
- and finally (iii) 11 transcripts of cluster 3 colored in blue: CYP2W1, GPX1, GRN, HLA-A, HLA-G, MTRNR2L6, MTRNR2L8, PPT1, PQLC2, PTMA, ZNF865 corresponding to the biological process ‘GO:0042270—protection from natural killer cell mediated cytotoxicity’ (2/11 counts).
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abraham, C.M. A brief historical perspective on dental implants, their surface coatings and treatments. Open Dent. J. 2014, 8, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Brånemark, P.I. Vital microscopy of bone marrow in rabbit. Scand. J. Clin. Lab. Investig. 1959, 38, 1–82. [Google Scholar]
- Baroli, B. From natural bone grafts to tissue engineering therapeutics: Brainstorming on pharmaceutical formulative requirements and challenges. J. Pharm. Sci. 2009, 98, 1317–1375. [Google Scholar] [CrossRef] [PubMed]
- Jemat, A.; Ghazali, M.J.; Razali, M.; Otsuka, Y. Surface Modifications and Their Effects on Titanium Dental Implants. Biomed. Res. Int. 2015, 2015, 791725. [Google Scholar] [CrossRef] [Green Version]
- Pérez-Díaz, L.; Anina DeDavid, B.; Gehrke, S.A. Evaluation of Fibroblasts Cells Viability and Adhesion on Six Different Titanium Surfaces: An in vitro Experimental Study. Recent Pat. Biotechnol. 2018, 12, 145–153. [Google Scholar] [CrossRef]
- Albrektsson, T.; Wennerberg, A. On osseointegration in relation to implant surfaces. Clin. Implant. Dent. Relat. Res. 2019, 21 (Suppl. 1), 4–7. [Google Scholar] [CrossRef] [Green Version]
- Thomsen, P.; Larsson, C.; Ericson, L.E.; Sennerby, L.; Lausmaa, J.; Kasemo, B. Structure of the interface between rabbit cortical bone and implants of gold, zirconium and titanium. J. Mater. Sci. Mater. Med. 1997, 8, 653–665. [Google Scholar] [CrossRef]
- Sidambe, A.T. Biocompatibility of Advanced Manufactured Titanium Implants-A Review. Materials 2014, 7, 8168–8188. [Google Scholar] [CrossRef] [Green Version]
- Suárez-López Del Amo, F.; Garaicoa-Pazmiño, C.; Fretwurst, T.; Castilho, R.M.; Squarize, C.H. Dental implants-associated release of titanium particles: A systematic review. Clin. Oral. Implants Res. 2018, 29, 1085–1100. [Google Scholar] [CrossRef]
- Dias Corpa Tardelli, J.; Lima da Costa Valente, M.; Theodoro de Oliveira, T.; Cândido Dos Reis, A. Influence of chemical composition on cell viability on titanium surfaces: A systematic review. J. Prosthet. Dent. 2021, 125, 421–425. [Google Scholar] [CrossRef]
- Nautiyal, V.P.; Mittal, A.; Agarwal, A.; Pandey, A. Tissue response to titanium implant using scanning electron microscope. Natl. J. Maxillofac. Surg. 2013, 4, 7–12. [Google Scholar] [PubMed] [Green Version]
- Ungersboeck, A.; Geret, V.; Pohler, O.; Schuetz, M.; Wuest, W. Tissue reaction to bone plates made of pure titanium: A prospective, quantitative clinical study. J. Mater. Sci. Mater. Med. 1995, 6, 223–229. [Google Scholar] [CrossRef]
- Cordeiro, J.M.; Barão, V.A.R. Is there scientific evidence favoring the substitution of commercially pure titanium with titanium alloys for the manufacture of dental implants? Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 71, 1201–1215. [Google Scholar] [CrossRef] [PubMed]
- Bozoglan, A.; Dundar, S. Comparison of osseointegration of Ti-Al6V4 and Ti-Al6Nb7 implants: An experimental study. J. Oral Biol. Craniofac. Res. 2021, 11, 624–627. [Google Scholar] [CrossRef]
- Wang, Y.; Okada, M.; Xie, S.C.; Jiao, Y.Y.; Hara, E.S.; Yanagimoto, H.; Fukumoto, T.; Matsumoto, T. Immediate soft-tissue adhesion and the mechanical properties of the Ti-6Al-4V alloy after long-term acid treatment. J. Mater. Chem. B 2021, 9, 8348–8354. [Google Scholar] [CrossRef]
- Nahle, S.; Cassidy, H.; Leroux, M.M.; Mercier, R.; Ghanbaja, J.; Doumandji, Z.; Matallanas, D.; Rihn, B.H.; Joubert, O.; Ferrari, L. Genes expression profiling of alveolar macrophages exposed to non-functionalized, anionic and cationic multi-walled carbon nanotubes shows three different mechanisms of toxicity. J. Nanobiotechnology 2020, 18, 36. [Google Scholar] [CrossRef] [Green Version]
- Chézeau, L.; Sébillaud, S.; Safar, R.; Seidel, C.; Dembélé, D.; Lorcin, M.; Langlais, C.; Grossmann, S.; Nunge, H.; Michaux, S.; et al. Short- and long-term gene expression profiles induced by inhaled TiO2 nanostructured aerosol in rat lung. Toxicol. Appl. Pharmacol. 2018, 356, 54–64. [Google Scholar] [CrossRef]
- Brazma, A.; Hingamp, P.; Quackenbush, J.; Sherlock, G.; Spellman, P.; Stoeckert, C.; Aach, J.; Ansorge, W.; Ball, C.A.; Causton, H.C.; et al. Minimum information about a microarray experiment (MIAME)-toward standards for microarray data. Nat. Genet. 2001, 29, 365–371. [Google Scholar] [CrossRef]
- Barrett, T.; Edgar, R. Gene expression omnibus: Microarray data storage, submission, retrieval, and analysis. Methods Enzymol. 2006, 411, 352–369. [Google Scholar]
- Dembélé, D.; Kastner, P. Fold change rank ordering statistics: A new method for detecting differentially expressed genes. BMC Bioinform. 2014, 15, 14. [Google Scholar] [CrossRef] [Green Version]
- Chézeau, L.; Kohlstaedt, L.A.; Le Faou, A.; Cosnier, F.; Rihn, B.; Gaté, L. Proteomic analysis of bronchoalveolar lavage fluid in rat exposed to TiO2 nanostructured aerosol by inhalation. J. Proteom. 2019, 207, 103451. [Google Scholar] [CrossRef] [PubMed]
- Szklarczyk, D.; Franceschini, A.; Wyder, S.; Forslund, K.; Heller, D.; Huerta-Cepas, J.; Simonovic, M.; Roth, A.; Santos, A.; Tsafou, K.P.; et al. STRING v10: Protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res. 2015, 43, D447–D452. [Google Scholar] [CrossRef] [PubMed]
- Baseri, M.; Radmand, F.; Hamedi, R.; Yousefi, M.; Kafil, H.S. Immunological Aspects of Dental Implant Rejection. Biomed. Res. Int. 2020, 2020, 7279509. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Marquez, D.; Delmar, Y.; Sun, S.; Stewart, R.A. Exploring Macroporosity of Additively Manufactured Titanium Metamaterials for Bone Regeneration with Quality by Design: A Systematic Literature Review. Materials 2020, 13, 4794. [Google Scholar] [CrossRef]
- Geetha, M.; Singh, A.K.; Asokamani, R.; Gogia, A.K. Ti based biomaterials, the ulti-mate choice for orthopaedic implants—A review. Prog. Mater. Sci. 2009, 54, 397–425. [Google Scholar] [CrossRef]
- Quirynen, M.; Abarca, M.; Van Assche, N.; Nevins, M.; Van Steenberghe, D. Impact of supportive periodontal therapy and implant surface roughness on implant outcome in patients with a history of periodontitis. J. Clin. Periodontol. 2007, 34, 805–815. [Google Scholar] [CrossRef]
- Esposito, M.; Murray-Curtis, L.; Grusovin, M.G.; Patel, S.; Coulthard, P.; Worthington, H.V. Interventions for replacing missing teeth: Different types of dental implants. Cochrane Database Syst. Rev. 2007, 17, CD003815. [Google Scholar]
- Le Guéhennec, L.; Soueidan, A.; Layrolle, P.; Amouriq, Y. Surface treatments of titanium dental implants for rapid osseointegration. Dent. Mater. 2007, 23, 844–854. [Google Scholar] [CrossRef]
- Matos, G.R.M. Surface Roughness of Dental Implant and Osseointegration. J. Maxillofac. Oral Surg. 2021, 20, 1–4. [Google Scholar] [CrossRef]
- Civantos, A.; Martínez-Campos, E.; Ramos, V.; Elvira, C.; Gallardo, A.; Abarrategi, A. Titanium Coatings and Surface Modifications: Toward Clinically Useful Bioactive Implants. ACS Biomater. Sci. Eng. 2017, 3, 1245–1261. [Google Scholar] [CrossRef]
- Wang, S.; Niu, L.; Chen, C.; Pang, Y.; Liao, B.; Zhong, Z.H.; Lu, P.; Li, P.; Wu, X.D.; Coenen, J.W.; et al. Size effects on the tensile properties and deformation mechanism of commercial pure titanium foils. Mater. Sci. Eng. A 2018, 730, 244–261. [Google Scholar] [CrossRef]
- Luo, H.; Wu, Y.; Diao, X.; Shi, W.; Feng, F.; Qian, F.; Umeda, J.; Kondoh, K.; Xin, H.; Shen, J. Mechanical properties and biocompatibility of titanium with a high oxygen concentration for dental implants. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 117, 111306. [Google Scholar] [CrossRef] [PubMed]
- Naganawa, T.; Ishihara, Y.; Iwata, T.; Koide, M.; Ohguchi, M.; Ohguchi, Y.; Murase, Y.; Kamei, H.; Sato, N.; Mizuno, M.; et al. In vitro biocompatibility of a new titanium-29niobium-13tantalum-4.6zirconium alloy with osteoblast-like MG63 cells. J. Periodontol. 2004, 75, 1701–1707. [Google Scholar] [CrossRef] [PubMed]
- Falanga, A.; Laheurte, P.; Vahabi, H.; Tran, N.; Khamseh, S.; Saeidi, H.; Khodadadi, M.; Zarrintaj, P.; Saeb, M.R.; Mozafari, M. Niobium-Treated Titanium Implants with Improved Cellular and Molecular Activities at the Tissue-Implant Interface. Materials 2019, 12, 3861. [Google Scholar] [CrossRef] [Green Version]
- Lin, X.; Xiao, X.; Wang, Y.; Gu, C.; Wang, C.; Chen, J.; Liu, H.; Luo, J.; Li, T.; Wang, D.; et al. Biocompatibility of Bespoke 3D-Printed Titanium Alloy Plates for Treating Acetabular Fractures. Biomed. Res. Int. 2018, 2018, 2053486. [Google Scholar] [CrossRef] [Green Version]
- Olesova, V.N.; Shashmurina, V.R.; Shugailov, I.A.; Olesov, E.E.; Mirgazizov, M.Z. Study of the Biocompatibility of Titanium-Niobium Implants by the Parameters of Their Osseointegration under Experimental Conditions. Bull. Exp. Biol. Med. 2019, 166, 686–688. [Google Scholar] [CrossRef]
- Chen, Y.; Han, P.; Dehghan-Manshadi, A.; Kent, D.; Ehtemam-Haghighi, S.; Jowers, C.; Bermingham, M.; Li, T.; Cooper-White, J.; Dargusch, M.S. Sintering and biocompatibilityof blended elemental Ti-xNb alloys. J. Mech. Behav. Biomed. Mater. 2020, 104, 103691. [Google Scholar] [CrossRef]
- Yolun, A.; Şimşek, M.; Kaya, M.; Annaç, E.E.; Köm, M.; Çakmak, Ö. Fabrication, characterization, and in vivo biocompatibility evaluation of titanium-niobium implants. Proc. Inst. Mech. Eng. H 2021, 235, 99–108. [Google Scholar] [CrossRef]
- Challa, V.S.A.; Mali, S.; Misra, R.D.K. Reduced toxicity and superior cellular response of preosteoblasts to Ti-6Al-7Nb alloy and comparison with Ti-6Al-4V. J. Biomed. Mater. Res. 2013, 101, 2083–2089. [Google Scholar] [CrossRef]
- Dika Nguea, H.; Rihn, B.; Mahon, D.; Bernard, J.L.; De Reydellet, A.; Le Faou, A. Effects of various man-made mineral fibers on cell apoptosis and viability. Arch. Toxicol. 2005, 79, 487–492. [Google Scholar] [CrossRef]
- Nguea, H.D.; de Reydellet, A.; Le Faou, A.; Zaiou, M.; Rihn, B. Macrophage culture as a suitable paradigm for evaluation of synthetic vitreous fibers. Crit. Rev. Toxicol. 2008, 38, 675–695. [Google Scholar] [CrossRef] [PubMed]
- Noronha Oliveira, M.; Schunemann, W.V.H.; Mathew, M.T.; Henriques, B.; Magini, R.S.; Teughels, W.; Souza, J.C.M. Can degradation products released from dental implants affect peri-implant tissues? J. Periodontal Res. 2018, 53, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, Y.; Feng, Y.; Cheng, H.; Li, D. Macrophage polarization in aseptic bone resorption around dental implants induced by Ti particles in a murine model. J. Periodontal Res. 2019, 54, 329–338. [Google Scholar] [CrossRef]
- Messous, R.; Henriques, B.; Bousbaa, H.; Silva, F.S.; Teughels, W.; Souza, J.C.M. Cytotoxic effects of submicron- and nano-scale titanium debris released from dental implants: An integrative review. Clin. Oral Investig. 2021, 25, 1627–1640. [Google Scholar] [CrossRef]
- Tchinda, A.; Pierson, G.; Chézeau, L.; Kouitat-Njiwa, R.; Rihn, B.H.; Bravetti, P. Desulfovibrio fairfieldensis adhesion on implantable titanium used in odontology: A preliminary study. Cell Mol Biol. 2021, 67, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Deng, Y.; Zheng, Y.; Li, Y.; Zhang, R.; Lv, Y.; Zhao, Q.; Wei, S. Characterization, corrosion behavior, cellular response and in vivo bone tissue compatibility of titanium niobium alloy with low Young’s modulus. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 59, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Correa, D.R.N.; Kuroda, P.A.B.; Lourenço, M.L.; Fernandes, C.J.C.; Buzalaf, M.A.R.; Zambuzzi, W.F.; Grandinibe, C.R. Development of Ti-15Zr-Mo alloys for applying as implantable biomedical devices. J. Alloys Compd. 2018, 749, 163–171. [Google Scholar] [CrossRef] [Green Version]
- Fisher, J.G.; Thuan, U.T.; Farooq, M.U.; Chandrasekaran, G.; Jung, Y.D.; Hwang, E.C.; Lee, J.J.; Lakshmanan, V.K. Prostate Cancer Cell-Specific Cytotoxicity of Sub-Micron Potassium Niobate Powder. J. Nanosci Nanotechnol. 2018, 18, 3141–3147. [Google Scholar] [CrossRef]
- Eger, M.; Hiram-Bab, S.; Liron, T.; Sterer, N.; Carmi, Y.; Kohavi, D.; Gabet, Y. Mechanism and Prevention of Titanium Particle-Induced Inflammation and Osteolysis. Front. Immunol. 2018, 9, 2963. [Google Scholar] [CrossRef] [Green Version]
- Dera, A.A.; Ranganath, L.; Barraclough, R.; Vinjamuri, S.; Hamill, S.; Barraclough, D.L. Cathepsin Z as a novel potential biomarker for osteoporosis. Sci. Rep. 2019, 9, 9752. [Google Scholar] [CrossRef]
- Doumandji, Z.; Safar, R.; Lovera-Leroux, M.; Nahle, S.; Cassidy, H.; Matallanas, D.; Rihn, B.; Ferrari, L.; Joubert, O. Protein and lipid homeostasis altered in rat macrophages after exposure to metallic oxide nanoparticles. Cell Biol. Toxicol. 2020, 36, 65–82. [Google Scholar] [CrossRef] [Green Version]
- Eidi, H.; Joubert, O.; Némos, C.; Grandemange, S.; Mograbi, B.; Foliguet, B.; Tournebize, J.; Maincent, P.; Le Faou, A.; Aboukhamis, I. Drug delivery by polymeric nanoparticles induces autophagy in macrophages. Int. J. Pharm. 2012, 422, 495–503. [Google Scholar] [CrossRef] [PubMed]
- do Prado, R.F.; Rabêlo, S.B.; de Andrade, D.P.; Nascimento, R.D.; Henriques, V.A.; Carvalho, Y.R.; Cairo, C.A.; de Vasconcellos, L.M. Porous titanium and Ti-35Nb alloy: Effects on gene expression of osteoblastic cells derived from human alveolar bone. J. Mater. Sci. Mater. Med. 2015, 26, 259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lai, B.W.; Chang, Y.Y.; Shieh, T.M.; Huang, H.L. Biocompatibility and Microstructure-Based Stress Analyses of TiNbZrTa Composite Films. Materials 2021, 15, 29. [Google Scholar] [CrossRef] [PubMed]
- do Prado, R.F.; Esteves, G.C.; Santos, E.L.S.; Bueno, D.A.G.; Cairo, C.A.A.; Vasconcellos, L.G.O.; Sagnori, R.S.; Tessarin, F.B.P.; Oliveira, F.E.; Oliveira, L.D. In vitro and in vivo biological performance of porous Ti alloys prepared by powder metallurgy. PLoS ONE 2018, 13, e0196169. [Google Scholar] [CrossRef] [Green Version]


| A_23_P336854 | Human Agilent Probe/RPL12L3-001 * |
| A_24_P204144 | Human Agilent Probe/Not recognized by Ensembl© |
| A_33_P3270581 | Human Agilent Probe/RP11-79L9.2-001 * |
| A_33_P3299436 | Human Agilent Probe/RP5-827C21.1-001 * |
| A_33_P3315027 | Human Agilent Probe/RP11-510I6.1-001 * |
| A_33_P3353259 | Human Agilent Probe/RP11-179H18.5-001 * |
| A_33_P3370515 | Human Agilent Probe/Not recognized by Ensembl© |
| A_33_P3375668 | Human Agilent Probe/AP000354.2-001 * |
| A_33_P3381429 | Human Agilent Probe/RP11-16L9.1-001* |
| A_33_P3396434 | Human Agilent Probe/No external name |
| ACTB | Actin, cytoplasmic 1 |
| AS3MT | Arsenite methyltransferase |
| CHI3L1 | Chitinase-3-like protein 1 |
| CYP2W1 | Cytochrome P450 2W1 |
| EEF1A1 | Elongation factor 1-alpha 1 |
| FTH1 | Ferritin heavy chain |
| FTL | Ferritin light chain |
| FUT6 | Alpha-(1,3)-fucosyltransferase 6 |
| GPR155 | Integral membrane protein GPR155 |
| GPX1 | Glutathione peroxidase 1 |
| GRN | Granulin precursor |
| H3F3A | H3 histone family member 3A |
| HLA-A | HLA class I histocompatibility antigen |
| HLA-G | HLA class I histocompatibility antigen, alpha chain G |
| HMGN2 | Non-histone chromosomal protein HMG-17 |
| MMP9 | Matrix metalloproteinase-9 |
| MTRNR2L8 | MT-RNR2 like 8 |
| MTRNR2L6 | Humanin-like 6 |
| OAZ1 | Ornithine decarboxylase antizyme 1 |
| PPIA | Peptidyl-prolyl cis-trans isomerase A; |
| PPT1 | Palmitoyl-protein thioesterase 1 |
| PQLC2 | Solute carrier family 66 (lysosomal lysine-arginine transporter) |
| PTMA | Prothymosin alpha |
| RPL5 | 60S ribosomal protein L5 |
| RPL6 | 60S ribosomal protein L6 |
| RPL7A | Large subunit ribosomal protein L7ae |
| RPL10A | 60S ribosomal protein L10a |
| RPL12 | Large subunit ribosomal protein L12e |
| RPL13 | Large subunit ribosomal protein L13e |
| RPL13A | Large subunit ribosomal protein L13ae |
| RPL19 | Large subunit ribosomal protein L19e |
| RPL21 | 60S ribosomal protein L21 |
| RPL23 | Large subunit ribosomal protein L23e |
| RPL23A | 60S ribosomal protein L23a |
| RPL30 | Large subunit ribosomal protein L30e |
| RPL32 | Large subunit ribosomal protein L32e |
| RPL35 | 60S ribosomal protein L35 |
| RPL35A | Large subunit ribosomal protein L35ae |
| RPL38 | Large subunit ribosomal protein L38e |
| RPL41 | Ribosomal protein L41 |
| RPLP0 | Ribosomal protein lateral stalk subunit p0 |
| RPLP1 | 60S acidic ribosomal protein P1 |
| RPLP2 | 60S acidic ribosomal protein P2 |
| RPS2 | Small subunit ribosomal protein S2e |
| RPS3A | 40S ribosomal protein S3a |
| RPS5 | Small subunit ribosomal protein S5e |
| RPS6 | 40S ribosomal protein S6 |
| RPS7 | 40S ribosomal protein S7 |
| RPS8 | Small subunit ribosomal protein S8e |
| RPS10 | 40S ribosomal protein S10 |
| RPS11 | Small subunit ribosomal protein S11e |
| RPS13 | Small subunit ribosomal protein S13e |
| RPS16 | Small subunit ribosomal protein S16e |
| RPS17 | Small subunit ribosomal protein S17e |
| RPS18 | 40S ribosomal protein S18 |
| RPS19 | 40S ribosomal protein S19 |
| RPS20 | Small subunit ribosomal protein S20e |
| RPS21 | Small subunit ribosomal protein S21e |
| RPS25 | Small subunit ribosomal protein S25e |
| RPS27 | 40S ribosomal protein S27 |
| RPS28 | Small subunit ribosomal protein S28e |
| RPS29 | Small subunit ribosomal protein S29e |
| S100A8 | Protein S100-A8 |
| TMSB4X | Thymosin beta 4, X-linked |
| UBC | Polyubiquitin-C |
| XLOC_014512 | RP11-54O7.1 (Clone-based (Vega) gene) |
| ZNF865 | Zinc finger protein 865 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chézeau, L.; Tchinda, A.; Pierson, G.; Bravetti, P.; Ferrari, L.; Joubert, O.; Zaiou, M.; Rihn, B.H. In Vitro Molecular Study of Titanium-Niobium Alloy Biocompatibility. Biomedicines 2022, 10, 1898. https://doi.org/10.3390/biomedicines10081898
Chézeau L, Tchinda A, Pierson G, Bravetti P, Ferrari L, Joubert O, Zaiou M, Rihn BH. In Vitro Molecular Study of Titanium-Niobium Alloy Biocompatibility. Biomedicines. 2022; 10(8):1898. https://doi.org/10.3390/biomedicines10081898
Chicago/Turabian StyleChézeau, Laëtitia, Alex Tchinda, Gaël Pierson, Pierre Bravetti, Luc Ferrari, Olivier Joubert, Mohamed Zaiou, and Bertrand H. Rihn. 2022. "In Vitro Molecular Study of Titanium-Niobium Alloy Biocompatibility" Biomedicines 10, no. 8: 1898. https://doi.org/10.3390/biomedicines10081898
APA StyleChézeau, L., Tchinda, A., Pierson, G., Bravetti, P., Ferrari, L., Joubert, O., Zaiou, M., & Rihn, B. H. (2022). In Vitro Molecular Study of Titanium-Niobium Alloy Biocompatibility. Biomedicines, 10(8), 1898. https://doi.org/10.3390/biomedicines10081898








