Sex Steroid Receptors in Polycystic Ovary Syndrome and Endometriosis: Insights from Laboratory Studies to Clinical Trials
Abstract
1. Introduction
2. Characteristics of Sex Steroid Receptors
2.1. Structure
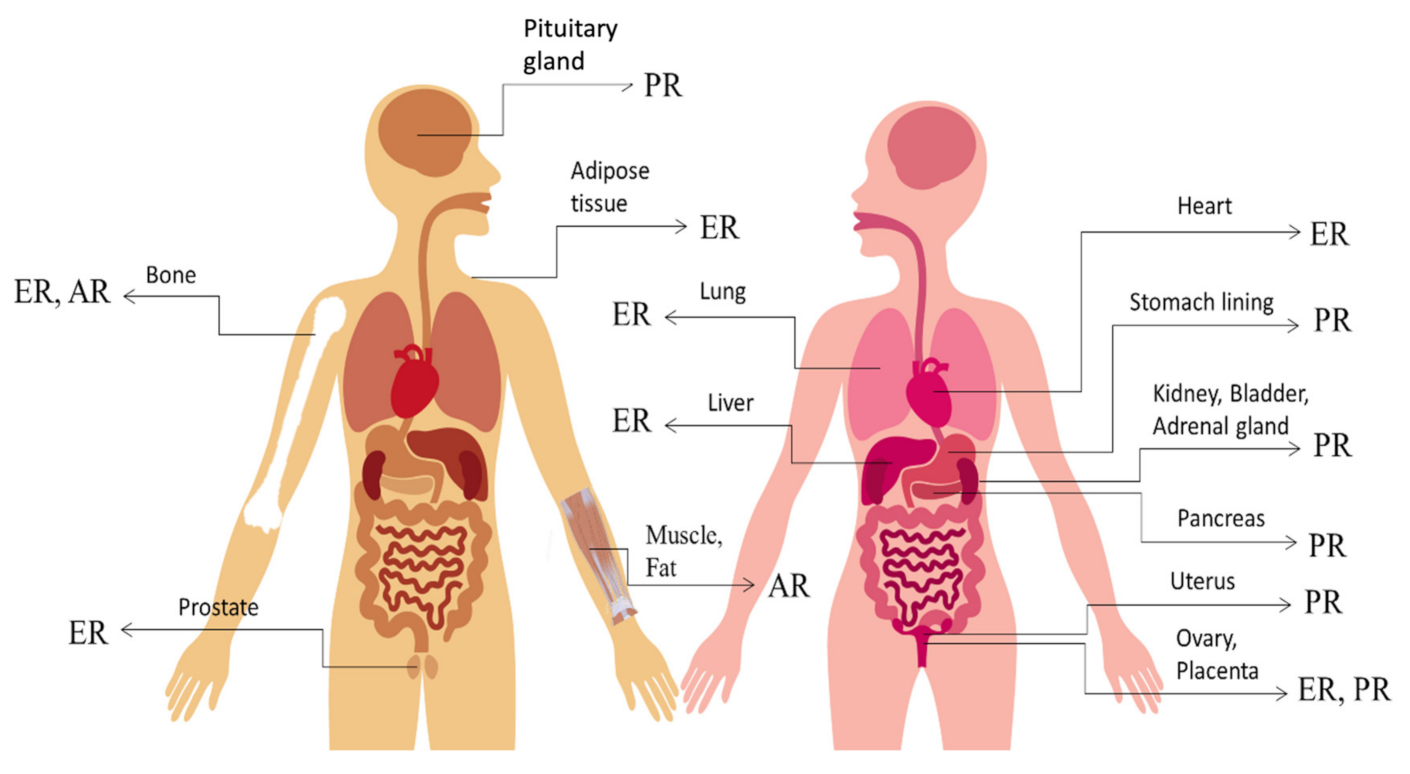
2.2. Distribution
2.3. Physiological Functions
2.3.1. Oestrogen Receptors
2.3.2. Progesterone Receptors
2.3.3. Androgen Receptor
3. Sex Steroid Receptors in PCOS and Endometriosis
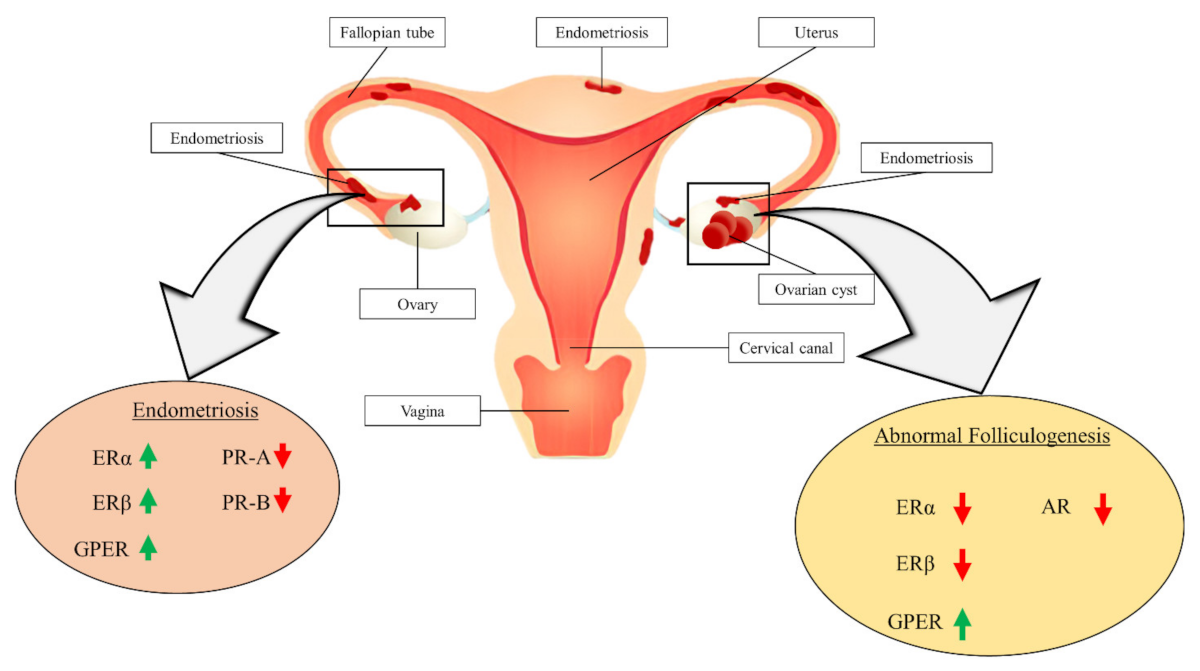
3.1. Abnormal Folliculogenesis in PCOS
3.2. Metabolic Disorders in PCOS
3.3. Endometriosis
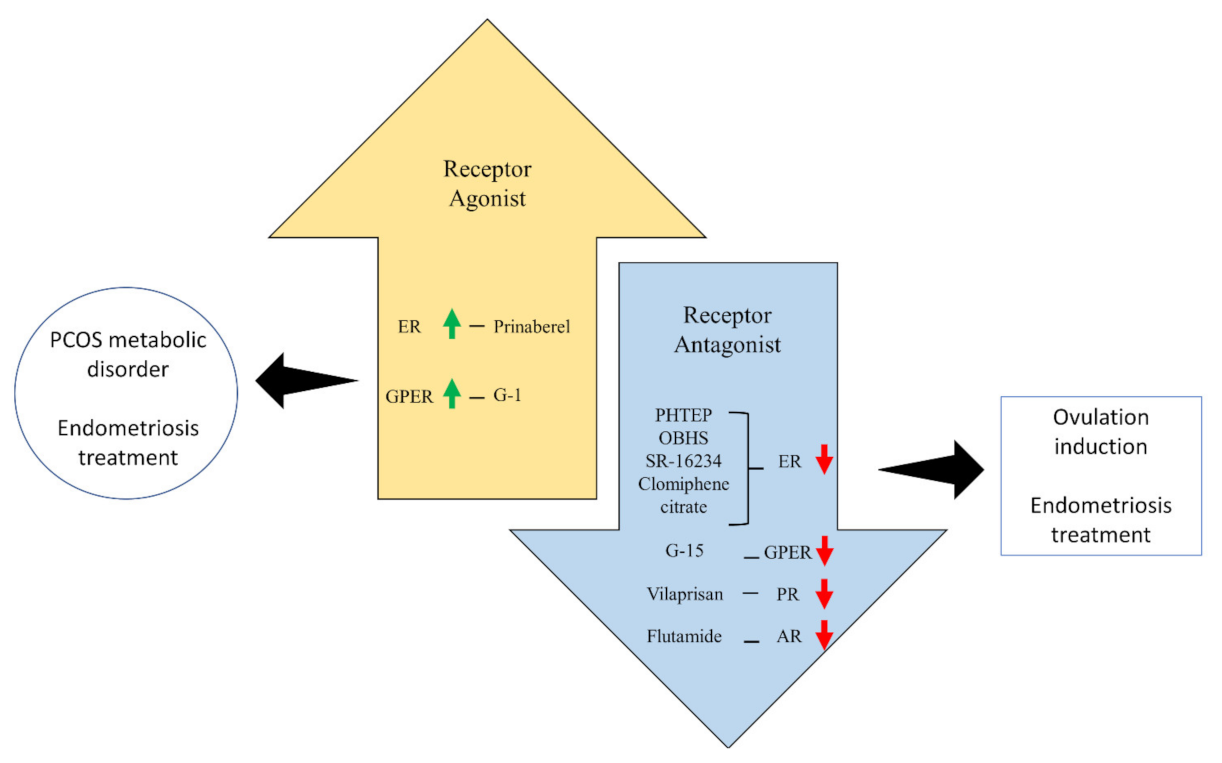
4. Potential Therapeutic Routes
4.1. Receptor Agonist
4.2. Receptor Antagonist
5. Limitations
6. Recommendations
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Azziz, R.; Carmina, E.; Chen, Z.; Dunaif, A.; Laven, J.S.; Legro, R.S.; Lizneva, D.; Natterson-Horowtiz, B.; Teede, H.J.; Yildiz, B.O. Polycystic ovary syndrome. Nat. Rev. Dis. Primers 2016, 2, 16057. [Google Scholar] [CrossRef] [PubMed]
- Swaroop, A.; Jaipuriar, A.S.; Gupta, S.K.; Bagchi, M.; Kumar, P.; Preuss, H.G.; Bagchi, D. Efficacy of a Novel Fenugreek Seed Extract (Trigonella foenum-graecum, Furocyst) in Polycystic Ovary Syndrome (PCOS). Int. J. Med. Sci. 2015, 12, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Teede, H.; Deeks, A.; Moran, L. Polycystic ovary syndrome: A complex condition with psychological, reproductive and metabolic manifestations that impacts on health across the lifespan. BMC Med. 2010, 8, 41. [Google Scholar] [CrossRef]
- Carmina, E. Diagnosis of polycystic ovary syndrome: From NIH criteria to ESHRE-ASRM guidelines. Minerva Ginecol. 2004, 56, 6. [Google Scholar]
- Gupta, M.; Singh, D.; Toppo, M.; Priya, A.; Sethia, S.; Gupta, P. A cross sectional study of polycystic ovarian syndrome among young women in Bhopal, Central India. Int. J. Community Med. Public Health 2018, 5, 95–100. [Google Scholar] [CrossRef]
- Sirmans, S.M.; Pate, K.A. Epidemiology, diagnosis, and management of polycystic ovary syndrome. Clin. Epidemiol. 2014, 6, 13. [Google Scholar] [CrossRef] [PubMed]
- Azziz, R.; Carmina, E.; Dewailly, D.; Diamanti-Kandarakis, E.; Escobar-Morreale, H.F.; Futterweit, W.; Janssen, O.E.; Legro, R.S.; Norman, R.J.; Taylor, A.E. Criteria for defining polycystic ovary syndrome as a predominantly hyperandrogenic syndrome: An androgen excess society guideline. J. Clin. Endocrinol. Metab. 2006, 91, 4237–4245. [Google Scholar] [CrossRef]
- Fauser, B.C.; Tarlatzis, B.C.; Rebar, R.W.; Legro, R.S.; Balen, A.H.; Lobo, R.; Carmina, E.; Chang, J.; Yildiz, B.O.; Laven, J.S. Consensus on women’s health aspects of polycystic ovary syndrome (PCOS): The Amsterdam ESHRE/ASRM-Sponsored 3rd PCOS Consensus Workshop Group. Fertil. Steril. 2012, 97, 28–38.e25. [Google Scholar] [CrossRef]
- Glintborg, D.; Andersen, M. An update on the pathogenesis, inflammation, and metabolism in hirsutism and polycystic ovary syndrome. Gynecol. Endocrinol. 2010, 26, 281–296. [Google Scholar] [CrossRef]
- Sourial, S.; Tempest, N.; Hapangama, D.K. Theories on the Pathogenesis of Endometriosis. Int. J. Reprod. Med. 2014, 2014, 179515. [Google Scholar] [CrossRef]
- Signorile, P.G.; Viceconte, R.; Baldi, A. New Insights in Pathogenesis of Endometriosis. Front. Med. 2022, 9, 879015. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.-Y.; Liu, Y.; Lv, Y.; Huang, T.; Lu, G.; Liu, H.-B.; Zhao, S.-G. Role of Androgen Receptor for Reconsidering the “true” polycystic ovarian Morphology in pcos. Sci. Rep. 2020, 10, 7. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-L.; Deng, S.-L.; Lian, Z.-X.; Yu, K. Estrogen Receptors in Polycystic Ovary Syndrome. Cells 2021, 10, 459. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Xiang, L.; Li, T.; Bai, Z. Cancer hallmarks, biomarkers and breast cancer molecular subtypes. J. Cancer 2016, 7, 1281. [Google Scholar] [CrossRef] [PubMed]
- Barton, M.; Filardo, E.J.; Lolait, S.J.; Thomas, P.; Maggiolini, M.; Prossnitz, E.R. Twenty years of the G protein-coupled estrogen receptor GPER: Historical and personal perspectives. J. Steroid Biochem. Mol. Biol. 2018, 176, 4–15. [Google Scholar] [CrossRef]
- Fuentes, N.; Silveyra, P. Estrogen receptor signaling mechanisms. Adv. Protein Chem. Struct. Biol. 2019, 116, 135–170. [Google Scholar]
- Hua, H.; Zhang, H.; Kong, Q.; Jiang, Y. Mechanisms for estrogen receptor expression in human cancer. Exp. Hematol. Oncol. 2018, 7, 11. [Google Scholar] [CrossRef]
- Luo, J.; Liu, D. Does GPER really function as a G protein-coupled estrogen receptor in vivo? Front. Endocrinol. 2020, 11, 148. [Google Scholar] [CrossRef]
- Vasquez, Y.; DeMayo, F. Role of nuclear receptors in blastocyst implantation. Semin. Cell Dev. Biol. 2013, 24, 724–735. [Google Scholar] [CrossRef]
- Kastner, P.; Krust, A.; Turcotte, B.; Stropp, U.; Tora, L.; Gronemeyer, H.; Chambon, P. Two distinct estrogen-regulated promoters generate transcripts encoding the two functionally different human progesterone receptor forms A and B. EMBO J. 1990, 9, 1603–1614. [Google Scholar] [CrossRef]
- Grimm, S.L.; Hartig, S.M.; Edwards, D.P. Progesterone Receptor Signaling Mechanisms. J. Mol. Biol. 2016, 428, 3831–3849. [Google Scholar] [CrossRef]
- Tsai, M.-J.; O’Malley, B.W. Molecular mechanisms of action of steroid/thyroid receptor superfamily members. Annu. Rev. Biochem. 1994, 63, 451–486. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Xie, Y.-J.; Liu, Y.-T.; Long, S.-L.; Mo, Z.-C. Polycystic ovarian syndrome: Correlation between hyperandrogenism, insulin resistance and obesity. Clin. Chim. Acta 2020, 502, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Burger, H.G. Androgen production in women. Fertil. Steril. 2002, 77, 3–5. [Google Scholar] [CrossRef]
- Hutson, D.D.; Gurrala, R.; Ogola, B.O.; Zimmerman, M.A.; Mostany, R.; Satou, R.; Lindsey, S.H. Estrogen receptor profiles across tissues from male and female Rattus norvegicus. Biol. Sex Differ. 2019, 10, 13. [Google Scholar] [CrossRef] [PubMed]
- Desmawati, D.; Sulastri, D. Phytoestrogens and their Health Effect. Open Access Maced. J. Med. Sci. 2019, 7, 495–499. [Google Scholar] [CrossRef]
- Asavasupreechar, T.; Saito, R.; Miki, Y.; Edwards, D.P.; Boonyaratanakornkit, V.; Sasano, H. Systemic distribution of progesterone receptor subtypes in human tissues. J. Steroid Biochem. Mol. Biol. 2020, 199, 105599. [Google Scholar] [CrossRef]
- Mhaouty-Kodja, S. Role of the androgen receptor in the central nervous system. Mol. Cell. Endocrinol. 2018, 465, 103–112. [Google Scholar] [CrossRef]
- Hughes, C.H.; Murphy, B.D. Nuclear receptors: Key regulators of somatic cell functions in the ovulatory process. Mol. Asp. Med. 2021, 78, 100937. [Google Scholar] [CrossRef]
- Richards, J.S.; Pangas, S.A. The ovary: Basic biology and clinical implications. J. Clin. Investig. 2010, 120, 963–972. [Google Scholar] [CrossRef]
- Prossnitz, E.R.; Barton, M. The G-protein-coupled estrogen receptor GPER in health and disease. Nat. Rev. Endocrinol. 2011, 7, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Enmark, E.; Pelto-Huikko, M.; Grandien, K.; Lagercrantz, S.; Lagercrantz, J.; Fried, G.; Nordenskjöld, M.; Gustafsson, J.-A.K. Human estrogen receptor β-gene structure, chromosomal localisation, and expression pattern. J. Clin. Endocrinol. Metab. 1997, 82, 4258–4265. [Google Scholar] [PubMed]
- Lecce, G.; Meduri, G.; Ancelin, M.; Bergeron, C.; Perrot-Applanat, M. Presence of estrogen receptor β in the human endometrium through the cycle: Expression in glandular, stromal, and vascular cells. J. Clin. Endocrinol. Metab. 2001, 86, 1379–1386. [Google Scholar] [CrossRef] [PubMed]
- Punyadeera, C.; Verbost, P.; Groothuis, P. Oestrogen and progestin responses in human endometrium. J. Steroid Biochem. Mol. Biol. 2003, 84, 393–410. [Google Scholar] [CrossRef]
- Pugach, E.K.; Blenck, C.L.; Dragavon, J.M.; Langer, S.J.; Leinwand, L.A. Estrogen receptor profiling and activity in cardiac myocytes. Mol. Cell. Endocrinol. 2016, 431, 62–70. [Google Scholar] [CrossRef]
- Wiik, A.; Hellsten, Y.; Berthelson, P.; Lundholm, L.; Fischer, H.; Jansson, E. Activation of estrogen response elements is mediated both via estrogen and muscle contractions in rat skeletal muscle myotubes. Am. J. Physiol. Cell Physiol. 2009, 296, C215–C220. [Google Scholar] [CrossRef]
- Klinge, C.M. Estrogens regulate life and death in mitochondria. J. Bioenerg. Biomembr. 2017, 49, 307–324. [Google Scholar] [CrossRef]
- Simpkins, J.W.; Yang, S.-H.; Sarkar, S.N.; Pearce, V. Estrogen actions on mitochondria—physiological and pathological implications. Mol. Cell. Endocrinol. 2008, 290, 51–59. [Google Scholar] [CrossRef]
- Hsieh, Y.-C.; Yang, S.; Choudhry, M.A.; Yu, H.-P.; Rue, L.W., III; Bland, K.I.; Chaudry, I.H. PGC-1 upregulation via estrogen receptors: A common mechanism of salutary effects of estrogen and flutamide on heart function after trauma-hemorrhage. Am. J. Physiol.-Heart Circ. Physiol. 2005, 289, H2665–H2672. [Google Scholar] [CrossRef]
- Mattingly, K.A.; Ivanova, M.M.; Riggs, K.A.; Wickramasinghe, N.S.; Barch, M.J.; Klinge, C.M. Estradiol stimulates transcription of nuclear respiratory factor-1 and increases mitochondrial biogenesis. Mol. Endocrinol. 2008, 22, 609–622. [Google Scholar] [CrossRef]
- Witt, H.; Schubert, C.; Jaekel, J.; Fliegner, D.; Penkalla, A.; Tiemann, K.; Stypmann, J.; Roepcke, S.; Brokat, S.; Mahmoodzadeh, S. Sex-specific pathways in early cardiac response to pressure overload in mice. J. Mol. Med. 2008, 86, 1013–1024. [Google Scholar] [CrossRef] [PubMed]
- Boland, R.; Vasconsuelo, A.; Milanesi, L.; Ronda, A.C.; de Boland, A.R. 17β-Estradiol signaling in skeletal muscle cells and its relationship to apoptosis. Steroids 2008, 73, 859–863. [Google Scholar] [CrossRef] [PubMed]
- Vasconsuelo, A.; Milanesi, L.; Boland, R. Participation of HSP27 in the antiapoptotic action of 17β-estradiol in skeletal muscle cells. Cell Stress Chaperones 2010, 15, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Bopassa, J.C.; Eghbali, M.; Toro, L.; Stefani, E. A novel estrogen receptor GPER inhibits mitochondria permeability transition pore opening and protects the heart against ischemia-reperfusion injury. Am. J. Physiol. Heart Circ. Physiol. 2010, 298, H16–H23. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.-C.; Yu, H.-P.; Suzuki, T.; Choudhry, M.A.; Schwacha, M.G.; Bland, K.I.; Chaudry, I.H. Upregulation of mitochondrial respiratory complex IV by estrogen receptor-β is critical for inhibiting mitochondrial apoptotic signaling and restoring cardiac functions following trauma–hemorrhage. J. Mol. Cell. Cardiol. 2006, 41, 511–521. [Google Scholar] [CrossRef] [PubMed]
- Campbell, S.; Febbraio, M. Effect of ovarian hormones on mitochondrial enzyme activity in the fat oxidation pathway of skeletal muscle. Am. J. Physiol. -Endocrinol. Metab. 2001, 281, E803–E808. [Google Scholar] [CrossRef]
- Vasconsuelo, A.; Milanesi, L.; Boland, R. Actions of 17β-estradiol and testosterone in the mitochondria and their implications in aging. Ageing Res. Rev. 2013, 12, 907–917. [Google Scholar] [CrossRef]
- Cerf, M.E. Beta cell dysfunction and insulin resistance. Front. Endocrinol. 2013, 4, 37. [Google Scholar] [CrossRef]
- Kumar, P.; Sait, S.F. Luteinizing hormone and its dilemma in ovulation induction. J. Hum. Reprod. Sci. 2011, 4, 2. [Google Scholar] [CrossRef]
- Sharma, G.; Hu, C.; Brigman, J.L.; Zhu, G.; Hathaway, H.J.; Prossnitz, E.R. GPER deficiency in male mice results in insulin resistance, dyslipidemia, and a proinflammatory state. Endocrinology 2013, 154, 4136–4145. [Google Scholar] [CrossRef]
- Prossnitz, E.R.; Hathaway, H.J. What have we learned about GPER function in physiology and disease from knockout mice? J. Steroid Biochem. Mol. Biol. 2015, 153, 114–126. [Google Scholar] [CrossRef] [PubMed]
- Longuet, C.; Broca, C.; Costes, S.; Hani, E.H.; Bataille, D.; Dalle, S.P. Extracellularly regulated kinases 1/2 (p44/42 mitogen-activated protein kinases) phosphorylate synapsin I and regulate insulin secretion in the MIN6 β-cell line and islets of Langerhans. Endocrinology 2005, 146, 643–654. [Google Scholar] [CrossRef] [PubMed]
- Mauvais-Jarvis, F. Estrogen and androgen receptors: Regulators of fuel homeostasis and emerging targets for diabetes and obesity. Trends Endocrinol. Metab. 2011, 22, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Zhu, P.; Yuen, J.M.; Sham, K.W.; Cheng, C.H. GPER mediates the inhibitory actions of estrogen on adipogenesis in 3T3-L1 cells through perturbation of mitotic clonal expansion. Gen. Comp. Endocrinol. 2013, 193, 19–26. [Google Scholar] [CrossRef]
- Liu, Y.-X.; Zhang, Y.; Li, Y.-Y.; Liu, X.-M.; Wang, X.-X.; Zhang, C.-L.; Hao, C.-F.; Deng, S.-L. Regulation of follicular development and differentiation by intra-ovarian factors and endocrine hormones. Front. Biosci. 2019, 24, 983–993. [Google Scholar] [CrossRef]
- Patel, B.; Elguero, S.; Thakore, S.; Dahoud, W.; Bedaiwy, M.; Mesiano, S. Role of nuclear progesterone receptor isoforms in uterine pathophysiology. Hum. Reprod. Update 2015, 21, 155–173. [Google Scholar] [CrossRef]
- Gellersen, B.; Brosens, J.J. Cyclic decidualisation of the human endometrium in reproductive health and failure. Endocr. Rev. 2014, 35, 851–905. [Google Scholar] [CrossRef]
- Wilkens, J.; Male, V.; Ghazal, P.; Forster, T.; Gibson, D.; Williams, A.; Brito-Mutunayagam, S.; Craigon, M.; Lourenco, P.; Cameron, I. Bleeding in women and are suppressed by uterine NK cells regulate endometrial the progesterone receptor modulator asoprisnil. J. Immunol. 2013, 191, 2226–2235. [Google Scholar] [CrossRef]
- Tan, H.; Yi, L.; Rote, N.S.; Hurd, W.W.; Mesiano, S. Progesterone receptor-A and-B have opposite effects on proinflammatory gene expression in human myometrial cells: Implications for progesterone actions in human pregnancy and parturition. J. Clin. Endocrinol. 2012, 97, E719–E730. [Google Scholar] [CrossRef]
- Cha, J.; Sun, X.; Dey, S.K. Mechanisms of implantation: Strategies for successful pregnancy. Nat. Med. 2012, 18, 1754–1767. [Google Scholar] [CrossRef]
- Stein, D. Wright DW, Kellermann AL. Does progesterone have neuroprotective properties. Ann. Emerg. Med. 2008, 51, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Cooke, P.S.; Nanjappa, M.K.; Yang, Z.; Wang, K.K. Therapeutic effects of progesterone and its metabolites in traumatic brain injury may involve non-classical signaling mechanisms. Front. Neurosci. 2013, 7, 108. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.M.; Irwin, R.W.; Liu, L.; Chen, S.; Brinton, R.D. Regeneration in a degenerating brain: Potential of allopregnanolone as a neuroregenerative agent. Curr. Alzheimer Res. 2007, 4, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, A.N.; Siddiqui, N.; Khan, R.A.; Kalam, A.; Jabir, N.R.; Kamal, M.A.; Firoz, C.K.; Tabrez, S. Neuroprotective role of steroidal sex hormones: An overview. CNS Neurosci. Ther. 2016, 22, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Wyse Jackson, A.C.; Roche, S.L.; Byrne, A.M.; Ruiz-Lopez, A.M.; Cotter, T.G. Progesterone receptor signalling in retinal photoreceptor neuroprotection. J. Neurochem. 2016, 136, 63–77. [Google Scholar] [CrossRef]
- Lee, S.R.; Choi, W.-Y.; Heo, J.H.; Huh, J.; Kim, G.; Lee, K.-P.; Kwun, H.-J.; Shin, H.-J.; Baek, I.-J.; Hong, E.-J. Progesterone increases blood glucose via hepatic progesterone receptor membrane component 1 under limited or impaired action of insulin. Sci. Rep. 2020, 10, 11. [Google Scholar] [CrossRef]
- Rebarber, A.; Istwan, N.B.; Russo-Stieglitz, K.; Cleary-Goldman, J.; Rhea, D.J.; Stanziano, G.J.; Saltzman, D.H. Increased incidence of gestational diabetes in women receiving prophylactic 17α-hydroxyprogesterone caproate for prevention of recurrent preterm delivery. Diabetes Care 2007, 30, 2277–2280. [Google Scholar] [CrossRef]
- Picard, F.; Wanatabe, M.; Schoonjans, K.; Lydon, J.; O’Malley, B.W.; Auwerx, J. Progesterone receptor knockout mice have an improved glucose homeostasis secondary to β-cell proliferation. Proc. Natl. Acad. Sci. USA 2002, 99, 15644–15648. [Google Scholar] [CrossRef]
- Boonyaratanakornkit, V.; Pateetin, P. The role of ovarian sex steroids in metabolic homeostasis, obesity, and postmenopausal breast cancer: Molecular mechanisms and therapeutic implications. BioMed Res. Int. 2015, 2015, 140196. [Google Scholar] [CrossRef]
- Saunders, P.T.; Millar, M.R.; Williams, K.; Macpherson, S.; Harkiss, D.; Anderson, R.A.; Orr, B.; Groome, N.P.; Scobie, G.; Fraser, H.M. Differential expression of estrogen receptor-α and-β and androgen receptor in the ovaries of marmosets and humans. Biol. Reprod. 2000, 63, 1098–1105. [Google Scholar] [CrossRef]
- Horne, A.W.; King, A.E.; Shaw, E.; McDonald, S.E.; Williams, A.R.; Saunders, P.T.; Critchley, H.O. Attenuated sex steroid receptor expression in fallopian tube of women with ectopic pregnancy. J. Clin. Endocrinol. Metab. 2009, 94, 5146–5154. [Google Scholar] [CrossRef] [PubMed]
- Marshall, E.; Lowrey, J.; MacPherson, S.; Maybin, J.A.; Collins, F.; Critchley, H.O.; Saunders, P.T. In silico analysis identifies a novel role for androgens in the regulation of human endometrial apoptosis. J. Clin. Endocrinol. Metab. 2011, 96, E1746–E1755. [Google Scholar] [CrossRef] [PubMed]
- Dart, D.A.; Waxman, J.; Aboagye, E.O.; Bevan, C.L. Visualising androgen receptor activity in male and female mice. PLoS ONE 2013, 8, e71694. [Google Scholar] [CrossRef] [PubMed]
- Hillier, S.G.; Tetsuka, M.; Fraser, H.M. Location and developmental regulation of androgen receptor in primate ovary. Hum. Reprod. 1997, 12, 107–111. [Google Scholar] [CrossRef]
- Weil, S.; Vendola, K.; Zhou, J.; Bondy, C.A. Androgen and follicle-stimulating hormone interactions in primate ovarian follicle development. J. Clin. Endocrinol. Metab. 1999, 84, 2951–2956. [Google Scholar] [CrossRef]
- Astapova, O.; Minor, B.M.; Hammes, S.R. Physiological and pathological androgen actions in the ovary. Endocrinology 2019, 160, 1166–1174. [Google Scholar] [CrossRef]
- Shang, K.; Jia, X.; Qiao, J.; Kang, J.; Guan, Y. Endometrial abnormality in women with polycystic ovary syndrome. Reprod. Sci. 2012, 19, 674–683. [Google Scholar] [CrossRef]
- Dennett, C.C.; Simon, J. The role of polycystic ovary syndrome in reproductive and metabolic health: Overview and approaches for treatment. Diabetes Spectr. 2015, 28, 116–120. [Google Scholar] [CrossRef]
- Hager, M.; Wenzl, R.; Riesenhuber, S.; Marschalek, J.; Kuessel, L.; Mayrhofer, D.; Ristl, R.; Kurz, C.; Ott, J. The Prevalence of Incidental Endometriosis in Women Undergoing Laparoscopic Ovarian Drilling for Clomiphene-Resistant Polycystic Ovary Syndrome: A Retrospective Cohort Study and Meta-Analysis. J. Clin. Med. 2019, 8, 1210. [Google Scholar] [CrossRef]
- Isobe, N.; Yoshimura, Y. Deficient proliferation and apoptosis in the granulosa and theca interna cells of the bovine cystic follicle. J. Reprod. Dev. 2007, 53, 1119–1124. [Google Scholar] [CrossRef]
- Hamilton, K.J.; Arao, Y.; Korach, K.S. Estrogen hormone physiology: Reproductive findings from estrogen receptor mutant mice. Reprod. Biol. 2014, 14, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Jakimiuk, A.J.; Weitsman, S.R.; Yen, H.W.; Bogusiewicz, M.; Magoffin, D.A. Estrogen receptor alpha and beta expression in theca and granulosa cells from women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2002, 87, 5532–5538. [Google Scholar] [CrossRef] [PubMed]
- Rumi, M.K.; Dhakal, P.; Kubota, K.; Chakraborty, D.; Lei, T.; Larson, M.A.; Wolfe, M.W.; Roby, K.F.; Vivian, J.L.; Soares, M.J. Generation of Esr1-knockout rats using zinc finger nuclease-mediated genome editing. Endocrinology 2014, 155, 1991–1999. [Google Scholar] [CrossRef]
- Quaynor, S.D.; Stradtman, E.W., Jr.; Kim, H.-G.; Shen, Y.; Chorich, L.P.; Schreihofer, D.A.; Layman, L.C. Delayed puberty and estrogen resistance in a woman with estrogen receptor α variant. New Engl. J. Med. 2013, 369, 164–171. [Google Scholar] [CrossRef]
- Tang, Z.-R.; Zhang, R.; Lian, Z.-X.; Deng, S.-L.; Yu, K. Estrogen-receptor expression and function in female reproductive disease. Cells 2019, 8, 1123. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, S.C.; Winuthayanon, W.; Korach, K.S. What’s new in estrogen receptor action in the female reproductive tract. J. Mol. Endocrinol. 2016, 56, R55–R71. [Google Scholar] [CrossRef] [PubMed]
- Harris, H.A.; Bruner-Tran, K.L.; Zhang, X.; Osteen, K.G.; Lyttle, C.R. A selective estrogen receptor-β agonist causes lesion regression in an experimentally induced model of endometriosis. Hum. Reprod. 2005, 20, 936–941. [Google Scholar] [CrossRef]
- Couse, J.; Yates, M.; Deroo, B.; Korach, K. Estrogen receptor beta augments gonadotropin-induced granulose cell differentiation and pre-ovulatory response to gonadotropins. Endocrinology 2005, 146, 3247–3262. [Google Scholar] [CrossRef]
- Zhang, H.; Yi, M.; Zhang, Y.; Jin, H.; Zhang, W.; Yang, J.; Yan, L.; Li, R.; Zhao, Y.; Qiao, J. High-fat diets exaggerate endocrine and metabolic phenotypes in a rat model of DHEA-induced PCOS. Reproduction 2016, 151, 431–441. [Google Scholar] [CrossRef]
- Majumder, S.; Das, S.; Moulik, S.R.; Mallick, B.; Pal, P.; Mukherjee, D. G-protein coupled estrogen receptor (GPER) inhibits final oocyte maturation in common carp, Cyprinus carpio. Gen. Comp. Endocrinol. 2015, 211, 28–38. [Google Scholar] [CrossRef]
- McPhaul, M.J. Androgen receptor mutations and androgen insensitivity. Mol. Cell. Endocrinol. 2002, 198, 61–67. [Google Scholar] [CrossRef]
- Walters, K.; Handelsman, D. Role of androgens in the ovary. Mol. Cell. Endocrinol. 2018, 465, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Haas, E.; Bhattacharya, I.; Brailoiu, E.; Damjanovic, M.; Brailoiu, G.C.; Gao, X.; Mueller-Guerre, L.; Marjon, N.A.; Gut, A.; Minotti, R.; et al. Regulatory role of G protein-coupled estrogen receptor for vascular function and obesity. Circ. Res. 2009, 104, 288–291. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Le May, C.; Wong, W.P.; Ward, R.D.; Clegg, D.J.; Marcelli, M.; Korach, K.S.; Mauvais-Jarvis, F. Importance of extranuclear estrogen receptor-α and membrane G protein–coupled estrogen receptor in pancreatic islet survival. Diabetes 2009, 58, 2292–2302. [Google Scholar] [CrossRef] [PubMed]
- Martensson, U.E.; Salehi, S.A.; Windahl, S.; Gomez, M.F.; Swärd, K.; Daszkiewicz-Nilsson, J.; Wendt, A.; Andersson, N.; Hellstrand, P.; Grände, P.-O. Deletion of the G protein-coupled receptor 30 impairs glucose tolerance, reduces bone growth, increases blood pressure, and eliminates estradiol-stimulated insulin release in female mice. Endocrinology 2009, 150, 687–698. [Google Scholar] [CrossRef]
- Lancaster, T.; Jefferson, S.J.; Hunter, J.C.; Lopez, V.; Van Eyk, J.; Lakatta, E.G.; Korzick, D.H. Quantitative proteomic analysis reveals novel mitochondrial targets of estrogen deficiency in the aged female rat heart. Physiol. Genom. 2012, 44, 957–969. [Google Scholar] [CrossRef]
- Meng, Z.; Jing, H.; Gan, L.; Li, H.; Luo, B. Resveratrol attenuated estrogen-deficient-induced cardiac dysfunction: Role of AMPK, SIRT1, and mitochondrial function. Am. J. Transl. Res. 2016, 8, 2641. [Google Scholar]
- Rattanasopa, C.; Phungphong, S.; Wattanapermpool, J.; Bupha-Intr, T. Significant role of estrogen in maintaining cardiac mitochondrial functions. J. Steroid Biochem. Mol. Biol. 2015, 147, 9. [Google Scholar] [CrossRef]
- Sbert-Roig, M.; Bauzá-Thorbrügge, M.; Galmés-Pascual, B.M.; Capllonch-Amer, G.; García-Palmer, F.J.; Lladó, I.; Proenza, A.M.; Gianotti, M. GPER mediates the effects of 17β-estradiol in cardiac mitochondrial biogenesis and function. Mol. Cell. Endocrinol. 2016, 420, 116–124. [Google Scholar] [CrossRef]
- Sun, L.-Y.; Wang, N.; Ban, T.; Sun, Y.-H.; Han, Y.; Sun, L.-L.; Yan, Y.; Kang, X.-H.; Chen, S.; Sun, L.-H. MicroRNA-23a mediates mitochondrial compromise in estrogen deficiency-induced concentric remodeling via targeting PGC-1α. J. Mol. Cell. Cardiol. 2014, 75, 11. [Google Scholar] [CrossRef]
- Sheppard, K.M.; Padmanabhan, V.; Coolen, L.M.; Lehman, M.N. Prenatal programming by testosterone of hypothalamic metabolic control neurones in the ewe. J. Neuroendocrinol. 2011, 23, 401–411. [Google Scholar] [CrossRef]
- Dumesic, D.A.; Akopians, A.L.; Madrigal, V.K.; Ramirez, E.; Margolis, D.J.; Sarma, M.K.; Thomas, A.M.; Grogan, T.R.; Haykal, R.; Schooler, T.A. Hyperandrogenism accompanies increased intra-abdominal fat storage in normal weight polycystic ovary syndrome women. J. Clin. Endocrinol. Metab. 2016, 101, 4178–4188. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, A.S.; Edwards, M.C.; Desai, R.; Jimenez, M.; Gilchrist, R.B.; Handelsman, D.J.; Walters, K.A. Neuroendocrine androgen action is a key extraovarian mediator in the development of polycystic ovary syndrome. Proc. Natl. Acad. Sci. USA 2017, 114, E3334–E3343. [Google Scholar] [CrossRef] [PubMed]
- Yanes, L.L.; Romero, D.G.; Moulana, M.; Lima, R.; Davis, D.D.; Zhang, H.; Lockhart, R.; Racusen, L.C.; Reckelhoff, J.F. Cardiovascular-renal and metabolic characterization of a rat model of polycystic ovary syndrome. Gend. Med. 2011, 8, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Mishra, J.S.; More, A.S.; Hankins, G.D.; Kumar, S. Hyperandrogenemia reduces endothelium-derived hyperpolarizing factor-mediated relaxation in mesenteric artery of female rats. Biol. Reprod. 2017, 96, 1221–1230. [Google Scholar] [CrossRef]
- Torres Fernandez, E.D.; Adams, K.V.; Syed, M.; Maranon, R.O.; Romero, D.G.; Yanes Cardozo, L.L. Long-Lasting Androgen-Induced Cardiometabolic Effects in Polycystic Ovary Syndrome. J. Endocr. Soc. 2018, 2, 949–964. [Google Scholar] [CrossRef]
- Davis, G.K.; Intapad, S.; Newsome, A.D.; Coats, L.E.; Bamrick, D.R.; Ojeda, N.B.; Alexander, B.T. Androgen receptor blockade differentially regulates blood pressure in growth-restricted versus ovarian deficient rats. Hypertension 2019, 74, 975–982. [Google Scholar] [CrossRef]
- van Hoesel, M.H.; Chen, Y.L.; Zheng, A.; Wan, Q.; Mourad, S.M. Selective oestrogen receptor modulators (SERMs) for endometriosis. Cochrane Database Syst. Rev. 2021, 5, CD011169. [Google Scholar]
- Bulun, S.E.; Monsavais, D.; Pavone, M.E.; Dyson, M.; Xue, Q.; Attar, E.; Tokunaga, H.; Su, E.J. Role of estrogen receptor-β in endometriosis. Semin. Reprod. Med. 2012, 30, 39–45. [Google Scholar] [CrossRef]
- Burns, K.A.; Rodriguez, K.F.; Hewitt, S.C.; Janardhan, K.S.; Young, S.L.; Korach, K.S. Role of estrogen receptor signaling required for endometriosis-like lesion establishment in a mouse model. Endocrinology 2012, 153, 3960–3971. [Google Scholar] [CrossRef]
- Burney, R.O.; Talbi, S.; Hamilton, A.E.; Vo, K.C.; Nyegaard, M.; Nezhat, C.R.; Lessey, B.A.; Giudice, L.C. Gene expression analysis of endometrium reveals progesterone resistance and candidate susceptibility genes in women with endometriosis. Endocrinology 2007, 148, 3814–3826. [Google Scholar] [CrossRef] [PubMed]
- Han, S.J.; Jung, S.Y.; Wu, S.-P.; Hawkins, S.M.; Park, M.J.; Kyo, S.; Qin, J.; Lydon, J.P.; Tsai, S.Y.; Tsai, M.-J. Estrogen receptor β modulates apoptosis complexes and the inflammasome to drive the pathogenesis of endometriosis. Cell 2015, 163, 960–974. [Google Scholar] [CrossRef] [PubMed]
- Plante, B.J.; Lessey, B.A.; Taylor, R.N.; Wang, W.; Bagchi, M.K.; Yuan, L.; Scotchie, J.; Fritz, M.A.; Young, S.L. G protein-coupled estrogen receptor (GPER) expression in normal and abnormal endometrium. Reprod. Sci. 2012, 19, 684–693. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, C.; Gori, I.; Achtari, C.; Hornung, D.; Chardonnens, E.; Wunder, D.; Fiche, M.; Canny, G.O. The expression of estrogen receptors as well as GREB1, c-MYC, and cyclin D1, estrogen-regulated genes implicated in proliferation, is increased in peritoneal endometriosis. Fertil. Steril. 2012, 98, 1200–1208. [Google Scholar] [CrossRef]
- Xue, Q.; Lin, Z.; Cheng, Y.-H.; Huang, C.-C.; Marsh, E.; Yin, P.; Milad, M.P.; Confino, E.; Reierstad, S.; Innes, J. Promoter methylation regulates estrogen receptor 2 in human endometrium and endometriosis. Biol. Reprod. 2007, 77, 681–687. [Google Scholar] [CrossRef]
- Montagna, P.; Capellino, S.; Villaggio, B.; Remorgida, V.; Ragni, N.; Cutolo, M.; Ferrero, S. Peritoneal fluid macrophages in endometriosis: Correlation between the expression of estrogen receptors and inflammation. Fertil. Steril. 2008, 90, 156–164. [Google Scholar] [CrossRef]
- Bedaiwy, M.A.; Dahoud, W.; Skomorovska-Prokvolit, Y.; Yi, L.; Liu, J.H.; Falcone, T.; Hurd, W.W.; Mesiano, S. Abundance and localization of progesterone receptor isoforms in endometrium in women with and without endometriosis and in peritoneal and ovarian endometriotic implants. Reprod. Sci. 2015, 22, 1153–1161. [Google Scholar] [CrossRef]
- Bukulmez, O.; Hardy, D.B.; Carr, B.R.; Word, R.A.; Mendelson, C.R. Inflammatory status influences aromatase and steroid receptor expression in endometriosis. Endocrinology 2008, 149, 1190–1204. [Google Scholar] [CrossRef]
- Vinci, G.; Arkwright, S.; Audebourg, A.; Radenen, B.; Chapron, C.; Borghese, B.; Dousset, B.; Mehats, C.; Vaiman, D.; Vacher-Lavenu, M.-C. Correlation between the clinical parameters and tissue phenotype in patients affected by deep-infiltrating endometriosis. Reprod. Sci. 2016, 23, 1258–1268. [Google Scholar] [CrossRef]
- Chaudhary, S.C.; Singh, T.; Talwelkar, S.S.; Srivastava, R.K.; Arumugam, A.; Weng, Z.; Elmets, C.A.; Afaq, F.; Kopelovich, L.; Athar, M. Erb-041, an estrogen receptor-β agonist, inhibits skin photocarcinogenesis in SKH-1 hairless mice by downregulating the WNT signaling pathway. Cancer Prev. Res. 2014, 7, 186–198. [Google Scholar] [CrossRef]
- Roman-Blas, J.A.; Castañeda, S.; Cutolo, M.; Herrero-Beaumont, G. Efficacy and safety of a selective estrogen receptor β agonist, ERB-041, in patients with rheumatoid arthritis: A 12-week, randomized, placebo-controlled, phase II study. Arthritis Care Res. 2010, 62, 1588–1593. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.; Prossnitz, E.R. GPER/GPR30 knockout mice: Effects of GPER on metabolism. In Estrogen Receptors; Springer: Berlin/Heidelberg, Germany, 2016; pp. 489–502. [Google Scholar]
- Kim, J.B. Dynamic cross talk between metabolic organs in obesity and metabolic diseases. Exp. Mol. Med. 2016, 48, e214. [Google Scholar] [CrossRef] [PubMed]
- Falconer, H.; Mwenda, J.M.; Chai, D.C.; Wagner, C.; Song, X.; Mihalyi, A.; Simsa, P.; Kyama, C.; Cornillie, F.; Bergqvist, A. Treatment with anti-TNF monoclonal antibody (c5N) reduces the extent of induced endometriosis in the baboon. Hum. Reprod. 2006, 21, 1856–1862. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Gong, P.; Chen, Y.; Nwachukwu, J.C.; Srinivasan, S.; Ko, C.; Bagchi, M.K.; Taylor, R.N.; Korach, K.S.; Nettles, K.W. Dual suppression of estrogenic and inflammatory activities for targeting of endometriosis. Sci. Transl. Med. 2015, 7, 271ra9. [Google Scholar] [CrossRef]
- Khine, Y.M.; Taniguchi, F.; Nagira, K.; Nakamura, K.; Ohbayashi, T.; Osaki, M.; Harada, T. New insights into the efficacy of SR-16234, a selective estrogen receptor modulator, on the growth of murine endometriosis-like lesions. Am. J. Reprod. Immunol. 2018, 80, e13023. [Google Scholar] [CrossRef]
- Tanbo, T.; Mellembakken, J.; Bjercke, S.; Ring, E.; Åbyholm, T.; Fedorcsak, P. Ovulation induction in polycystic ovary syndrome. Acta Obstet. Et Gynecol. Scand. 2018, 97, 1162–1167. [Google Scholar] [CrossRef]
- Jones, T.; Ho, J.R.; Gualtieri, M.; Bruno-Gaston, J.; Chung, K.; Paulson, R.J.; Bendikson, K.A. Clomiphene stair-step protocol for women with polycystic ovary syndrome. Obstet. Gynecol. 2018, 131, 91–95. [Google Scholar] [CrossRef]
- Chaabane, S.; Sheehy, O.; Monnier, P.; Fraser, W.; Bissonnette, F.; Trasler, J.M.; Muanda, F.T.; Boukhris, T.; Karam, F.; Santos, F. Ovarian stimulation, intrauterine insemination, multiple pregnancy and major congenital malformations: A systematic review and meta-analysis-The ART_Rev Study. Curr. Drug Saf. 2016, 11, 222–261. [Google Scholar] [CrossRef]
- Coughlan, C.; Fitzgerald, J.; Milne, P.; Wingfield, M. Is it safe to prescribe clomiphene citrate without ultrasound monitoring facilities? J. Obstet. Gynaecol. 2010, 30, 393–396. [Google Scholar] [CrossRef]
- Perales-Puchalt, A.; Legro, R.S. Ovulation induction in women with polycystic ovary syndrome. Steroids 2013, 78, 767–772. [Google Scholar] [CrossRef]
- Sharma, G.; Prossnitz, E.R. Mechanisms of estradiol-induced insulin secretion by the G protein-coupled estrogen receptor GPR30/GPER in pancreatic β-cells. Endocrinology 2011, 152, 3030–3039. [Google Scholar] [CrossRef] [PubMed]
- Tica, A.A.; Dun, E.C.; Tica, O.S.; Gao, X.; Arterburn, J.B.; Brailoiu, G.C.; Oprea, T.I.; Brailoiu, E. G protein-coupled estrogen receptor 1-mediated effects in the rat myometrium. Am. J. Physiol. Cell Physiol. 2011, 301, C1262–C1269. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wagenfeld, A.; Bone, W.; Schwede, W.; Fritsch, M.; Fischer, O.M.; Moeller, C. BAY 1002670: A novel, highly potent and selective progesterone receptor modulator for gynaecological therapies. Hum. Reprod. 2013, 28, 2253–2264. [Google Scholar] [CrossRef]
- Bradley, L.; Ren, X.; Groettrup-Wolfers, E.; Petersdorf, K.; Seitz, C. Results of the asteroid (assess safety and efficacy of vilaprisan in patients with uterine fibroids) 1 study: A phase 2, placebo-controlled dose finding study. Fertil. Steril. 2016, 106, e95–e96. [Google Scholar] [CrossRef]
- Schütt, B.; Schultze-Mosgau, M.H.; Draeger, C.; Chang, X.; Löwen, S.; Kaiser, A.; Rohde, B. Effect of the novel selective progesterone receptor modulator vilaprisan on ovarian activity in healthy women. J. Clin. Pharmacol. 2018, 58, 228–239. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Afrin, S.; Jones, S.I.; Segars, J. Selective progesterone receptor modulators—Mechanisms and therapeutic utility. Endocr. Rev. 2020, 41, 643–694. [Google Scholar] [CrossRef]
- Schultze-Mosgau, M.-H.; Schuett, B.; Hafner, F.-T.; Zollmann, F.; Kaiser, A.; Hoechel, J.; Rohde, B. Pharmacokinetics and safety of the selective progesterone receptor modulator vilaprisan in healthy postmenopausal women. Int. J. Clin. Pharmacol. Ther. 2017, 55, 16. [Google Scholar] [CrossRef]
- Calaf, J.; Lopez, E.; Millet, A.; Alcañiz, J.; Fortuny, A.; Vidal, O.; Callejo, J.; Escobar-Jimenez, F.; Torres, E.; Espinos, J. Long-term efficacy and tolerability of flutamide combined with oral contraception in moderate to severe hirsutism: A 12-month, double-blind, parallel clinical trial. J. Clin. Endocrinol. Metab. 2007, 92, 3446–3452. [Google Scholar] [CrossRef]
- Paradisi, R.; Fabbri, R.; Battaglia, C.; Venturoli, S. Ovulatory effects of flutamide in the polycystic ovary syndrome. Gynecol. Endocrinol. 2013, 29, 391–395. [Google Scholar] [CrossRef]
- Diamanti-Kandarakis, E.; Mitrakou, A.; Raptis, S.; Tolis, G.; Duleba, A.J. The effect of a pure antiandrogen receptor blocker, flutamide, on the lipid profile in the polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 1998, 83, 2699–2705. [Google Scholar] [CrossRef][Green Version]
- Conneely, O.M.; Lydon, J.P. Progesterone receptors in reproduction: Functional impact of the A and B isoforms. Steroids 2000, 65, 571–577. [Google Scholar] [CrossRef]
- Kaya, H.S.; Hantak, A.M.; Stubbs, L.J.; Taylor, R.N.; Bagchi, I.C.; Bagchi, M.K. Roles of progesterone receptor A and B isoforms during human endometrial decidualization. Mol. Endocrinol. 2015, 29, 882–895. [Google Scholar] [CrossRef] [PubMed]
- Babayev, S.N.; Park, C.W.; Keller, P.W.; Carr, B.R.; Word, R.A.; Bukulmez, O. Androgens upregulate endometrial epithelial progesterone receptor expression: Potential implications for endometriosis. Reprod. Sci. 2017, 24, 1454–1461. [Google Scholar] [CrossRef] [PubMed]
- Iwai, M.; Kanzaki, H.; Fujimoto, M.; Kojima, K.; Hatayama, H.; Inoue, T.; Higuchi, T.; Nakayama, H.; Mori, T.; Fujita, J. Regulation of sex steroid receptor gene expression by progesterone and testosterone in cultured human endometrial stromal cells. J. Clin. Endocrinol. Metab. 1995, 80, 450–454. [Google Scholar] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdul Hamid, F.; Abu, M.A.; Abdul Karim, A.K.; Ahmad, M.F.; Abd. Aziz, N.H.; Mohd Kamal, D.A.; Mokhtar, M.H. Sex Steroid Receptors in Polycystic Ovary Syndrome and Endometriosis: Insights from Laboratory Studies to Clinical Trials. Biomedicines 2022, 10, 1705. https://doi.org/10.3390/biomedicines10071705
Abdul Hamid F, Abu MA, Abdul Karim AK, Ahmad MF, Abd. Aziz NH, Mohd Kamal DA, Mokhtar MH. Sex Steroid Receptors in Polycystic Ovary Syndrome and Endometriosis: Insights from Laboratory Studies to Clinical Trials. Biomedicines. 2022; 10(7):1705. https://doi.org/10.3390/biomedicines10071705
Chicago/Turabian StyleAbdul Hamid, Fazilah, Muhammad Azrai Abu, Abdul Kadir Abdul Karim, Mohd Faizal Ahmad, Nor Haslinda Abd. Aziz, Datu Agasi Mohd Kamal, and Mohd Helmy Mokhtar. 2022. "Sex Steroid Receptors in Polycystic Ovary Syndrome and Endometriosis: Insights from Laboratory Studies to Clinical Trials" Biomedicines 10, no. 7: 1705. https://doi.org/10.3390/biomedicines10071705
APA StyleAbdul Hamid, F., Abu, M. A., Abdul Karim, A. K., Ahmad, M. F., Abd. Aziz, N. H., Mohd Kamal, D. A., & Mokhtar, M. H. (2022). Sex Steroid Receptors in Polycystic Ovary Syndrome and Endometriosis: Insights from Laboratory Studies to Clinical Trials. Biomedicines, 10(7), 1705. https://doi.org/10.3390/biomedicines10071705






