A Narrative Review of 99mTc-Aprotinin in the Diagnosis of Cardiac Amyloidosis and a New Life for an Unfairly Abandoned Drug
Abstract
1. Introduction
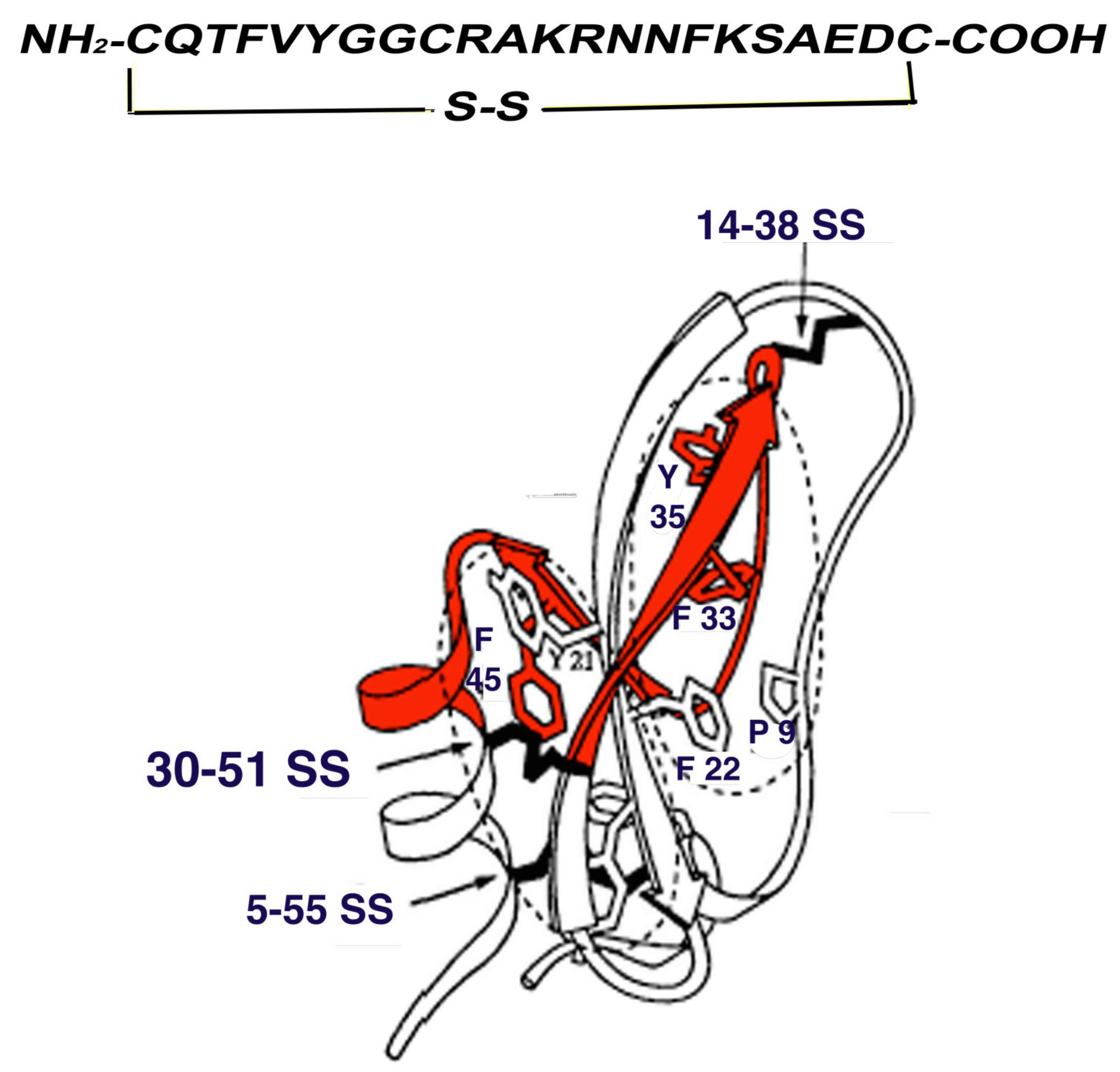
2. Binding Mechanism and Specificity
3. Radiopharmacokinetics
4. Endomyocardial Biopsy
5. Diagnosis, Risk Stratification, and Long-Term Follow-Up
5.1. Referring Centers and Technical Approach
5.2. Clinical Cases Reports
5.3. Sensitivity and Specificity
5.4. Unanswered Questions Arising from Clinical Studies
5.5. Limits of TcA
6. Safety
7. Dosimetry
8. Future Directions
8.1. Recombinant Aprotinin
8.2. Cyclic Peptide 30–51 SS
8.3. Ga-NOTA-Aprotinin
9. Labeled Aprotinin in the Present Amyloid Cardiac Imaging Landscape
10. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hawkins, P.N.; Levender, P.; Pepys, M.B. Evaluation of systemic amyloidosis by scintigraphy with I-123—labelled serum amyloid P component. N. Engl. J. Med. 1990, 325, 508–513. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, C.; Donadio, C.; Tramonti, G.; Lorusso, P.; Bellitto, L.; Lunghi, F. 99mTc-aprotinin: A new tracer for kidney morphology and function. Eur. J. Nucl. Med. 1984, 9, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Aprile, C.; Marinone, G.; Saponaro, R.; Bonino, C.; Merlini, G. Cardiac and pleuropulmonary AL amyloid imaging with technetium-99m labeled aprotinin. Eur. J. Nucl. Med. 1995, 22, 1393–1401. [Google Scholar] [CrossRef] [PubMed]
- Fergusson, D.; Hébert, P.C.; Mazer, C.D.; Fremes, S.; MacAdams, C.; Murkin, J.M.; Teoh, K.; Duke, P.C.; Arellano, R.; Blajchman, M.A.; et al. A comparison of aprotinin and lysine analogues in high-risk cardiac surgery. N. Engl. J. Med. 2008, 358, 2319–2331. [Google Scholar] [CrossRef] [PubMed]
- Howell, N.; Senanayake, E.; Freemantle, N.; Pagano, D.J. Putting the record straight on aprotinin as safe and effective: Results from a mixed treatment meta-analysis of trials of aprotinin. Thorac. Cardiovasc. Surg. 2013, 145, 234–240. [Google Scholar] [CrossRef]
- EMA/CHMP/119704/2012 European Medicines Agency Recommends Lifting Suspension of Aprotinin. Available online: https://www.ema.europa.eu/en/documents/press-release/european-medicines-agency-recommends-lifting-suspension-aprotinin_en.pdf (accessed on 15 March 2022).
- Marcoux, J.; Mangione, P.P.; Porcari, R.; Degiacomi, M.; Verona, G.; Taylor, G.W.; Giorgetti, S.; Raimondi, S.; Cianferani, S.; Benesch, J.; et al. A novel mechano-enzymatic cleavage mechanism underlies transthyretin amyloidogenesis. EMBO Mol. Med. 2015, 7, 1337–1349. [Google Scholar] [CrossRef]
- Morgan, G.J.; Kelly, J.W. The kinetic stability of a full-length antibody light chain dimer determines whether endoproteolysis can release amyloidogenic variable domains. J. Mol. Biol. 2016, 428, 4280–4297. [Google Scholar] [CrossRef]
- Cardoso, I.; Pereira, P.J.; Damas, A.M.; Saraiva, M.J. Aprotinin binding to amyloid fibrils. Eur. J. Biochem. 2000, 267, 2307–2311. [Google Scholar] [CrossRef]
- Rusckowski, M.; Qu, T.; Chang, F.; Hnatowich, D.J. Technetium-99m labeled epidermal growth factor-tumor imaging in mice. J. Pept. Res. 1997, 50, 393–401. [Google Scholar] [CrossRef]
- Komarek, P.; Kleisner, I.; Komarkova, I.; Konopkova, M. Accumulation of radiolabelled low molecular peptides and proteins in experimental inflammation. Int. J. Pharm. 2005, 291, 119–125. [Google Scholar] [CrossRef]
- Aprile, C.; Saponaro, R.; Villa, G.; Lunghi, F. 99mTc-aprotinin: Comparison with 99mTc-DMSA in normal and diseased kidneys. Nuklearmedizin 1984, 23, 22–26. [Google Scholar]
- Sojan, S.M.; Smyth, D.R.; Tsopelas, C.; Mudge, D.; Collins, P.J.; Chatterton, B.E. Pharmacokinetics and normal scintigraphic appearance of 99mTc aprotinin. Nucl. Med. Commun. 2005, 26, 535–539. [Google Scholar] [CrossRef]
- Bellitto, L.; Donadio, C.; Tramonti, G.; Lorusso, P.; Lunghi, F.; Zito, F.; Bianchi, C. Aprotinin-99 m Tc: A new radiopharmaceutical for the study of kidney morphology and function. In Technetium in Chemistry and Nuclear Medicine; Deutsch, E., Nicolini, M., Wagner, H.N., Jr., Eds.; Cortina International: Verona, Italy, 1983; pp. 171–173. [Google Scholar]
- Smyth, D.R.; Tsopelas, C. An improved (99m)Tc-aprotinin kit formulation: Quality control analysis of radiotracer stability and cold kit shelf life. Nucl. Med. Biol. 2005, 32, 885–889. [Google Scholar] [CrossRef]
- Benson, M.D.; Berk, J.L.; Dispenzieri, A.; Damy, T.; Gillmore, J.D.; Hazenberg, B.P.; Lavatelli, F.; Picken, M.M.; Röcken, C.; Schönland, S.; et al. Tissue biopsy for the diagnosis of amyloidosis: Experience from some centres. Amyloid 2022, 29, 8–13. [Google Scholar] [CrossRef]
- Schaadt, B.K.; Hendel, H.W.; Gimsing, P.; Jonsson, V.; Pedersen, H.; Hesse, B. 99mTc-Aprotinin scintigraphy in amyloidosis. J. Nucl. Med. 2003, 44, 177–183. [Google Scholar]
- Han, S.; Chong, V.; Murray, T.; McDonagh, T.; Hunter, J.; Poon, F.W.; Gray, H.W.; Neilly, J.B. Preliminary experience of 99mTc-Aprotinin scintigraphy in amyloidosis. Eur. J. Haematol. 2007, 79, 494–500. [Google Scholar] [CrossRef]
- Merlini, G.; Palladini, G.; Obici, L.; Anesi, E.; Saponaro, R.; Cannizzaro, G.; Aprile, C. Accuracy of 99mTc-Aprotinin scintigraphy for the detection of myocardial amyloidosis: Long term follow-up of 78 patients. In Amyloid and Amyloidosis 2001. The Proceedings of the IXth International Symposium on Amyloidosis. Budapest, Hungary; Bély, M., Ágnes Apáthy, Á., Eds.; PXP Első Magyar Digitális Nyomda Rt.: Budapest, Hungary, 2001; pp. 174–175. [Google Scholar]
- Awaya, T.; Minamimoto, R.; Iwama, K.; Kubota, S.; Hotta, M.; Hirai, R.; Yamamoto, M.; Okazaki, O.; Hara, H.; Hiroi, Y.; et al. Performance of 99mTc-aprotinin scintigraphy for diagnosing light chain (AL) cardiac amyloidosis confirmed by endomyocardial biopsy. J. Nucl. Cardiol. 2020, 27, 1145–1153. [Google Scholar] [CrossRef]
- Johnston, A.; Ho Shon, I.; Coyle, L.; Tan, C.W.; Snowdon, G.; Cooper, B.; Schembri, G.; Roach, P. Technetium-99m Aprotinin Scintigraphy in Amyloidosis: A Pilot Study. Intern. Med. J. 2006, 36 (Suppl 3), A117. [Google Scholar] [CrossRef]
- Aprile, C.; Merlini, G.; Saponaro, R.; Cannizzaro, G.; Calsamiglia, G.; Anesi, E.; Garini, P. Aprotinin 99m Tc myocardial scan: Risk stratification of cardiac events in patients with AL/ATTR amyloidosis. In Radioactive Isotopes in Clinical Medicine and Research XXIII; Bergmann, H., Kohn, H., Sinziger, H., Eds.; Birkhauser Verlag: Basel, Switzerland, 1999; pp. 479–482. [Google Scholar]
- Minamimoto, R.; Kubota, K.; Ishii, K.; Morooka, M.; Okasaki, M.; Miyata, Y.; Nakajima, K.; Sato, T.; Igari, T.; Hirai, R.; et al. Re-evaluating the potentials and limitations of (99m)Tc-aprotinin scintigraphy for amyloid imaging. Am. J. Nucl. Med. Mol. Imaging 2013, 3, 261–271. [Google Scholar]
- Moore, S.C.; Southekal, S.; Park, M.A.; McQuaid, S.J.; Kijewski, M.F.; Müller, S.P. Improved regional activity quantitation in nuclear medicine using a new approach to correct for tissue partial volume and spillover effects. IEEE Trans. Med. Imaging 2012, 31, 405–416. [Google Scholar] [CrossRef]
- Pretorius, P.H.; King, M.A. Diminishing the impact of the partial volume effect in cardiac SPECT perfusion imaging. Med. Phys. 2009, 36, 105–115. [Google Scholar] [CrossRef]
- Pretorius, P.H.; King, M.A. Spillover Compensation in the Presence of Respiratory Motion Embedded in SPECT Perfusion Data. IEEE Trans. Nucl. Sci. 2008, 55, 537–542. [Google Scholar] [CrossRef]
- Aprile, C.; Villa, G.; Saponaro, R.; Piazza, V.; Suppi, A.; Jurisic, M.; Buonocore, M.; Marinone, M.G.; Poggio, F.; Salvadeo, A.; et al. A new approach to the detection of dialysis related amyloidosis (DRA) with 99 m Tc-aprotinin. Nephrol Dial Transplant. 1996, 11, A235. [Google Scholar] [CrossRef]
- Hnatowich, D.J.; Qu, T.; Chang, F.; Ley, A.C.; Ladner, R.C.; Rusckowski, M. Labeling peptides with technetium-99m using a bifunctional chelator of a N-hydroxysuccinimide ester of mercaptoacetyltriglycine. J. Nucl. Med. 1998, 39, 56–64. [Google Scholar]
- SMPC. Aprotinin 10,000 KIU/mL Injection BP-SPC 19th November 2020. Available online: https://www.medicines.org.uk/emc/product/2472/smpc/print (accessed on 15 March 2022).
- Kozak, R.W.; Golker, C.F.; Stadler, P. Transmissible spongiform encephalopathies (TSE): Minimizing the risk of transmission by biological/biopharmaceutical products: An industry perspective. Dev. Biol Stand. 1996, 88, 257–264. [Google Scholar]
- Berger, C.N.; Le Donne, P.; Windemann, H. Use of substances of animal origin in pharmaceutics and compliance with the TSE-risk guideline—A market survey. Biologicals 2005, 33, 1–7. [Google Scholar] [CrossRef]
- Aprile, C.; Saponaro, R.; Raimondi, S.; Galeone, M.; Bellotti, V.; Merlini, G. Recombinant Aprotinin (tobacco): Technetium Labelling and Amyloid Fibrils Affinity. Eur. J. Nucl. Med. Mol. Imaging 2007, 34, S148–S149. [Google Scholar] [CrossRef]
- Aprile, C.; Saponaro, R.; Buroni, F.E.; Lodola, L.; Raimondi, S.; Bellotti, V.; Cavenaghi, G.; Lavatelli, F.; Merlini, G.; Cassani, G. A 99mTc-Labeled Synthetic Cyclic Peptide (Aprotinin Analogue) with High Affinity for Amyloid Fibrils. Eur. J. Nucl. Med. Mol. Imaging 2011, 38, S145. [Google Scholar] [CrossRef]
- Buroni, F.E.; Lodola, L.; Aprile, C. A new PET radiopharmaceutical with high affinity for amyloid fibrils. Clin. Transl. Imaging 2013, 1, S26–S27. [Google Scholar] [CrossRef][Green Version]
- Slart, R.H.J.A.; Glaudemans, A.W.J.M.; Noordzij, W.; Bijzet, J.; Hazenberg, B.P.C.; Nienhuis, H.L.A. Time for new imaging and therapeutic approaches in cardiac amyloidosis. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1402–1406. [Google Scholar] [CrossRef] [PubMed]
- Saeed, S.; Saad, J.M.; Ahmed, A.I.; Han, Y.; Al-Mallah, M.H. The utility of positron emission tomography in cardiac amyloidosis. Heart Fail. Rev. 2021. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Castaño, A.; DeLuca, A.; Weinberg, R.; Pozniakoff, T.; Blaner, W.S.; Pirmohamed, A.; Bettencourt, B.; Gollob, J.; Karsten, V.; Vest, J.A.; et al. Serial scanning with technetium pyrophosphate (99mTc-PYP) in advanced ATTR cardiac amyloidosis. J. Nucl. Cardiol. 2016, 23, 1355–1363. [Google Scholar] [CrossRef] [PubMed]
- Buroni, F.E.; Persico, M.G.; Lodola, L.; Concardi, M.; Aprile, C. In vitro study: Binding of 99mTc-DPD to synthetic amyloid fibrils. Curr. Issues Pharm. Med. Sci. 2015, 28, 231–235. [Google Scholar] [CrossRef]
- Thelander, U.; Westermark, G.T.; Antoni, G.; Estrada, S.; Zancanaro, A.; Ihse, E.; Westermark, P. Cardiac microcalcifications in transthyretin (ATTR) amyloidosis. Int. J. Cardiol. 2022, 352, 84–91. [Google Scholar] [CrossRef]
- Minamimoto, R.; Awaya, T.; Iwama, K.; Hotta, M.; Nakajima, K.; Hirai, R.; Okazaki, O.; Hiroi, Y. Significance of 11C-PIB PET/CT in cardiac amyloidosis compared with 99mTc-aprotinin scintigraphy: A pilot study. J. Nucl. Cardiol. 2020, 27, 202–209. [Google Scholar] [CrossRef]
- Wall, J.S.; Martin, E.B.; Endsley, A.; Stuckey, A.C.; Williams, A.D.; Powell, D.; Whittle, B.; Hall, S.; Lambeth, T.R.; Julian, R.R.; et al. First in Human Evaluation and Dosimetry Calculations for Peptide (124) I-p5+14-a Novel Radiotracer for the Detection of Systemic Amyloidosis Using PET/CT Imaging. Mol. Imaging Biol. 2021. Online ahead of print. [Google Scholar] [CrossRef]
- Wall, J.S.; Heidel, R.E.; Martin, E.B.; Stuckey, A.; Powell, D.; Guthrie, S.; Lands, R.; Kennel, S.J. Detection of Cardiac Amyloidosis In Patients With Systemic AlL and ATTR Amyloidosis Using Iodine Evuzamitide PET/CT Imaging and The Correlation With Serum NT-proBNP. JACC 2022, 79, 1184. [Google Scholar] [CrossRef]
- Slart, R.H.J.A.; Glaudemans, A.W.J.M.; Noordzij, W.; Nienhuis, H.L.A.; Hazenberg, B.P.C. 99mTc-aprotinin imaging in cardiac amyloidosis. Make an old tool new again? J. Nucl. Cardiol. 2020, 27, 1155–1157. [Google Scholar] [CrossRef]

| Endomyocardial Biopsy | |||
|---|---|---|---|
| + | − | ||
| Tc-A scan | + | 16 | 0 |
| − | 1 | 11 | |
| Pre-Test Score (n) | Evaluable | Scan | Alive | Non Cardiac Death | Cardiac Death | CF Onset or Prog. | Lost at f.u. |
|---|---|---|---|---|---|---|---|
| A (34) | 32 | − | 26 | 3 | 2 | ||
| + | 1 | 1 | 1 | ||||
| B (15) | 13 | − | 2 | 2 | 2 | 1 | |
| + | 1 | 3 | 3 | 1 | |||
| C (12) | 12 | − | 1 | 1 | |||
| + | 5 | 1 | 4 | ||||
| D (17) | 15 | − | |||||
| + | 4 | 8 | 3 | 2 |
| Scan Results | Subjects under Study | [Ref] | |||
|---|---|---|---|---|---|
| TP | FP | TN | FN | ||
| 36 | 1 | 35 | 2 | n.. 74 AL biopsy-proven and long-term f.u. (3 localized, 2 MGUS) | [19] |
| 10 | - | 12 | - | n. 22 with known or suspected amyloidosis (14 AL, 2 hereditary form, 3 localized, 3 MM) | [17] |
| 18 | - | 17 | - | n. 35: n.18 biopsy-proven (14 AL, 3 AA, 1 ATTR), n 17 control. | [18] |
| 3 | - | 6 | 1 | n. 10 patients with biopsy-proven primary amyloidosis | [21] |
| - | - | 12 | - | n. 12 control subjects with cardiac non-amyloidosis pathology | [3] ^ |
| 4 | - | 5 | 1 | n. 10: 5 AL type, 5 non-amyloidosis patients, planar scans | [20] * |
| 5 | 3 | 2 | - | Results of the same study with SPECT/CT | |
| 6 | - | 4 | - | n. 10 subjects with familial ATTR | [22] ¶ |
| Label | Compound | Kd | Fibrils | [Ref] |
|---|---|---|---|---|
| 125I | Aprotinin native | 2.9 × 10−6 mol | h r-insulin | [9] |
| 99mTc | r-Aprotinin | 9.9 × 10−8 mol | h r-insulin | [32] |
| 99mTc | 30-51SS | 5.2 × 10−12 mol | h r-insulin | [33] |
| 67Ga | r-ANGa | 8.59 × 10−8 mol | h r-insulin | [34] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aprile, C.; Lodola, L. A Narrative Review of 99mTc-Aprotinin in the Diagnosis of Cardiac Amyloidosis and a New Life for an Unfairly Abandoned Drug. Biomedicines 2022, 10, 1377. https://doi.org/10.3390/biomedicines10061377
Aprile C, Lodola L. A Narrative Review of 99mTc-Aprotinin in the Diagnosis of Cardiac Amyloidosis and a New Life for an Unfairly Abandoned Drug. Biomedicines. 2022; 10(6):1377. https://doi.org/10.3390/biomedicines10061377
Chicago/Turabian StyleAprile, Carlo, and Lorenzo Lodola. 2022. "A Narrative Review of 99mTc-Aprotinin in the Diagnosis of Cardiac Amyloidosis and a New Life for an Unfairly Abandoned Drug" Biomedicines 10, no. 6: 1377. https://doi.org/10.3390/biomedicines10061377
APA StyleAprile, C., & Lodola, L. (2022). A Narrative Review of 99mTc-Aprotinin in the Diagnosis of Cardiac Amyloidosis and a New Life for an Unfairly Abandoned Drug. Biomedicines, 10(6), 1377. https://doi.org/10.3390/biomedicines10061377







