OncoPan®: An NGS-Based Screening Methodology to Identify Molecular Markers for Therapy and Risk Assessment in Pancreatic Ductal Adenocarcinoma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients and Tissues
2.2. NGS Analysis
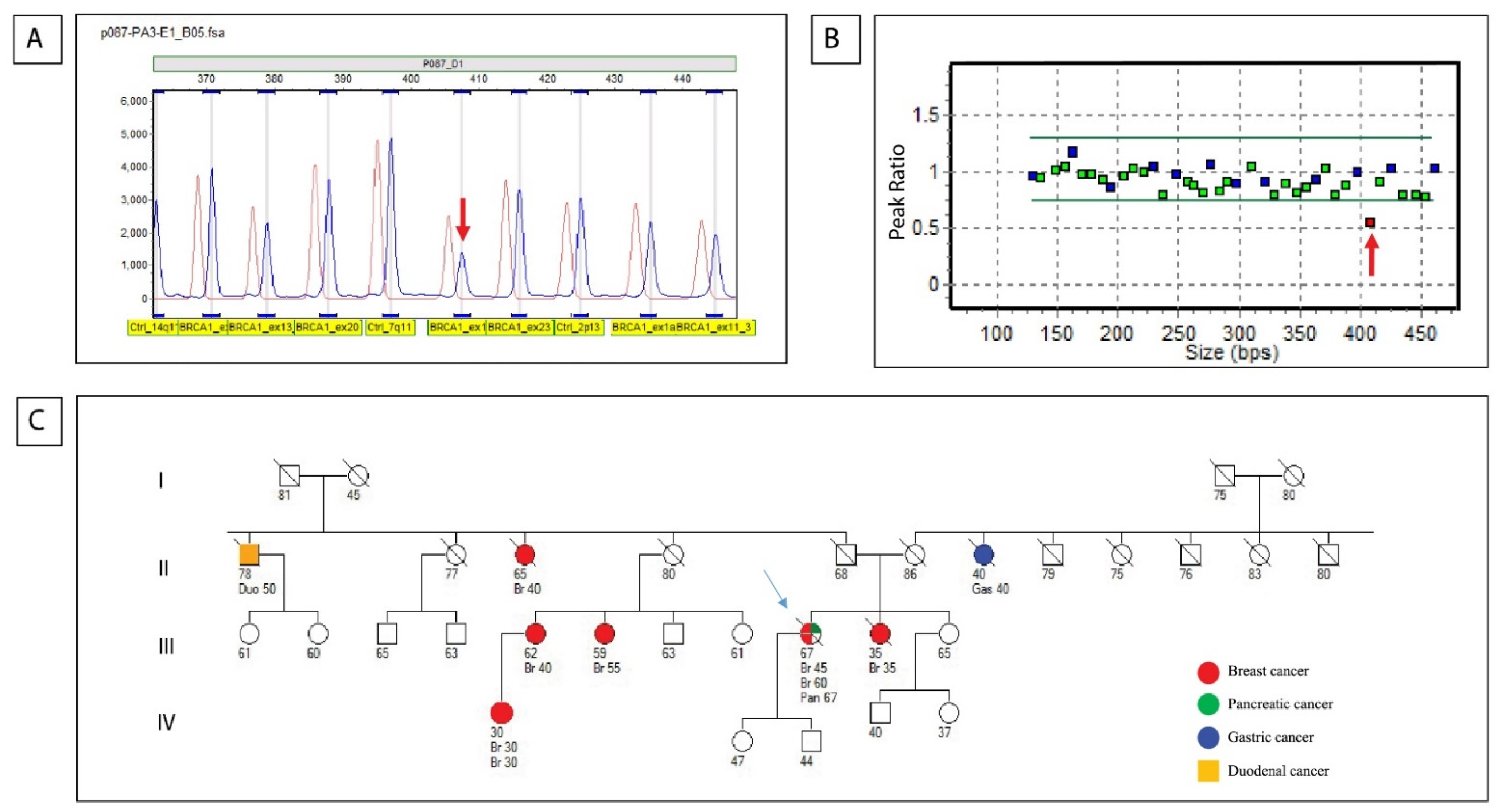
2.3. Sanger Sequencing
2.4. Multiplex Ligation-Dependent Probe Amplification (MLPA)
2.5. Variant Classification
2.6. FISH Analysis
2.7. Immunohistochemical Analysis
3. Results
3.1. NGS Results
3.2. Germline Results (Sanger Sequencing and MLPA)
3.3. FISH Results
3.4. IHC Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Waddell, N.; Pajic, M.; Patch, A.M.; Chang, D.K.; Kassahn, K.S.; Bailey, P.; Johns, A.L.; Miller, D.; Nones, K.; Quek, K.; et al. Whole genomes redefine the mutational landscape of pancreatic cancer. Nature 2015, 518, 495–501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diaz, K.E.; Lucas, A.L. Familial Pancreatic Ductal Adenocarcinoma. Am. J. Pathol. 2019, 189, 36–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goggins, M.; Overbeek, K.A.; Brand, R.; Syngal, S.; Chiaro, M.D.; Bartsch, D.K.; Bassi, C.; Carrato, A.; Farrell, J.; Fishman, E.K.; et al. Management of patients with increased risk for familial pancreatic cancer: Updated recommendations from the International Cancer of the Pancreas Screening (CAPS) Consortium. Gut 2020, 69, 7–17. [Google Scholar] [CrossRef] [Green Version]
- Rebelatto, T.F.; Falavigna, M.; Pozzari, M.; Spada, F.; Cella, C.A.; Laffi, A.; Pellicori, S.; Fazio, N. Should platinum-based chemotherapy be preferred for germline BReast CAncer genes (BRCA) 1 and 2-mutated pancreatic ductal adenocarcinoma (PDAC) patients? A systematic review and meta-analysis. Cancer Treat. Rev. 2015, 80, 101895. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, D.J.; Vijai, J.; Cronin, A.M.; Bhatia, J.; Vickers, A.J.; Gaudet, M.M.; Fine, S.; Reuter, V.; Scher, H.I.; Hallden, C.; et al. Susceptibility loci associated with prostate cancer progression and mortality. Clin. Cancer Res. 2010, 16, 2819–2832. [Google Scholar] [CrossRef] [Green Version]
- Golan, T.; Hammel, P.; Reni, M.; Cutsem, E.V.; Macarulla, T.; Hall, M.J.; Park, J.O.; Hochhauser, D.; Arnold, D.; Oh, D.Y.; et al. Maintenance Olaparib for Germline BRCA-Mutated Metastatic Pancreatic Cancer. N. Engl. J. Med. 2019, 381, 317–327. [Google Scholar] [CrossRef]
- Czink, E.; Kloor, M.; Goeppert, B.; Frohling, S.; Uhrig, S.; Weber, T.F.; Meinel, J.; Sutter, C.; Weiss, K.H.; Schirmacher, P.; et al. Successful immune checkpoint blockade in a patient with advanced stage microsatellite-unstable biliary tract cancer. Mol. Case Stud. 2017, 3, a001974. [Google Scholar] [CrossRef]
- Dunnen, J.T.D.; Dalgleish, R.; Maglott, D.R.; Hart, R.K.; Greenblatt, M.S.; McGowan-Jordan, J.; Roux, A.F.; Smith, T.; Antonarakis, S.E.; Taschner, P.E. HGVS Recommendations for the Description of Sequence Variants: 2016 Update. Hum. Mutat. 2016, 37, 564–569. [Google Scholar] [CrossRef] [Green Version]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–424. [Google Scholar] [CrossRef] [Green Version]
- Rosa, S.L.; Bernasconi, B.; Frattini, M.; Tibiletti, M.G.; Molinari, F.; Furlan, D.; Sahnane, N.; Vanoli, A.; Albarello, L.; Zhang, L.; et al. TP53 alterations in pancreatic acinar cell carcinoma: New insights into the molecular pathology of this rare cancer. Virchows Arch. 2016, 468, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Wolff, A.C.; Hammond, M.E.H.; Allison, K.H.; Harvey, B.E.; Mangu, P.B.; Bartlett, J.M.S.; Bilous, M.; Ellis, I.O.; Fitzgibbons, P.; Hanna, W.; et al. Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update. Arch. Pathol. Lab. Med. 2018, 142, 1364–1382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartley, A.N.; Washington, M.K.; Colasacco, C.; Ventura, C.B.; Ismaila, N.; Benson, A.B., 3rd; Carrato, A.; Gulley, M.L.; Jain, D.; Kakar, S.; et al. HER2 Testing and Clinical Decision Making in Gastroesophageal Adenocarcinoma: Guideline From the College of American Pathologists, American Society for Clinical Pathology, and the American Society of Clinical Oncology. J. Clin. Oncol. 2017, 35, 446–464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hofmann, M.; Stoss, O.; Shi, D.; Buttner, R.; Vijver, M.V.D.; Kim, W.; Ochiai, A.; Ruschoff, J.; Henkel, T. Assessment of a HER2 scoring system for gastric cancer: Results from a validation study. Histopathology 2008, 52, 797–805. [Google Scholar] [CrossRef]
- Chiaravalli, A.M.; Carnevali, I.; Sahnane, N.; Leoni, E.; Furlan, D.; Berselli, M.; Sessa, F.; Tibiletti, M.G. Universal screening to identify Lynch syndrome: Two years of experience in a Northern Italian Center. Eur. J. Cancer Prev. 2020, 29, 281–288. [Google Scholar] [CrossRef]
- Cicenas, J.; Zalyte, E.; Rimkus, A.; Dapkus, D.; Noreika, R.; Urbonavicius, S. JNK, p38, ERK, and SGK1 Inhibitors in Cancer. Cancers 2017, 10, 1. [Google Scholar] [CrossRef] [Green Version]
- Nadkarni, A.; Furda, A.; Rajesh, C.; McInnes, C.; Ruch, R.J.; Pittman, D.L. Functional characterization of the RAD51D E233G genetic variant. Pharmacogenet. Genom. 2009, 19, 153–160. [Google Scholar] [CrossRef]
- Pishvaian, M.J.; Petricoin, E., 3rd. Molecular Profiling of Pancreatic Cancer Patients-Response. Clin. Cancer Res. 2018, 24, 6612. [Google Scholar] [CrossRef] [Green Version]
- Wartenberg, M.; Cibin, S.; Zlobec, I.; Vassella, E.; Eppenberger-Castori, S.; Terracciano, L.; Eichmann, M.D.; Worni, M.; Gloor, B.; Perren, A.; et al. Integrated Genomic and Immunophenotypic Classification of Pancreatic Cancer Reveals Three Distinct Subtypes with Prognostic/Predictive Significance. Clin. Cancer Res. 2018, 24, 4444–4454. [Google Scholar] [CrossRef] [Green Version]
- The Cancer Genome Atlas Research Network. Integrated Genomic Characterization of Pancreatic Ductal Adenocarcinoma. Cancer Cell 2017, 32, 185–203. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Mao, T.; Zhang, B.; Xu, H.; Cui, J.; Jiao, F.; Chen, D.; Wang, Y.; Hu, J.; Xia, Q.; et al. Characterization of the genomic landscape in large-scale Chinese patients with pancreatic cancer. EBioMedicine 2022, 77, 103897. [Google Scholar] [CrossRef] [PubMed]
- Varghese, A.M.; Singh, I.; Singh, R.; Kunte, S.; Chou, J.F.; Capanu, M.; Wong, W.; Lowery, M.A.; Stadler, Z.K.; Salo-Mullen, E.; et al. Early-Onset Pancreas Cancer: Clinical Descriptors, Genomics, and Outcomes. J. Natl. Cancer Inst. 2021, 113, 1194–1202. [Google Scholar] [CrossRef] [PubMed]
- Young, E.L.; Thompson, B.A.; Neklason, D.W.; Firpo, M.A.; Werner, T.; Bell, R.; Berger, J.; Fraser, A.; Gammon, A.; Koptiuch, C.; et al. Pancreatic cancer as a sentinel for hereditary cancer predisposition. BMC Cancer 2018, 18, 697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirokawa, Y.S.; Iwata, T.; Okugawa, Y.; Tanaka, K.; Sakurai, H.; Watanabe, M. HER2-positive adenocarcinoma arising from heterotopic pancreas tissue in the duodenum: A case report. World J. Gastroenterol. 2021, 27, 4738–4745. [Google Scholar] [CrossRef]
- Peretti, U.; Cavaliere, A.; Niger, M.; Tortora, G.; Marco, M.C.D.; Rodriquenz, M.G.; Centonze, F.; Rapposelli, I.G.; Giordano, G.; Vita, F.D.; et al. Germinal BRCA1-2 pathogenic variants (gBRCA1-2pv) and pancreatic cancer: Epidemiology of an Italian patient cohort. ESMO Open 2021, 6, 100032. [Google Scholar] [CrossRef]
- Ghiorzo, P.; Fornarini, G.; Sciallero, S.; Battistuzzi, L.; Belli, F.; Bernard, L.; Bonelli, L.; Borgonovo, G.; Bruno, W.; Cian, F.D.; et al. CDKN2A is the main susceptibility gene in Italian pancreatic cancer families. J. Med. Genet. 2012, 49, 164–170. [Google Scholar] [CrossRef]
- Xiao, J.; Li, W.; Huang, Y.; Huang, M.; Li, S.; Zhai, X.; Zhao, J.; Gao, C.; Xie, W.; Qin, H.; et al. A next-generation sequencing-based strategy combining microsatellite instability and tumor mutation burden for comprehensive molecular diagnosis of advanced colorectal cancer. BMC Cancer 2021, 21, 282. [Google Scholar] [CrossRef]



| Case | NGS Variant | HGVS Class | Immunohistochemical Expression | ||||
|---|---|---|---|---|---|---|---|
| MSH2 | MSH6 | MSH3 | MLH1 | PMS2 | |||
| PA1 | MSH3: c.1088C>A p.(Thr363Asn) | 3 | POS | POS | POS | POS | POS |
| PA7 | PMS2: c.2380C>T p.(Pro794Ser) | 3 | POS | POS | POS | POS | POS |
| PA9 | MSH2: c.301G>T p.(Glu101 *) | 5 | POS | POS | POS | POS | POS |
| MSH2: c.2703A>C p.(Glu901Asp) | 3 | ||||||
| PA12 | MSH6: c.3126_3172 + 38del p.? | 4 | POS | POS | POS | POS | POS |
| PA13 | MSH2: c.435T>G p.(Ile145Met) | 3 | POS | POS | POS/NEG | POS | POS/NEG * |
| PMS2: c.1148A>T p.(Asn383Ile) | 3 | ||||||
| PA18 | MSH2: c.4_78del p.(Ala2_Met26del) | 3 | POS | POS | POS | POS | POS |
| PMS2: c.1004A>G p.(Asn335Ser) | 3 | ||||||
| MSH6: c.1957_2010del p.(Val653_Gly670del) | 3 | ||||||
| PA19 | MSH6: c.866G>C p.(Gly289Ala) | 3 | POS | POS | POS | POS | POS |
| Case | NGS Copy Number | FISH Copy Number | Ratio HER2/CEN 17 | IHC Expression (% of Tumor Cell) | IHC Expression (Score) |
|---|---|---|---|---|---|
| PA1 | 4 | 4.92 | 1.26 * | 10 | 2+ |
| PA3 | 4 | 3.80 | 1.31 | NEG | 0 |
| PA4 | 36 | 15.33 | 3.83 | 90 | 3+ |
| PA15 | 4 | 2.54 | 1.11 | NEG | 0 |
| PA21 | 4 | 2.77 | 1.13 | NEG | 0 |
| PA20 | ** | NEG | 0 | ||
| PA8 | ** | NEG | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tibiletti, M.G.; Carnevali, I.; Pensotti, V.; Chiaravalli, A.M.; Facchi, S.; Volorio, S.; Mariette, F.; Mariani, P.; Fortuzzi, S.; Pierotti, M.A.; et al. OncoPan®: An NGS-Based Screening Methodology to Identify Molecular Markers for Therapy and Risk Assessment in Pancreatic Ductal Adenocarcinoma. Biomedicines 2022, 10, 1208. https://doi.org/10.3390/biomedicines10051208
Tibiletti MG, Carnevali I, Pensotti V, Chiaravalli AM, Facchi S, Volorio S, Mariette F, Mariani P, Fortuzzi S, Pierotti MA, et al. OncoPan®: An NGS-Based Screening Methodology to Identify Molecular Markers for Therapy and Risk Assessment in Pancreatic Ductal Adenocarcinoma. Biomedicines. 2022; 10(5):1208. https://doi.org/10.3390/biomedicines10051208
Chicago/Turabian StyleTibiletti, Maria Grazia, Ileana Carnevali, Valeria Pensotti, Anna Maria Chiaravalli, Sofia Facchi, Sara Volorio, Frederique Mariette, Paolo Mariani, Stefano Fortuzzi, Marco Alessandro Pierotti, and et al. 2022. "OncoPan®: An NGS-Based Screening Methodology to Identify Molecular Markers for Therapy and Risk Assessment in Pancreatic Ductal Adenocarcinoma" Biomedicines 10, no. 5: 1208. https://doi.org/10.3390/biomedicines10051208
APA StyleTibiletti, M. G., Carnevali, I., Pensotti, V., Chiaravalli, A. M., Facchi, S., Volorio, S., Mariette, F., Mariani, P., Fortuzzi, S., Pierotti, M. A., & Sessa, F. (2022). OncoPan®: An NGS-Based Screening Methodology to Identify Molecular Markers for Therapy and Risk Assessment in Pancreatic Ductal Adenocarcinoma. Biomedicines, 10(5), 1208. https://doi.org/10.3390/biomedicines10051208







