Serum-Based Biomarkers in Neurodegeneration and Multiple Sclerosis
Abstract
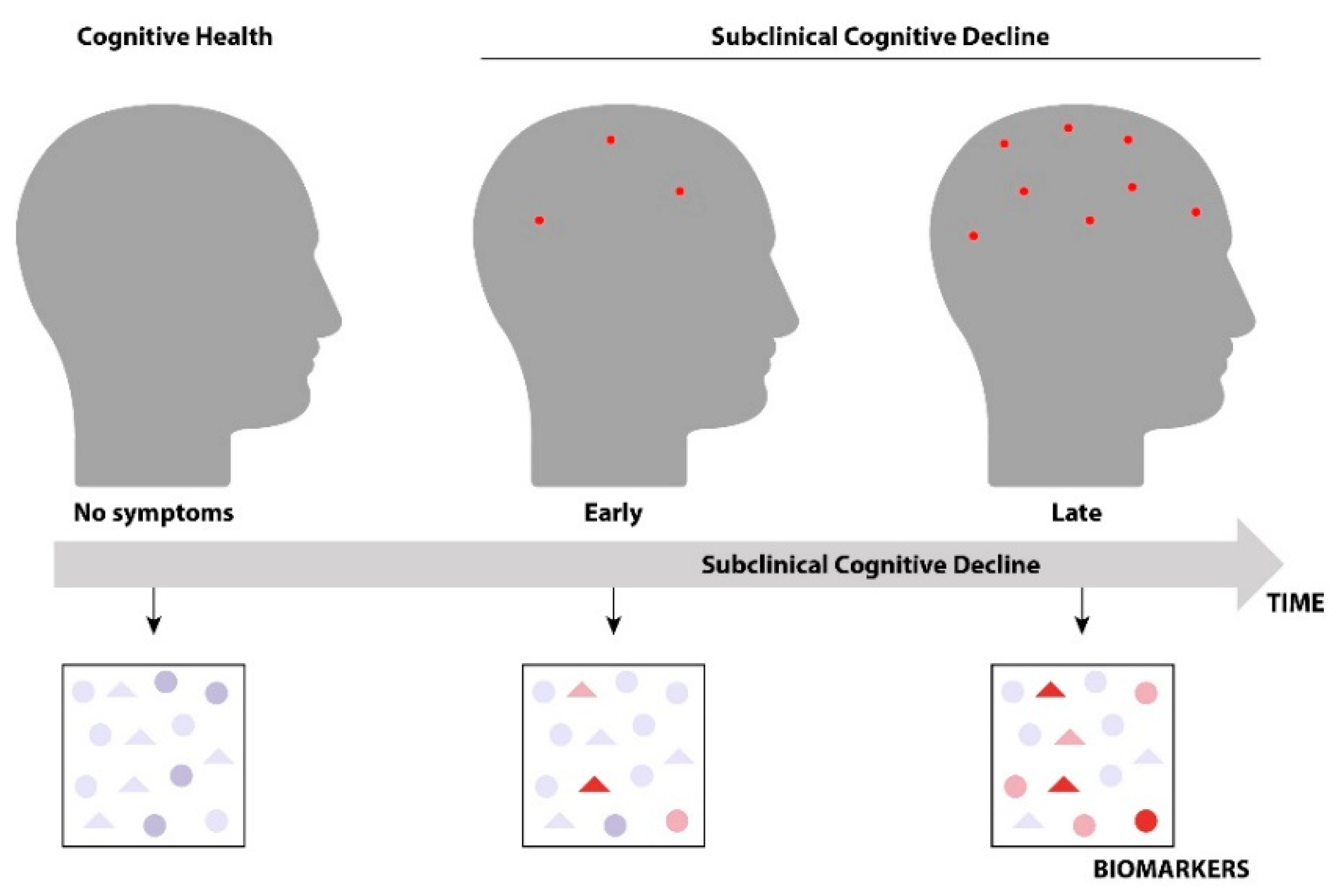
:1. Introduction
2. Neurofilaments
3. Total and Phosphorylated Tau
4. Amyloid-Peptide-β, Bri2, and Bri2-23
5. N-Acetylaspartate
6. 14-3-3 Family Proteins
7. Additional Biomarkers of Neurodegeneration
8. Osteopontin
9. Future Directions
10. Summary
11. Outstanding Questions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AA: | Amino acid |
| AD: | Alzheimer’s disease |
| ALS: | Amyotrophic lateral sclerosis |
| APP: | Amyloid precursor protein |
| CNS: | Central nervous system |
| CSF: | Cerebrospinal fluid |
| EAE: | Experimental autoimmune encephalomyelitis |
| ECL: | Electro chemi luminescence |
| ELISA: | Enzyme-linked immunosorbent assay |
| GFAP: | Glial fibrillary acidic protein |
| IFN: | Interferon |
| LBD: | Lewy body dementia |
| MCI: | Mild cognitive impairment |
| MRI: | Magnetic resonance imaging |
| MS: | Multiple sclerosis |
| NAA: | N-Acetylaspartate |
| NFs: | Neurofilaments |
| NFL: | Neurofilament light |
| PET: | Positron emission tomography |
| PP-MS: | Primary progressive multiple sclerosis |
| RR-MS: | Relapsing-remitting multiple sclerosis |
| SIMOA: | Single-molecule array |
| sNF: | Serum neurofilament |
| SP-MS: | Secondary progressive multiple sclerosis |
References
- GBD 2016 Multiple Sclerosis Collaborators. Global, regional, and national burden of multiple sclerosis 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 269–285. [Google Scholar] [CrossRef] [Green Version]
- Hogancamp, W.E.; Rodriguez, M.; Weinshenker, B.G. The epidemiology of multiple sclerosis. Mayo Clin. Proc. 1997, 72, 871–878. [Google Scholar] [CrossRef] [PubMed]
- Compston, A.; Coles, A. Multiple sclerosis. Lancet 2002, 359, 1221–1231. [Google Scholar] [CrossRef]
- Bjornevik, K.; Cortese, M.; Healy, B.C.; Kuhle, J.; Mina, M.J.; Leng, Y.; Elledge, S.J.; Niebuhr, D.W.; Scher, A.I.; Munger, K.L.; et al. Longitudinal analysis reveals a high prevalence of Epstein-Barr virus-associated with multiple sclerosis. Science 2022, 375, 296–301. [Google Scholar] [CrossRef]
- Kuchroo, V.K.; Weiner, H.L. How does Epstein-Barr virus trigger MS? Immunity 2022, 55, 390–392. [Google Scholar] [CrossRef]
- Kurtzke, J.F. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 1983, 33, 1444–1452. [Google Scholar] [CrossRef] [Green Version]
- Tsang, B.K.; Macdonell, R. Multiple sclerosis- diagnosis, management and prognosis. Aust. Fam. Physician 2011, 40, 948–955. [Google Scholar]
- Banwell, B.; Giovannoni, G.; Hawkes, C.; Lublin, F. Multiple Sclerosis is a multifaceted disease. Mult. Scler. Relat. Disord. 2014, 3, 553–554. [Google Scholar] [CrossRef]
- Bonzano, L.; Bove, M.; Sormani, M.P.; Stromillo, M.L.; Giorgio, A.; Amato, M.P.; Tacchino, A.; Mancardi, G.L.; De Stefano, N. Subclinical motor impairment assessed with an engineered glove correlates with magnetic resonance imaging tissue damage in radiologically isolated syndrome. Eur. J. Neurol. 2019, 26, 162–167. [Google Scholar] [CrossRef] [Green Version]
- Benedict, R.H.; Pol, J.; Yasin, F.; Hojnacki, D.; Kolb, C.; Eckert, S.; Tacca, B.; Drake, A.; Wojcik, C.; Morrow, S.A.; et al. Recovery of cognitive function after relapse in multiple sclerosis. Mult. Scler. J. 2021, 27, 71–78. [Google Scholar] [CrossRef]
- LoPresti, P. Silent Free Fall at Disease Onset: A Perspective on Therapeutics for Progressive Multiple Sclerosis. Front. Neurol. 2018, 9, 973. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cree, B.A.C.; Hollenbach, J.A.; Bove, R.; Kirkish, G.; Sacco, S.; Caverzasi, E.; Bischof, A.; Gundel, T.; Zhu, A.H.; Papinutto, N.; et al. Silent progression in disease activity-free relapsing multiple sclerosis. Ann. Neurol. 2019, 85, 653–666. [Google Scholar] [PubMed]
- Dujmovic, I. Cerebrospinal fluid and blood biomarkers of neuroaxonal damage in multiple sclerosis. Mult. Scler. Int. 2011, 2011, 767083. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Comabella, M.; Montalban, X. Body fluid biomarkers in multiple sclerosis. Lancet Neurol. 2014, 13, 113–126. [Google Scholar] [CrossRef]
- Gafson, A.; Craner, M.J.; Matthews, P.M. Personalised medicine for multiple sclerosis care. Mult. Scler. J. 2017, 23, 362–369. [Google Scholar] [CrossRef] [Green Version]
- Fox, R.J. Tissue Markers for Acute Multiple Sclerosis Treatment Response-A Step toward Personalized Medicine. JAMA Neurol. 2018, 75, 406–407. [Google Scholar] [CrossRef]
- Shinomoto, M.; Kasai, T.; Tatebe, H.; Kondo, M.; Ohmichi, T.; Morimoto, M.; Chiyonobu, T.; Terada, N.; Allsop, D.; Yokota, I.; et al. Plasma neurofilament light chain: A potential prognostic biomarker of dementia in adult Down syndrome patients. PLoS ONE 2019, 14, e0211575. [Google Scholar] [CrossRef]
- Preische, O.; Schultz, S.A.; Apel, A.; Kuhle, J.; Kaeser, S.A.; Barro, C.; Gräber, S.; Kuder-Buletta, E.; LaFougere, C.; Laske, C.; et al. Serum neurofilament dynamics predicts neurodegeneration and clinical progression in presymptomatic Alzheimer’s disease. Nat. Med. 2019, 25, 277–283. [Google Scholar] [CrossRef]
- Lee, J.Y.; Taghian, K.; Petratos, S. Axonal degeneration in multiple sclerosis: Can we predict and prevent permanent disability? Acta Neuropathol. Commun. 2014, 2, 97. [Google Scholar] [CrossRef]
- Cree, B.A.; Gourraud, P.A.; Oksenberg, J.R.; Bevan, C.; Crabtree-Hartman, E.; Gelfand, J.; Goodin, D.; Graves, J.; Green, A.; Mowry, E.; et al. Long-term evolution of multiple sclerosis disability in the treatment era. Ann. Neurol. 2016, 80, 499–510. [Google Scholar]
- Virgilio, E.; Vecchio, D.; Crespi, I.; Serino, R.; Cantello, R.; Dianzani, U.; Comi, C. Cerebrospinal Tau levels as a predictor of early disability in multiple sclerosis. Mult. Scler. Relat. Disord. 2021, 56, 103231. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, R.; Smith, K.E.; Allegretta, M.; Arnold, D.L.; Carroll, W.; Comabella, M.; Furlan, R.; Harp, C.; Kuhle, J.; Leppert, D.; et al. Serum neurofilament light as a biomarker in progressive multiple sclerosis. Neurology 2020, 95, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Hein Née Maier, K.; Köhler, A.; Diem, R.; Sättler, M.B.; Demmer, I.; Lange, P.; Bähr, M.; Otto, M. Biological markers for axonal degeneration in CSF and blood of patients with the first event indicative for multiple sclerosis. Neurosci. Lett. 2008, 436, 72–76. [Google Scholar] [CrossRef]
- Spitzer, P.; Lang, R.; Oberstein, T.J.; Lewczuk, P.; Ermann, N.; Huttner, H.B.; Masouris, I.; Kornhuber, J.; Ködel, U.; Maler, J.M. A Specific Reduction in Aβ1-42 vs. a Universal Loss of Aβ Peptides in CSF Differentiates Alzheimer’s Disease From Meningitis and Multiple Sclerosis. Front. Aging Neurosci. 2018, 10, 152. [Google Scholar] [CrossRef] [PubMed]
- Harris, V.K.; Diamanduros, A.; Good, P.; Zakin, E.; Chalivendra, V.; Sadiq, S.A. Bri2-23 is a potential cerebrospinal fluid biomarker in multiple sclerosis. Neurobiol. Dis. 2010, 40, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Harris, V.K.; Sadiq, S.A. Disease biomarkers in multiple sclerosis: Potential for use in therapeutic decision making. Mol. Diagn. Ther. 2009, 13, 225–244. [Google Scholar] [CrossRef]
- Miller, D.H. Biomarkers and surrogate outcomes in neurodegenerative disease: Lessons from multiple sclerosis. NeuroRx 2004, 1, 284–294. [Google Scholar] [CrossRef]
- Agah, E.; Zardoui, A.; Saghazadeh, A.; Ahmadi, M.; Tafakhori, A.; Rezaei, N. Osteopontin (OPN) as a CSF and blood biomarker for multiple sclerosis: A systematic review and meta-analysis. PLoS ONE 2018, 13, e0190252. [Google Scholar] [CrossRef]
- Housley, W.J.; Pitt, D.; Hafler, D.A. Biomarkers in multiple sclerosis. Clin. Immunol. 2015, 161, 51–58. [Google Scholar] [CrossRef]
- Ferreira-Atuesta, C.; Reyes, S.; Giovanonni, G.; Gnanapavan, S. The Evolution of Neurofilament Light Chain in Multiple Sclerosis. Front. Neurosci. 2021, 15, 642384. [Google Scholar] [CrossRef]
- Traka, M.; Podojil, J.R.; Mccarthy, D.P.; Miller, S.D.; Popko, B. Oligodendrocyte death results in immune-mediated CNS demyelination. Nat. Neurosci. 2016, 19, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Basso, A.S.; Frenkel, D.; Quintana, F.J.; Costa-Pinto, F.A.; Petrovic-Stojkovic, S.; Puckett, L.; Monsonego, A.; Bar-Shir, A.; Engel, Y.; Gozin, M.; et al. Reversal of axonal loss and disability in a mouse model of progressive multiple sclerosis. J. Clin. Investig. 2008, 118, 1532–1543. [Google Scholar] [CrossRef]
- LoPresti, P. The Selective HDAC6 Inhibitor ACY-738 Impacts Memory and Disease Regulation in an Animal Model of Multiple Sclerosis. Front. Neurol. 2019, 10, 519. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trapp, B.D.; Nave, K.A. Multiple sclerosis: An immune or neurodegenerative disorder? Annu. Rev. Neurosci. 2008, 31, 247–269. [Google Scholar] [CrossRef]
- Di Filippo, M.; Mancini, A.; Bellingacci, L.; Gaetani, L.; Mazzocchetti, P.; Zelante, T.; La Barbera, L.; De Luca, A.; Tantucci, M.; Tozzi, A.; et al. Interleukin-17 affects synaptic plasticity and cognition in an experimental model of multiple sclerosis. Cell Rep. 2021, 37, 110094. [Google Scholar] [CrossRef] [PubMed]
- Bourel, J.; Planche, V.; Dubourdieu, N.; Oliveira, A.; Séré, A.; Ducourneau, E.G.; Tible, M.; Maitre, M.; Lesté-Lasserre, T.; Nadjar, A.; et al. Complement C3 mediates early hippocampal neurodegeneration and memory impairment in experimental multiple sclerosis. Neurobiol. Dis. 2021, 160, 105533. [Google Scholar] [CrossRef] [PubMed]
- Mandolesi, G.; Gentile, A.; Musella, A.; Fresegna, D.; De Vito, F.; Bullitta, S.; Sepman, H.; Marfia, G.A.; Centonze, D. Synaptopathy connects inflammation and neurodegeneration in multiple sclerosis. Nat. Rev. Neurol. 2015, 11, 711–724. [Google Scholar] [CrossRef]
- LoPresti, P. Glatiramer acetate guards against rapid memory decline during relapsing-remitting experimental autoimmune encephalomyelitis. Neurochem. Res. 2015, 40, 473–479. [Google Scholar] [CrossRef]
- Buffolo, F.; Petrosino, V.; Albini, M.; Moschetta, M.; Carlini, F.; Floss, T.; Kerlero de Rosbo, N.; Cesca, F.; Rocchi, A.; Uccelli, A.; et al. Neuroinflammation induces synaptic scaling through IL-1β-mediated activation of the transcriptional repressor REST/NRSF. Cell Death Dis. 2021, 12, 180. [Google Scholar] [CrossRef]
- Bruno, A.; Dolcetti, E.; Rizzo, F.R.; Fresegna, D.; Musella, A.; Gentile, A.; De Vito, F.; Caioli, S.; Guadalupi, L.; Bullitta, S.; et al. Inflammation-Associated Synaptic Alterations as Shared Threads in Depression and Multiple Sclerosis. Front. Cell. Neurosci. 2020, 14, 169. [Google Scholar] [CrossRef]
- Nasios, G.; Bakirtzis, C.; Messinis, L. Cognitive Impairment and Brain Reorganization in MS: Underlying Mechanisms and the Role of Neurorehabilitation. Front. Neurol. 2020, 11, 147. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, F.R.; Musella, A.; De Vito, F.; Fresegna, D.; Bullitta, S.; Vanni, V.; Guadalupi, L.; Stampanoni Bassi, M.; Buttari, F.; Mandolesi, G.; et al. Tumor Necrosis Factor and Interleukin-1β Modulate Synaptic Plasticity during Neuroinflammation. Neural. Plast. 2018, 10, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Teunissen, C.E.; Dijkstra, C.; Polman, C. Biological markers in CSF and blood for axonal degeneration in multiple sclerosis. Lancet Neurol. 2005, 4, 32–41. [Google Scholar] [CrossRef]
- Kappos, L.; Butzkueven, H.; Wiendl, H.; Spelman, T.; Pellegrini, F.; Chen, Y.; Dong, Q.; Koendgen, H.; Belachew, S.; Trojano, M.; et al. Greater sensitivity to multiple sclerosis disability worsening and progression events using a roving versus a fixed reference value in a prospective cohort study. Mult. Scler. J. 2018, 24, 963–973. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brady, S.T. Motor neurons and neurofilaments in sickness and in health. Cell 1993, 73, 1–3. [Google Scholar] [CrossRef]
- Yuan, A.; Nixon, R.A. Neurofilament Proteins as Biomarkers to Monitor Neurological Diseases and the Efficacy of Therapies. Front. Neurosci. 2021, 15, 689938. [Google Scholar] [CrossRef]
- Khalil, M.; Teunissen, C.E.; Otto, M.; Piehl, F.; Sormani, M.P.; Gattringer, T.; Barro, C.; Kappos, L.; Comabella, M.; Fazekas, F.; et al. Neurofilaments as biomarkers in neurological disorders. Nat. Rev. Neurol. 2018, 14, 577–589. [Google Scholar] [CrossRef]
- Teunissen, C.E.; Petzold, A.; Bennett, J.L.; Berven, F.S.; Brundin, L.; Comabella, M.; Franciotta, D.; Frederiksen, J.L.; Fleming, J.O.; Furlan, R.; et al. A consensus protocol for the standardization of cerebrospinal fluid collection and biobanking. Neurology 2009, 73, 1914–1922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, M.K.; Cleveland, D.W. Neuronal intermediate filaments. Ann. Rev. Neurosci. 1996, 19, 187–217. [Google Scholar] [CrossRef]
- Semra, Y.K.; Seidi, O.A.; Sharief, M.K. Heightened intrathecal release of axonal cytoskeletal proteins in multiple sclerosis is associated with progressive disease and clinical disability. J. Neuroimmunol. 2002, 122, 132–139. [Google Scholar] [CrossRef]
- Malmestrom, C.; Haghighi, S.; Rosengren, L.; Andersen, O.; Lycke, J. Neurofilament light protein and glial fibrillary acidic protein as biological markers in MS. Neurology 2003, 61, 1720–1725. [Google Scholar] [CrossRef] [PubMed]
- Feneberg, E.; Oeckl, P.; Steinacker, P.; Verde, F.; Barro, C.; Van Damme, P.; Gray, E.; Grosskreutz, J.; Jardel, C.; Kuhle, J.; et al. Multicenter evaluation of neurofilaments in early symptom onset amyotrophic lateral sclerosis. Neurology 2018, 90, e22–e30. [Google Scholar] [CrossRef] [PubMed]
- Barro, C.; Benkert, P.; Disanto, G.; Tsagkas, C.; Amann, M.; Naegelin, Y.; Leppert, D.; Gobbi, C.; Granziera, C.; Yaldizli, Ö.; et al. Serum neurofilament as a predictor of disease worsening and brain and spinal cord atrophy in multiple sclerosis. Brain 2018, 141, 2382–2391. [Google Scholar] [CrossRef] [PubMed]
- Budelier, M.M.; He, Y.; Barthelemy, N.R.; Jiang, H.; Li, Y.; Park, E.; Henson, R.L.; Schindler, S.E.; Holtzman, D.M.; Bateman, R.J. A map of neurofilament light chain species in brain and cerebrospinal fluid and alterations in Alzheimer’s disease. Brain Commun. 2022, 4, fcac045. [Google Scholar] [CrossRef] [PubMed]
- Gunnarsson, M.; Malmeström, C.; Axelsson, M.; Sundström, P.; Dahle, C.; Vrethem, M.; Olsson, T.; Piehl, F.; Norgren, N.; Rosengren, L.; et al. Axonal damage in relapsing multiple sclerosis is markedly reduced by natalizumab. Ann. Neurol. 2011, 69, 83–89. [Google Scholar] [CrossRef]
- Jakimovski, D.; Zivadinov, R.; Ramanthan, M.; Hagemeier, J.; Weinstock-Guttman, B.; Tomic, D.; Kropshofer, H.; Fuchs, T.A.; Barro, C.; Leppert, D.; et al. Serum neurofilament light chain level associations with clinical and cognitive performance in multiple sclerosis: A longitudinal retrospective 5-year study. Mult. Scler. J. 2020, 26, 1670–1681. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Therriault, J.; Luo, J.; Ba, M.; Zhang, H.; Initiative, A. Neurofilament light as a biomarker of axonal degeneration in patients with mild cognitive impairment and Alzheimer’s disease. J. Integr. Neurosci. 2021, 20, 861–870. [Google Scholar] [CrossRef]
- Lee, E.H.; Kwon, H.S.; Koh, S.H.; Choi, S.H.; Jin, J.H.; Jeong, J.H.; Jang, J.W.; Park, K.W.; Kim, E.J.; Kim, H.J.; et al. Serum neurofilament light chain level as a predictor of cognitive stage transition. Alzheimer’s Res. Ther. 2022, 14, 6. [Google Scholar] [CrossRef]
- Silva-Spínola, A.; Lima, M.; Leitão, M.J.; Durães, J.; Tábuas-Pereira, M.; Almeida, M.R.; Santana, I.; Baldeiras, I. Serum neurofilament light chain as a surrogate of cognitive decline in sporadic and familial frontotemporal dementia. Eur. J. Neurol. 2022, 29, 36–46. [Google Scholar] [CrossRef]
- Fialová, L.; Bartos, A.; Švarcová, J.; Zimova, D.; Kotoucova, J. Serum and cerebrospinal fluid heavy neurofilaments and antibodies against them in early multiple sclerosis. J. Neuroimmunol. 2013, 259, 81–87. [Google Scholar] [CrossRef]
- Disanto, G.; Barro, C.; Benkert, P.; Naegelin, Y.; Schädelin, S.; Giardiello, A.; Zecca, C.; Blennow, K.; Zetterberg, H.; Leppert, D.; et al. Serum Neurofilament light: A biomarker of neuronal damage in multiple sclerosis. Ann. Neurol. 2017, 81, 857–870. [Google Scholar] [CrossRef]
- Kuhle, J.; Nourbakhsh, B.; Grant, D.; Morant, S.; Barro, C.; Yaldizli, Ö.; Pelletier, D.; Giovannoni, G.; Waubant, E.; Gnanapavan, S. Serum neurofilament is associated with progression of brain atrophy and disability in early MS. Neurology 2017, 88, 826–831. [Google Scholar] [CrossRef] [Green Version]
- Brureau, A.; Blanchard-Bregeon, V.; Pech, C.; Hamon, S.; Chaillou, P.; Guillemot, J.C.; Barneoud, P.; Bertrand, P.; Pradier, L.; Rooney, T.; et al. NF-L in cerebrospinal fluid and serum is a biomarker of neuronal damage in an inducible mouse model of neurodegeneration. Neurobiol. Dis. 2017, 104, 73–84. [Google Scholar] [CrossRef]
- Martin, S.J.; McGlasson, S.; Hunt, D.; Overell, J. Cerebrospinal fluid neurofilament light chain in multiple sclerosis and its subtypes: A meta-analysis of case-control studies. J. Neurol. Neurosurg. Psychiatry 2019, 90, 1059–1067. [Google Scholar] [CrossRef] [Green Version]
- Siller, N.; Kuhle, J.; Muthuraman, M.; Barro, C.; Uphaus, T.; Groppa, S.; Kappos, L.; Zipp, F.; Bittner, S. Serum neurofilament light chain is a biomarker of acute and chronic neuronal damage in early multiple sclerosis. Mult. Scler. J. 2019, 25, 678–686. [Google Scholar] [CrossRef]
- Sellebjerg, F.; Royen, L.; Soelberg Sørensen, P.; Oturai, A.B.; Jensen, P. Prognostic value of cerebrospinal fluid neurofilament light chain and chitinase-3-like-1 in newly diagnosed patients with multiple sclerosis. Mult. Scler. J. 2019, 25, 1444–1451. [Google Scholar] [CrossRef]
- Norgren, N.; Sundström, P.; Svenningsson, A.; Rosengren, L.; Stigbrand, T.; Gunnarsson, M. Neurofilament and glial fibrillary acidic protein in multiple sclerosis. Neurology 2004, 63, 1586–1590. [Google Scholar] [CrossRef]
- Kuhle, J.; Kropshofer, H.; Hearing, D.A.; Kundu, U.; Meinert, R.; Barro, C.; Dahlke, F.; Tomic, D.; Leppert, D.; Kappos, L. Blood neurofilament light chain as a biomarker of MS disease activity and treatment response. Neurology 2019, 92, e1007–e1015. [Google Scholar] [CrossRef]
- Bhan, A.; Jacobsen, C.; Dalen, I.; Bergsland, N.; Zivadinov, R.; Alves, G.; Myhr, K.M.; Farbu, E. CSF neurofilament light chain predicts 10-year clinical and radiologic worsening in multiple sclerosis. Mult. Scler. J. Exp. Transl. Clin. 2021, 7, 20552173211060337. [Google Scholar] [CrossRef]
- Lycke, J.N.; Karlsson, J.E.; Andersen, O.; Rosengren, L.E. Neurofilament protein in cerebrospinal fluid: A potential marker of activity in multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 1998, 64, 402–404. [Google Scholar] [CrossRef] [Green Version]
- Cai, L.; Huang, J. Neurofilament light chain as a biological marker for multiple sclerosis: A meta-analysis study. Neuropsychiatr. Dis. Treat. 2018, 14, 2241–2254. [Google Scholar] [CrossRef] [Green Version]
- Vavasour, I.M.; Becquart, P.; Gill, J.; Zhao, G.; Yik, J.T.; Traboulsee, A.; Carruthers, R.L.; Kolind, S.H.; Schabas, A.J.; Sayao, A.L.; et al. Diffusely abnormal white matter in clinically isolated syndrome is associated with parenchymal loss and elevated neurofilament levels. Mult. Scler. Relat. Disord. 2021, 57, 103422. [Google Scholar] [CrossRef]
- Maggi, P.; Kuhle, J.; Schädelin, S.; van der Meer, F.; Weigel, M.; Galbusera, R.; Mathias, A.; Lu, P.J.; Rahmanzadeh, R.; Benkert, P.; et al. Chronic White Matter Inflammation and Serum Neurofilament Levels in Multiple Sclerosis. Neurology 2021, 97, e543–e553. [Google Scholar] [CrossRef]
- Puentes, F.; Benkert, P.; Amor, S.; Kuhle, J.; Giovannoni, G. Antibodies to neurofilament light as potential biomarkers in multiple sclerosis. BMJ Neurol. Open 2021, 3, e000192. [Google Scholar] [CrossRef]
- Calabresi, P.A.; Arnold, D.L.; Sangurdekar, D.; Singh, C.M.; Altincatal, A.; de Moor, C.; Engle, B.; Goyal, J.; Deykin, A.; Szak, S.; et al. Temporal profile of serum neurofilament light in multiple sclerosis: Implications for patient monitoring. Mult. Scler. J. 2021, 27, 1497–1505. [Google Scholar] [CrossRef]
- Jakimovski, D.; Dwyer, M.G.; Bergsland, N.; Weinstock-Guttman, B.; Zivadinov, R. Disease biomarkers in multiple sclerosis: Current serum neurofilament light chain perspectives. Neurodegener. Dis. Manag. 2021, 11, 329–340. [Google Scholar] [CrossRef]
- Bergman, J.; Dring, A.; Zetterberg, H.; Blennow, K.; Norgren, N.; Gilthorpe, J.; Bergenheim, T.; Svenningsson, A. Neurofilament light in CSF and serum is a sensitive marker for axonal white matter injury in MS. Neurol. Neuroimmunol. Neuroinflamm. 2016, 3, e271. [Google Scholar] [CrossRef] [Green Version]
- Bridel, C.; Leurs, C.E.; van Lierop, Z.; van Kempen, Z.; Dekker, I.; Twaalfhoven, H.; Moraal, B.; Barkhof, F.; Uitdehaag, B.; Killestein, J.; et al. Serum Neurofilament Light Association with Progression in Natalizumab-Treated Patients With Relapsing-Remitting Multiple Sclerosis. Neurology 2021, 97, e1898–e1905. [Google Scholar] [CrossRef]
- Kuhle, J.; Daizadeh, N.; Benkert, P.; Maceski, A.; Barro, C.; Michalak, Z.; Sormani, M.P.; Godin, J.; Shankara, S.; Samad, T.A.; et al. Sustained reduction of serum neurofilament light chain over 7 years by alemtuzumab in early relapsing-remitting MS. Mult. Scler. J. 2021, 28, 13524585211032348. [Google Scholar] [CrossRef]
- Cantó, E.; Barro, C.; Zhao, C.; Caillier, S.J.; Michalak, Z.; Bove, R.; Tomic, D.; Santaniello, A.; Häring, D.A.; Hollenbach, J.; et al. Association Between Serum Neurofilament Light Chain Levels and Long-term Disease Course Among Patients With Multiple Sclerosis Followed up for 12 Years. JAMA Neurol. 2019, 76, 1359–1366. [Google Scholar] [CrossRef]
- Kuhle, J.; Plavina, T.; Barro, C.; Disanto, G.; Sangurdekar, D.; Singh, C.M.; de Moor, C.; Engle, B.; Kieseier, B.C.; Fisher, E.; et al. Neurofilament light levels are associated with long-term outcomes in multiple sclerosis. Mult. Scler. J. 2020, 26, 1691–1699. [Google Scholar] [CrossRef] [Green Version]
- Disanto, G.; Adiutori, R.; Dobson, R.; Martinelli, V.; Dalla Costa, G.; Runia, T.; Evdoshenko, E.; Thouvenot, E.; Trojano, M.; Norgren, N.; et al. Serum neurofilament light chain levels are increased in patients with a clinically isolated syndrome. J. Neurol. Neurosurg. Psychiatry 2016, 87, 126–129. [Google Scholar] [CrossRef]
- Dalla Costa, G.; Martinelli, V.; Sangalli, F.; Moiola, L.; Colombo, B.; Radaelli, M.; Letizia, L.; Roberto, F.; Comi, G. Prognostic value of serum neurofilaments in patients with clinically isolated syndromes. Neurology 2019, 92, e733–e741. [Google Scholar] [CrossRef] [Green Version]
- Jakimovski, D.; Kuhle, J.; Ramanathan, M.; Barro, C.; Tomic, D.; Hagemeier, J.; Kropshofer, H.; Bergsland, N.; Leppert, D.; Dwyer, M.G. Serum neurofilament light chain levels associations with gray matter pathology: A 5-year longitudinal study. Ann. Clin. Transl. Neurol. 2019, 6, 1757–1770. [Google Scholar] [CrossRef] [Green Version]
- Bjornevik, K.; Munger, K.L.; Cortese, M.; Barro, C.; Healy, B.C.; Niebuhr, D.W.; Scher, A.I.; Kuhle, J.; Ascherio, A. Serum Neurofilament Light Chain Levels in Patients With Presymptomatic Multiple Sclerosis. JAMA Neurol. 2020, 77, 58–64. [Google Scholar] [CrossRef]
- Bittner, S.; Steffen, F.; Uphaus, T.; Muthuraman, M.; Fleischer, V.; Salmen, A.; Luessi, F.; Berthele, A.; Klotz, L.; Meuth, S.G.; et al. Clinical implications of serum neurofilament in newly diagnosed MS patients: A longitudinal multicentre cohort study. EBioMedicine 2020, 56, 102807. [Google Scholar] [CrossRef]
- Novakova, L.; Axelsson, M.; Khademi, M.; Zetterberg, H.; Blennow, K.; Malmeström, C.; Piehl, F.; Olsson, T.; Lycke, J. Cerebrospinal fluid biomarkers as a measure of disease activity and treatment efficacy in relapsing-remitting multiple sclerosis. J. Neurochem. 2017, 141, 296–304. [Google Scholar] [CrossRef] [Green Version]
- Håkansson, I.; Tisell, A.; Cassel, P.; Blennow, K.; Zetterberg, H.; Lundberg, P.; Dahle, C.; Vrethem, M.; Ernerudh, J. Neurofil ament light chain in cerebrospinal fluid and prediction of disease activity in clinically isolated syndrome and relapsing-remitting multiple sclerosis. Eur. J. Neurol. 2017, 24, 703–712. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Li, X.; Qiao, J. Neurofilament protein light in multiple sclerosis. Zhonghua Yi Xue Za Zhi 2007, 87, 2745–2749. [Google Scholar]
- Haghighi, S.; Andersen, O.; Odén, A.; Rosengren, L. Cerebrospinal fluid markers in MS patients and their healthy siblings. Acta Neurol. Scand. 2004, 109, 97–99. [Google Scholar] [CrossRef]
- Norgren, N.; Rosengren, L.; Stigbrand, T. Elevated neurofilament levels in neurological diseases. Brain Res. 2003, 987, 25–31. [Google Scholar] [CrossRef]
- Gil-Perotin, S.; Castillo-Villalba, J.; Cubas-Nuñez, L.; Gasque, R.; Hervas, D.; Gomez-Mateu, J.; Alcala, C.; Perez-Miralles, F.; Gascon, F.; Dominguez, J.A.; et al. Combined cerebrospinal fluid neurofilament light chain protein and chitinase-3 like-1 levels in defining disease course and prognosis in multiple sclerosis. Front. Neurol. 2019, 10, 1008. [Google Scholar] [CrossRef]
- Gaetani, L.; Salvadori, N.; Lisetti, V.; Eusebi, P.; Mancini, A.; Gentili, L.; Borrelli, A.; Portaccio, E.; Sarchielli, P.; Blennow, K.; et al. Cerebrospinal fluid neurofilament light chain tracks cognitive impairment in multiple sclerosis. J. Neurol. 2019, 266, 2157–2163. [Google Scholar] [CrossRef]
- Gaetani, L.; Eusebi, P.; Mancini, A.; Gentili, L.; Borrelli, A.; Parnetti, L.; Calabresi, P.; Sarchielli, P.; Blennow, K.; Zetterberg, H.; et al. Cerebrospinal fluid neurofilament light chain predicts disease activity after the first demyelinating event suggestive of multiple sclerosis. Mult. Scler. Relat. Disord. 2019, 35, 228–232. [Google Scholar] [CrossRef]
- Olesen, M.N.; Soelberg, K.; Debrabant, B.; Nilsson, A.C.; Lillevang, S.T.; Grauslund, J.; Brandslund, I.; Madsen, J.S.; Paul, F.; Smith, T.J.; et al. Cerebrospinal fluid biomarkers for predicting development of multiple sclerosis in acute optic neuritis: A population-based prospective cohort study. J. Neuroinflamm. 2019, 16, 59. [Google Scholar] [CrossRef]
- Bhan, A.; Jacobsen, C.; Myhr, K.M.; Dalen, I.; Lode, K.; Farbu, E. Neurofilaments and 10-year follow-up in multiple sclerosis. Mult. Scler. J. 2018, 24, 1301–1307. [Google Scholar] [CrossRef]
- Quintana, E.; Coll, C.; Salavedra-Pont, J.; Muñoz-San Martín, M.; Robles-Cedeño, R.; Tomàs-Roig, J.; Buxó, M.; Matute-Blanch, C.; Villar, L.M.; Montalban, X.; et al. Cognitive impairment in early stages of multiple sclerosis is associated with high cerebrospinal fluid levels of chitinase 3-like 1 and neurofilament light chain. Eur. J. Neurol. 2018, 25, 1189–1191. [Google Scholar] [CrossRef]
- Van der Vuurst de Vries, R.M.; Wong, Y.Y.M.; Mescheriakova, J.Y.; van Pelt, E.D.; Runia, T.F.; Jafari, N.; Siepman, T.A.; Melief, M.J.; Wierenga-Wolf, A.F.; van Luijn, M.M.; et al. High neurofilament levels are associated with clinically definite multiple sclerosis in children and adults with clinically isolated syndrome. Mult. Scler. J. 2019, 25, 958–967. [Google Scholar] [CrossRef] [Green Version]
- Novakova, L.; Axelsson, M.; Malmeström, C.; Imberg, H.; Elias, O.; Zetterberg, H.; Nerman, O.; Lycke, J. Searching for neurodegeneration in multiple sclerosis at clinical onset:Diagnostic value of biomarkers. PLoS ONE 2018, 13, e0194828. [Google Scholar] [CrossRef] [Green Version]
- Tortorella, C.; Direnzo, V.; Ruggieri, M.; Zoccolella, S.; Mastrapasqua, M.; D’onghia, M.; Paolicelli, D.; Cuonzo, F.D.; Gasperini, C.; Trojano, M. Cerebrospinal fluid neurofilament light levels mark grey matter volume in clinically isolated syndrome suggestive of multiple sclerosis. Mult. Scler. J. 2018, 241, 039–1045. [Google Scholar] [CrossRef]
- Arrambide, G.; Espejo, C.; Eixarch, H.; Villar, L.M.; Alvarez-Cermeño, J.C.; Picón, C.; Kuhle, J.; Disanto, G.; Kappos, L.; Sastre-Garriga, J.; et al. Neurofilament light chain level is a weak risk factor for the development of MS. Neurology 2016, 87, 1076–1084. [Google Scholar] [CrossRef] [Green Version]
- Reyes, S.; Smets, I.; Holden, D.; Carrillo-Loza, K.; Christmas, T.; Bianchi, L.; Ammoscato, F.; Turner, B.; Marta, M.; Schmierer, K.; et al. CSF neurofilament light chain testing as an aid to determine treatment strategies in MS. Neurol. Neuroimmunol. Neuroinflamm. 2020, 7, e880. [Google Scholar] [CrossRef]
- Modvig, S.; Degn, M.; Roed, H.; Sørensen, T.L.; Larsson, H.B.; Langkilde, A.R.; Frederiksen, J.L.; Sellebjerg, F. Cerebrospinal fluid levels of chitinase 3-like 1 and neurofilament light chain predict multiple sclerosis development and disability after optic neuritis. Mult. Scler. J. 2015, 21, 1761–1770. [Google Scholar] [CrossRef]
- Manouchehrinia, A.; Stridh, P.; Khademi, M.; Leppert, D.; Barro, C.; Michalak, Z.; Benkert, P.; Lycke, J.; Alfredsson, L.; Kappos, L.; et al. Plasma neurofilament light levels are associated with risk of disability in multiple sclerosis. Neurology 2020, 94, e2457–e2467. [Google Scholar] [CrossRef]
- Novakova, L.; Zetterberg, H.; Sundström, P.; Axelsson, M.; Khademi, M.; Gunnarsson, M.; Malmeström, C.; Svenningsson, A.; Olsson, T.; Piehl, F.; et al. Monitoring disease activity in multiple sclerosis using serum neurofilament light protein. Neurology 2017, 89, 2230–2237. [Google Scholar] [CrossRef] [Green Version]
- Engel, S.; Friedrich, M.; Muthuraman, M.; Steffen, F.; Poplawski, A.; Groppa, S.; Bittner, S.; Zipp, F.; Luessi, F. Intrathecal B-cell accumulation and axonal damage distinguish MRI-based benign from aggressive onset in MS. Neurol. Neuroimmunol. Neuroinflamm. 2019, 6, e595. [Google Scholar] [CrossRef] [Green Version]
- Kuhle, J.; Barro, C.; Disanto, G.; Mathias, A.; Soneson, C.; Bonnier, G.; Yaldizli, Ö.; Regeniter, A.; Derfuss, T.; Canales, M.; et al. Serum neurofilament light chain in early relapsing remitting MS is increased and correlates with CSF levels and with MRI measures of disease severity. Mult. Scler. J. 2016, 22, 1550–1559. [Google Scholar] [CrossRef]
- Piehl, F.; Kockum, I.; Khademi, M.; Blennow, K.; Lycke, J.; Zetterberg, H.; Olsson, T. Plasma neurofilament light chain levels in patients with MS switching from injectable therapies to fingolimod. Mult. Scler. 2018, 24, 1046–1054. [Google Scholar] [CrossRef]
- Ayrignac, X.; Le Bars, E.; Duflos, C.; Hirtz, C.; Maleska Maceski, A.; Carra-Dallière, C.; Charif, M.; Pinna, F.; Prin, P.; de Champfleur, N.M.; et al. Serum GFAP in multiple sclerosis: Correlation with disease type and MRI markers of disease severity. Sci. Rep. 2020, 10, 10923. [Google Scholar] [CrossRef]
- Watanabe, M.; Nakamura, Y.; Michalak, Z.; Isobe, N.; Barro, C.; Leppert, D.; Matsushita, T.; Hayashi, F.; Yamasaki, R.; Kuhle, J.; et al. Serum GFAP and neurofilament light as biomarkers of disease activity and disability in NMOSD. Neurology 2019, 93, e1299–e1311. [Google Scholar] [CrossRef]
- Wong, Y.Y.M.; Bruijstens, A.L.; Barro, C.; Michalak, Z.; Melief, M.J.; Wierenga, A.F.; van Pelt, E.D.; Neuteboom, R.F.; Kuhle, J.; Hintzen, R.Q. Serum neurofilament light chain in pediatric MS and other acquired demyelinating syndromes. Neurology 2019, 93, e968–e974. [Google Scholar] [CrossRef] [PubMed]
- Håkansson, I.; Tisell, A.; Cassel, P.; Blennow, K.; Zetterberg, H.; Lundberg, P.; Dahle, C.; Vrethem, M.; Ernerudh, J. Neurofilament levels, disease activity and brain volume during follow-up in multiple sclerosis. J. Neuroinflamm. 2018, 15, 209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kouchaki, E.; Dashti, F.; Mirazimi, S.; Alirezaei, Z.; Jafari, S.H.; Hamblin, M.R.; Mirzaei, H. Neurofilament light chain as a biomarker for diagnosis of multiple sclerosis. EXCLI J. 2021, 20, 1308–1325. [Google Scholar] [CrossRef]
- Binder, L.I.; Frankfurter, A.; Rebhun, L.I. The distribution of tau in the mammalian central nervous system. J. Cell Biol. 1985, 101, 1371–1378. [Google Scholar] [CrossRef] [Green Version]
- LoPresti, P.; Szuchet, S.; Papasozomenos, S.C.; Zinkowski, R.P.; Binder, L.I. Functional implications for the microtubule-associated protein tau: Localization in oligodendrocytes. Proc. Natl. Acad. Sci. USA 1995, 92, 1036973. [Google Scholar] [CrossRef] [Green Version]
- LoPresti, P. Inducible Expression of a Truncated Form of Tau in Oligodendrocytes Elicits Gait Abnormalities and a Decrease in Myelin: Implications for Selective CNS Degenerative Diseases. Neurochem. Res. 2015, 40, 2188–2199. [Google Scholar] [CrossRef]
- LoPresti, P. Tau in Oligodendrocytes Takes Neurons in Sickness and in Health. Int. J. Mol. Sci. 2018, 19, 2408. [Google Scholar] [CrossRef] [Green Version]
- Anderson, J.M.; Hampton, D.W.; Patani, R.; Pryce, G.; Crowther, R.A.; Reynolds, R.; Franklin, R.J.; Giovannoni, G.; Compston, D.A.; Baker, D.; et al. Abnormally phosphorylated tau is associated with neuronal and axonal loss in experimental autoimmune encephalomyelitis and multiple sclerosis. Brain 2008, 131, 1736–1748. [Google Scholar] [CrossRef]
- Anderson, J.M.; Patani, R.; Reynolds, R.; Nicholas, R.; Compston, A.; Spillantini, M.G.; Chandran, S. Evidence for abnormal tau phosphorylation in early aggressive multiple sclerosis. Acta Neuropathol. 2009, 117, 583–589. [Google Scholar] [CrossRef]
- Ballatore, C.; Lee, V.M.; Trojanowski, J.Q. Tau-mediated neurodegeneration in Alzheimer’s disease and related disorders. Nature reviews. Neuroscience 2007, 8, 663–672. [Google Scholar]
- Mandelkow, E.M.; Mandelkow, E. Biochemistry and cell biology of tau protein in neurofibrillary degeneration. Cold Spring Harbor Perspect. Med. 2012, 2, a006247. [Google Scholar] [CrossRef] [PubMed]
- Bartosik-Psujek, H.; Stelmasiak, Z. The CSF levels of total-tau and phosphotau in patients with relapsing-remitting multiple sclerosis. J. Neural Transm. 2006, 113, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Schneider, A.; Araújo, G.W.; Trajkovic, K.; Herrmann, M.M.; Merkler, D.; Mandelkow, E.M.; Weissert, R.; Simons, M. Hyperphosphorylation and aggregation of tau in experimental autoimmune encephalomyelitis. J. Biol. Chem. 2004, 279, 55833–55839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rojas, J.C.; Bang, J.; Lobach, I.V.; Tsai, R.M.; Rabinovici, G.D.; Miller, B.L.; Boxer, A.L.; AL-108-231 Investigators. CSF neurofilament light chain and phosphorylated tau 181 predict disease progression in PSP. Neurology 2018, 90, e273–e281. [Google Scholar] [CrossRef] [PubMed]
- Momtazmanesh, S.; Shobeiri, P.; Saghazadeh, A.; Teunissen, C.E.; Burman, J.; Szalardy, L.; Klivenyi, P.; Bartos, A.; Fernandes, A.; Rezaei, N. Neuronal and glial CSF biomarkers in multiple sclerosis: A systematic review and meta-analysis. Rev. Neurosci. 2021, 32, 573–595. [Google Scholar] [CrossRef]
- Gehrmann, J.; Banati, R.B.; Cuzner, M.L.; Kreutzberg, G.W.; Newcombe, J. Amyloid precursor protein (APP) expression in multiple sclerosis lesions. Glia 1995, 15, 141–151. [Google Scholar] [CrossRef]
- Mattsson, N.; Axelsson, M.; Haghighi, S.; Malmeström, C.; Wu, G.; Anckarsäter, R.; Sankaranarayanan, S.; Andreasson, U.; Fredrikson, S.; Gundersen, A.; et al. Reduced cerebrospinal fluid BACE1 activity in multiple sclerosis. Multiple Sclerosis 2009, 15, 448–454. [Google Scholar] [CrossRef]
- Mathur, D.; Mishra, B.K.; Rout, S.; Lopez-Iranzo, F.J.; Lopez-Rodas, G.; Vallamkondu, J.; Kandimalla, R.; Casanova, B. Potential Biomarkers Associated with Multiple Sclerosis Pathology. Int. J. Mol. Sci. 2021, 22, 10323. [Google Scholar] [CrossRef]
- Fagan, A.M.; Mintun, M.A.; Shah, A.R.; Aldea, P.; Roe, C.M.; Mach, R.H.; Marcus, D.; Morris, J.C.; Holtzman, D.M. Cerebrospinal fluid tau and ptau (181) increase with cortical amyloid deposition in cognitively normal individuals: Implications for future clinical trials of Alzheimer’s disease. EMBO Mol. Med. 2009, 1, 371–380. [Google Scholar] [CrossRef]
- Bos, I.; Vos, S.; Verhey, F.; Scheltens, P.; Teunissen, C.; Engelborghs, S.; Sleegers, K.; Frisoni, G.; Blin, O.; Richardson, J.C.; et al. Cerebrospinal fluid biomarkers of neurodegeneration, synaptic integrity, and astroglial activation across the clinical Alzheimer’s disease spectrum. Alzheimers Dement. 2019. [Google Scholar] [CrossRef]
- Tarawneh, R. Biomarkers: Our Path Towards a Cure for Alzheimer Disease. Biomarker Insights 2020, 15, 1177271920976367. [Google Scholar] [CrossRef]
- Li, X.; Li, T.Q.; Andreasen, N.; Wiberg, M.K.; Westman, E.; Wahlund, L.O. Ratio of Aβ42/P-tau181p in CSF is associated with aberrant default mode network in AD. Sci. Rep. 2013, 3, 1339. [Google Scholar] [CrossRef] [PubMed]
- Janelidze, S.; Stomrud, E.; Smith, R.; Palmqvist, S.; Mattsson, N.; Airey, D.C.; Proctor, N.K.; Chai, X.; Shcherbinin, S.; Sims, J.R.; et al. Cerebrospinal fluid p-tau217 performs better than p-tau181 as a biomarker of Alzheimer’s disease. Nat. Commun. 2020, 11, 1683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Telser, J.; Risch, L.; Saely, C.H.; Grossmann, K.; Werner, P. P-tau217 in Alzheimer’s disease. Clin. Chim. Acta 2022, 531, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Moffett, J.R.; Ross, B.; Arun, P.; Madhavarao, C.N.; Namboodiri, A.M.A. N-Acetylaspartate in the CNS: From neurodiagnostics to neurobiology. Prog. Neurobiol. 2007, 81, 89–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, O.; Shen, Y.; Caon, C.; Bao, F.; Ching, W.; Reznar, M.; Buccheister, A.; Hu, J.; Latif, Z.; Tselis, A.; et al. Axonal metabolic recovery and potential neuroprotective effect of Glatiramer acetate in relapsing-remitting multiple sclerosis. Multiple Sclerosis 2005, 11, 646–651. [Google Scholar] [CrossRef]
- Narayanan, S.; De Stefano, N.; Francis, G.S.; Arnaoutelis, R.; Caramanos, Z.; Collins, D.L.; Pelletier, D.; Arnason, B.G.W.; Antel, J.P.; Arnold, D.L. Axonal metabolic recovery in multiple sclerosis patients treated with interferon beta-1b. J. Neurol. 2001, 248, 979–986. [Google Scholar] [CrossRef]
- Solanky, B.S.; John, N.A.; DeAngelis, F.; Stutters, J.; Prados, F.; Schneider, T.; Parker, R.A.; Weir, C.J.; Monteverdi, A.; Plantone, D.; et al. NAA is a Marker of Disability in Secondary-Progressive MS: A Proton MR Spectroscopic Imaging Study. Am. J. Neuroradiol. 2020, 41, 2209–2218. [Google Scholar] [CrossRef]
- Giovannoni, G. Multiple sclerosis cerebrospinal fluid biomarkers. Dis. Markers 2006, 22, 187–196. [Google Scholar] [CrossRef]
- Martínez-Yélamos, A.; Saiz, A.; Sanchez-Valle, R.; Casado, V.; Ramón, J.M.; Graus, F.; Arbizu, T. 14-3-3 Protein in the CSF as prognostic marker in early multiple sclerosis. Neurology 2001, 57, 722–724. [Google Scholar] [CrossRef]
- Martínez-Yélamos, A.; Rovira, A.; Sánchez-Valle, R.; Martínez-Yélamos, S.; Tintoré, M.; Blanco, Y.; Graus, F.; Montalban, X.; Arbizu, T.; Saiz, A. CSF 14-3-3 protein assay and MRI as prognostic markers in patients with a clinically isolated syndrome suggestive of MS. J. Neurol. 2004, 251, 1278–1279. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Cui, L.; Zeng, Y.; Song, W.; Gaur, U.; Yang, M. 14-3-3 Proteins Are on the Crossroads of Cancer, Aging, and Age-Related Neurodegenerative Disease. Int. J. Mol. Sci. 2019, 20, 3518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chatterjee, M.; Koel-Simmelink, M.J.; Verberk, I.M.; Killestein, J.; Vrenken, H.; Enzinger, C.; Ropele, S.; Fazekas, F.; Khalil, M.; Teunissen, C.E. Contactin-1 and contactin-2 in cerebrospinal fluid as potential biomarkers for axonal domain dysfunction in multiple sclerosis. Mult. Scler. J. 2018, 4, 2055217318819535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Lierop, Z.Y.; Wieske, L.; Koel-Simmelink, M.J.; Chatterjee, M.; Dekker, I.; Leurs, C.E.; Willemse, E.A.; Moraal, B.; Barkhof, F.; Eftimov, F.; et al. Serum contactin-1 as a biomarker of long-term disease progression in natalizumab-treated multiple sclerosis. Mult. Scler. J. 2022, 28, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.H.; Yu, J.W.; Wu, J.G.; Wang, S.L.; Jiang, B.J. Significances of contactin-1 expression in human gastric cancer and knockdown of contactin-1 expression inhibits invasion and metastasis of MKN45 gastric cancer cells. J. Cancer Res. Clin. Oncol. 2015, 141, 2109–2120. [Google Scholar] [CrossRef] [Green Version]
- Pallante, B.A.; Giovannone, S.; Fang-Yu, L.; Zhang, J.; Liu, N.; Kang, G.; Dun, W.; Boyden, P.A.; Fishman, G.I. Contactin-2 expression in the cardiac Purkinje fiber network. Circulation. Arrhythmia Electrophysiol. 2010, 3, 186–194. [Google Scholar] [CrossRef] [Green Version]
- De Vito, F.; Musella, A.; Fresegna, D.; Rizzo, F.R.; Gentile, A.; Stampanoni Bassi, M.; Gilio, L.; Buttari, F.; Procaccini, C.; Colamatteo, A.; et al. MiR-142-3p regulates synaptopathy-driven disease progression in multiple sclerosis. Neuropathol. Appl. Neurobiol. 2021, 48, e12765. [Google Scholar] [CrossRef]
- Axelsson, M.; Malmeström, C.; Nilsson, S.; Haghighi, S.; Rosengren, L.; Lycke, J. Glial fibrillary acidic protein: A potential biomarker for progression in multiple sclerosis. J. Neurol. 2011, 258, 882–888. [Google Scholar] [CrossRef]
- Abdelhak, A.; Hottenrott, T.; Morenas-Rodríguez, E.; Suárez-Calvet, M.; Zettl, U.K.; Haass, C.; Meuth, S.G.; Rauer, S.; Otto, M.; Tumani, H.; et al. Glial Activation Markers in CSF and Serum from Patients With Primary Progressive Multiple Sclerosis: Potential of Serum GFAP as Disease Severity Marker? Front. Neurol. 2019, 10, 280. [Google Scholar] [CrossRef]
- Tanaka, M.; Vécsei, L. Monitoring the Redox Status in Multiple Sclerosis. Biomedicines 2020, 8, 406. [Google Scholar] [CrossRef]
- Arslan, B.; Arslan, G.A.; Tuncer, A.; Karabudak, R.; Dinçel, A.S. Evaluation of Thiol Homeostasis in Multiple Sclerosis and Neuromyelitis Optica Spectrum Disorders. Front. Neurol. 2021, 12, 716195. [Google Scholar] [CrossRef] [PubMed]
- Bivona, G.; Gambino, C.M.; Lo Sasso, B.; Scazzone, C.; Giglio, R.V.; Agnello, L.; Ciaccio, M. Serum Vitamin D as a Biomarker in Autoimmune, Psychiatric and Neurodegenerative Diseases. Diagnostics 2022, 12, 130. [Google Scholar] [CrossRef] [PubMed]
- Börnsen, L.; Khademi, M.; Olsson, T.; Sørensen, P.S.; Sellebjerg, F. Osteopontin concentrations are increased in cerebrospinal fluid during attacks of multiple sclerosis. Mult. Scler. J. 2011, 17, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Jakovac, H.; Grubić Kezele, T.; Šućurović, S.; Mulac-Jeričević, B.; Radošević-Stašić, B. Osteopontin-metallothionein I/II interactions in experimental autoimmune encephalomyelitis. Neuroscience 2017, 350, 133–145. [Google Scholar] [CrossRef]
- LoPresti, P. HDAC6 in Diseases of Cognition and of Neurons. Cells 2020, 10, 12. [Google Scholar] [CrossRef]
- Strebl, M.G.; Campbell, A.J.; Zhao, W.N.; Schroeder, F.A.; Riley, M.M.; Chindavong, P.S.; Morin, T.M.; Haggarty, S.J.; Wagner, F.F.; Ritter, T.; et al. HDAC6 Brain Mapping with [18F] Bavarostat Enabled by a Ru-Mediated Deoxyfluorination. ACS Central Sci. 2017, 3, 1006–1014. [Google Scholar] [CrossRef] [Green Version]
- Joilin, G.; Gray, E.; Thompson, A.G.; Bobeva, Y.; Talbot, K.; Weishaupt, J.; Ludolph, A.; Malaspina, A.; Leigh, P.N.; Newbury, S.F.; et al. Identification of a potential non-coding RNA biomarker signature for amyotrophic lateral sclerosis. Brain Commun. 2020, 2, fcaa053. [Google Scholar] [CrossRef]

| Sample | Detection | References |
|---|---|---|
| Serum | ELISA | [65] |
| ECL | [82,83] | |
| SIMOA | [84,85,86] | |
| Blood | SIMOA | [68] |
| CSF | ELISA | [87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103] |
| Plasma | SIMOA | [104] |
| Serum + CSF | ELISA | [105,106] |
| ECL | [107] | |
| SIMOA | [108,109,110,111,112,113] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
LoPresti, P. Serum-Based Biomarkers in Neurodegeneration and Multiple Sclerosis. Biomedicines 2022, 10, 1077. https://doi.org/10.3390/biomedicines10051077
LoPresti P. Serum-Based Biomarkers in Neurodegeneration and Multiple Sclerosis. Biomedicines. 2022; 10(5):1077. https://doi.org/10.3390/biomedicines10051077
Chicago/Turabian StyleLoPresti, Patrizia. 2022. "Serum-Based Biomarkers in Neurodegeneration and Multiple Sclerosis" Biomedicines 10, no. 5: 1077. https://doi.org/10.3390/biomedicines10051077
APA StyleLoPresti, P. (2022). Serum-Based Biomarkers in Neurodegeneration and Multiple Sclerosis. Biomedicines, 10(5), 1077. https://doi.org/10.3390/biomedicines10051077





