Ammonium Glycyrrhizinate and Bergamot Essential Oil Co-Loaded Ultradeformable Nanocarriers: An Effective Natural Nanomedicine for In Vivo Anti-Inflammatory Topical Therapies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Ultradeformable Nanocarriers Preparation and Purification
2.3. Physicochemical Characterization
2.4. High Performance Liquid Chromatography (HPLC) Instrument
2.5. Evaluation of Entrapment Efficiency of Natural Compounds
2.6. Freeze-Drying Stability Studies
2.7. In Vitro Ammonium Glycyrrhizinate Release Kinetic
2.8. Deformability Assay
2.9. In Vivo Evaluation of Topical Tolerability
2.10. In Vivo Anti-Inflammatory Evaluation of Vesicles
2.11. Statistical Analysis
3. Results and Discussion
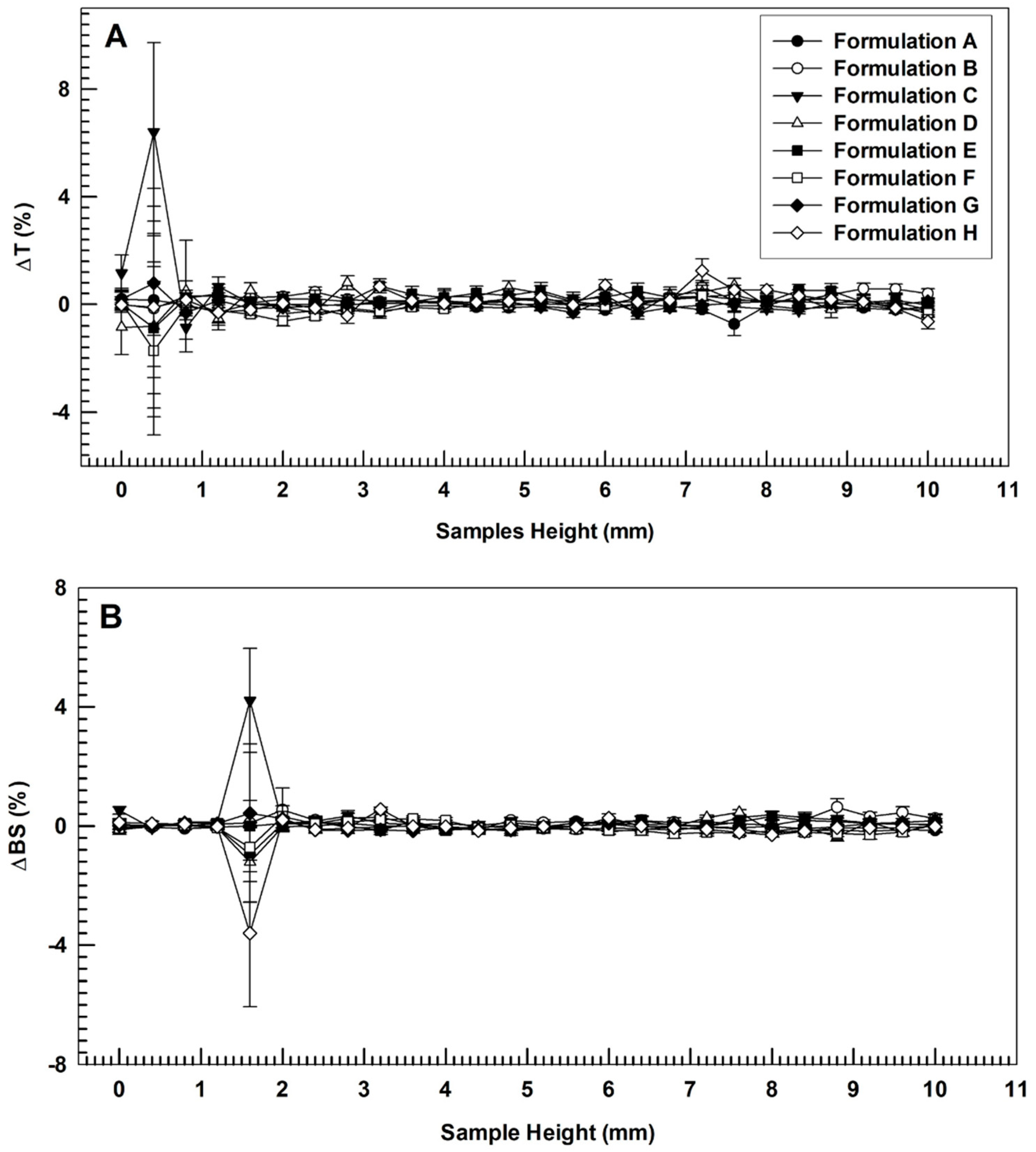
3.1. Physicochemical and Technological Characterization of Ultradeformable Nanocarriers
3.2. Entrapment Efficiency Evaluation
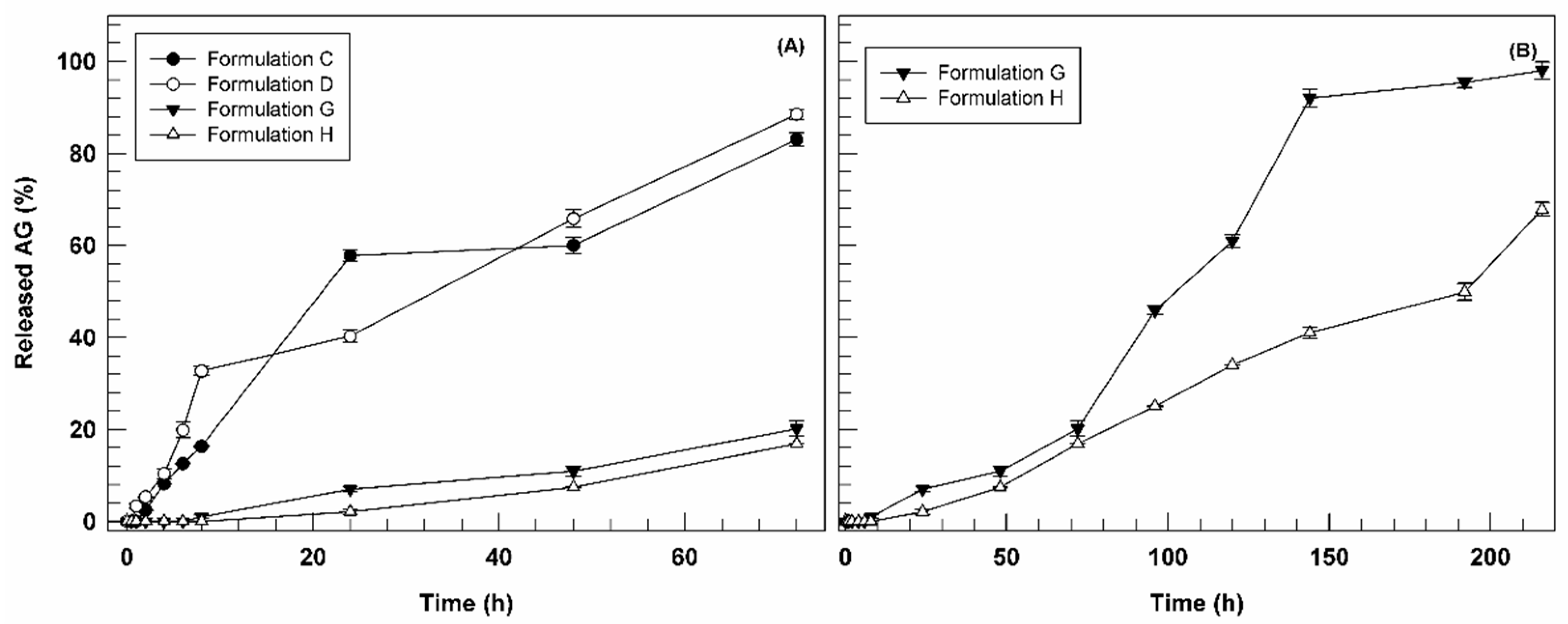
3.3. In Vitro Ammonium Glycyrrhizinate Release Studies
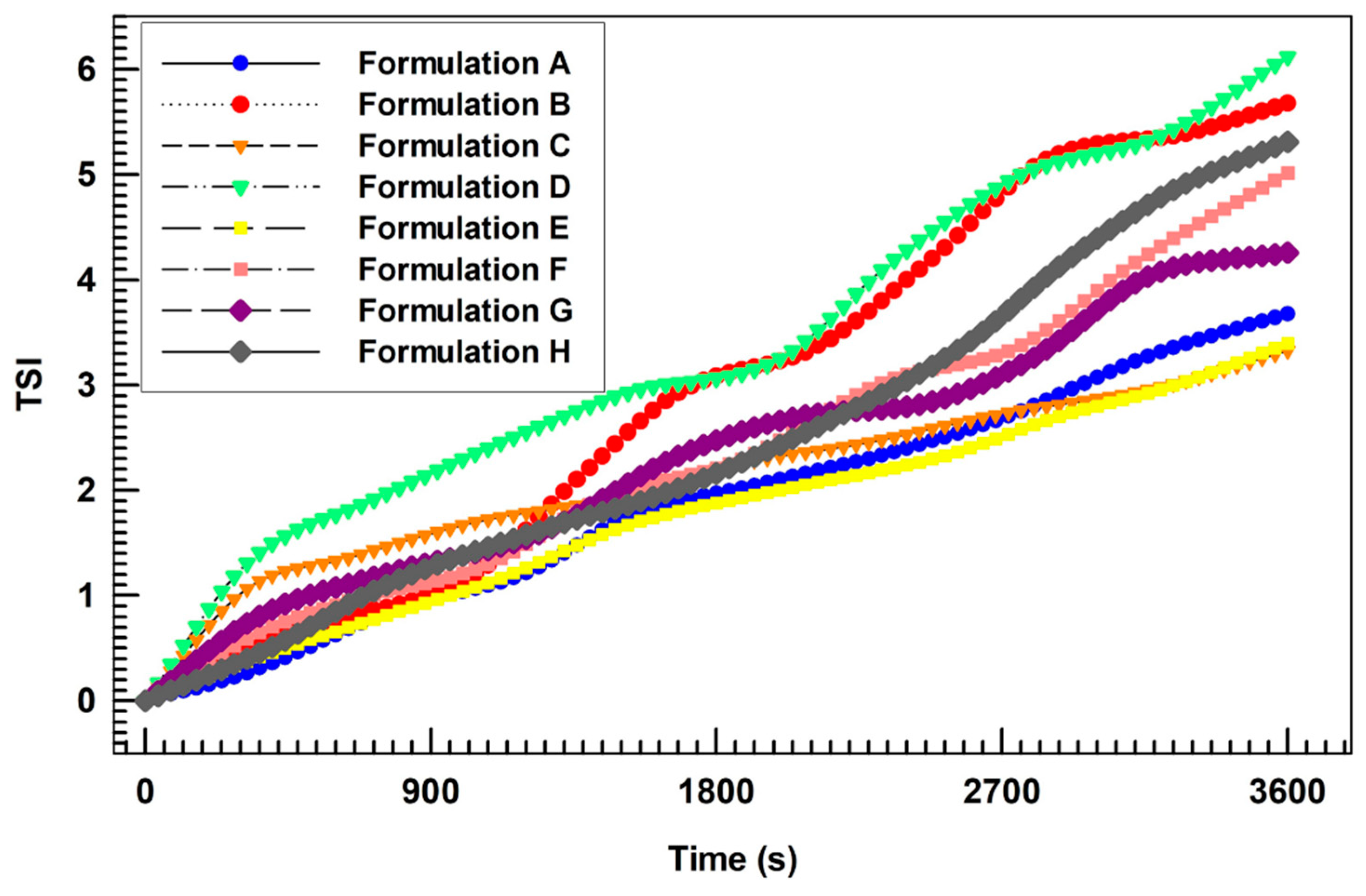
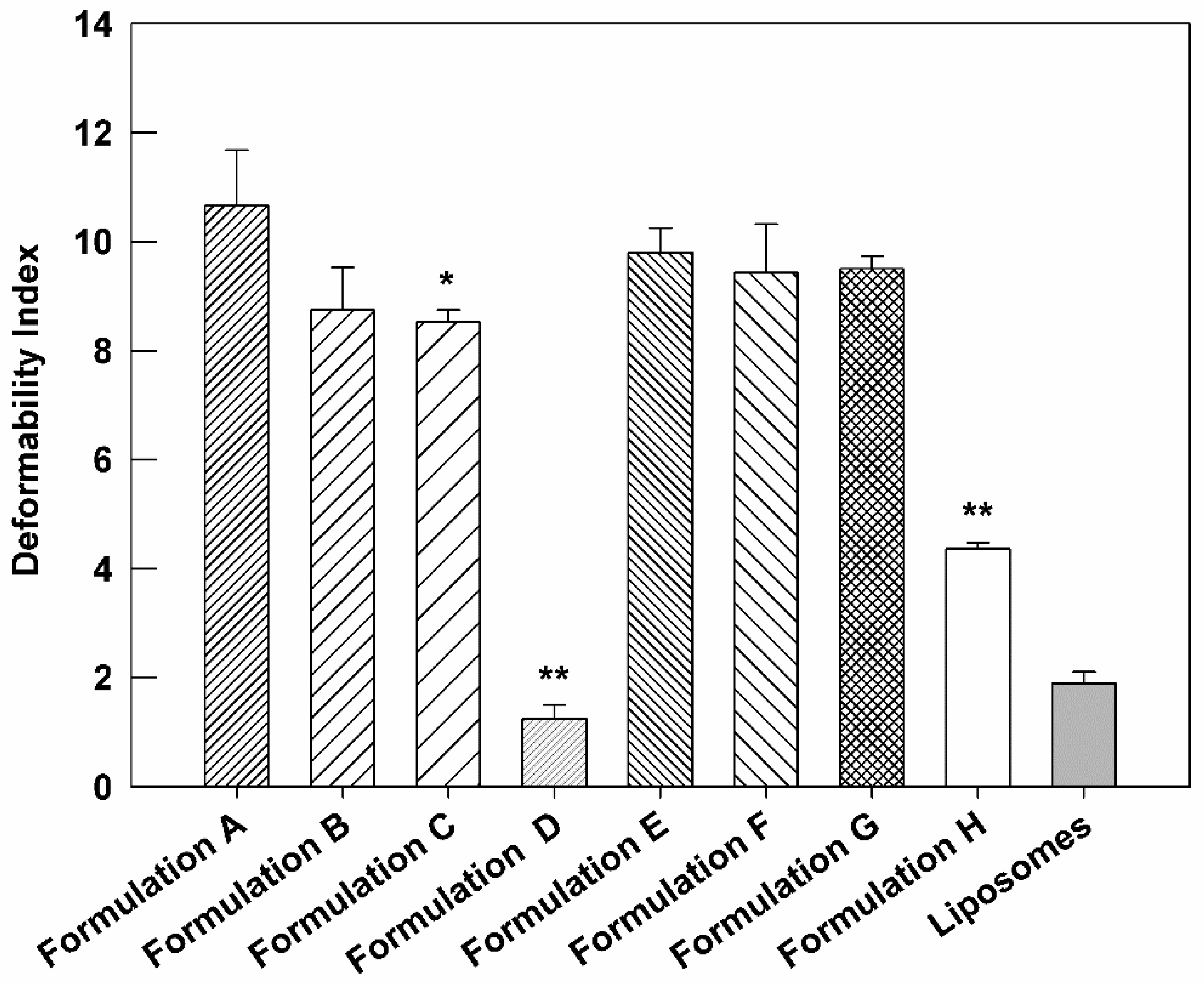
3.4. Deformability Index
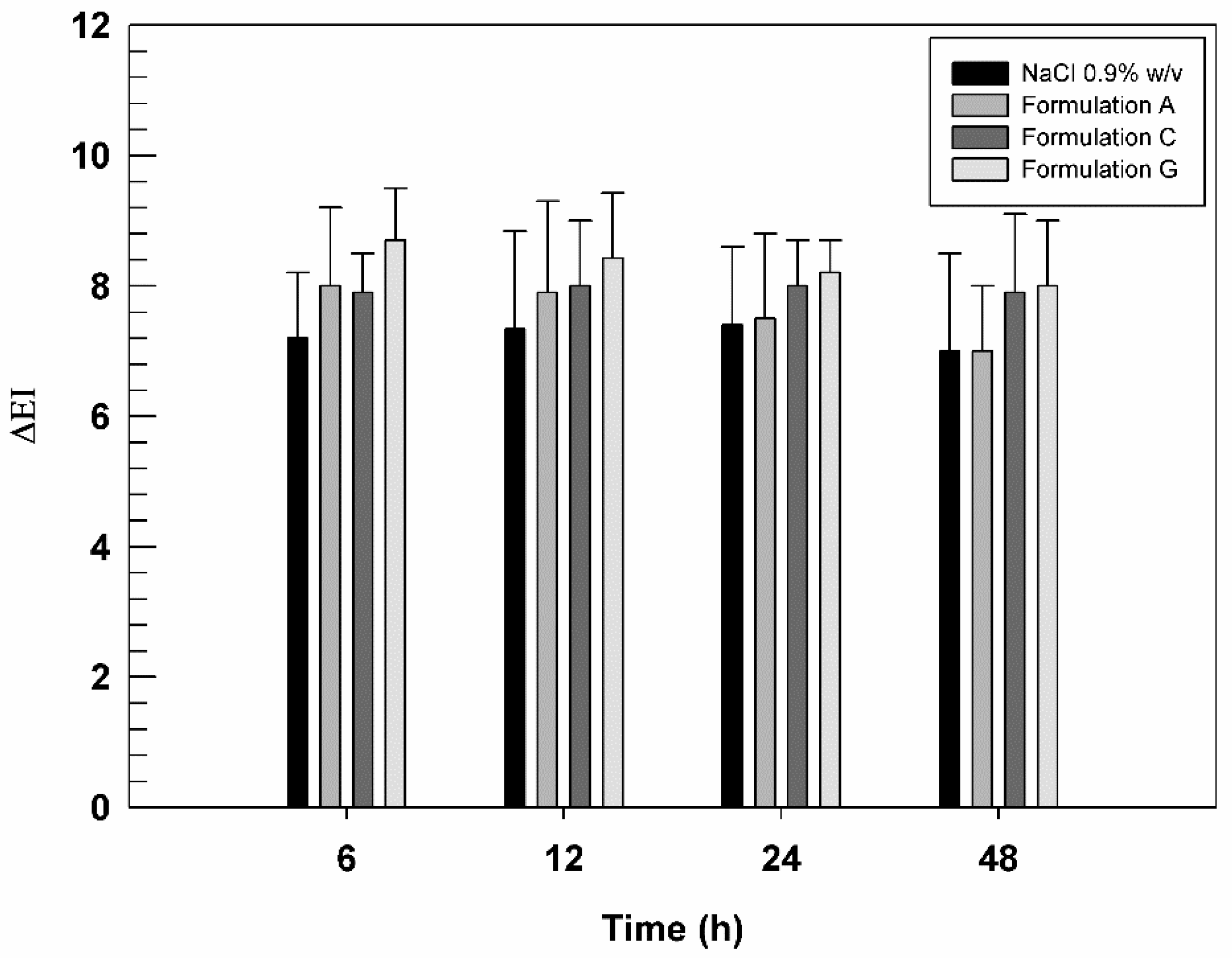
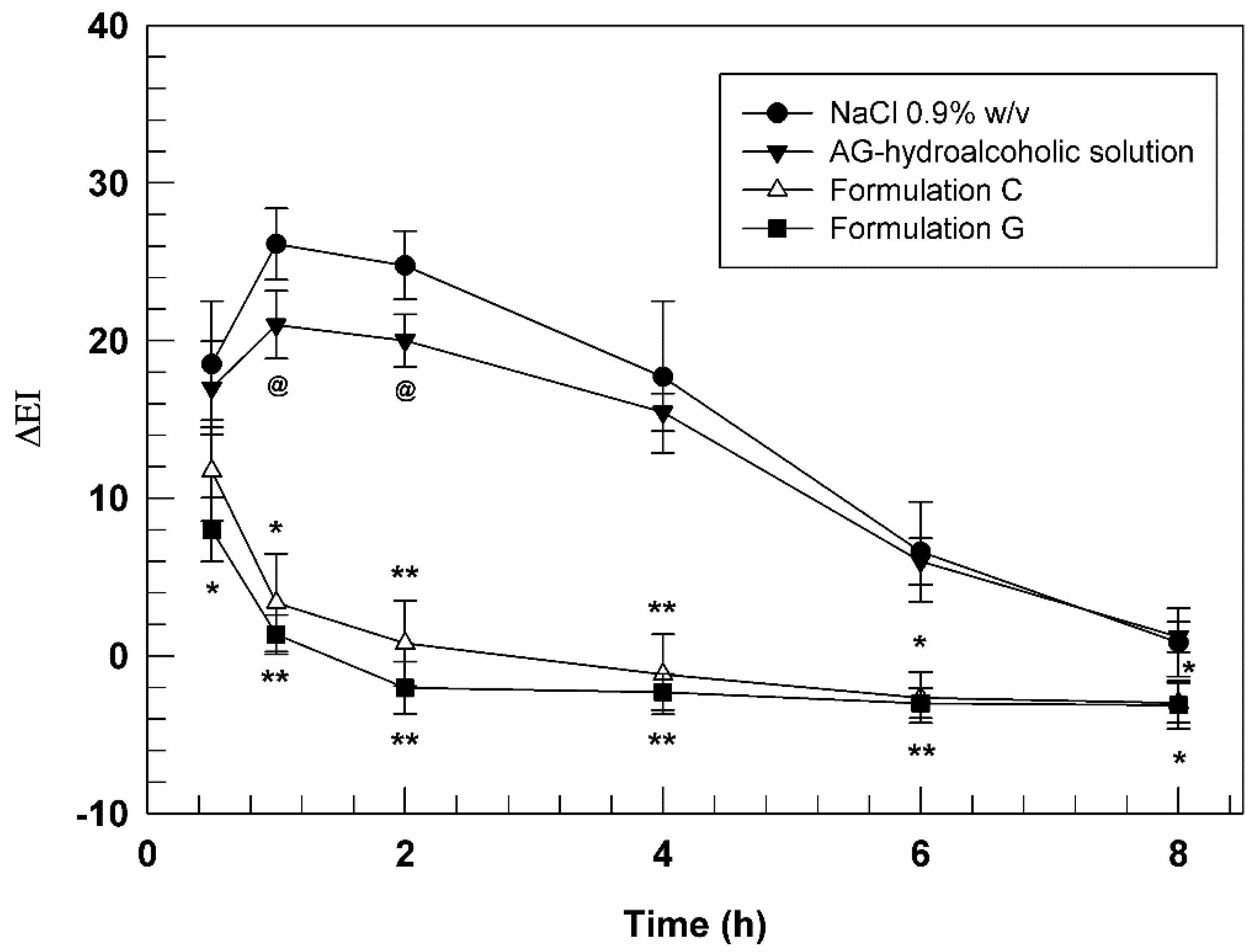
3.5. In Vivo Studies on Human Health Volunteers
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Allijn, I.E.; Brinkhuis, R.P.; Storm, G.; Schiffelers, R.M. Anti-inflammatory properties of plant derived natural products—A systematic review. Curr. Med. Chem. 2019, 26, 4506–4536. [Google Scholar] [CrossRef] [PubMed]
- Azab, A.; Nassar, A.; Azab, A.N. Anti-inflammatory activity of natural products. Molecules 2016, 21, 1321. [Google Scholar] [CrossRef] [PubMed]
- Raskin, I.; Ribnicky, D.M.; Komarnytsky, S.; Ilic, N.; Poulev, A.; Borisjuk, N.; Brinker, A.; Moreno, D.A.; Ripoll, C.; Yakoby, N. Plants and human health in the twenty-first century. Trends Biotechnol. 2002, 20, 522–531. [Google Scholar] [CrossRef]
- Yeo, S.K.; Ali, A.Y.; Hayward, O.A.; Turnham, D.; Jackson, T.; Bowen, I.D.; Clarkson, R. β-Bisabolene, a sesquiterpene from the essential oil extract of opoponax (Commiphora guidottii), exhibits cytotoxicity in breast cancer cell lines. Phytother. Res. 2016, 30, 418–425. [Google Scholar] [CrossRef]
- De Oliveira, E.F.; Paula, H.C.; de Paula, R.C. Alginate/cashew gum nanoparticles for essential oil encapsulation. Colloids Surf. B Biointerfaces 2014, 113, 146–151. [Google Scholar] [CrossRef]
- Elkordy, A.A.; Haj-Ahmad, R.R.; Awaad, A.S.; Zaki, R.M. An overview on natural product drug formulations from conventional medicines to nanomedicines: Past, present and future. J. Drug Deliv. Sci. Technol. 2021, 63, 102459. [Google Scholar] [CrossRef]
- Ghodrati, M.; Farahpour, M.R.; Hamishehkar, H. Encapsulation of Peppermint essential oil in nanostructured lipid carriers: In-vitro antibacterial activity and accelerative effect on infected wound healing. Colloids Surf. Physicochem. Eng. Asp. 2019, 564, 161–169. [Google Scholar] [CrossRef]
- Martins, J.P.; Das Neves, J.; de la Fuente, M.; Celia, C.; Florindo, H.; Günday-Türeli, N.; Popat, A.; Santos, J.L.; Sousa, F.; Schmid, R. The solid progress of nanomedicine. Drug Deliv. Transl. Res. 2020, 10, 726–729. [Google Scholar] [CrossRef] [Green Version]
- Dosoky, N.S.; Setzer, W.N. Biological activities and safety of Citrus spp. essential oils. Int. J. Mol. Sci. 2018, 19, 1966. [Google Scholar] [CrossRef] [Green Version]
- Forlot, P.; Pevet, P. Bergamot (Citrus bergamia Risso et Poiteau) essential oil: Biological properties, cosmetic and medical use. A review. J. Essent. Oil Res. 2012, 24, 195–201. [Google Scholar] [CrossRef]
- Nauman, M.C.; Johnson, J.J. Clinical application of bergamot (Citrus bergamia) for reducing high cholesterol and cardiovascular disease markers. Integr. Food Nutr. Metab. 2019, 6, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhia, M.; Motallebi, M.; Abadi, B.; Zarepour, A.; Pereira-Silva, M.; Saremnejad, F.; Santos, A.C.; Zarrabi, A.; Melero, A.; Jafari, S.M.; et al. Naringenin nano-delivery systems and their therapeutic applications. Pharmaceutics 2021, 13, 291. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.-C.; Tang, N.; Liu, J.-Y.; Zhou, A.-M.; Huang, K.-X.; Chen, S.-X. Preparation and Antioxidant Activity of Bergamot Essential Oil Liposomes. Mod. Food Sci. Technol. 2020, 36, 210–221. [Google Scholar] [CrossRef]
- Lombardo, G.E.; Cirmi, S.; Musumeci, L.; Pergolizzi, S.; Maugeri, A.; Russo, C.; Mannucci, C.; Calapai, G.; Navarra, M. Mechanisms underlying the anti-inflammatory activity of bergamot essential oil and its antinociceptive effects. Plants 2020, 9, 704. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Kaur, A.; Kaur, J.; Bhatti, M.S.; Singh, P.; Bhatti, R. Bergapten inhibits chemically induced nociceptive behavior and inflammation in mice by decreasing the expression of spinal PARP, iNOS, COX-2 and inflammatory cytokines. Inflammopharmacology 2019, 27, 749–760. [Google Scholar] [CrossRef]
- Zheng, D.-S.; Chen, L.-S. Bergapten Attenuates Allergic Inflammation in Mast Cells Through Inhibition of Histamine Release and Pro-Inflammatory Cytokines. Lat. Am. J. Pharm. 2016, 35, 1855–1860. [Google Scholar]
- Zhou, Y.; Wang, J.; Yang, W.; Qi, X.; Lan, L.; Luo, L.; Yin, Z. Bergapten prevents lipopolysaccharide-induced inflammation in RAW264. 7 cells through suppressing JAK/STAT activation and ROS production and increases the survival rate of mice after LPS challenge. Int. Immunopharmacol. 2017, 48, 159–168. [Google Scholar] [CrossRef]
- Hasan, M.K.; Ara, I.; Mondal, M.S.A.; Kabir, Y. Phytochemistry, pharmacological activity, and potential health benefits of Glycyrrhiza glabra. Heliyon 2021, 7, e07240. [Google Scholar] [CrossRef]
- Maione, F.; Minosi, P.; Di Giannuario, A.; Raucci, F.; Chini, M.G.; De Vita, S.; Bifulco, G.; Mascolo, N.; Pieretti, S. Long-lasting anti-inflammatory and antinociceptive effects of acute ammonium glycyrrhizinate administration: Pharmacological, biochemical, and docking studies. Molecules 2019, 24, 2453. [Google Scholar] [CrossRef] [Green Version]
- Bhattacharjee, S.; Bhattacharjee, A.; Majumder, S.; Majumdar, S.B.; Majumdar, S. Glycyrrhizic acid suppresses Cox-2-mediated anti-inflammatory responses during Leishmania donovani infection. J. Antimicrob. Chemother. 2012, 67, 1905–1914. [Google Scholar] [CrossRef] [Green Version]
- Karlberg, A.T.; Magnusson, K.; Nilsson, U. Air oxidation of d-limonene (the citrus solvent) creates potent allergens. Contact Dermat. 1992, 26, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Navarra, M.; Mannucci, C.; Delbò, M.; Calapai, G. Citrus bergamia essential oil: From basic research to clinical application. Front. Pharmacol. 2015, 6, 36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Opatha, S.A.T.; Titapiwatanakun, V.; Chutoprapat, R. Transfersomes: A promising nanoencapsulation technique for transdermal drug delivery. Pharmaceutics 2020, 12, 855. [Google Scholar] [CrossRef] [PubMed]
- Zeb, A.; Qureshi, O.S.; Kim, H.-S.; Cha, J.-H.; Kim, H.-S.; Kim, J.-K. Improved skin permeation of methotrexate via nanosized ultradeformable liposomes. Int. J. Nanomed. 2016, 11, 3813. [Google Scholar] [CrossRef] [Green Version]
- Cevc, G. Lipid vesicles and other colloids as drug carriers on the skin. Adv. Drug Deliv. Rev. 2004, 56, 675–711. [Google Scholar] [CrossRef]
- El Zaafarany, G.M.; Awad, G.A.; Holayel, S.M.; Mortada, N.D. Role of edge activators and surface charge in developing ultradeformable vesicles with enhanced skin delivery. Int. J. Pharm. 2010, 39, 164–172. [Google Scholar] [CrossRef]
- Cevc, G.; Schatzlein, A.G.; Richardsen, H. Ultradeformable lipid vesicles can penetrate the skin and other semi-permeable barriers unfragmented. Evidence from double label CLSM experiments and direct size measurements. Biochim. Biophys. Acta (BBA)-Biomembr. 2002, 1564, 21–30. [Google Scholar] [CrossRef] [Green Version]
- Elsayed, M.M.; Abdallah, O.Y.; Naggar, V.F.; Khalafallah, N.M. Lipid vesicles for skin delivery of drugs: Reviewing three decades of research. Int. J. Pharm. 2007, 332, 1–16. [Google Scholar] [CrossRef]
- Cristiano, M.C.; Froiio, F.; Spaccapelo, R.; Mancuso, A.; Nisticò, S.P.; Udongo, B.P.; Fresta, M.; Paolino, D. Sulforaphane-Loaded Ultradeformable Vesicles as A Potential Natural Nanomedicine for the Treatment of Skin Cancer Diseases. Pharmaceutics 2019, 12, 6. [Google Scholar] [CrossRef] [Green Version]
- Di Francesco, M.; Celia, C.; Cristiano, M.C.; d’Avanzo, N.; Ruozi, B.; Mircioiu, C.; Cosco, D.; Di Marzio, L.; Fresta, M. Doxorubicin hydrochloride-loaded nonionic surfactant vesicles to treat metastatic and non-metastatic breast cancer. ACS omega 2021, 6, 2973–2989. [Google Scholar] [CrossRef]
- Barone, A.; Cristiano, M.C.; Cilurzo, F.; Locatelli, M.; Iannotta, D.; Di Marzio, L.; Celia, C.; Paolino, D. Ammonium glycyrrhizate skin delivery from ultradeformable liposomes: A novel use as an anti-inflammatory agent in topical drug delivery. Colloids Surf. B 2020, 193, 111152. [Google Scholar] [CrossRef] [PubMed]
- D’Avanzo, N.; Torrieri, G.; Figueiredo, P.; Celia, C.; Paolino, D.; Correia, A.; Moslova, K.; Teesalu, T.; Fresta, M.; Santos, H.A. LinTT1 peptide-functionalized liposomes for targeted breast cancer therapy. Int. J. Pharm. 2021, 597, 120346. [Google Scholar] [CrossRef] [PubMed]
- Kirjavainen, M.; Mönkkönen, J.; Saukkosaari, M.; Valjakka-Koskela, R.; Kiesvaara, J.; Urtti, A. Phospholipids affect stratum corneum lipid bilayer fluidity and drug partitioning into the bilayers. J. Control Release 1999, 58, 207–214. [Google Scholar] [CrossRef]
- Cristiano, M.C.; Mancuso, A.; Giuliano, E.; Cosco, D.; Paolino, D.; Fresta, M. EtoGel for Intra-Articular Drug Delivery: A New Challenge for Joint Diseases Treatment. J. Funct. Biomater. 2021, 12, 34. [Google Scholar] [CrossRef]
- Müller-Goymann, C. Physicochemical characterization of colloidal drug delivery systems such as reverse micelles, vesicles, liquid crystals and nanoparticles for topical administration. Eur. J. Pharm. Biopharm. 2004, 58, 343–356. [Google Scholar] [CrossRef]
- Ahad, A.; Al-Saleh, A.A.; Al-Mohizea, A.M.; Al-Jenoobi, F.I.; Raish, M.; Yassin, A.E.B.; Alam, M.A. Formulation and characterization of novel soft nanovesicles for enhanced transdermal delivery of eprosartan mesylate. Saudi Pharm. J. 2017, 25, 1040–1046. [Google Scholar] [CrossRef]
- Marwah, H.; Garg, T.; Goyal, A.K.; Rath, G. Permeation enhancer strategies in transdermal drug delivery. Drug Deliv. 2016, 23, 564–578. [Google Scholar] [CrossRef]
- Fang, J.-Y.; Lin, Y.-K.; Wang, P.-W.; Alalaiwe, A.; Yang, Y.-C.; Yang, S.-C. The droplet-size effect of squalene@ cetylpyridinium chloride nanoemulsions on antimicrobial potency against planktonic and biofilm MRSA. Int. J. Nanomed. 2019, 14, 8133. [Google Scholar] [CrossRef] [Green Version]
- Yu, H.-P.; Liu, F.-C.; Umoro, A.; Lin, Z.-C.; Elzoghby, A.O.; Hwang, T.-L.; Fang, J.-Y. Oleic acid-based nanosystems for mitigating acute respiratory distress syndrome in mice through neutrophil suppression: How the particulate size affects therapeutic efficiency. J. Nanobiotech. 2020, 18, 1–16. [Google Scholar] [CrossRef]
- Calienni, M.N.; Prieto, M.J.; Couto, V.M.; de Paula, E.; Alonso, S.d.V.; Montanari, J. 5-Fluorouracil-loaded ultradeformable liposomes for skin therapy. In AIP Conference Proceedings; AIP Publishing LLC: Melville, NY, USA, 2018; p. 020024. [Google Scholar] [CrossRef] [Green Version]
- Cristiano, M.C.; Froiio, F.; Mancuso, A.; Cosco, D.; Dini, L.; Di Marzio, L.; Fresta, M.; Paolino, D. Oleuropein-laded ufasomes improve the nutraceutical efficacy. Nanomaterials 2021, 11, 105. [Google Scholar] [CrossRef]
- Charoenviriyakul, C.; Takahashi, Y.; Nishikawa, M.; Takakura, Y. Preservation of exosomes at room temperature using lyophilization. Int. J. Pharm. 2018, 553, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Jiao, Y.; Zhang, Z. Calcium-carboxymethyl chitosan hydrogel beads for protein drug delivery system. J. Appl. Polym. Sci. 2007, 103, 3164–3168. [Google Scholar] [CrossRef]
- Natsheh, H.; Touitou, E. Phospholipid vesicles for dermal/transdermal and nasal administration of active molecules: The effect of surfactants and alcohols on the fluidity of their lipid bilayers and penetration enhancement properties. Molecules 2020, 25, 2959. [Google Scholar] [CrossRef] [PubMed]
- Maritim, S.; Boulas, P.; Lin, Y. Comprehensive analysis of liposome formulation parameters and their influence on encapsulation, stability and drug release in glibenclamide liposomes. Int. J. Pharm. 2021, 592, 120051. [Google Scholar] [CrossRef]
- Stark, B.; Pabst, G.; Prassl, R. Long-term stability of sterically stabilized liposomes by freezing and freeze-drying: Effects of cryoprotectants on structure. Eur. J. Pharm. Sci. 2010, 41, 546–555. [Google Scholar] [CrossRef]
- Matsuoka, K.; Miyajima, R.; Ishida, Y.; Karasawa, S.; Yoshimura, T. Aggregate formation of glycyrrhizic acid. Colloids Surf. Physicochem. Eng. Asp. 2016, 500, 112–117. [Google Scholar] [CrossRef]
- Ma, X.; Santiago, N.; Chen, Y.-S.; Chaudhary, K.; Milstein, S.J.; Baughman, R.A. Stability study of drug-loaded proteinoid microsphere formulations during freeze-drying. J. Drug Target. 1994, 2, 9–21. [Google Scholar] [CrossRef]
- El Kateb, N.; Cynober, L.; Chaumeil, J.C.; Dumortier, G. L-cysteine encapsulation in liposomes: Effect of phospholipids nature on entrapment efficiency and stability. J. Microencaps. 2008, 25, 399–413. [Google Scholar] [CrossRef]
- Nahak, P.; Karmakar, G.; Roy, B.; Guha, P.; Sapkota, M.; Koirala, S.; Chang, C.-H.; Panda, A.K. Physicochemical studies on local anaesthetic loaded second generation nanolipid carriers. RSC Adv. 2015, 5, 26061–26070. [Google Scholar] [CrossRef]
- Dayan, N.; Touitou, E. Carriers for skin delivery of trihexyphenidyl HCl: Ethosomes vs. liposomes. Biomaterials 2000, 21, 1879–1885. [Google Scholar] [CrossRef]
- Tisserand, R.; Young, R. Essential Oil Safety, 2nd ed.; Elsevier: New York, NY, USA, 2014. [Google Scholar]
- Jain, S.; Jain, P.; Umamaheshwari, R.B.; Jain, N.K. Transfersomes—A Novel Vesicular Carrier for Enhanced Transdermal Delivery: Development, Characterization, and Performance Evaluation. Drug Dev. Ind. Pharm. 2003, 29, 1013–1026. [Google Scholar] [CrossRef] [PubMed]
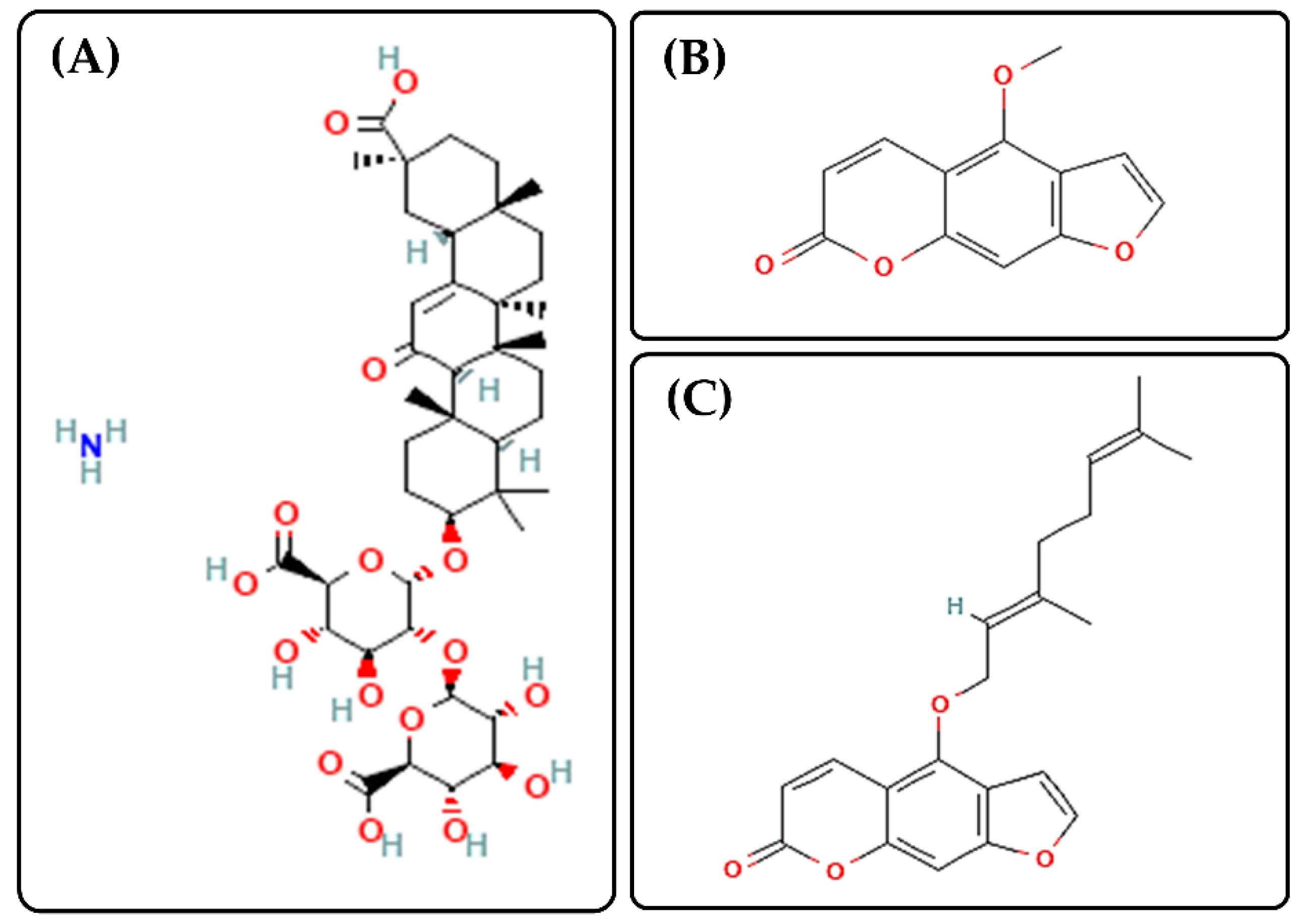
| Sample | AG (mg/mL) | BEO (% w/v) |
|---|---|---|
| A | - | - |
| B | 3 | - |
| C | 5 | - |
| D | 7.5 | - |
| E | - | 1 |
| F | 3 | 1 |
| G | 5 | 1 |
| H | 7.5 | 1 |
| Sample | Mean Size (nm) | PdI a | Net Surface Charge (mV) |
|---|---|---|---|
| A | 135 ± 2 | 0.109 ± 0.019 | −35.9 ± 0.2 |
| B | 134 ± 1 | 0.110 ± 0.001 | −32.6 ± 0.4 |
| C | 119 ± 1 | 0.117 ± 0.008 | −37.3 ± 1.3 |
| D | 105 ± 1 | 0.168 ± 0.003 | −39.0 ± 1.3 |
| E | 133 ±1 | 0.082 ± 0.026 | −30.5 ± 0.8 |
| F | 126 ± 1 | 0.091 ± 0.007 | −32.8 ± 0.6 |
| G | 129 ± 1 | 0.120 ± 0.024 | −43.2 ± 1.6 |
| H | 124 ± 1 | 0.139 ± 0.022 | −46.7 ± 0.4 |
| Sample | AG E.E. (%) | BEO E.E. (%) |
|---|---|---|
| B | 39.5 ± 0.6 | - |
| C | 77.8 ± 1.0 | - |
| D | 75.1 ± 1.8 | - |
| E | - | 33.1 ± 0.2 |
| F | 45.2 ± 0.6 | 28.1 ± 0.7 |
| G | 65.0 ± 2.0 | 26.4 ± 1.3 |
| H | 74.2 ± 1.0 | 24.0 ± 0.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cristiano, M.C.; d’Avanzo, N.; Mancuso, A.; Tarsitano, M.; Barone, A.; Torella, D.; Paolino, D.; Fresta, M. Ammonium Glycyrrhizinate and Bergamot Essential Oil Co-Loaded Ultradeformable Nanocarriers: An Effective Natural Nanomedicine for In Vivo Anti-Inflammatory Topical Therapies. Biomedicines 2022, 10, 1039. https://doi.org/10.3390/biomedicines10051039
Cristiano MC, d’Avanzo N, Mancuso A, Tarsitano M, Barone A, Torella D, Paolino D, Fresta M. Ammonium Glycyrrhizinate and Bergamot Essential Oil Co-Loaded Ultradeformable Nanocarriers: An Effective Natural Nanomedicine for In Vivo Anti-Inflammatory Topical Therapies. Biomedicines. 2022; 10(5):1039. https://doi.org/10.3390/biomedicines10051039
Chicago/Turabian StyleCristiano, Maria Chiara, Nicola d’Avanzo, Antonia Mancuso, Martine Tarsitano, Antonella Barone, Daniele Torella, Donatella Paolino, and Massimo Fresta. 2022. "Ammonium Glycyrrhizinate and Bergamot Essential Oil Co-Loaded Ultradeformable Nanocarriers: An Effective Natural Nanomedicine for In Vivo Anti-Inflammatory Topical Therapies" Biomedicines 10, no. 5: 1039. https://doi.org/10.3390/biomedicines10051039
APA StyleCristiano, M. C., d’Avanzo, N., Mancuso, A., Tarsitano, M., Barone, A., Torella, D., Paolino, D., & Fresta, M. (2022). Ammonium Glycyrrhizinate and Bergamot Essential Oil Co-Loaded Ultradeformable Nanocarriers: An Effective Natural Nanomedicine for In Vivo Anti-Inflammatory Topical Therapies. Biomedicines, 10(5), 1039. https://doi.org/10.3390/biomedicines10051039












