Time Course of Neurobehavioral Disruptions and Regional Brain Metabolism Changes in the Rotenone Mice Model of Parkinson’s Disease
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Experimental Procedures
2.2. Complex Evaluation of Animal Behavior
2.3. Dynamics of the Food-Getting Behavior Formation in the ShuttleBox Test
2.4. Tissue Collection and Processing
2.5. Immunohistochemistry
2.6. Cytochrome-C-Oxidase (COX) Histochemistry
2.7. Lipid Peroxidation and Endogenous Antioxidant System
2.8. High-Performance Liquid Chromatography with Electrochemical Detection
2.9. Statistical Analysis
3. Results
3.1. Time Course of Behavioral Changes in Rotenone-Treated Animals
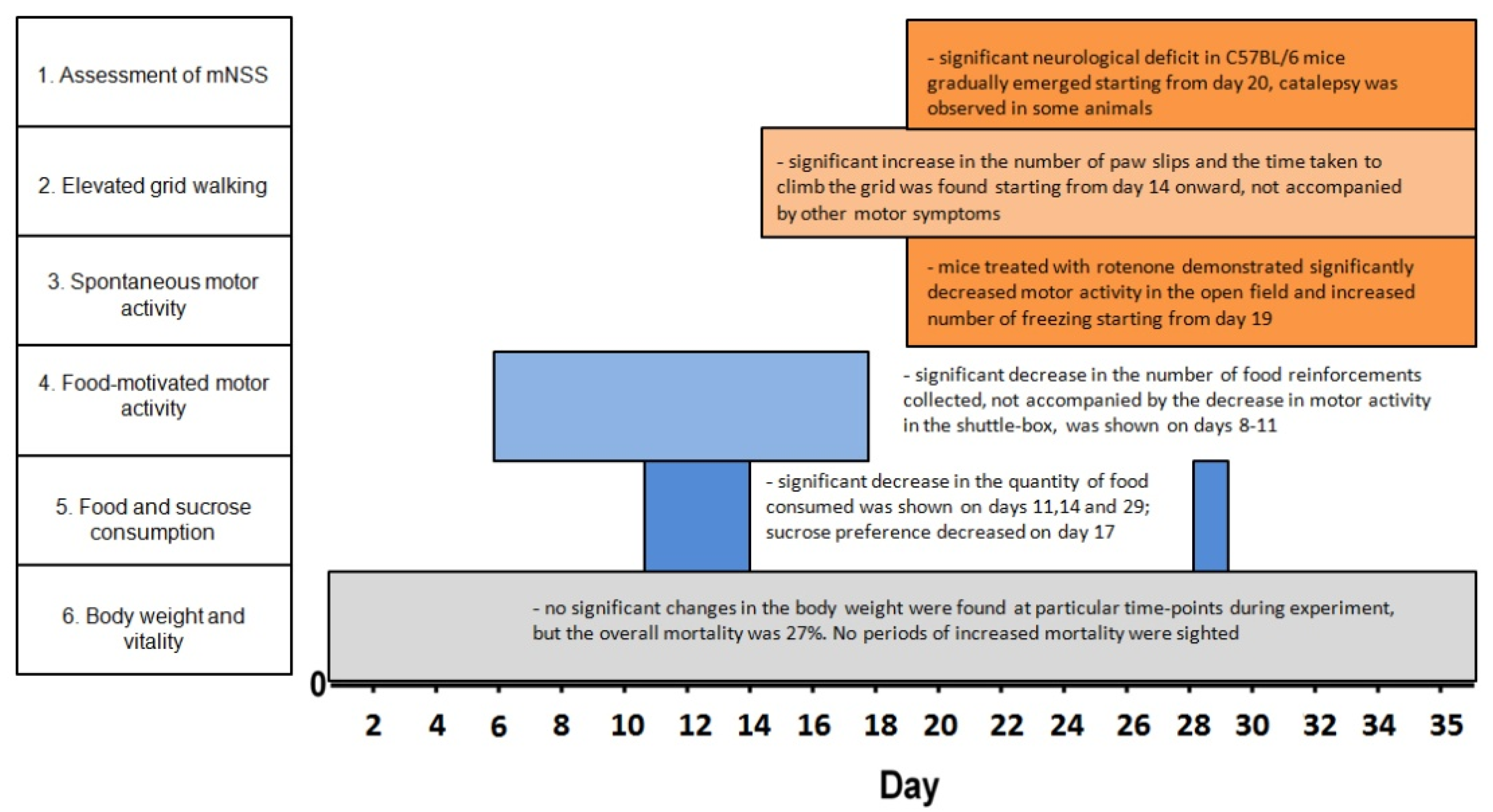
3.1.1. Survival, Vitality and Food Consumption
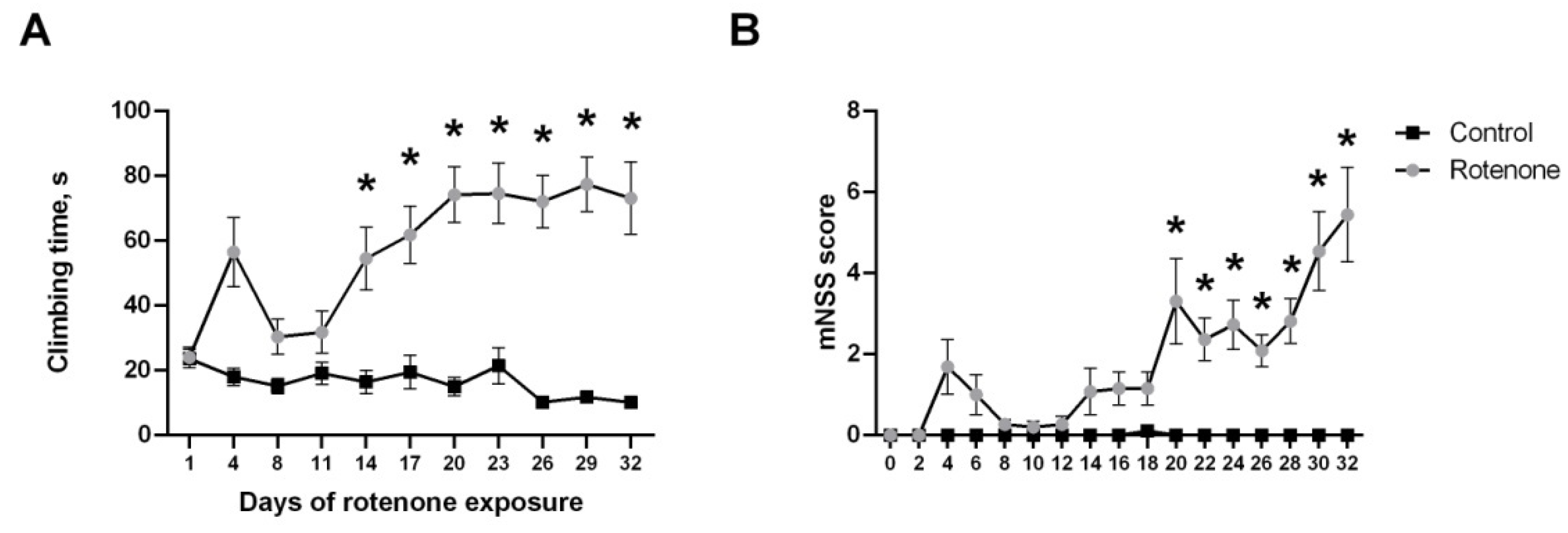
3.1.2. Motor Symptoms
3.1.3. Spontaneous and Food-Motivated Motor Activity
3.2. Neuropathological Effects of Rotenone
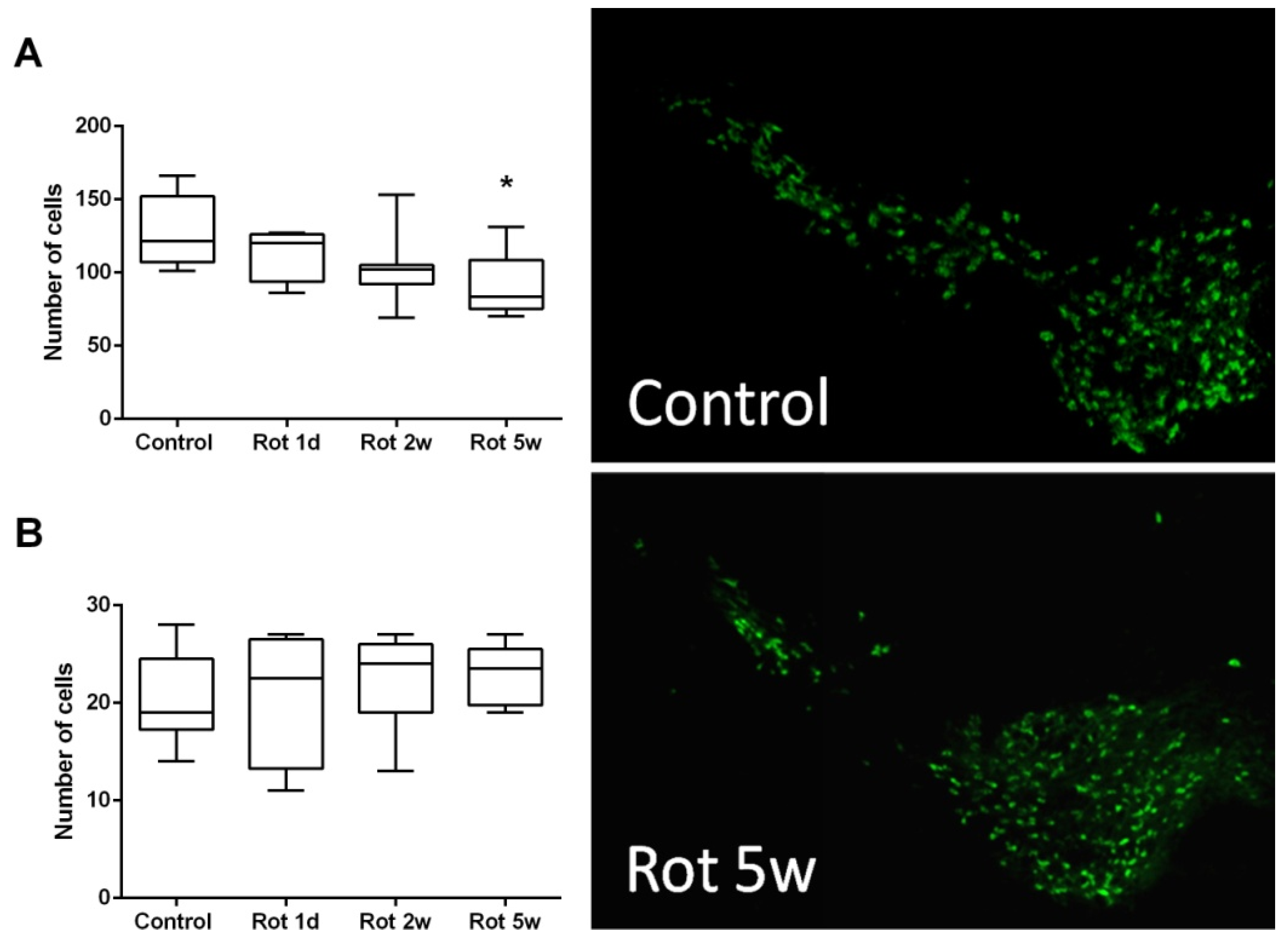
3.2.1. Time Course of Histological Changes in Rotenone-Treated Animals
3.2.2. Time Course of Biochemical Changes in Rotenone-Treated Animals
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PD | Parkinson’s disease |
| SNpc | substantia nigra pars compacta |
| SNpr | substantia nigra pars reticulata |
| VTA | ventral tegmental area |
| V1 | visual cortex |
| M1 | motor cortex |
| PFC | prefrontal cortex |
| DLStr | dorsolateral striatum |
| VMStr | ventromedial striatum |
| Amy | amygdala |
| Thal | Thalamus |
| NR | nucleus ruber |
| DHip | dorsal hippocampus |
| VHip | ventral hippocampus |
| MPTP | 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine |
| 6-OHDA | 6-hydroxydopamine |
| VTA | ventral tegmental area |
| DMSO | dimethyl sulfoxide |
| mNSS | modified neurological severity scores |
| PBS | phosphate-buffered saline |
| LPO | lipid peroxidation |
| CL | Chemiluminescence |
| DA | Dopamine |
| 5-HT | Serotonin |
| NA | Noradrenaline |
| DOPAC | 3,4-dihydroxyphenylacetic acid |
| 3-MT | 3-methoxytyramine |
| HVA | homovanillic acid |
| 5-HIAA | 5-hydroxyindoleacetic acid |
| HPLC-ED | high-performance liquid chromatography with electrochemical detection |
| TH | tyrosine hydroxylase |
| DAPI | 4′,6-diamidino-2-phenylindole |
| NADH | nicotinamide adenine dinucleotide |
References
- Braak, H.; Ghebremedhin, E.; Rub, U.; Bratzke, H.; Del Tredici, K. Stages in the development of Parkinson’s disease-related pathology. Cell Tissue Res. 2004, 318, 121–134. [Google Scholar] [CrossRef] [PubMed]
- Mahlknecht, P.; Seppi, K.; Poewe, W. The Concept of Prodromal Parkinson’s Disease. J. Parkinsons Dis. 2015, 5, 681–697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xia, R.; Mao, Z.H. Progression of motor symptoms in Parkinson’s disease. Neurosci. Bull. 2012, 28, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Vingill, S.; Connor-Robson, N.; Wade-Martins, R. Are rodent models of Parkinson’s disease behaving as they should? Behav. Brain Res. 2018, 15, 133–141. [Google Scholar] [CrossRef]
- Hornykiewicz, O.; Kish, S.J. Biochemical pathophysiology of Parkinson’s disease. Adv. Neurol. 1987, 45, 19–34. [Google Scholar]
- von Bohlen, O.; Schober, A.; Krieglstein, K. Genes, proteins, and neurotoxins involved in Parkinson’s disease. Prog. Neurobiol. 2004, 73, 151–177. [Google Scholar]
- Anderson, K.E. Behavioral disturbances in Parkinson’s disease. Dialogues Clin. Neurosci. 2004, 6, 323–332. [Google Scholar]
- Agid, Y. Parkinson’s disease: Pathophysiology. Lancet 1991, 337, 1321–1323. [Google Scholar] [CrossRef]
- Cacabelos, R. Parkinson’s Disease: From Pathogenesis to Pharmacogenomics. Int. J. Mol. Sci. 2017, 18, 551. [Google Scholar] [CrossRef]
- Jagmag, S.A.; Tripathi, N.; Shukla, S.D.; Maiti, S.; Khurana, S. Evaluation of models of Parkinson’s disease. Front. Neurosci. 2016, 9, 503. [Google Scholar] [CrossRef] [Green Version]
- Tieu, K. A guide to neurotoxic animal models of Parkinson’s disease. Cold Spring Harb. Perspect. Med. 2011, 1, a009316. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Zhao, T.T.; Lee, M.K. Animal models of Parkinson’s disease and their applications. J. Parkinsonism Restless Legs Syndr. 2016, 6, 73–82. [Google Scholar]
- Johnson, M.E.; Bobrovskaya, L. An update on the rotenone models of Parkinson’s disease: Their ability to reproduce the features of clinical disease and model gene—Environment interactions. NeuroToxicology 2005, 46, 101–116. [Google Scholar] [CrossRef] [PubMed]
- Betarbet, R.; Sherer, T.B.; MacKenzie, G.; Garcia-Osuna, M.; Panov, A.V.; Greenamyre, J.T. Chronic systemic pesticide exposure reproduces features of Parkinson’s disease. Nat. Neurosci. 2000, 3, 1301–1306. [Google Scholar] [CrossRef]
- Sherer, T.B.; Kim, J.H.; Betarbet, R.; Greenamyre, J.T. Subcutaneous rotenone exposure causes highly selective dopaminergic degeneration and alpha-synuclein aggregation. Exp. Neurol. 2003, 179, 9–16. [Google Scholar] [CrossRef]
- Inden, M.; Kitamura, Y.; Abe, M.; Tamaki, A.; Takata, K.; Taniguchi, T. Parkinsonian rotenone mouse model: Reevaluation of long-term administration of rotenone in C57BL/6 mice. Biol. Pharm. Bull. 2011, 34, 92–96. [Google Scholar] [CrossRef] [Green Version]
- Dhillon, A.S.; Tarbutton, G.L.; Levin, J.L.; Plotkin, G.M.; Lowry, L.K.; Nalbone, J.T.; Shepherd, S. Pesticide/environmental exposures and Parkinson’s disease in East Texas. J. Agromed. 2008, 13, 37–48. [Google Scholar] [CrossRef]
- Richter, F.; Hamann, M.; Richter, A. Chronic rotenone treatment induces behavioral effects but no pathological signs of parkinsonism in mice. J. Neurosci. Res. 2007, 85, 681–691. [Google Scholar] [CrossRef]
- Inden, M.; Kitamura, Y.; Takeuchi, H.; Yanagida, T.; Takata, K.; Kobayashi, Y.; Taniguchi, T.; Yoshimoto, K.; Kaneko, M.; Okuma, Y.; et al. Neurodegeneration of mouse nigrostriatal dopaminergic system induced by repeated oral administration of rotenone is prevented by 4-phenylbutyrate, a chemical chaperone. J. Neurochem. 2007, 101, 1491–1504. [Google Scholar] [CrossRef]
- Rojo, A.I.; Cavada, C.; De Sagarra, M.R.; Cuadrado, A. Chronic inhalation of rotenone or paraquat does not induce Parkinson’s disease symptoms in mice or rats. Exp. Neurol. 2007, 208, 120–126. [Google Scholar] [CrossRef]
- Miyazaki, I.; Isooka, N.; Imafuku, F.; Sun, J.; Kikuoka, R.; Furukawa, C.; Asanuma, M. Chronic Systemic Exposure to Low-Dose Rotenone Induced Central and Peripheral Neuropathology and Motor Deficits in Mice: Reproducible Animal Model of Parkinson’s Disease. Int. J. Mol. Sci. 2020, 21, 3254. [Google Scholar] [CrossRef] [PubMed]
- Pan-Montojo, F.; Anichtchik, O.; Dening, Y.; Knels, L.; Pursche, S.; Jung, R.; Jackson, S.; Gille, G.; Spillantini, M.G.; Reichmann, H.; et al. Progression of Parkinson’s disease pathology is reproduced by intragastric administration of rotenone in mice. PLoS ONE 2010, 5, e8762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berezhnoy, D.S.; Troshev, D.V.; Nalobin, D.S.; Fedorova, T.N. Changes in COX histochemistry in the brain of mice and rats exposed to chronic subcutaneous rotenone. J. Chem. Neuroanat. 2020, 110, 101880. [Google Scholar] [CrossRef] [PubMed]
- Berezhnoy, D.S.; Troshev, D.V.; Kulikova, O.I.; Abaimov, D.V.; Muzychuk, O.A.; Stvolinsky, S.L.; Fedorova, T.N. Comparison of neurobehavioral changes in mice treated with mitochondrial toxins, rotenone and MPTP. Hum. Physiol. 2021, 47, 821–830. [Google Scholar] [CrossRef]
- National Research Council. Guide for the Care and Use of Laboratory Animals; Russian Version; National Academy Press: Washington, DC, USA, 1996. [Google Scholar]
- Chen, J.; Zhang, C.; Jiang, H.; Li, Y.; Zhang, L.; Robin, A.; Chopp, M. Atorvastatin Induction of VEGF and BDNF Promotes Brain Plasticity after Stroke in Mice. J. Cereb. Blood Flow Metab. 2005, 25, 281–290. [Google Scholar] [CrossRef] [Green Version]
- Blume, S.R.; Cass, D.K.; Tseng, K.Y. Stepping test in mice: A reliable approach in determining forelimb akinesia in MPTP-induced Parkinsonism. Exp. Neurol. 2009, 219, 208–211. [Google Scholar] [CrossRef]
- Troshev, D.; Berezhnoy, D.; Kulikova, O.; Abaimov, D.; Muzychuk, O.; Nalobin, D.; Stvolinsky, S.; Fedorova, T. The dynamics of nigrostriatal system damage and neurobehavioral changes in the rotenone rat model of Parkinson’s disease. Brain Res. Bull. 2021, 173, 1–13. [Google Scholar] [CrossRef]
- Liu, M.Y.; Yin, C.Y.; Zhu, L.J.; Zhu, X.H.; Xu, C.; Luo, C.X.; Chen, H.; Zhu, D.Y.; Zhou, Q.G. Sucrose preference test for measurement of stress-induced anhedonia in mice. Nat. Protoc. 2018, 13, 1686–1698. [Google Scholar] [CrossRef]
- Franklin, K.; Paxinos, G. The Mouse Brain in Stereotaxic Coordinates; Academic Press: New York, NY, USA, 2008. [Google Scholar]
- Strazielle, C.; Jazi, R.; Verdier, Y.; Qian, S.; Lalonde, R. Regional brain metabolism with cytochrome c oxidase histochemistry in a PS1/A246E mouse model of autosomal dominant Alzheimer’s disease: Correlations with behavior and oxidative stress. Neurochem. Int. 2009, 55, 806–814. [Google Scholar] [CrossRef]
- Schmidt, W.J.; Alam, M. Controversies on new animal models of Parkinson’s disease pro and con: The rotenone model of Parkinson’s disease (PD). Parkinson’s Dis. Relat. Disord. 2006, 70, 273–276. [Google Scholar]
- Bhurtel, S.; Katila, N.; Srivastav, S.; Neupane, S.; Choi, D.Y. Mechanistic comparison between MPTP and rotenone neurotoxicity in mice. Neurotoxicology 2019, 71, 113–121. [Google Scholar] [CrossRef]
- Abhilash, P.; Bharti, U.; Haorei, Y.; Mariamma, P.; Rashmi, K.; Raju, T.R.; Kutty, B.M.; Sagar, B.K.C.; AlladiP, A. Ageing and MPTP-sensitivity depend on molecular and ultrastructural signatures of Astroglia and Microglia in mice nigra. bioRxiv, 2021; in press. [Google Scholar]
- Gallistel, C.R.; Fairhurst, S.; Balsam, P. The learning curve: Implications of a quantitative analysis. Proc. Natl. Acad. Sci. USA 2004, 101, 13124–13131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santiago, R.M.; Barbieiro, J.; Lima, M.M.; Dombrowski, P.A.; Andreatini, R.; Vital, M.A. Depressive-like behaviors alterations induced by intranigral MPTP, 6-OHDA, LPS and rotenone models of Parkinson’s disease are predominantly associated with serotonin and dopamine. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2010, 34, 1104–1114. [Google Scholar] [CrossRef] [PubMed]
- Fields, H.L.; Hjelmstad, G.O.; Margolis, E.B.; Nicola, S.M. Ventral tegmental area neurons in learned appetitive behavior and positive reinforcement. Annu. Rev.Neurosci. 2007, 30, 289–316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McHugh, P.C.; Buckley, D.A. The Structure and Function of the Dopamine Transporter and Its Role in CNS Diseases. Vitam. Horm. 2015, 98, 339–369. [Google Scholar]
- Dreyer, J.K.; Herrik, K.F.; Berg, R.W.; Hounsgaard, J.D. Influence of Phasic and Tonic Dopamine Release on Receptor Activation. J. Neurosci. 2010, 30, 14273–14283. [Google Scholar] [CrossRef]
- Tillerson, J.L.; Caudle, W.M.; Reverón, M.E.; Miller, G.W. Detection of behavioral impairments correlated to neurochemical deficits in mice treated with moderate doses of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. Exp. Neurol. 2002, 178, 80–90. [Google Scholar] [CrossRef]
- Van den Berg, C.; Beek, P.J.; Wagenaar, R.C.; van Wieringen, P.C. Coordination disorders in patients with Parkinson’s disease: A study of paced rhythmic forearm movements. Exp. Brain Res. 2000, 134, 174–186. [Google Scholar] [CrossRef]
- Stoyka, L.E.; Arrant, A.E.; Thrasher, D.R.; Russell, D.L.; Freire, J.; Mahoney, C.L.; Narayanan, A.; Dib, A.G.; Standaert, D.G.; Volpicelli-Daley, L.A. Behavioral defects associated with amygdala and cortical dysfunction in mice with seeded α-synuclein inclusions. Neurobiol. Dis. 2020, 134, 104708. [Google Scholar] [CrossRef]
- Giordano, S.; Lee, J.; Darley-Usmar, V.M.; Zhang, J. Distinct effects of rotenone, 1- methyl-4-phenylpyridinium and 6-hydroxydopamine on cellular bioenergetics and cell death. PLoS ONE 2012, 7, e44610. [Google Scholar] [CrossRef] [PubMed]
- Fowler, A.K.; Thompson, J.; Chen, L.; Dagda, M.; Dertien, J.; Dossou, K.S.; Moaddel, R.; Bergeson, S.E.; Kruman, I.I. Differential sensitivity of prefrontal cortex and hippocampus to alcohol-induced toxicity. PLoS ONE 2014, 9, e106945. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Test | Days |
|---|---|
| Neurological scale evaluation | 0, 2, 4, 6, 8, 10, 12, 14, 16, 18, 20, 22, 24, 26, 28, 30, 32 |
| Catalepsy test | 0, 1, 4, 8, 11, 14, 17, 20, 23, 26, 29, 32 |
| Elevated grid walking | 0, 1, 4, 8, 11, 14, 17, 20, 23, 26, 29, 32 |
| Open field | 1, 5, 12, 19, 26, 33 |
| Operant learning | 0, 2, 8, 11, 14, 17 |
| Sucrose, food consumption testing | 0, 2, 5, 8, 11, 14, 17 |
| Structures | Control | Rot 1 d | Rot 2 w | Rot 5 w | |
|---|---|---|---|---|---|
| Striatum | NA | 1.16± 0.05 | 1.25 ± 0.06 | 1.2 ± 0.06 | 1.02 ± 0.04 |
| DA | 70.6 ± 1.18 | 65.6 ± 0.74 | 68.9 ± 0.78 | 73.4 ± 2.01 | |
| HVA | 5.24 ± 0.11 | 7.21 ± 0.21 | 6.37 ± 0.3 | 10.42 ± 0.76 | |
| DOPAC | 3.01 ± 0.04 | 3.46 ± 0.15 | 4.5 ± 0.32 | 7.9 ± 0.78 | |
| 3-MT | 3.6 ± 0.11 | 3.65 ± 0.15 | 2.7 ± 0.16 | 2 ± 0.125 ** | |
| 5-HT | 4.25 ± 0.05 | 4.66 ± 0.16 | 4.66 ± 0.09 | 4.25 ± 0.09 | |
| 5-HIAA | 3.24 ± 0.06 | 4.12 ± 0.09 | 5.83 ± 0.37 | 5.85 ± 0.33 | |
| Cortex | NA | 4.39 ± 0.07 | 3.61 ± 0.14 | 4.55 ± 0.08 | 5.26 ± 0.15 |
| DA | 4.1 ± 0.81 | 4.86 ± 1.14 | 1.12 ± 0.17 | 2.75 ± 0.44 | |
| HVA | 1.66 ± 0.06 | 1.84 ± 0.09 | 2.11 ± 0.3 | 3.546 ± 0.45 | |
| 3-MT | - | 0.81 ± 0.34 | 0.17 ± 0.03 | - | |
| 5-HT | 6.65 ± 0.16 | 6 ± 0.22 | 8.4 ± 0.21 | 6.53 ± 0.43 | |
| 5-HIAA | 3.65 ± 0.16 | 4.96 ± 0.45 | 5.22 ± 0.41 | 4.69 ± 0.25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Troshev, D.; Voronkov, D.; Pavlova, A.; Abaimov, D.; Latanov, A.; Fedorova, T.; Berezhnoy, D. Time Course of Neurobehavioral Disruptions and Regional Brain Metabolism Changes in the Rotenone Mice Model of Parkinson’s Disease. Biomedicines 2022, 10, 466. https://doi.org/10.3390/biomedicines10020466
Troshev D, Voronkov D, Pavlova A, Abaimov D, Latanov A, Fedorova T, Berezhnoy D. Time Course of Neurobehavioral Disruptions and Regional Brain Metabolism Changes in the Rotenone Mice Model of Parkinson’s Disease. Biomedicines. 2022; 10(2):466. https://doi.org/10.3390/biomedicines10020466
Chicago/Turabian StyleTroshev, Dmitry, Dmitry Voronkov, Anastasia Pavlova, Denis Abaimov, Alexander Latanov, Tatiana Fedorova, and Daniil Berezhnoy. 2022. "Time Course of Neurobehavioral Disruptions and Regional Brain Metabolism Changes in the Rotenone Mice Model of Parkinson’s Disease" Biomedicines 10, no. 2: 466. https://doi.org/10.3390/biomedicines10020466
APA StyleTroshev, D., Voronkov, D., Pavlova, A., Abaimov, D., Latanov, A., Fedorova, T., & Berezhnoy, D. (2022). Time Course of Neurobehavioral Disruptions and Regional Brain Metabolism Changes in the Rotenone Mice Model of Parkinson’s Disease. Biomedicines, 10(2), 466. https://doi.org/10.3390/biomedicines10020466






