Abstract
Kidney disease is a multifactorial problem, with a growing prevalence and an increasing global burden. With the latest worldwide data suggesting that chronic kidney disease (CKD) is the 12th leading cause of death, it is no surprise that CKD remains a public health problem that requires urgent attention. Multiple factors contribute to kidney disease, each with its own pathophysiology and pathogenesis. Furthermore, microRNAs (miRNAs) have been linked to several types of kidney diseases. As dysregulation of miRNAs is often seen in some diseases, there is potential in the exploitation of this for therapeutic applications. In addition, uptake of interference RNA has been shown to be rapid in kidneys making them a good candidate for RNA therapy. The latest advancements in RNA therapy and lipid-based nanocarriers have enhanced the effectiveness and efficiency of RNA-related drugs, thereby making RNA therapy a viable treatment option for renal disease. This is especially useful for renal diseases, for which a suitable treatment is not yet available. Moreover, the high adaptability of RNA therapy combined with the low risk of lipid-based nanocarriers make for an attractive treatment choice. Currently, there are only a small number of RNA-based drugs related to renal parenchymal disease, most of which are in different stages of clinical trials. We propose the use of miRNAs or short interfering RNAs coupled with a lipid-based nanocarrier as a delivery vehicle for managing renal disease.
1. Introduction
The global prevalence of chronic kidney disease (CKD) is estimated at approximately 13.4%, while estimates suggest that acute kidney injury (AKI) affects approximately 13.3 million people globally every year [1,2]. According to the Global Burden of Disease study in 2017, 1.2 million deaths were attributed to CKD, and it was the 12th leading cause of death worldwide [3]. CKD is defined as the presence of renal damage (usually detected as a urinary albumin excretion of >30 mg/day) or decreased renal function (characterised by an estimated glomerular filtration rate (eGFR) of <60 mL/min/1.73 m2) for over three months, irrespective of the cause [4]. The persistence of renal damage or decreased function for at least three months is necessary to distinguish CKD from AKI. Conversely, the occurrence of AKI is abrupt and the decline in GFR is usually reversible [5]. Taking into consideration the aforementioned concerns, the employment of RNA therapy is being looked at for the management of renal diseases as its use in other diseases such as cancer and in vaccinations has gained traction. Some of the major advantages of RNA therapy over other therapies include its capacity to target an otherwise non-targetable small molecule and its ability to rapidly interact with messenger RNA (mRNA) constructs [6].
Over the years, studies on the diverse functions and mechanisms of action of RNA molecules have enabled its use in therapy. From the discovery of mRNA in 1961 [7] to the approval and use of RNA-based drugs, the research on RNA has indeed come far. Along the same timeline, but in a different field, is the progression of nanotechnology. Following the initial introduction of the concept of nanotechnology by American physicist and Nobel Prize laureate Richard Feynman in 1959, the use of nanotechnology is now seen in almost every field of science, including the field of medicine [8]. One of the current uses of nanotechnology in medicine is its application in drug delivery.
Both the RNA-based drug development and efficient drug delivery methods have advanced substantially and simultaneously to allow for the clinical use of RNA therapy. Most recently and notably in this regard, are two authorised vaccines used against the coronavirus disease 2019 (COVID-19) [9]. Both the BNT162b2 and mRNA-1273 vaccines employ lipid nanocarriers to facilitate the delivery of antigenic mRNA [10,11]. In this review, we aim to examine the role of RNA in renal pathophysiology and its putative role in the pathogenesis of renal parenchymal diseases. We also discuss various strategies for using short interfering RNA (siRNA) and microRNA (miRNA) as therapies and the process of drug delivery. The advantages of using lipid-based nanocarriers as efficient delivery systems of RNA molecules are elaborated. The review will focus mainly on the clinical applications of RNA therapy for renal diseases.
2. RNA Therapy
It is widely accepted that RNAs are important molecules involved in nearly all biological pathways [12]. While the diverse roles of RNA were being discovered, it was also noticed that the use of RNA as a form of therapy had substantial potential. The current principle of RNA therapy involves the use of both coding and non-coding RNAs. There are five known types of therapeutic RNAs that are classified by their mode of action: (1) RNAs that inhibit RNA activity (miRNAs, siRNAs, antisense RNAs), (2) RNAs that target proteins (RNA aptamers), (3) RNAs that reprogram genetic information (trans-splicing ribozyme), (4) RNAs that encode therapeutic proteins (mRNAs) and (5) DNA-modifying CRISPR guide RNAs (gRNAs) [13]. The initial use of RNA as a form of therapy employed chemically synthesised antisense oligonucleotides [12]. In addition, the discovery and research of interference RNA (RNAi) has led to vast possibilities in the field of RNA therapeutics. RNAi uses siRNAs or miRNAs. Both antisense RNA and RNAi work by suppressing the expression of a target RNA [12].
Of particular interest here are the RNAs that relate to protein downregulation, which are miRNA and siRNA. As miRNAs and siRNAs have emerged as a class of therapeutics with great potential, especially for the treatment of cancer, there has been some interest in using them as a therapy for renal diseases [14,15]. Both miRNAs and siRNAs are powerful tools that can silence post-transcriptional gene expression. For example, a specifically designed miRNA/siRNA targets an mRNA via sequence-specific binding, resulting in degradation of mRNA translation [14]. Current application in cancer therapy uses synthetic siRNA to target oncogenes and genes involved in cancer cell proliferation, survival, invasion, angiogenesis, metastasis and drug resistance [14]. A look into how these applications of siRNAs translate to similar renal therapies would prove highly useful. Studies in animal models have shown that siRNA uptake is rapid and effective in kidneys without affecting basic renal function parameters [16]. Furthermore, miRNAs have been discovered to be implicated in many diseases, including renal disease, as these components are often dysregulated in the disease process [17]. Thus, there is potential in exploiting the involvement of miRNAs in the pathogenesis of disease towards discovering new treatment options. Moreover, as miRNAs are highly effective tools for gene silencing, the possibility of developing gene-specific RNA drugs is vast, including for genes with no currently known pharmacological antagonists or inhibitors [15]. It is, therefore, not surprising that the application of miRNAs and siRNAs would be worth tapping into and should be extensively researched and applied to renal therapy.
3. Rationale of RNA-Based Strategy in Renal Diseases
3.1. Renal Fibrosis
In various progressive renal diseases, TGFβ signalling plays an essential role. In human studies, TGFβ1 expression is majorly upregulated in fibrotic tissues [18]. Accumulating evidence has demonstrated that miRNAs regulate the TGFβ pathways [19,20]. Conversely, TGFβ also affects miRNA expression [21,22,23]. Micro-150 promoted renal fibrosis in a renal ischemia/reperfusion model [24]. Similarly, increased miR-668 [25] and miR-489 [26] expression was observed in acute kidney injury (AKI) in humans, mice, and renal tubular cells by hypoxia-inducible factor-1 (HIF-1) signalling. In addition, abundant extracellular matrix protein deposition is a key feature of fibrosis. The expression of collagen types I and III was affected by mmu-miR-29-5p [27]. TGFβ reduces miR-29-5p expression, leading to the feedforward upregulation of TGFβ expression [28]. In addition, it has been documented that miR-21 promotes epithelial-to-mesenchymal transition (EMT) induced by TGFβ1 by inhibiting the target Smad7 [29].
3.2. Diabetic Nephropathy
Podocyte apoptosis has been documented to be associated with diabetic nephropathy, and miRNAs have been implicated in the pathogenesis of diabetic nephropathy. The expression of mmu-miR-29c increased in glomeruli and microvascular endothelial cells in a mouse model [30]. Studies of renal tissues of patients with diabetes revealed that miR-192 expression was inversely related to renal fibrosis and was associated with eGFR [31,32]. Other studies have revealed that TGFβ is involved in miR-192 regulation in proximal convoluted tubule cells exposed to high-glucose conditions [31]. TGFβ upregulated the expression of miR-192 and consequently inhibited the expression of Zeb1/2 which sequentially increased the expression of miR200 [33]. Hyperglycaemia induced cell hypertrophy and increased matrix protein levels in mouse diabetic models, and miR-21 manipulated this process by reducing tumour suppressor protein phosphatase and tensin homologue deleted on chromosome 10 (PTEN) expression [34]. Overall, the impact of miRNAs on these functions is important.
Downstream effects of TGF-β are mediated by connective tissue growth factor (CTGF), whose expression is upregulated in several renal cells and secreted in the urine in the diabetic state [35]. In addition, the interaction of advanced glycation end products (AGEs) with their receptors (RAGE) generates oxidative stress and stimulates fibrotic reactions in diabetic kidneys [36,37]. Furthermore, PTEN is a tumour suppressor protein that is recognised as a negative factor of the phosphoinositide-3-kinase (PI3K) Akt signalling pathway [38]. Evidence has revealed that miR-214 regulates PTEN expression via Akt activation, leading to high glucose-induced mesangial and proximal tubular cell hypertrophy and fibronectin deposition [39].
3.3. Glomerulonephritis
Glomerulonephritis includes a group of diverse disorders that may present as proteinuria or microscopic haematuria with or without renal dysfunctions. Accumulating evidence has revealed that RNA-related pathophysiology and potential molecules or pathways may be therapeutically useful for treating glomerulonephritis.
IgA nephropathy is caused by abnormal O-galactosylation of IgA, resulting in the deposition of IgA complexes in the kidneys and activation of the complement system. Various miRNAs play roles in IgA nephropathy as observed in a genome-wide analysis [40]. Members of the miR-200 and miR-29 families, which regulate EMT and progressive renal fibrosis, caused prominent expression changes in IgA nephropathy and were associated with renal fibrosis and proteinuria. Patients with IgA nephropathy presented increased expression of miR-146a and miR-155 compared to controls. These two factors were inversely associated with eGFR, and proteinuria and the latter were related to renal fibrosis [41]. miR-155 has been reported to affect T cell development and reduce regulatory T cell number and T cell dysfunction, which is associated with the pathogenesis of IgA [42,43,44]. Another study reported that the inhibition of miR-21 expression potentially reduced fibrogenic processes in podocytes and tubular cells by preventing PTEN/Akt pathway activation [45].
Systemic lupus erythematosus (SLE) is a systemic disease caused by abnormal immune system activities. Kidney biopsy analysis of patients with lupus nephritis revealed upregulated miR-146 and miR-198 expression in glomerular lesions and increased miR-638 expression in tubulointerstitial lesions [46]. Interstitial miR-638 expression was associated with clinical markers of proteinuria, while miR-146 expression was associated with eGFR. Thus, these two factors are pathogenic factors for the development of clinical lupus nephritis. In addition, miR26a and miR-30b were reported to be involved in cell cycle regulation and their reduced expression was observed in renal tissues as well as in a human mesangial cell model [47]. In lupus nephritis, increased expression of human epithelial growth factor receptor (HER-2) and the degree of HER-2 amplification was associated with lupus activity and the severity of proteinuria. It was revealed that INFꭤ enhanced HER-2 expression, which led to miR-26a and miR-30b upregulation and proliferative lupus nephritis.
Focal segmental glomerulosclerosis (FSGS) is characterised by the obliteration of the capillary lumen and increase in mesangial matrix amount in the involved glomeruli. Podocyte injury is a factor affecting the development of FSGS, and molecules associated with increased permeability of glomerular basement membranes are potentially related to the pathogenesis of FSGS [48,49]. MicroRNA-30 has been postulated to maintain cytoskeletal integrity and podocyte health [50]. The growth of podocyte cells is reported to be regulated by the expression of miR-132 and miR-134 [51]. In addition, serum and urine miRNAs have been used as biomarkers to evaluate FSGS disease activity. Lower concentrations of miR-125 and miR-186 have been detected in patients with disease remissions [52]. FSGS disease activity is reported to be associated with miR-196 and miR-30 expression [53]. The latter was a potential predictor of steroid responsiveness in patients with active FSGS.
3.4. Hypertension
Hypertension is a significant risk factor for coronary artery disease, congestive heart failure, sudden death, left ventricular hypertrophy and stroke [54,55,56]. Hypertension is the second most common cause of end-stage kidney disease in the US and in a study, the prevalence of hypertension in CKD group was higher than that in control group [57]. It is a complex disease with different factors involved in its pathogenesis, including genetic, environmental, haemodynamic, and renal factors. MiRNAs are involved in pathophysiological alterations related to the progression of hypertension and cardiovascular and renal complications.
Activation of the renin-angiotensin-aldosterone system (RAS) plays an essential role in the pathophysiology of hypertension. The expression of many miRNAs, including miR-29b, miR-129-3p, miR-132, and miR-212, are upregulated by Ang II in HEK293N cells [58]. In addition, Ang II type 1 receptor (AT1R)-regulated miR-483 serves as a potential negative regulator of RAS components in vascular smooth muscle cells (VSMCs) [59].
Oxidative stress potentially induces endothelial cell apoptosis and atherosclerosis development. Overexpression of miR-210 inhibits apoptosis and reduces ROS levels [60]. Inhibition of miR-1555 increases endothelial nitric oxide synthase (eNOS) expression and NO production, restoring endothelium-dependent vasorelaxation in human internal mammary arteries [61].
In addition, sympathetic nervous system overactivity has a significant role in hypertension development and persistence. In genetically hypertensive mice (BPH/2J), the roles of the sympathetic nervous system (SNS) and RAS are important. Hypertension in BPH/2J is mediated by SNS during the active period and RAS during the inactive period [62]. During the active periods, the hypertensive mice had higher Ren1 (renin) mRNA and lower miR-181 expression. Similarly, in a human study, miRNA-181 expression inversely regulated Ren1 mRNA expression in the renal tissue of patients with hypertension [63].
3.5. Autosomal Dominant Polycystic Kidney Disease (ADPKD)
ADPKD is a genetic disorder caused by mutations in either PKD or PKD2 genes, resulting in impaired ciliary functions, leading to renal and hepatic cyst formations. In a rat model of ADPKD, the expression of miR-21 was found to be upregulated [64]. Inhibition of miR-21 expression reduces cyst growth in a mouse model of ADPKD [65]. In addition, the expression of genes belonging to the miR-17 cluster is upregulated in mouse models and deletion of miR-17 leads to the resolution of cysts and better renal and animal survival [66].
3.6. Alport Syndrome
Mutation in genes coding ꭤ3, ꭤ4 or ꭤ5 chains of collagen Type IV leads to abnormal basement membrane formations in the kidneys, eyes, and inner ears. miR-21 is typically expressed in the tubulointerstitial compartment instead of the glomerular compartments in normal mice, while it is expressed equally in both compartments in Col4 ꭤ3-/-mice [67].
4. Nanocarriers for Systemic Delivery
The stability of RNA, delivery efficiency, and activation of the innate immune system are the various challenges for the widespread use of RNA therapy. The limitations of RNA-based therapies are attributed mainly to their short half-life and rapid renal clearance [14]. However, recent advancements in nanotechnology have opened more avenues for improving RNA therapies. Nanocarriers have been shown to overcome many challenges associated with the use of systemic RNAs.
Nanotechnology is an interdisciplinary field that can be defined in various ways. Yet, a reasonable general consensus exists, where nanotechnology is concerned with materials and processes taking place in the nanoscale [68]. According to the National Nanotechnology Initiative (NNI) in the United States, nanotechnology is defined as a science, engineering or technology conducted at the nanoscale level which is typically cited between 1 to 100 nanometre (nm) [8]. Nanotechnology has broad applications in medicine, such as drug delivery, gene therapy, drug synthesis, imaging, and diagnostics of diseases [69]. In the field of nanotechnology, one of the materials produced is known as a nanoparticle. Nanoparticles are a wide class of particulate substance ranging between 1 and 100 nm in size [70]. Depending on their size and surface properties, nanoparticles exhibit different functionalities [71]. Nanoparticles are divided into various broad categories based on their morphology, size, and chemical properties. Some of the well-known classes of nanoparticles include carbon-based nanoparticles, metal nanoparticles, ceramic nanoparticles, semiconductor nanoparticles, polymeric nanoparticles and lipid-based nanoparticles [72].
Nanocarriers use agents like nanoparticles as a transport carrier. A carrier system is defined as technology that optimises the therapeutic efficiency of a bioactive compound [73]. This is particularly useful especially in the drug delivery system. Of the various nanocarriers available out there, the one that is of greatest interest is the use of lipid-based nanocarriers as a means of drug delivery. Lipid-based nanocarriers or nanoparticles (LNPs) have various favourable properties, making them an ideal choice. LNPs are characteristically spherical and possess a lipid solid core with a matrix containing soluble lipophilic molecules. As cell membranes are mainly composed of lipids and phospholipids, lipid-based nanocarriers as a drug transport have the natural advantage of better interaction with cells, and therefore facilitating better cellular uptake of transported drug [74]. Furthermore, lipid nanoparticle materials are biocompatible and biodegradable [75]. Several notable classes of lipid-based nanocarriers have been studied for RNA delivery, including liposomes, solid lipid-based nanoparticles, nanostructured lipid carriers, lipidoid nanoparticles, lipophilic conjugates, transfersomes, lipospheres and nanoemulsions. [14,76,77]. It is important to note that there are many other lipid-based carrier systems out there; however, Table 1 summarises some of them and their advantages.

Table 1.
Summary of the types of lipid-based nanocarriers.
LNPs no doubt aid in the efficient delivery of RNA drugs to their target. Nevertheless, there are still considerations and precautions to be taken. Lipotoxicity has been one of the concerns with using LNPs. Newer generations of LNPs have managed to overcome this issue [82]. Another major consideration is the targeting selectivity of LNPs. Unmodified LNPs have limitations in terms of being able to efficiently reach their intended target to deliver their payload [83]. As LNPs naturally accumulate in the liver, effective targeting strategies have to be employed to ensure successful uptake in other cell types [84]. Addition of specific ligands to the surface of LNPs have so far aided in their selective targeting followed by uptake by intended cells [85]. In the process of formulation design, production, and characterisation of LNPs, some parameters to consider include type of LNP, size, thermal properties, encapsulation efficiency, loading capacity and long-term storage stability [86]. With regards to kidneys, certain properties of LNPs have a huge bearing on targeting, such as size and charge [87]. For example, smaller LNPs penetrated the kidneys more readily; however, they were associated with higher clearance and lower retention [88]. As the glomerular filtration barrier is charge selective, charges of LNPs can influence their interaction with kidneys. Modifications to LNPs have to be carefully formulated as some additions could have both advantages and disadvantages. For example, PEGylation was used to stabilise LNPs and reduce rapid clearance, yet the presence of PEG on the surface of LNPs activated the immune system via induction of anti-PEG IgM production [89]. In short, for a successful delivery of a drug to occur, the LNP has to protect the RNA from degradation, traffic the drug to its intended target and then facilitate the uptake of said drug by target cells to achieve its desired effect.
5. Clinical Therapeutic Application of RNA in Renal Diseases
In the previous sections, we discussed the role of RNA and its types in renal diseases, as well as potential RNA dysregulation. MiRNA-based therapies either inhibit deleterious miRNA or replace a deficient one with a beneficial miRNA. Antagomirs or antimiRs and miRNA antagonists, are single-stranded molecules that can bind to mature miRNAs and block their action. Nanoparticle-based viral vectors express the desired miRNA or siRNAs, which are delivered to the target site [90]. In addition, designing an effective RNA-related therapeutic agent is required to avoid degradation and unintended effects, as well as to deliver its payload efficiently to the intended target tissues.
Some phase I and phase II trials are ongoing or have been completed to evaluate the efficacy of RNA-based therapeutic agents for the management and treatment of chronic hepatitis C, diabetes mellitus type 2 with fatty liver and cancers [90,91,92]. Table 2 summarises key studies investigating RNA therapy in renal diseases. In addition, some are used as therapeutic agents for renal diseases. Some studies have revealed the feasible use of siRNA-based therapy to reduce glomerular sclerosis in a mouse model of glomerulonephritis by manipulating TGFβ signalling, resulting in Mapk1 silencing [93]. Furthermore, siRNA against Smad4 prevented renal fibrosis in an in vivo study [94]. Other studies have revealed the efficacy of siRNA-based therapies targeting p53 and meprin-1β, which are essential for apoptosis and depolarisation, in cisplatin-related AKI models [95]. In unilateral ureteral obstruction models, siRNA therapy against Cd40 significantly reduced inflammation and enhanced repair [96]. siRNA-based therapies in the kidney serve as fantastic platforms with promising results, especially for the prevention of AKI, and various clinical trials are ongoing [15].

Table 2.
Summary of clinical trials of RNA therapy in kidney disease.
In contrast to siRNAs, miRNAs typically bind with partial complementarity and usually promote translational repression by triggering mRNA decay, and different miRNAs can interact with the same transcript by overlapping or non-overlapping sites [103]. Importantly, miRNAs play a significant role in the negative regulation of post-transcriptional gene expression. We have previously discussed various miRNAs that have been reported in AKI, renal fibrosis, ADPKD and progression to CKD [38,50,51,52,53,104]. In animal models, evidence has revealed that miRNA dysregulation is a therapeutic target for renal diseases. Chau et al. [105] reported that the inhibition of miR-21 with complementary oligonucleotides reduced renal fibrosis after injury. In a diabetic nephropathy model, an anti-miR-192-based translational approach reduced renal fibrosis and improved proteinuria and is a potential therapy [32]. Several studies have shown that the clinical utility of miRNA mimetics and inhibitors is a promising future intervention for the pathogenesis of renal disease. However, translation of preclinical findings is complicated, and enhancing efficient targeting to the intended tissues is difficult. In addition, various miRNAs are regulated in a cell-type- or organ-specific manner, so off-target and undesired influences in unrelated tissues are high. Some clinical trials are ongoing to investigate miRNA modulators in renal diseases.
Currently, only a few RNA therapies are associated with renal diseases. At the time of writing this article, they are at different stages of clinical trials and have not been approved for clinical use. These include QPI-1002 and NOX-E36 (Emamticap pegol). QPI-1002 uses an siRNA to target the p53 gene and is currently in phase I and II trials for acute renal failure and AKI, respectively [13]. NOX-E36 is an RNA aptamer targeting the C-C motif ligand 2, currently in phase II trials for type 2 diabetes mellitus and albuminuria [6,13]. Another drug is one developed for Alport nephropathy, a genetic disorder characterised by chronic glomerulonephritis that progresses to end-stage renal disease in young adults. An antagomir against miR21 was effectively evaluated in a mouse model, where the animals displayed substantially milder disease and significantly improved survival after treatment [67]. The drug RG012 targeting miR21 is currently undergoing a phase II clinical trial. Another drug is RGLS4326, an antagomir-inhibiting miR17, developed for the treatment of ADPKD, in which hyperproliferation of the renal tubular cells and cyst formation is observed [66]. Treatment with RGLS4326 attenuated cyst growth in several PKD mouse models and human ADPKD models in vitro and is now in a phase I clinical trial. Although also related to other cancers, APN401 is a drug trialled for renal cancer [106]. Patients were treated with siRNA-transfected peripheral blood mononuclear cells to aid in killing cancer cells. The treatment was targeted at patients with metastatic tumours or tumours that could not be surgically removed. This drug has completed phase I clinical trials [102].
6. Conclusions and Future Prospects
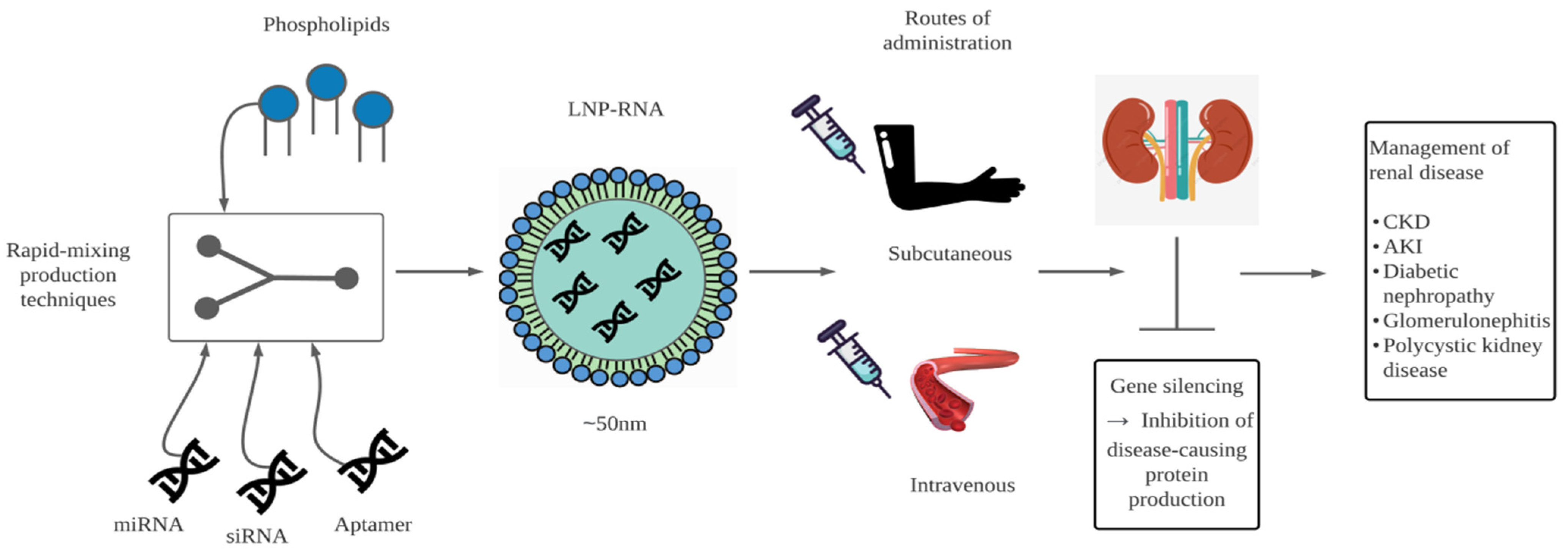
It is evident in our time that advancements in medicine and technology go hand in hand. As previously mentioned, the world of medicine and therapeutics have benefitted from both advancements in RNA research and the development of nanocarriers. With the constant evolution and mutation of diseases, treatment methods must likewise keep up with disease progression. Therefore, new approaches for managing current diseases are vital. In terms of nephrology, RNA therapy appears to be a novel approach, opening various avenues for the treatment of renal disease (Figure 1).

Figure 1.
Diagrammatic representation of the packaging and delivery of RNA for the management of renal disease. LNP-RNA, lipid nanocarrier-RNA; CKD, chronic kidney disease; AKI, acute kidney injury.
In this review, we discussed the difficulty in delivering RNA molecules to the intended target. Naked RNAs are unstable in the blood and are rapidly degraded by circulating monocytes and macrophages in the kidneys and liver. Thus, effective delivery methods to prevent miRNA degradation must be developed. The designed delivery vehicles must be non-toxic, have low immunogenicity, and deliver efficiently to the intended targets [107,108]. With this in mind, we propose using LNPs to achieve this goal. With the right formulation in the process of designing LNPs, it is possible to overcome the challenges of using RNA as a viable treatment. As with all treatments, careful attention has to be paid with regards to potential adverse reactions that may occur in RNA therapy. For example, each cellular miRNA is able to regulate the expression of several hundred genes; hence, minor changes anywhere along the miRNA pathway may result in major consequences [109]. In short, off-target effects should ideally be avoided. With the right modifications and appropriate delivery systems, this can be overcome.
Our recommendation is that more emphasis and attention be put on the research of combining RNA with LNPs when it comes to renal disease. There are many types of renal disease out there that could benefit from this research. The use of lipid nanocarrier-based RNA delivery holds a promising future for the treatment of renal disease. We expect to see broad applications of this therapeutics across the board in renal disease in the coming years.
Author Contributions
Writing—draft preparation, C.-T.S. and D.H.W.S.; Writing—review and editing, C.-T.S.; Supervision, J.-W.H. All authors have read and agreed to the published version of the manuscript.
Funding
The work was funded by the National Taiwan University Hospital Yunlin Branch, Taiwan (NTUHYL 111.S022), the National Taiwan University Cancer Centre (NTUCCS-111–02), and the Ministry of Science and Technology, Taiwan (MOST 109–2311-B-002–006- and MOST 108-2314-B-002-062-MY3).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lv, J.-C.; Zhang, L.-X. Prevalence and Disease Burden of Chronic Kidney Disease. Adv. Exp. Med. Biol. 2019, 1165, 3–15. [Google Scholar] [PubMed]
- Bjornstad, E.C.; Muronya, W.; Smith, Z.H.; Gibson, K.; Mottl, A.K.; Charles, A.; Marshall, S.W.; Golightly, Y.M.; Munthali, C.K.; Gower, E.W. Incidence and epidemiology of acute kidney injury in a pediatric Malawian trauma cohort: A prospective observational study. BMC Nephrol. 2020, 21, 98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bikbov, B.; Purcell, C.A.; Levey, A.S.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.M.; Afarideh, M.; Agarwal, S.K.; Agudelo-Botero, M.; et al. Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef] [Green Version]
- Levey, A.S.; Levin, A.; Kellum, J.A. Definition and classification of kidney diseases. Am. J. Kidney Dis. 2013, 61, 686–688. [Google Scholar] [CrossRef]
- Duff, S.; Murray, P.T. Defining Early Recovery of Acute Kidney Injury. Clin. J. Am. Soc. Nephrol. 2020, 15, 1358–1360. [Google Scholar] [CrossRef] [Green Version]
- Damase, T.R.; Sukhovershin, R.; Boada, C.; Taraballi, F.; Pettigrew, R.I.; Cooke, J.P. The Limitless Future of RNA Therapeutics. Front. Bioeng. Biotechnol. 2021, 9. [Google Scholar] [CrossRef]
- Brenner, S.; Jacob, F.; Meselson, M. An Unstable Intermediate Carrying Information from Genes to Ribosomes for Protein Synthesis. Nat. Cell Biol. 1961, 190, 576–581. [Google Scholar] [CrossRef]
- Bayda, S.; Adeel, M.; Tuccinardi, T.; Cordani, M.; Rizzolio, F. The History of Nanoscience and Nanotechnology: From Chemical–Physical Applications to Nanomedicine. Molecules 2020, 25, 112. [Google Scholar] [CrossRef] [Green Version]
- Hou, X.; Zaks, T.; Langer, R.; Dong, Y. Lipid nanoparticles for mRNA delivery. Nat. Rev. Mater. 2021, 6, 1078–1094. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and efficacy of the BNT162b2 mRNA COVID-19 vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-K. RNA Therapy: Current Status and Future Potential. Chonnam. Med. J. 2020, 56, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Feng, R.; Patil, S.; Zhao, X.; Miao, Z.; Qian, A. RNA Therapeutics—Research and Clinical Advancements. Front. Mol. Biosci. 2021, 8, 710738. [Google Scholar] [CrossRef] [PubMed]
- Ozpolat, B.; Sood, A.K.; Lopez-Berestein, G. Liposomal siRNA nanocarriers for cancer therapy. Adv. Drug Deliv. Rev. 2014, 66, 110–116. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.; Zhang, C.; Zhao, Z.; Zhu, T.; Yang, B. Fighting against kidney diseases with small interfering RNA: Opportunities and challenges. J. Transl. Med. 2015, 13, 39. [Google Scholar] [CrossRef] [Green Version]
- Van De Water, F.M.; Boerman, O.C.; Wouterse, A.C.; Peters, J.G.P.; Russel, F.G.M.; Masereeuw, R. Intravenously Administered Short Interfering RNA Accumulates in the Kidney and Selectively Suppresses Gene Function in Renal Proximal Tubules. Drug Metab. Dispos. 2006, 34, 1393–1397. [Google Scholar] [CrossRef]
- Rupaimoole, R.; Han, H.-D.; Lopez-Berestein, G.; Sood, A.K. MicroRNA therapeutics: Principles, expectations, and challenges. Chin. J. Cancer 2011, 30, 368–370. [Google Scholar] [CrossRef]
- Böttinger, E.P. TGF-β in Renal Injury and Disease. Semin. Nephrol. 2007, 27, 309–320. [Google Scholar] [CrossRef]
- Martin, J.; Jenkins, R.H.; Bennagi, R.; Krupa, A.; Phillips, A.O.; Bowen, T.; Fraser, N.J. Post-Transcriptional Regulation of Transforming Growth Factor Beta-1 by MicroRNA-744. PLoS ONE 2011, 6, e25044. [Google Scholar] [CrossRef] [Green Version]
- Wang, B.; Koh, P.; Winbanks, C.; Coughlan, M.T.; McClelland, A.; Watson, A.; Jandeleit-Dahm, K.; Burns, W.C.; Thomas, M.C.; Cooper, M.E.; et al. miR-200a Prevents Renal Fibrogenesis Through Repression of TGF-β2 Expression. Diabetes 2010, 60, 280–287. [Google Scholar] [CrossRef] [Green Version]
- Wang, B.; Herman-Edelstein, M.; Koh, P.; Burns, W.; Jandeleit-Dahm, K.; Watson, A.; Saleem, M.; Goodall, G.J.; Twigg, S.M.; Cooper, M.E.; et al. E-Cadherin Expression Is Regulated by miR-192/215 by a Mechanism That Is Independent of the Profibrotic Effects of Transforming Growth Factor-β. Diabetes 2010, 59, 1794–1802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, A.C.; Huang, X.R.; Meng, X.; Lan, H.Y. miR-192 Mediates TGF-β/Smad3-Driven Renal Fibrosis. J. Am. Soc. Nephrol. 2010, 21, 1317–1325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, L.; Zhang, D.; Liu, F.; Xiang, X.; Ling, G.; Xiao, L.; Liu, Y.; Zhu, X.; Zhan, M.; Yang, Y.; et al. Low-dose paclitaxel ameliorates fibrosis in the remnant kidney model by down-regulating miR-192. J. Pathol. 2011, 225, 364–377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guan, H.; Peng, R.; Mao, L.; Fang, F.; Xu, B.; Chen, M. Injured tubular epithelial cells activate fibroblasts to promote kidney fibrosis through miR-150-containing exosomes. Exp. Cell Res. 2020, 392, 112007. [Google Scholar] [CrossRef]
- Wei, Q.; Sun, H.; Song, S.; Liu, Y.; Liu, P.; Livingston, M.J.; Wang, J.; Liang, M.; Mi, Q.-S.; Huo, Y.; et al. MicroRNA-668 represses MTP18 to preserve mitochondrial dynamics in ischemic acute kidney injury. J. Clin. Investig. 2018, 128, 5448–5464. [Google Scholar] [CrossRef] [Green Version]
- Wei, Q.; Liu, Y.; Liu, P.; Hao, J.; Liang, M.; Mi, Q.-S.; Chen, J.-K.; Dong, Z. MicroRNA-489 Induction by Hypoxia–Inducible Factor–1 Protects against Ischemic Kidney Injury. J. Am. Soc. Nephrol. 2016, 27, 2784–2796. [Google Scholar] [CrossRef] [Green Version]
- Maurer, B.; Stanczyk, J.; Jüngel, A.; Akhmetshina, A.; Trenkmann, M.; Brock, M.; Kowal-Bielecka, O.; Gay, R.E.; Michel, B.A.; Distler, J.H.W.; et al. MicroRNA-29, a key regulator of collagen expression in systemic sclerosis. Arthritis Rheumatol. 2010, 62, 1733–1743. [Google Scholar] [CrossRef]
- Liu, Y.; Taylor, N.; Lu, L.; Usa, K.; Cowley, A.W.; Ferreri, N.R.; Yeo, N.C.; Liang, M. Renal Medullary MicroRNAs in Dahl Salt-Sensitive Rats. Hypertension 2010, 55, 974–982. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.-Y.; Gao, Y.-B.; Zhang, N.; Zou, D.-W.; Wang, P.; Zhu, Z.-Y.; Li, J.-Y.; Zhou, S.-N.; Wang, S.-C.; Wang, Y.-Y.; et al. miR-21 overexpression enhances TGF-β1-induced epithelial-to-mesenchymal transition by target smad7 and aggravates renal damage in diabetic nephropathy. Mol. Cell. Endocrinol. 2014, 392, 163–172. [Google Scholar] [CrossRef]
- Long, J.; Wang, Y.; Wang, W.; Chang, B.H.J.; Danesh, F.R. MicroRNA-29c Is a Signature MicroRNA under High Glucose Conditions That Targets Sprouty Homolog 1, and Its in Vivo Knockdown Prevents Progression of Diabetic Nephropathy. J. Biol. Chem. 2011, 286, 11837–11848. [Google Scholar] [CrossRef] [Green Version]
- Krupa, A.; Jenkins, R.; Luo, D.D.; Lewis, A.; Phillips, A.; Fraser, D. Loss of MicroRNA-192 Promotes Fibrogenesis in Diabetic Nephropathy. J. Am. Soc. Nephrol. 2010, 21, 438–447. [Google Scholar] [CrossRef] [PubMed]
- Putta, S.; Lanting, L.; Sun, G.; Lawson, G.; Kato, M.; Natarajan, R. Inhibiting MicroRNA-192 Ameliorates Renal Fibrosis in Diabetic Nephropathy. J. Am. Soc. Nephrol. 2012, 23, 458–469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kato, M.; Arce, L.; Wang, M.; Putta, S.; Lanting, L.; Natarajan, R. A microRNA circuit mediates transforming growth factor-β1 autoregulation in renal glomerular mesangial cells. Kidney Int. 2011, 80, 358–368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dey, N.; Das, F.; Mariappan, M.M.; Mandal, C.; Ghosh-Choudhury, N.; Kasinath, B.S.; Choudhury, G.G. MicroRNA-21 Orchestrates High Glucose-induced Signals to TOR Complex 1, Resulting in Renal Cell Pathology in Diabetes. J. Biol. Chem. 2011, 286, 25586–25603. [Google Scholar] [CrossRef] [Green Version]
- Guha, M.; Xu, Z.-G.; Tung, D.; Lanting, L.; Natarajan, R. Specific down-regulation of connective tissue growth factor attenuates progression of nephropathy in mouse models of type 1 and type 2 diabetes. FASEB J. 2007, 21, 3355–3368. [Google Scholar] [CrossRef]
- Ramasamy, R.; Yan, S.F.; Schmidt, A.M. Receptor for AGE (RAGE): Signaling mechanisms in the pathogenesis of diabetes and its complications. Ann. N. Y. Acad. Sci. 2011, 1243, 88–102. [Google Scholar] [CrossRef] [Green Version]
- Matsui, T.; Higashimoto, Y.; Nishino, Y.; Nakamura, N.; Fukami, K.; Yamagishi, S.-I. RAGE-Aptamer Blocks the Development and Progression of Experimental Diabetic Nephropathy. Diabetes 2017, 66, 1683–1695. [Google Scholar] [CrossRef] [Green Version]
- Fan, Y.; Chen, H.; Huang, Z.; Zheng, H.; Zhou, J. Emerging role of miRNAs in renal fibrosis. RNA Biol. 2020, 17, 1–12. [Google Scholar] [CrossRef]
- Bera, A.; Das, F.; Ghosh-Choudhury, N.; Mariappan, M.M.; Kasinath, B.S.; Choudhury, G.G. Reciprocal regulation of miR-214 and PTEN by high glucose regulates renal glomerular mesangial and proximal tubular epithelial cell hypertrophy and matrix expansion. Am. J. Physiol. Physiol. 2017, 313, C430–C447. [Google Scholar] [CrossRef] [Green Version]
- Tan, K.; Chen, J.; Li, W.; Chen, Y.; Sui, W.; Zhang, Y.; Dai, Y. Genome-wide analysis of microRNAs expression profiling in patients with primary IgA nephropathy. Genome 2013, 56, 161–169. [Google Scholar] [CrossRef]
- Wang, G.; Kwan, B.C.-H.; Lai, F.M.-M.; Chow, K.-M.; Li, P.K.-T.; Szeto, C.-C. Elevated levels of miR-146a and miR-155 in kidney biopsy and urine from patients with IgA nephropathy. Dis. Markers 2011, 30, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Josefowicz, S.; Kas, A.; Chu, T.-T.; Gavin, M.A.; Rudensky, A.Y. Genome-wide analysis of Foxp3 target genes in developing and mature regulatory T cells. Nat. 2007, 445, 936–940. [Google Scholar] [CrossRef] [PubMed]
- Kohlhaas, S.; Garden, O.A.; Scudamore, C.; Turner, M.; Okkenhaug, K.; Vigorito, E. Cutting Edge: The Foxp3 Target miR-155 Contributes to the Development of Regulatory T Cells. J. Immunol. 2009, 182, 2578–2582. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Peng, Y.; Liu, F.; Lei, H. Is IgA nephropathy induced by abnormalities of CD4+CD25+Treg cells in the tonsils? Med. Hypotheses 2007, 69, 410–413. [Google Scholar] [CrossRef] [PubMed]
- Bao, H.; Hu, S.; Zhang, C.; Shi, S.; Qin, W.; Zeng, C.; Zen, K.; Liu, Z. Inhibition of miRNA-21 prevents fibrogenic activation in podocytes and tubular cells in IgA nephropathy. Biochem. Biophys. Res. Commun. 2014, 444, 455–460. [Google Scholar] [CrossRef]
- Lu, J.; Kwan, B.C.; Lai, F.M.-M.; Tam, L.-S.; Li, E.K.-M.; Chow, K.-M.; Wang, G.; Li, P.K.-T.; Szeto, C.-C. Glomerular and tubulointerstitial miR-638, miR-198 and miR-146a expression in lupus nephritis. Nephrology 2012, 17, 346–351. [Google Scholar] [CrossRef]
- Costa-Reis, P.; Russo, P.A.; Zhang, Z.; Colonna, L.; Maurer, K.; Gallucci, S.; Schulz, S.W.; Kiani, A.N.; Petri, M.; Sullivan, K.E. The Role of MicroRNAs and Human Epidermal Growth Factor Receptor 2 in Proliferative Lupus Nephritis. Arthritis Rheumatol. 2015, 67, 2415–2426. [Google Scholar] [CrossRef]
- McCarthy, E.T.; Sharma, M.; Savin, V.J. Circulating Permeability Factors in Idiopathic Nephrotic Syndrome and Focal Segmental Glomerulosclerosis. Clin. J. Am. Soc. Nephrol. 2010, 5, 2115–2121. [Google Scholar] [CrossRef] [Green Version]
- Shaffi, S.K.; Galas, D.; Etheridge, A.; Argyropoulos, C. Role of MicroRNAs in Renal Parenchymal Diseases—A New Dimension. Int. J. Mol. Sci. 2018, 19, 1797. [Google Scholar] [CrossRef] [Green Version]
- Brandenburger, T.; Lorenzen, J.M. Diagnostic and Therapeutic Potential of microRNAs in Acute Kidney Injury. Front. Pharmacol. 2020, 11, 657. [Google Scholar] [CrossRef]
- Ramalingam, H.; Yheskel, M.; Patel, V. Modulation of polycystic kidney disease by non-coding RNAs. Cell. Signal. 2020, 71, 109548. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Sun, L. MicroRNAs and Polycystic Kidney Disease. Kidney Med. 2020, 2, 762–770. [Google Scholar] [CrossRef] [PubMed]
- Lv, W.; Fan, F.; Wang, Y.; Gonzalez-Fernandez, E.; Wang, C.; Yang, L.; Booz, G.W.; Roman, R.J. Therapeutic potential of microRNAs for the treatment of renal fibrosis and CKD. Physiol. Genom. 2018, 50, 20–34. [Google Scholar] [CrossRef]
- Kannel, W.B. Blood pressure as a cardiovascular risk factor: Prevention and treatment. JAMA J. Am. Med. Assoc. 1996, 275, 1571–1576. [Google Scholar] [CrossRef]
- Schmieder, R.E.; Messerli, F.H. Hypertension and the heart. J. Hum. Hypertens. 2000, 14, 597–604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beauchet, O.; Celle, S.; Roche, F.; Bartha, R.; Montero-Odasso, M.; Allali, G.; Annweiler, C. Blood pressure levels and brain volume reduction. J. Hypertens. 2013, 31, 1502–1516. [Google Scholar] [CrossRef] [Green Version]
- Saran, R.; Robinson, B.; Abbott, K.C.; Agodoa, L.Y.; Albertus, P.; Ayanian, J.; Balkrishnan, R.; Bragg-Gresham, J.; Cao, J.; Chen, J.L.T.; et al. US Renal Data System 2016 Annual Data Report: Epidemiology of Kidney Disease in the United States. Am. J. Kidney Dis. 2017, 69, A7–A8, Erratum in Am. J. Kidney Dis. 2017, 69, 712. [Google Scholar] [CrossRef]
- Jeppesen, P.L.; Christensen, G.L.; Schneider, M.; Nossent, A.Y.; Jensen, H.B.; Andersen, D.C.; Eskildsen, T.; Gammeltoft, S.; Hansen, J.L.; Sheikh, S.P. Angiotensin II type 1 receptor signalling regulates microRNA differentially in cardiac fibroblasts and myocytes. J. Cereb. Blood Flow Metab. 2011, 164, 394–404. [Google Scholar] [CrossRef] [Green Version]
- Kemp, J.R.; Unal, H.; Desnoyer, R.; Yue, H.; Bhatnagar, A.; Karnik, S.S. Angiotensin II-regulated microRNA 483-3p directly targets multiple components of the renin–angiotensin system. J. Mol. Cell. Cardiol. 2014, 75, 25–39. [Google Scholar] [CrossRef] [Green Version]
- Li, T.; Song, X.; Zhang, J.; Zhao, L.; Shi, Y.; Li, Z.; Liu, J.; Liu, N.; Yan, Y.; Xiao, Y.; et al. Protection of Human Umbilical Vein Endothelial Cells against Oxidative Stress by MicroRNA-210. Oxidative Med. Cell. Longev. 2017, 2017, 3565613. [Google Scholar] [CrossRef]
- Sun, H.-X.; Zeng, D.-Y.; Li, R.-T.; Pang, R.-P.; Yang, H.; Hu, Y.-L.; Zhang, Q.; Jiang, Y.; Huang, L.-Y.; Tang, Y.-B.; et al. Essential Role of MicroRNA-155 in Regulating Endothelium-Dependent Vasorelaxation by Targeting Endothelial Nitric Oxide Synthase. Hypertension 2012, 60, 1407–1414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jackson, K.L.; Marques, F.Z.; Watson, A.M.; Palma-Rigo, K.; Nguyen-Huu, T.-P.; Morris, B.J.; Charchar, F.J.; Davern, P.J.; Head, G.A. A Novel Interaction Between Sympathetic Overactivity and Aberrant Regulation of Renin by miR-181a in BPH/2J Genetically Hypertensive Mice. Hypertension 2013, 62, 775–781. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marques, F.; Campaign, A.E.; Tomaszewski, M.; Zukowska-Szczechowska, E.; Yang, J.; Charchar, F.; Morris, B.J. Gene Expression Profiling Reveals Renin mRNA Overexpression in Human Hypertensive Kidneys and a Role for MicroRNAs. Hypertension 2011, 58, 1093–1098. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pandey, P.; Brors, B.; Srivastava, P.K.; Bott, A.; Boehn, S.N.; Groene, H.-J.; Gretz, N. Microarray-based approach identifies microRNAs and their target functional patterns in polycystic kidney disease. BMC Genom. 2008, 9, 624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hajarnis, S.; Lakhia, R.; Patel, V. MicroRNAs and Polycystic Kidney Disease. In Polycystic Kidney Disease; Codon Publications: Singapore, 2015; pp. 313–334. [Google Scholar]
- Lee, E.C.; Valencia, T.; Allerson, C.; Schairer, A.; Flaten, A.; Yheskel, M.; Kersjes, K.; Li, J.; Gatto, S.; Takhar, M.; et al. Discovery and preclinical evaluation of anti-miR-17 oligonucleotide RGLS4326 for the treatment of polycystic kidney disease. Nat. Commun. 2019, 10, 4148. [Google Scholar] [CrossRef] [Green Version]
- Gomez, I.G.; MacKenna, D.A.; Johnson, B.G.; Kaimal, V.; Roach, A.M.; Ren, S.; Nakagawa, N.; Xin, C.; Newitt, R.; Pandya, S.; et al. Anti–microRNA-21 oligonucleotides prevent Alport nephropathy progression by stimulating metabolic pathways. J. Clin. Investig. 2015, 125, 141–156. [Google Scholar] [CrossRef]
- Ramsden, J.J. What is nanotechnology? In Nanotechnology; Elsevier: Amsterdam, The Netherlands, 2016; pp. 1–18. [Google Scholar]
- Mozafari, M.R. (Ed.) Nanomaterials and Nanosystems for Biomedical Applications; Springer: Dordrecht, The Netherlands, 2007. [Google Scholar]
- Ganguly, R.; Singh, A.K.; Kumar, R.; Gupta, A.; Pandey, A.K.; Pandey, A.K. Nanoparticles as Modulators of Oxidative Stress. In Nanotechnology in Modern Animal Biotechnology; Elsevier: Amsterdam, The Netherlands, 2019; pp. 29–35. [Google Scholar] [CrossRef]
- Najahi-Missaoui, W.; Arnold, R.D.; Cummings, B.S. Safe Nanoparticles: Are We There Yet? Int. J. Mol. Sci. 2020, 22, 385. [Google Scholar] [CrossRef]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Mozafari, M.R. (Ed.) Nanocarrier Technologies; Springer: Dordrecht, The Netherlands, 2006. [Google Scholar]
- Xue, H.; Guo, P.; Wen, W.-C.; Wong, H. Lipid-Based Nanocarriers for RNA Delivery. Curr. Pharm. Des. 2015, 21, 3140–3147. [Google Scholar] [CrossRef]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Solid lipid nanoparticles and nanostructured lipid carriers as novel drug delivery systems: Applications, advantages and disadvantages. Res. Pharm. Sci. 2018, 13, 288–303. [Google Scholar] [CrossRef]
- Montoto, S.S.; Muraca, G.; Ruiz, M.E. Solid Lipid Nanoparticles for Drug Delivery: Pharmacological and Biopharmaceutical Aspects. Front. Mol. Biosci. 2020, 7, 587997. [Google Scholar] [CrossRef] [PubMed]
- Mishra, D.K.; Shandilya, R.; Mishra, P.K. Lipid based nanocarriers: A translational perspective. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 2023–2050. [Google Scholar] [CrossRef] [PubMed]
- Osborn, M.F.; Khvorova, A. Improving siRNA Delivery In Vivo through Lipid Conjugation. Nucleic Acid Ther. 2018, 28, 128–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Opatha, S.A.T.; Titapiwatanakun, V.; Chutoprapat, R. Transfersomes: A Promising Nanoencapsulation Technique for Transdermal Drug Delivery. Pharmaceutics 2020, 12, 855. [Google Scholar] [CrossRef]
- Yalavarthi, P.; Dudala, T.; Mudumala, N.; Pasupati, V.; Thanniru, J.; Vadlamudi, H.; Yaga, G. A perspective overview on lipospheres as lipid carrier systems. Int. J. Pharm. Investig. 2014, 4, 149–155. [Google Scholar] [CrossRef] [Green Version]
- Jaiswal, M.; Dudhe, R.; Sharma, P.K. Nanoemulsion: An advanced mode of drug delivery system. 3 Biotech 2015, 5, 123–127. [Google Scholar] [CrossRef] [Green Version]
- Duan, Y.; Dhar, A.; Patel, C.; Khimani, M.; Neogi, S.; Sharma, P.; Kumar, N.S.; Vekariya, R.L. A brief review on solid lipid nanoparticles: Part and parcel of contemporary drug delivery systems. RSC Adv. 2020, 10, 26777–26791. [Google Scholar] [CrossRef]
- Tenchov, R.; Bird, R.; Curtze, A.E.; Zhou, Q. Lipid Nanoparticles─From Liposomes to mRNA Vaccine Delivery, a Landscape of Research Diversity and Advancement. ACS Nano 2021, 15, 16982–17015. [Google Scholar] [CrossRef]
- Dammes, N.; Goldsmith, M.; Ramishetti, S.; Dearling, J.L.J.; Veiga, N.; Packard, A.B.; Peer, D. Conformation-sensitive targeting of lipid nanoparticles for RNA therapeutics. Nat. Nanotechnol. 2021, 16, 1030–1038. [Google Scholar] [CrossRef]
- Blanco, E.; Shen, H.; Ferrari, M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 2015, 33, 941–951. [Google Scholar] [CrossRef]
- Sakellari, G.I.; Zafeiri, I.; Batchelor, H.; Spyropoulos, F. Formulation design, production and characterisation of solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) for the encapsulation of a model hydrophobic active. Food Hydrocoll. Health 2021, 1, 100024. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Cai, F.; Li, Y.; Chen, J.; Han, F.; Lin, W. A review of the application of nanoparticles in the diagnosis and treatment of chronic kidney disease. Bioact. Mater. 2020, 5, 732–743. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Wang, J.; Jiang, K.; Chung, E.J. Improving kidney targeting: The influence of nanoparticle physicochemical properties on kidney interactions. J. Control. Release 2021, 334, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Żak, M.M.; Zangi, L. Lipid Nanoparticles for Organ-Specific mRNA Therapeutic Delivery. Pharm. 2021, 13, 1675. [Google Scholar] [CrossRef] [PubMed]
- Rupaimoole, R.; Slack, F.J. MicroRNA therapeutics: Towards a new era for the management of cancer and other diseases. Nat. Rev. Drug Discov. 2017, 16, 203–222. [Google Scholar] [CrossRef]
- Roberts, T.C.; Langer, R.; Wood, M.J.A. Advances in oligonucleotide drug delivery. Nat. Rev. Drug Discov. 2020, 19, 673–694. [Google Scholar] [CrossRef]
- Dhuri, K.; Bechtold, C.; Quijano, E.; Pham, H.; Gupta, A.; Vikram, A.; Bahal, R. Antisense Oligonucleotides: An Emerging Area in Drug Discovery and Development. J. Clin. Med. 2020, 9, 2004. [Google Scholar] [CrossRef]
- Shimizu, H.; Hori, Y.; Kaname, S.; Yamada, K.; Nishiyama, N.; Matsumoto, S.; Miyata, K.; Oba, M.; Yamada, A.; Kataoka, K.; et al. siRNA-Based Therapy Ameliorates Glomerulonephritis. J. Am. Soc. Nephrol. 2010, 21, 622–633. [Google Scholar] [CrossRef] [Green Version]
- Morishita, Y.; Yoshizawa, H.; Watanabe, M.; Ishibashi, K.; Muto, S.; Kusano, E.; Nagata, D. siRNAs targeted to Smad4 prevent renal fibrosis in vivo. Sci. Rep. 2014, 4, 6424. [Google Scholar] [CrossRef] [Green Version]
- Alidori, S.; Akhavein, N.; Thorek, D.L.J.; Behling, K.; Romin, Y.; Queen, D.; Beattie, B.J.; Manova-Todorova, K.; Bergkvist, M.; Scheinberg, D.A.; et al. Targeted fibrillar nanocarbon RNAi treatment of acute kidney injury. Sci. Transl. Med. 2016, 8, 331ra39. [Google Scholar] [CrossRef] [Green Version]
- Narváez, A.; Guiteras, R.; Sola, A.; Manonelles, A.; Morote, J.; Torras, J.; Grinyó, J.M.; Cruzado, J.M. siRNA-silencing of CD40 attenuates unilateral ureteral obstruction-induced kidney injury in mice. PLoS ONE 2019, 14, e0215232. [Google Scholar] [CrossRef] [PubMed]
- A Study to Evaluate Efficacy and Safety of QPI-1002 for Prevention of Acute Kidney Injury Following Cardiac Surgery. 2015. Available online: https://www.cochranelibrary.com/central/doi/10.1002/central/CN-01553862/full (accessed on 27 December 2021).
- QPI-1002 for Prevention of Delayed Graft Function in Recipients of an Older Donor Kidney Transplant (ReGIFT). Available online: https://clinicaltrials.gov/ct2/show/NCT02610296 (accessed on 27 December 2021).
- NOX-E36 in Patients with Type 2 Diabetes Mellitus and Albuminuria. Available online: https://clinicaltrials.gov/ct2/show/NCT01547897 (accessed on 27 December 2021).
- Study of Lademirsen (SAR339375) in Patients with Alport Syndrome (HERA). Available online: https://clinicaltrials.gov/ct2/show/NCT02855268 (accessed on 27 December 2021).
- Bajan, S.; Hutvagner, G. RNA-Based Therapeutics: From Antisense Oligonucleotides to miRNAs. Cells 2020, 9, 137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ledeganck, K.J.; Gielis, E.M.; Abramowicz, D.; Stenvinkel, P.; Shiels, P.G.; Van Craenenbroeck, A.H. MicroRNAs in AKI and Kidney Transplantation. Clin. J. Am. Soc. Nephrol. 2019, 14, 454–468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chau, B.N.; Xin, C.; Hartner, J.; Ren, S.; Castano, A.P.; Linn, G.; Li, J.; Tran, P.T.; Kaimal, V.; Huang, X.; et al. MicroRNA-21 Promotes Fibrosis of the Kidney by Silencing Metabolic Pathways. Sci. Transl. Med. 2012, 4, 121ra18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loibner, H.; Lametschwandtner, G.; Westritschnig, K.; Mutschlechner, O.; Dohnal, A.; Salzberg, M.O.; Triozzi, P.L. Adoptive cellular immunotherapy with APN401, autologous cbl-b silenced peripheral blood mononuclear cells: Data from a phase I study in patients with solid tumors. J. Clin. Oncol. 2018, 36, 3055. [Google Scholar] [CrossRef] [Green Version]
- APN401 in Treating Patients with Melanoma, Kidney Cancer, Pancreatic Cancer, or Other Solid Tumors That Are Metastatic or Cannot Be Removed by Surgery. Available online: https://clinicaltrials.gov/ct2/show/NCT02166255 (accessed on 27 December 2021).
- A Study of RGLS4326 in Patients with Autosomal Dominant Polycystic Kidney Disease. Available online: https://clinicaltrials.gov/ct2/show/NCT04536688 (accessed on 27 December 2021).
- Kumar, R.; Conklin, D.S.; Mittal, V. High-Throughput Selection of Effective RNAi Probes for Gene Silencing. Genome Res. 2003, 13, 2333–2340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marquet, R.; Davins, J.; Casas, J.; Fernández, R.M. Quality assurance at a health center: 2 years’ experience. Atención Primaria 1991, 8, 746–752. [Google Scholar]
- Castanotto, D.; Rossi, J.J. The promises and pitfalls of RNA-interference-based therapeutics. Nature 2009, 457, 426–433. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).