Biochemistry of Antioxidants: Mechanisms and Pharmaceutical Applications
Abstract
1. Introduction—Antioxidants as Pharmacological Agents
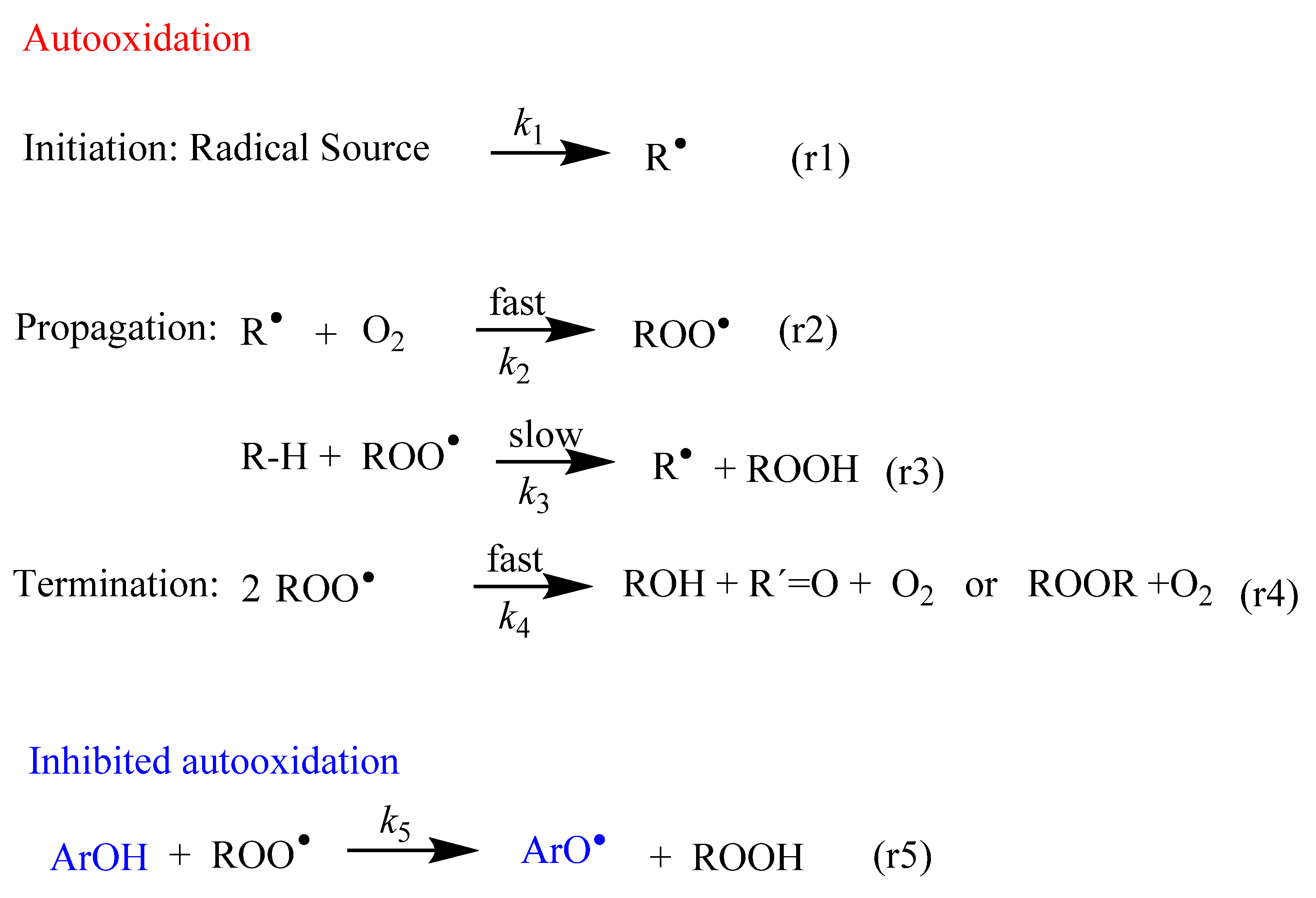
2. Oxidative Reactions of Free Radicals
3. Antioxidants
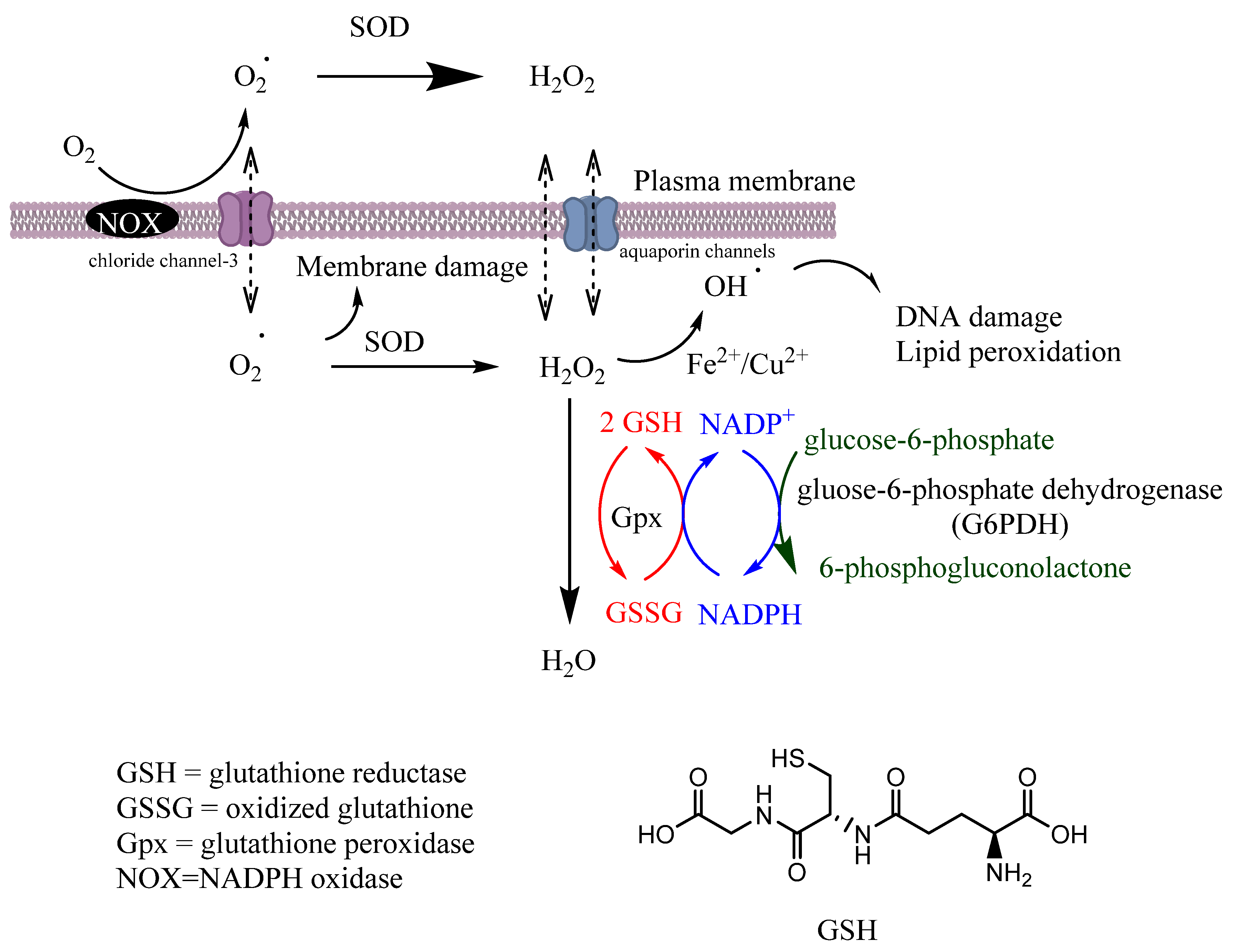
3.1. Endogenous Antioxidant Enzymes
3.1.1. Glutathione Peroxidase (Gpx)
3.1.2. Catalase (CAT)
3.1.3. Superoxide Dismutase (SODs)
3.2. Non-Enzymatic Endogenous Antioxidants
3.3. Exogenous Non-Enzymatic AOs—Dietary Sources and Classification
- Hydroxybenzoic acids include protocatechuic, vanillic and gallic acids, among others. They frequently can be found glycosylated, bound to small organic acids such as quinic, maleic or tartaric acids, or linked to structural components of plant cells (cellulose, proteins). Hydrolyzable tannins (gallotannins, ellagitannins) can be formed from gallic acid and carbohydrates.
- The most abundant hydroxycinnamic acids are caffeic, p-coumaric and ferulic acids, along with ester derivatives of caffeic acid such as chlorogenic acid.
- Flavones are characterized by the presence of a double bond between carbon 2 and 3, a keto group in carbon 4 and a B ring linked to carbon 2. The most common flavones include apigenin, luteolin and their glycosides.
- Isoflavonoids are flavones in which the B ring is linked to carbon 3 instead of carbon 2. This change causes isoflavonoids to resemble estrogens which are well-known as phytoestrogens, presenting a possible mild estrogenic activity. The most abundant isoflavones are glycitein and daidzein, among others, and their 7-O glycosides.
- Flavonols are flavones that have a hydroxyl group at carbon 3. Examples of flavonols are quercetin, myricetin, kaempferol and their glycosides.
- In flavanones and flavononols, ring C is a saturated pyrane ring with a ketone group in carbon 4. Flavononols also contain a –OH group in carbon atom 3. They are not the most abundant flavonoids in nature, but the most common flavonones are naringenin, hesperetin and eriodictyol, and the most common flavononols are taxifolin and its glycosides.
- Flavanols hold a ketone group at carbon 4 and a hydroxyl group at position 3. The most important flavanols are epicatechin, catechin, pirogallocatechin, gallocatechin and their oligomers, polymers and 3-O-gallates.
- Anthocyanidins are ionic flavonoids and differ from the other flavonoids by possessing an –OH group in carbon 3 and two double bonds in ring C. They are the major group of hydrophilic plant pigments, and their color changes with the pH. The most common are malvidin, peonidin, cyaniding, etc., which provide different colors to vegetables.
- Chalcones are characterized by the absence of the C ring of the basic flavonoid skeleton structure and are referred to as open-chain flavonoids. Major examples of chalcones include phloridzin, arbutin, phloretin and chalconaringenin. Chalcones have a common chemical scaffold of 1,3-diaryl-2-propen-1-one (α-, β- unsaturated ketones, also known as chalconoid) that exists as trans and cis isomers, with the trans isomer being thermodynamically more stable.
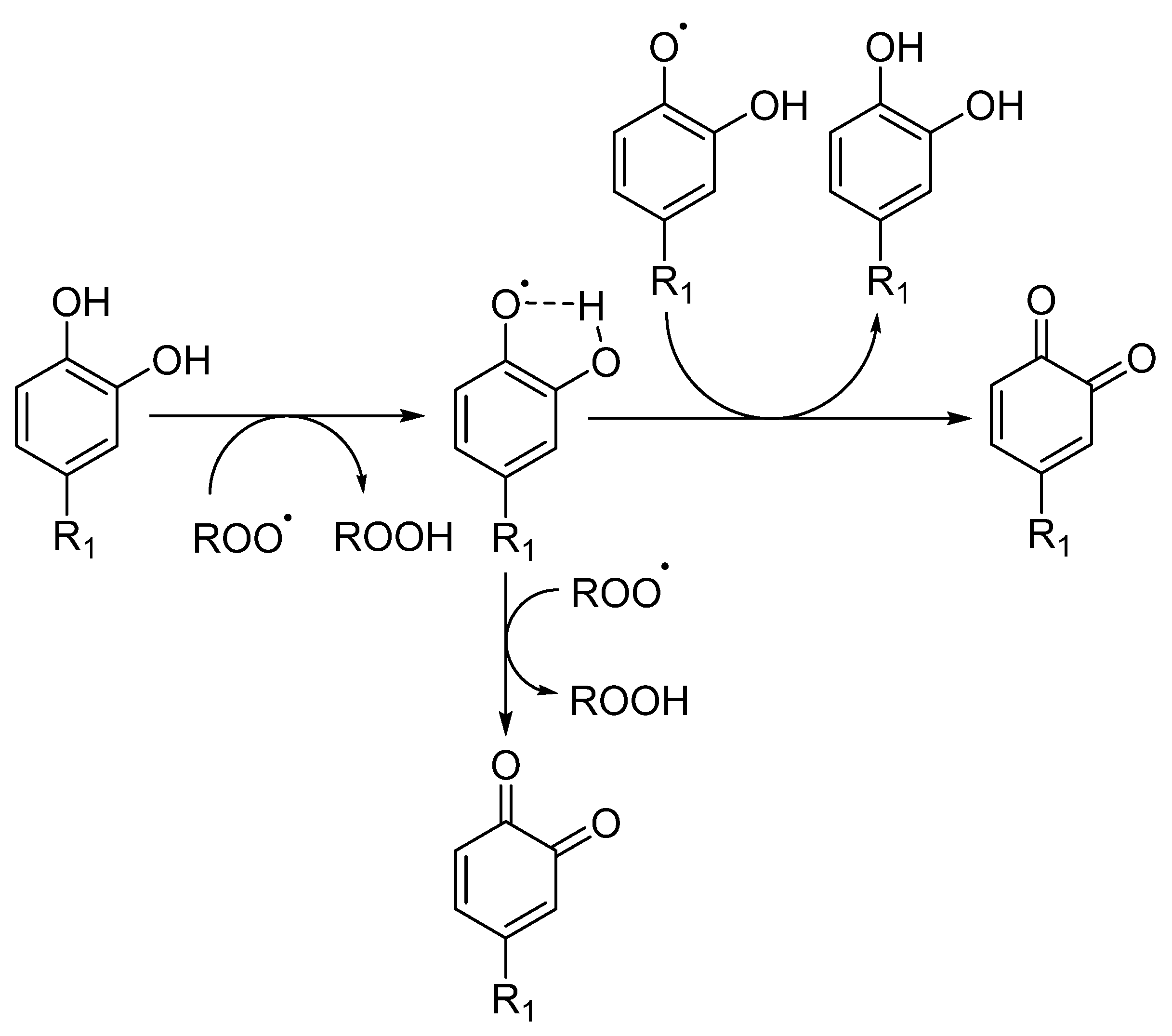
4. Control of Oxidative Reactions by Antioxidants
5. Importance of Partitioning of Antioxidants
5.1. Properties of Antioxidants Affecting their Solubility in Bulk Solvents
5.2. Properties of Solvents Affecting the Solubility of Solutes
5.3. Modifying the Distribution of Antioxidants in Partitioning Systems—A Brief Theoretical Background
5.4. Modifications to the Solvent Phase Affecting Partitioning (Table 4)
- -
- Cosolvents: The addition of cosolvents has the ability to alter the dielectric constant of a given solvent, affecting, therefore, the energy required to overcome hydrogen bonding forces in aqueous media and reducing the amount of energy required to create cavities in the mixture to accommodate the solute. Moreover, these changes in the solvent properties can greatly alter the degree of solvation of the solute, eventually affecting the partitioning of the molecule.
- -
- Hydrotropes: The addition of hydrotropes is commonly employed to increase the water solubility of poorly soluble drugs, and, in many cases, the water solubility is increased by several orders of magnitude [47]. Examples of hydrotropic agents include urea, caffeine, tryptophan, etc.
- -
- Micellar systems: Surfactants are amphiphilic molecules having both hydrophobic and hydrophilic portions that, when added to solution, associate spontaneously to form spherical aggregates—micelles—once the surfactant concentration reaches a critical value, called the critical micelle concentration. The hydrophobic portions of surfactants are chains with 8–22 carbon atoms. Common hydrophilic groups of ionic surfactants are carboxylate (–COO−), sulfate (–OSO3−), sulfonate (SO3−), carboxybetaine (–NR2CH2COO−), sulfobetaine (–N(CH3)2C3H6SO3−) and quaternary ammonium (–R4N+). The hydrophilic group of nonionic surfactants is usually a polyoxyethylene group, but glyceryl groups or sorbitol groups are also common, and their use depends on the particular application. Surfactants commonly used in food and pharmaceutical products include, but are not limited to, non-ionic surfactants such as polysorbates, polyoxyethylenes, triton X, Cremophor EL and Chremopor RH60.
- Micellar aggregates contain a hydrophobic core (e.g., in O/W systems) that allows solubilization of hydrophobic molecules that otherwise cannot be dissolved in the aqueous matrix. The partitioning effect is usually quantified in terms of the association constant of the molecule to the aggregate, Equation (6), where SW stands for the solute in water, SM the solute in the micelle, Dn is the micellized surfactant (Dn = [Surf]total – CMC) and KS stands for the association constant of the molecule to the micellar aggregate.
- -
- Liposomes: Liposomes are closed spherical vesicles composed of one or more bilayers of amphipathic lipid molecules enclosing one or more aqueous core compartments [48]. Hydrophobic antioxidants can be solubilized by liposomes, becoming an integral part of the lipid bilayer. Water-soluble drugs reside within the aqueous inner core and are released as the liposome erodes in vivo or by leakage. A typical liposome formulation contains water with phospholipid at ~5–20 mg/mL, an isotonicifier and a pH 5–8 buffer. The phospholipids normally used include phosphatidylcholine, phosphatidic acid, phosphatidylglycerol and saturated lipids such as l-a-dimyristoylphosphatidylcholine (DMPC), dipalmitoyl phosphatidylcholine (DPPC), dipalmitoyl phosphatidic acid (DPPA) and l-a-dimyristoylphosphatidylglycerol (DMPG).
- -
- Microemulsions: Microemulsions are a thermodynamically stable isotropically clear dispersion composed of a polar solvent (usually water), an oil, a surfactant and a cosurfactant. Microemulsions offer many advantages compared to macroemulsions; they have smaller particles (often <100 nm), require less energy to be produced and have higher physical stability, due to the high levels of emulsifiers used in their preparation [49]. Microemulsions generally have very low interfacial tension at the water–oil interface and form a highly fluid interfacial surfactant film. Due to the numerous small droplets, the surface area-to-volume ratio of microemulsions is very high.
- The presence of surfactant and cosurfactant could make microemulsion supersolvents for drugs relatively insoluble in both aqueous and hydrophobic solvents [50]. Sometimes mixed oils and/or mixed surfactants are employed in the preparation of microemulsions, offering some advantages over pure single component materials [50]. Quantifying the distribution of solutes in microemulsions is difficult. Nevertheless, once the interfacial polarity and microstructure are known, along with how compounds may interact with the different phase solvents, qualitative predictions can be made, Figure 6.

- -
- Emulsions: Emulsions are, in contrast with microemulsions, thermodynamically unstable mixtures of oil and water in which one liquid is dispersed as small spherical droplets in the other one. Since the molecules of the liquids are in direct contact with each other, they tend to separate and, on standing, emulsions phase separate through different processes to minimize the contact between them. Emulsions, however, can be stabilized kinetically by adding a surfactant or emulsifier. The added surfactants are arranged on the interface between the oil and water phases so that the hydrophilic part of the surfactant is located in the polar phase; meanwhile, the hydrophobic portion tends to be located in the oily phase, so that surfactants create a film surrounding the surface of the droplets, resulting in a decrease in the interfacial tension and ensuring kinetic stability for some time [49,51].
- When antioxidants are added to emulsions, they distribute, as in microemulsions, thermodynamically between the oil, interfacial and aqueous regions, so that two partition constants are necessary to describe its distribution, that between the oil–interfacial, POI (Equation (7)) and that between the aqueous–interfacial, PWI (Equation (8)) regions, Figure 6.
- -
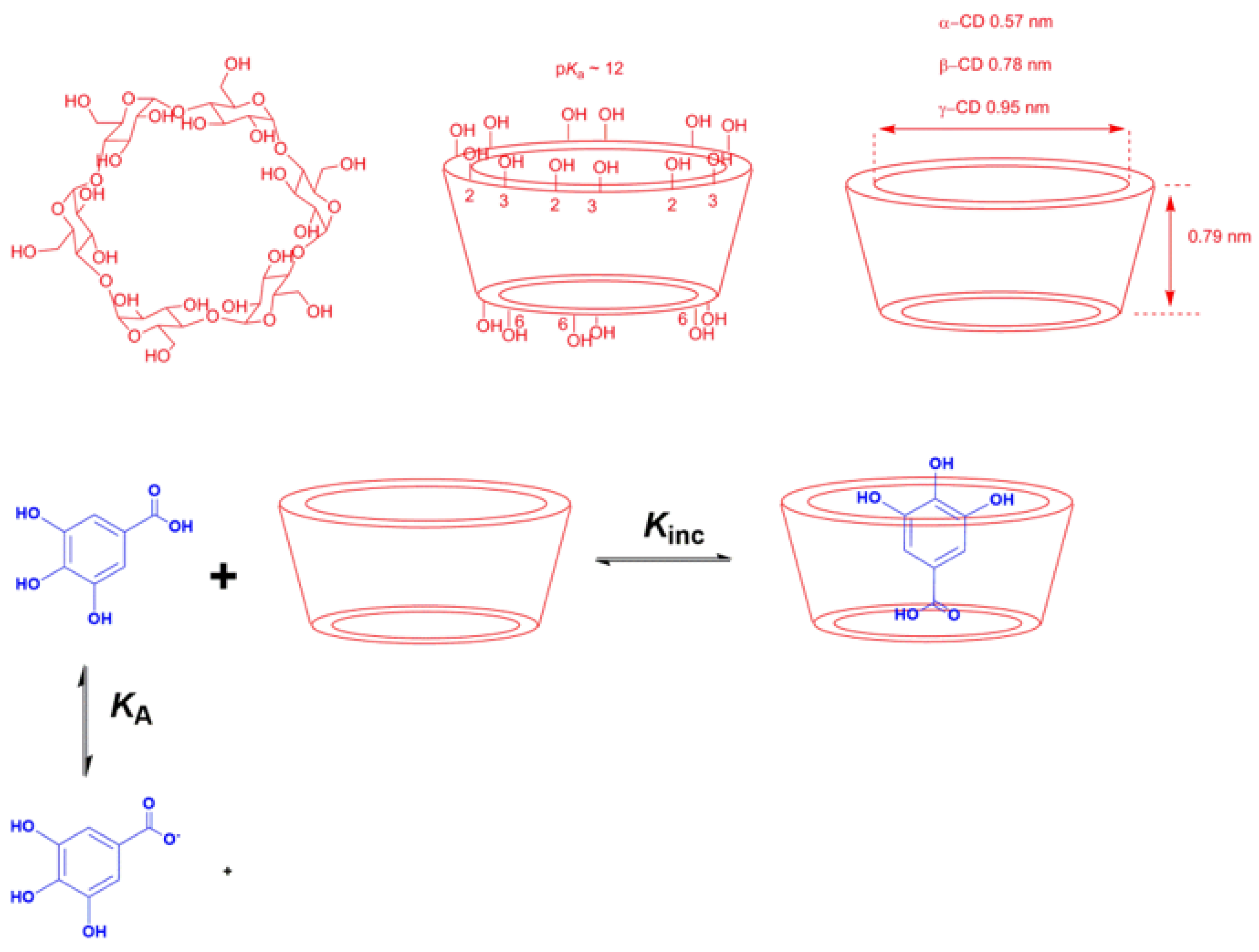
- Complexation: The main strategy of complexation is to reduce the exposure of the molecule’s hydrophobic region to water. This can be achieved, for instance, by adding water-soluble agents capable of hosting water-insoluble molecules. Crown-ethers and curcubiturils contain hydrophobic cavities capable of hosting hydrophobic molecules, but probably the most widely employed hosting agents are the cyclodextrins (CDs) because of their high water-solubility and in vivo safety margin, Figure 7 [52].
- -
- Combined strategies: Decisions regarding the optimal strategy to modify the partitioning of antioxidants often lie in modifying, in some way, the intrinsic solubility of the antioxidants in a given phase. Thus, combinations of methods, such as variations in the acidity or temperature of the solution together with the use of surfactant-based systems and cyclodextrins, have been widely used. Technologies such as cosolvency and pH modifications are also commonly employed to shift equilibria. Increasing the salt concentration affects partitioning due to the ion-pair association, salting-in or -out effects and because the composition of phases changes.
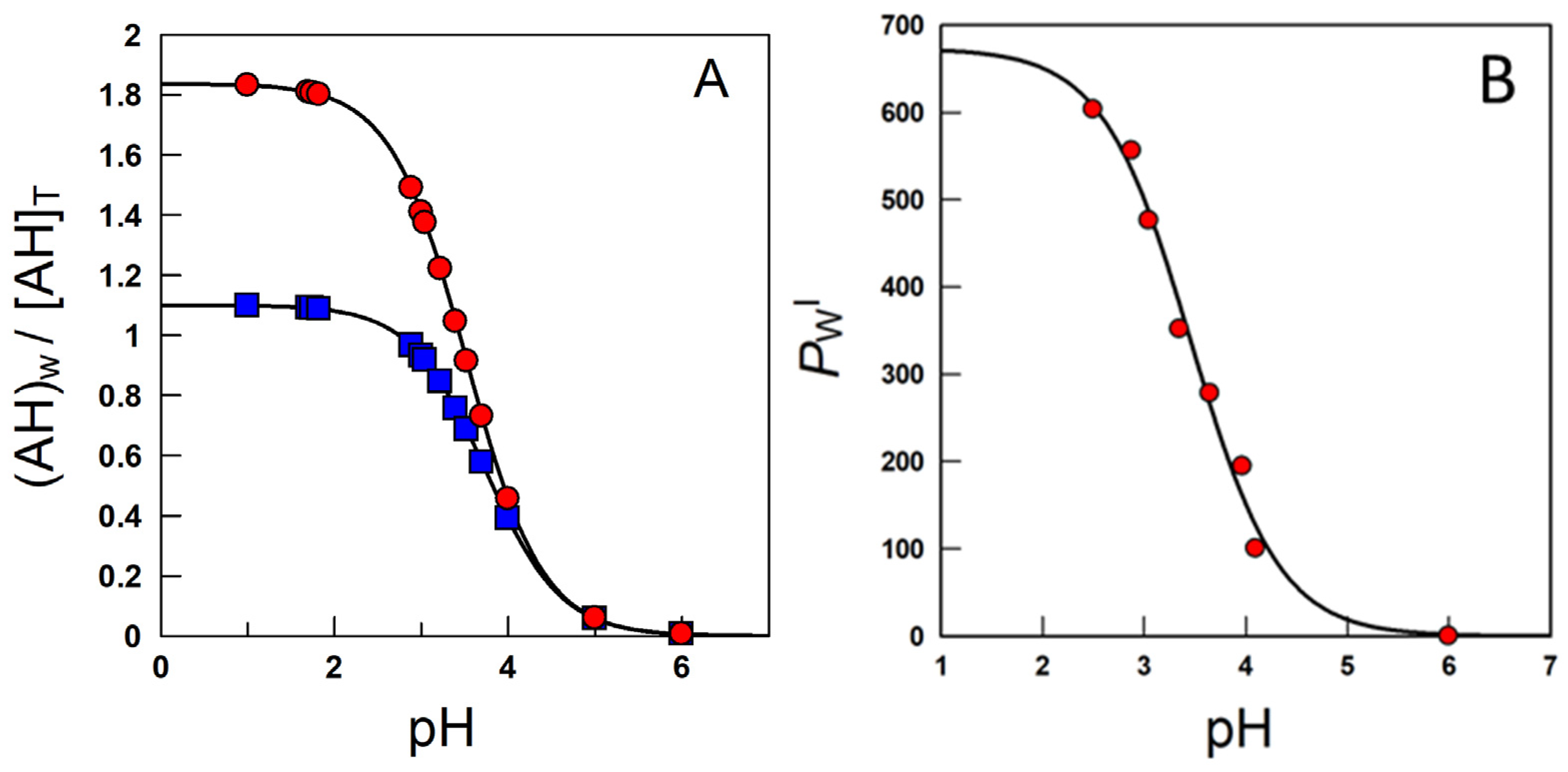
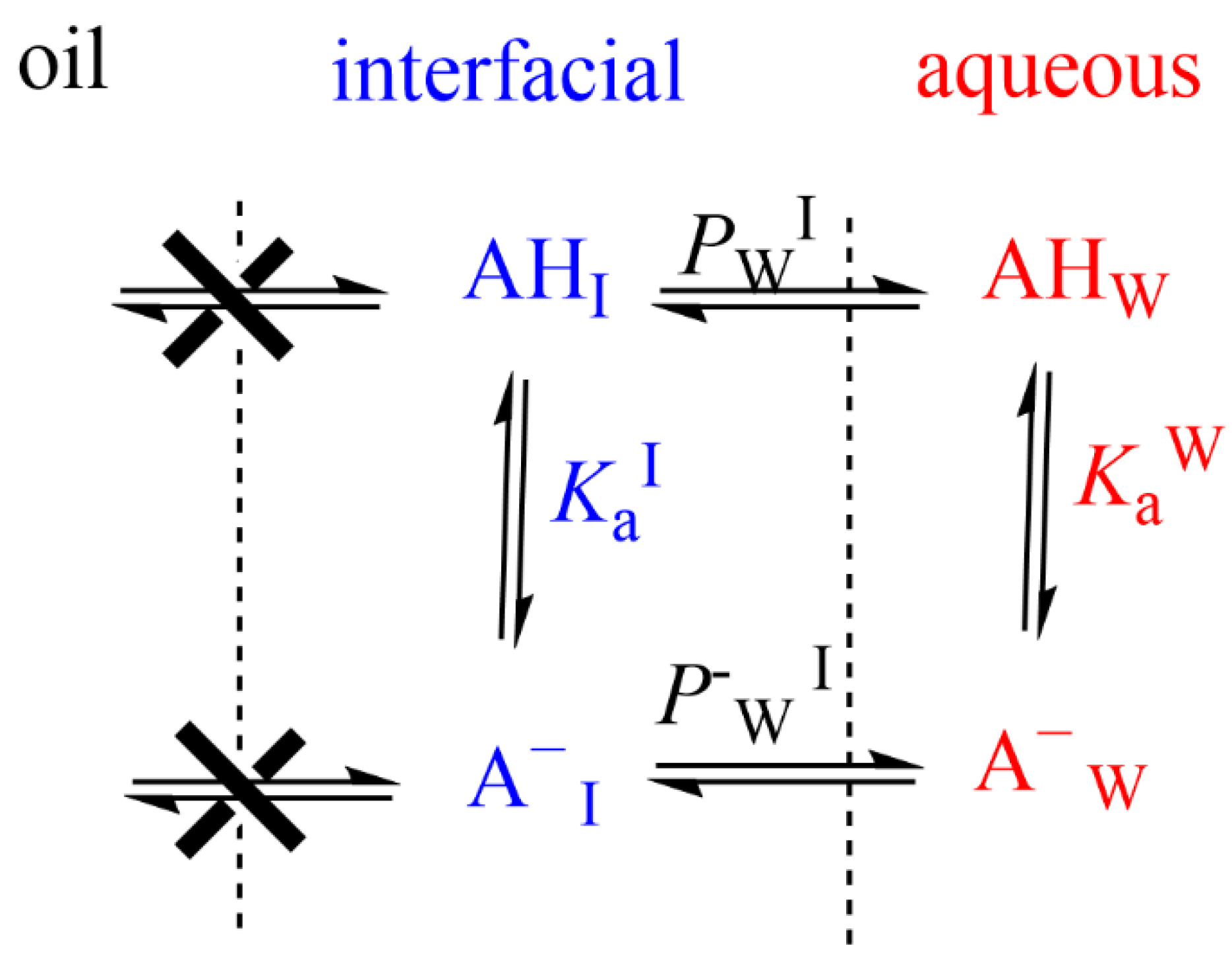
5.5. Modeling the pH-Dependent Partition Constants in Binary and Multiphasic Systems
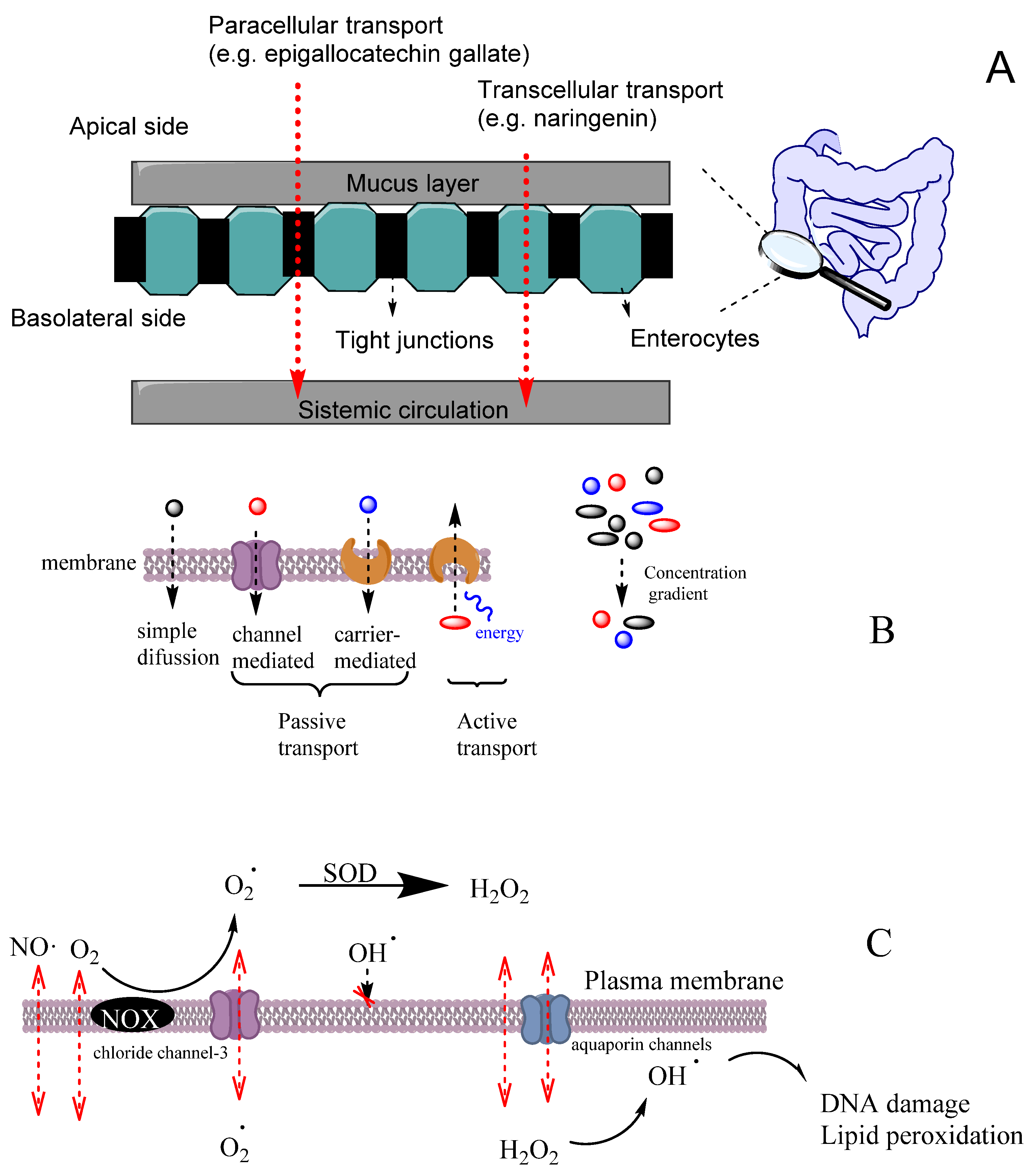
6. Diffusion and Transport of Reactive Species and Antioxidants
7. Bioactivity of Antioxidants: Mechanism(s) of Action
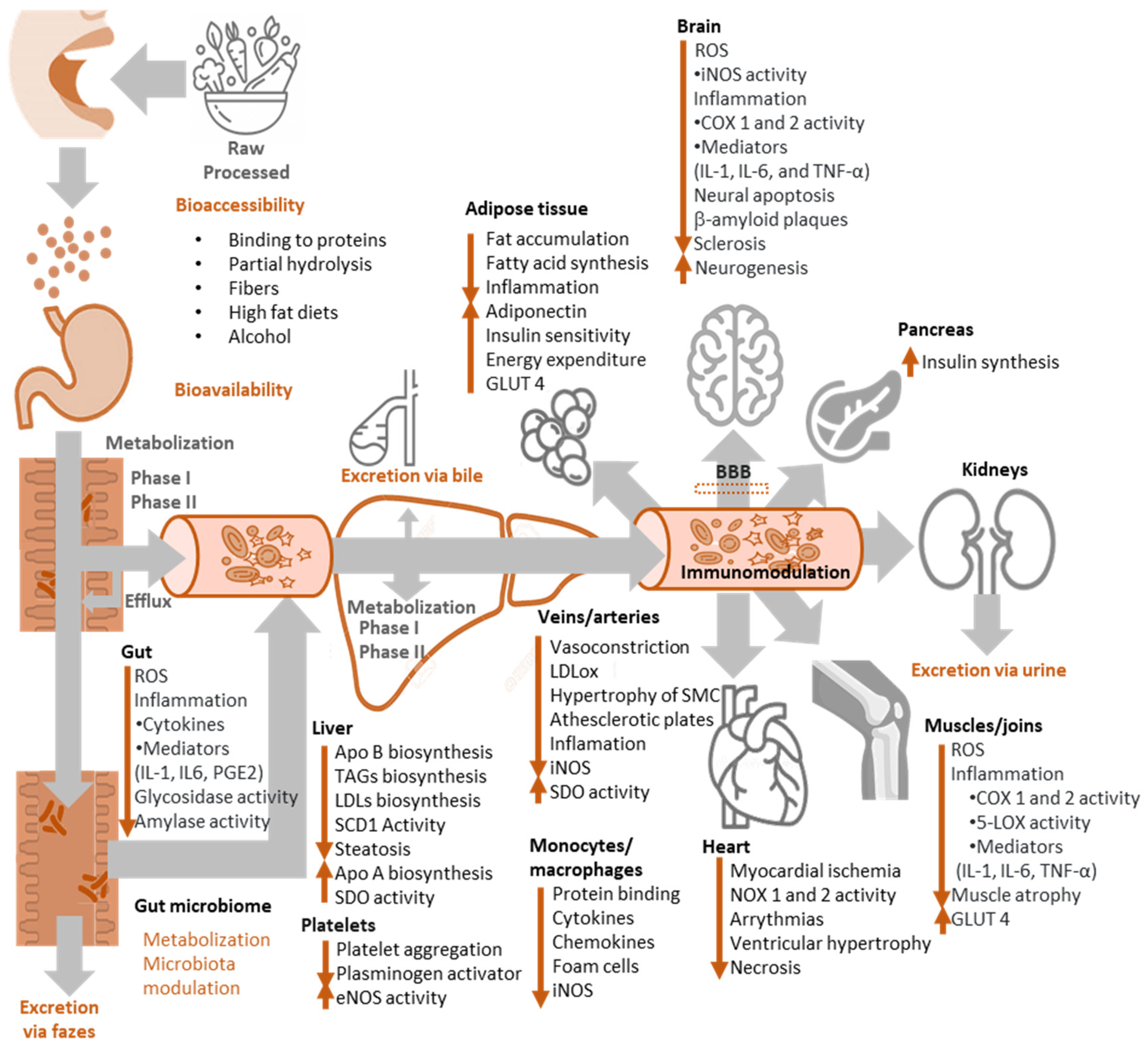
8. Bioavailability of Antioxidants
9. Nano Antioxidant Delivery Systems (NDDS): Antioxidant Bioavailability Improvement
9.1. Targeted Delivery of Antioxidants via NDDS
| NDDS | Advantages | Disadvantages |
|---|---|---|
| Nanoparticles: Spherical drug carriers fabricated from synthetic or natural components in the range of 10–1000 nm [113,114] |
|
|
| Liposomes: Nanosized (50–1000 nm) spherical vesicular drug delivery systems made of bilayered phospholipids in an aqueous medium [115] |
|
|
| Solid Lipid Nanoparticles: Nanocarriers (50–1000 nm) that are composed of solid lipids at body temperature. Drugs can be embedded to their core or attached to their surface [116] |
|
|
| Proliposomes: Provesicular systems in dry granular form that assemble liposomes upon hydration with water or body fluids. They are composed of carrier powder, phospholipids and cholesterol [117] |
|
|
| Polymeric micelles: Spherical nano-drug carriers, ranging in size from 10 to 100 nm and consist upon exposure of amphiphilic molecules to water [118] |
|
|
| Phytosomes: Liposome-like lipid-based NDDS manufactured by the interaction between plant extracts and phospholipids [119] |
|
|
9.2. Improved Bioavailability via NDDS
10. Antioxidants in Diseases: General Evidence
10.1. Effects on Cardiovascular Diseases, CVDs
10.2. Effects on Neurodegenerative Diseases
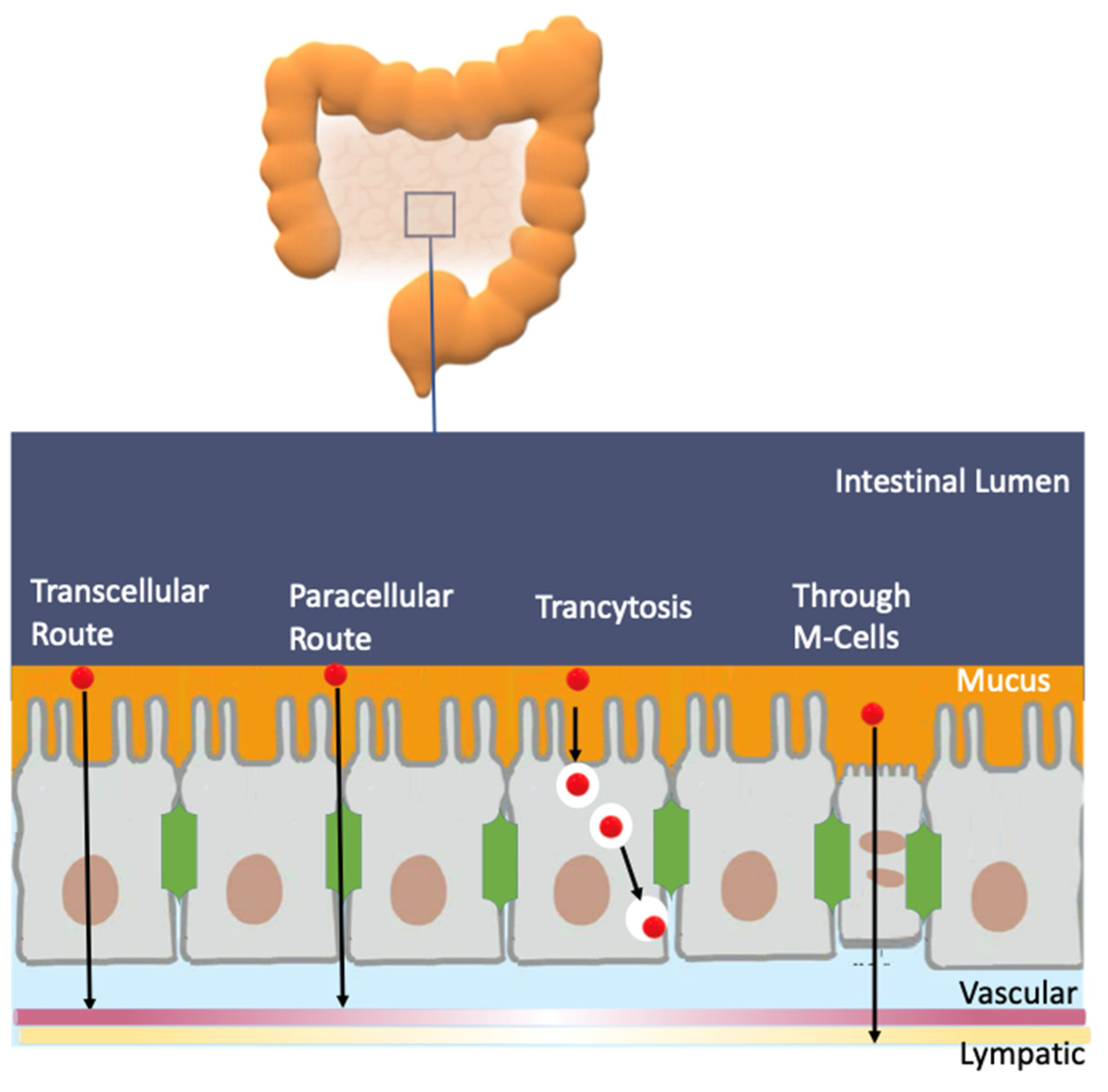
10.3. Effects on Digestive Diseases
10.4. Effects on Cancer
10.5. Effects on Inflammation
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative Stress: Harms and Benefits for Human Health. Oxid. Med. Cell. Longev. 2017, 2017, 8416763. [Google Scholar] [CrossRef]
- Forman, H.J.; Zhang, H. Targeting oxidative stress in disease: Promise and limitations of antioxidant therapy. Nat. Rev. Drug Discov. 2021, 20, 689–709. [Google Scholar] [CrossRef]
- Rudrapal, M.; Khairnar, S.J.; Khan, J.; Bin Dukhyil, A.; Ansari, M.A.; Alomary, M.N.; Alshabrmi, F.M.; Palai, S.; Deb, P.K.; Devi, R. Dietary Polyphenols and Their Role in Oxidative Stress-Induced Human Diseases: Insights into Protective Effects, Antioxidant Potentials and Mechanism(s) of Action. Front. Pharmacol. 2022, 13, 806470. [Google Scholar] [CrossRef]
- Kurutas, E.B. The importance of antioxidants which play the role in cellular response against oxidative/nitrosative stress: Current state. Nutr. J. 2016, 15, 71. [Google Scholar] [CrossRef]
- Adwas, A.A.; Elsayed, A.; Azab, A.E.; Quwaydir, F.A. Oxidative stress and antioxidant mechanisms in human body. J. Appl. Biotechnol. Bioeng. 2019, 6, 43–47. [Google Scholar] [CrossRef]
- Costa, M.; Losada-Barreiro, S.; Paiva-Martins, F.; Bravo-Díaz, C. Polyphenolic Antioxidants in Lipid Emulsions: Partitioning Effects and Interfacial Phenomena. Foods 2021, 10, 539. [Google Scholar] [CrossRef] [PubMed]
- Stromsnes, K.; Lagzdina, R.; Olaso-Gonzalez, G.; Gimeno-Mallench, L.; Gambini, J. Pharmacological Properties of Polyphenols: Bioavailability, Mechanisms of Action, and Biological Effects in In Vitro Studies, Animal Models, and Humans. Biomedicines 2021, 9, 1074. [Google Scholar] [CrossRef]
- Saso, L.; Firuzi, O. Pharmacological applications of antioxidants: Lights and shadows. Curr. Drug Targets 2014, 15, 1177–1199. [Google Scholar] [CrossRef] [PubMed]
- Schaich, K.M. CHAPTER 1—Challenges in Elucidating Lipid Oxidation Mechanisms: When, Where, and How Do Products Arise? In Lipid Oxidation; Logan, A., Nienaber, U., Pan, X., Eds.; AOCS Press: Champaign, IL, USA, 2013; pp. 1–52. [Google Scholar]
- Bravo-Díaz, C. Advances in the control of lipid peroxidation in oil-in-water emulsions: Kinetic approaches. Crit. Rev. Food Sci. Nutr. 2022, 1, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Schaich, K.M. Lipid Oxidation: Theoretical Aspects. In Bailey’s Industrial Oil and Fat Products; Shahidi, F., Ed.; John Wiley & Sons: New York, NY, USA, 2005; pp. 269–355. [Google Scholar]
- Schroën, K.; Berton-Carabin, C.C. A unifying approach to lipid oxidation in emulsions: Modelling and experimental validation. Food Res. Int. 2022, 160, 111621. [Google Scholar] [CrossRef] [PubMed]
- Hunyadi, A. The mechanism(s) of action of antioxidants: From scavenging reactive oxygen/nitrogen species to redox signaling and the generation of bioactive secondary metabolites. Med. Res. Rev. 2019, 39, 2505–2533. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, A.; Mondal, S.; Darbar, S.; Pal, S.K. Role of Nanomedicine in Redox Mediated Healing at Molecular Level. Biomol. Concepts 2019, 10, 160–174. [Google Scholar] [CrossRef] [PubMed]
- Bertelli, A.; Biagi, M.; Corsini, M.; Baini, G.; Cappellucci, G.; Miraldi, E. Polyphenols: From Theory to Practice. Foods 2021, 10, 2595. [Google Scholar] [CrossRef]
- Ighodaro, O.M.; Akinloye, O.A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Anil Kumar, N.V.; Zucca, P.; Varoni, E.M.; Dini, L.; Panzarini, E.; Rajkovic, J.; Tsouh Fokou, P.V.; Azzini, E.; Peluso, I.; et al. Lifestyle, Oxidative Stress, and Antioxidants: Back and Forth in the Pathophysiology of Chronic Diseases. Front. Physiol. 2020, 11, 694. [Google Scholar] [CrossRef] [PubMed]
- Sangouni, A.A.; Taghdir, M.; Mirahmadi, J.; Sepandi, M.; Parastouei, K. Effects of curcumin and/or coenzyme Q10 supplementation on metabolic control in subjects with metabolic syndrome: A randomized clinical trial. Nutr. J. 2022, 21, 62. [Google Scholar] [CrossRef]
- Iacobini, C.; Vitale, M.; Haxhi, J.; Pesce, C.; Pugliese, G.; Menini, S. Food-Related Carbonyl Stress in Cardiometabolic and Cancer Risk Linked to Unhealthy Modern Diet. Nutrients 2022, 14, 1061. [Google Scholar] [CrossRef]
- Burton-Freeman, B. Postprandial metabolic events and fruit-derived phenolics: A review of the science. Br. J. Nutr. 2010, 104, S1–S14. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, J.-L.; Zhou, Q. Targets and mechanisms of dietary anthocyanins to combat hyperglycemia and hyperuricemia: A comprehensive review. Crit. Rev. Food Sci. Nutr. 2022, 62, 1119–1143. [Google Scholar] [CrossRef]
- Nivoit, P.; Wiernsperger, N.; Moulin, P.; Lagarde, M.; Renaudin, C. Effect of glycated LDL on microvascular tone in mice: A comparative study with LDL modified in vitro or isolated from diabetic patients. Diabetologia 2003, 46, 1550–1558. [Google Scholar] [CrossRef]
- Diwadkar, V.A.; Anderson, J.W.; Bridges, S.R.; Gowri, M.S.; Oelgten, P.R. Postprandial Low-Density Lipoproteins in Type 2 Diabetes are Oxidized More Extensively Than Fasting Diabetes and Control Samples. Proc. Soc. Exp. Boil. Med. 1999, 222, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Marrocco, I.; Altieri, F.; Peluso, I. Measurement and Clinical Significance of Biomarkers of Oxidative Stress in Humans. Oxidative Med. Cell. Longev. 2017, 2017, 6501046. [Google Scholar] [CrossRef] [PubMed]
- Roy, J.; Galano, J.-M.; Durand, T.; Le Guennec, J.Y.; Lee, J.C.Y. Physiological role of reactive oxygen species as promoters of natural defenses. FASEB J. 2017, 31, 3729–3745. [Google Scholar] [CrossRef] [PubMed]
- Ristow, M.; Schmeisser, S. Extending life span by increasing oxidative stress. Free. Radic. Biol. Med. 2011, 51, 327–336. [Google Scholar] [CrossRef]
- Hackman, R.M.; Polagruto, J.A.; Zhu, Q.Y.; Sun, B.; Fujii, H.; Keen, C.L. Flavanols: Digestion, absorption and bioactivity. Phytochem. Rev. 2008, 7, 195–208. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects—A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Munekata, P.E.; Pateiro, M.; Zhang, W.; Dominguez, R.; Xing, L.; Fierro, E.M.; Lorenzo, J.M. Health benefits, extraction and development of functional foods with curcuminoids. J. Funct. Foods 2021, 79, 104392. [Google Scholar] [CrossRef]
- Costa, M.; Costa, V.; Lopes, M.; Paiva-Martins, F. A biochemical perspective on the fate of virgin olive oil phenolic compounds in vivo. Crit. Rev. Food Sci. Nutr. 2022, 12, 1–26. [Google Scholar] [CrossRef]
- Foti, M.C. Antioxidant properties of phenols. J. Pharm. Pharmacol. 2007, 59, 1673–1685. [Google Scholar] [CrossRef]
- Gülçin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef]
- Ross, L.; Barclay, C.; Vinqvist, M.R. Phenols as antioxidants, in The Chemistry of Phenols; Rappoport, Z., Ed.; John Wiley & Sons Ltd.: West Sussex, UK, 2003. [Google Scholar]
- Neves, A.R.; Nunes, C.; Reis, S. New Insights on the Biophysical Interaction of Resveratrol with Biomembrane Models: Relevance for Its Biological Effects. J. Phys. Chem. B 2015, 119, 11664–11672. [Google Scholar] [CrossRef] [PubMed]
- Meleleo, D. Study of Resveratrol’s Interaction with Planar Lipid Models: Insights into Its Location in Lipid Bilayers. Membranes 2021, 11, 132. [Google Scholar] [CrossRef] [PubMed]
- Košinová, P.; Berka, K.; Wykes, M.; Otyepka, M.; Trouillas, P. Positioning of Antioxidant Quercetin and Its Metabolites in Lipid Bilayer Membranes: Implication for Their Lipid-Peroxidation Inhibition. J. Phys. Chem. B 2012, 116, 1309–1318. [Google Scholar] [CrossRef] [PubMed]
- Karonen, M. Insights into Polyphenol–Lipid Interactions: Chemical Methods, Molecular Aspects and Their Effects on Membrane Structures. Plants 2022, 11, 1809. [Google Scholar] [CrossRef] [PubMed]
- Phan, H.T.; Yoda, T.; Chahal, B.; Morita, M.; Takagi, M.; Vestergaard, M.C. Structure-dependent interactions of polyphenols with a biomimetic membrane system. Biochim. Biophys. Acta (BBA) Biomembr. 2014, 1838, 2670–2677. [Google Scholar] [CrossRef] [PubMed]
- Lopes, R.; Costa, M.; Ferreira, M.; Gameiro, P.; Fernandes, S.; Catarino, C.; Santos-Silva, A.; Paiva-Martins, F. Caffeic acid phenolipids in the protection of cell membranes from oxidative injuries. Interaction with the membrane phospholipid bilayer. Biochim. Biophys. Acta (BBA) Biomembr. 2021, 1863, 183727. [Google Scholar] [CrossRef]
- Losada-Barreiro, S.; Bravo-Díaz, C.; Romsted, L.S. Distributions of phenolic acid antioxidants between the interfacial and aqueous regions of corn oil emulsions: Effects of pH and emulsifier concentration. Eur. J. Lipid Sci. Technol. 2015, 117, 1801–1813. [Google Scholar] [CrossRef]
- Galan, A.; Losada-Barreiro, S.; Bravo-Díaz, C. A Physicochemical Study of the Effects of Acidity on the Distribution and Antioxidant Efficiency of Trolox in Olive Oil-in-Water Emulsions. ChemPhysChem 2016, 17, 296–304. [Google Scholar] [CrossRef]
- Losada-Barreiro, S.; Sánchez-Paz, V.; Bravo-Díaz, C. Effects of emulsifier hydrophile-lipophile balance and emulsifier concentration on the distributions of gallic acid, propyl gallate, and α-tocopherol in corn oil emulsions. J. Colloid. Int. Sci. 2013, 389, 1–9. [Google Scholar] [CrossRef]
- Freiría-Gándara, J.; Losada-Barreiro, S.; Paiva-Martins, F.; Bravo-Díaz, C. Differential Partitioning of Bioantioxidants in Edible Oil–Water and Octanol–Water Systems: Linear Free Energy Relationships. J. Chem. Eng. Data 2018, 63, 2999–3007. [Google Scholar] [CrossRef]
- Jodko-Piórecka, K.; Cedrowski, J.; Litwinienko, G. Physico-chemical principles of antioxidant action, including solvent and matrix dependence and interfacial phenomena. In Measurement of Antioxidant Activity & Capacity; John Wiley & Sons, Inc.: New York, NY, USA, 2018; pp. 225–272. [Google Scholar]
- Litwinienko, G.; Ingold, K.U. Solvent Effects on the Rates and Mechanisms of Reaction of Phenols with Free Radicals. Acc. Chem. Res. 2007, 40, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Šegatin, N.; Žontar, T.P.; Ulrih, N.P. Dielectric Properties and Dipole Moment of Edible Oils Subjected to ‘Frying’ Thermal Treatment. Foods 2020, 9, 900. [Google Scholar] [CrossRef] [PubMed]
- Hopkins Hatzopoulos, M.; Eastoe, J.; Dowding, P.J.; Rogers, S.E.; Heenan, R.; Dyer, R. Are Hydrotropes Distinct from Surfactants? Langmuir 2011, 27, 12346–12353. [Google Scholar] [CrossRef] [PubMed]
- Andra, V.V.S.N.L.; Pammi, S.V.N.; Bhatraju, L.V.K.P.; Ruddaraju, L.K. A Comprehensive Review on Novel Liposomal Methodologies, Commercial Formulations, Clinical Trials and Patents. BioNanoScience 2022, 12, 274–291. [Google Scholar] [CrossRef] [PubMed]
- Pascual, G.-P. Plant Antioxidants in Food Emulsions. In Some New Aspects of Colloidal Systems in Foods; Jafar, M.M., Ed.; IntechOpen: Rijeka, Croatia, 2018; p. Ch. 2. [Google Scholar]
- McClements, D.J.; Jafari, S.M. Improving emulsion formation, stability and performance using mixed emulsifiers: A review. Adv. Colloid Interface Sci. 2018, 251, 55–79. [Google Scholar] [CrossRef]
- Sezgin-Bayindir, Z.; Losada-Barreiro, S.; Bravo-Díaz, C.; Sova, M.; Kristl, J.; Saso, L. Nanotechnology-Based Drug Delivery to Improve the Therapeutic Benefits of NRF2 Modulators in Cancer Therapy. Antioxidants 2021, 10, 685. [Google Scholar] [CrossRef]
- Martínez-Alonso, A.; Losada-Barreiro, S.; Bravo-Díaz, C. Encapsulation and solubilization of the antioxidants gallic acid and ethyl, propyl and butyl gallate with β-cyclodextrin. J. Mol. Liq. 2015, 210, 143–150. [Google Scholar] [CrossRef]
- Saenger, W.; Jacob, J.; Gessler, K.; Steiner, T.; Hoffmann, D.; Sanbe, H.; Koizumi, K.; Smith, A.S.M.; Takaha, T. Structures of the Common Cyclodextrins and Their Larger Analogues Beyond the Doughnut. Chem. Rev. 1998, 98, 1787–1802. [Google Scholar] [CrossRef]
- Connors, K.A. Measurement of Cyclodextrin Complex Stability Constants, in Comprehensive Supramolecular Chemistry; Szejtli, J., Osa, T., Eds.; Elsevier Sci. Ltd.: Cornwall, UK, 1996. [Google Scholar]
- Connors, K.A. The Stability of Cyclodextrin Complexes in Solution. Chem. Rev. 1997, 97, 1325–1358. [Google Scholar] [CrossRef]
- Lucarini, M.; Luppi, B.; Pedulli, G.F.; Roberts, B.P. Dynamic Aspects of Cyclodextrin Host-Guest inclusion as studied nu an EPR Spin-Probe Technique. Chem. Eur. J. 1999, 5, 2048. [Google Scholar] [CrossRef]
- García-Pérez, P.; Losada-Barreiro, S.; Gallego, P.P.; Bravo-Díaz, C. Cyclodextrin-Elicited Bryophyllum Suspension Cultured Cells: Enhancement of the Production of Bioactive Compounds. Int. J. Mol. Sci. 2019, 20, 5180. [Google Scholar] [CrossRef] [PubMed]
- Freiría-Gándara, J.; Bravo-Díaz, C. Effects of pH and surfactant concentration on the local concentrations of antioxidants in binary oil-water mixtures and in oil-in water emulsions. Bulg. Chem. Commun. 2018, 50, 260–267. [Google Scholar]
- Möller, M.N.; Cuevasanta, E.; Orrico, F.; Lopez, A.C.; Thomson, L.; Denicola, A. Diffusion and Transport of Reactive Species Across Cell Membranes. In Bioactive Lipids in Health and Disease; Trostchansky, A., Rubbo, H., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 3–19. [Google Scholar]
- Stillwell, W. Membrane Transport. In An Introduction to Biological Membranes, 2nd ed.; Stillwell, W., Ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 423–451. [Google Scholar] [CrossRef]
- Möller, M.N.; Cuevasanta, E.; Orrico, F.; Lopez, A.C.; Thomson, L.; Denicola, A. Diffusion and Transport of Reactive Species Across Cell Membranes. Adv. Exp. Med. Biol. 2019, 1127, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Stillwell, W. Chapter 9—Basic Membrane Properties of the Fluid Mosaic Model. In An Introduction to Biological Membranes; Stillwell, W., Ed.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 131–174. [Google Scholar] [CrossRef]
- Carecho, R.; Carregosa, D.; dos Santos, C.N. Low Molecular Weight (poly)Phenol Metabolites Across the Blood-Brain Barrier: The Underexplored Journey. Brain Plast. 2020, 6, 193–214. [Google Scholar] [CrossRef] [PubMed]
- Subczynski, W.K.; Hopwood, L.E.; Hyde, J.S. Is the mammalian cell plasma membrane a barrier to oxygen transport? J. Gen. Physiol. 1992, 100, 69–87. [Google Scholar] [CrossRef]
- Denicola, A.; Souza, J.M.; Radi, R.; Lissi, E. Nitric Oxide Diffusion in Membranes Determined by Fluorescence Quenching. Arch. Biochem. Biophys. 1996, 328, 208–212. [Google Scholar] [CrossRef]
- Signorelli, S.; Möller, M.N.; Coitiño, E.L.; Denicola, A. Nitrogen dioxide solubility and permeation in lipid membranes. Arch. Biochem. Biophys. 2011, 512, 190–196. [Google Scholar] [CrossRef]
- Makino, N.; Sasaki, K.; Hashida, K.; Sakakura, Y. A metabolic model describing the H2O2 elimination by mammalian cells including H2O2 permeation through cytoplasmic and peroxisomal membranes: Comparison with experimental data. Biochim. Biophys. Acta (BBA) Gen. Subj. 2004, 1673, 149–159. [Google Scholar] [CrossRef]
- Gus’Kova, R.A.; Ivanov, I.I.; Kol’Tover, V.K.; Akhobadze, V.V.; Rubin, A.B. Permeability of bilayer lipid membranes for superoxide (O2−) radicals. Biochim. Biophys. Acta (BBA) Biomembr. 1984, 778, 579–585. [Google Scholar] [CrossRef]
- Rekha, M.R.; Sharma, C.P. Chapter 8—Nanoparticle Mediated Oral Delivery of Peptides and Proteins: Challenges and Perspectives. In Peptide and Protein Delivery; Van Der Walle, C., Ed.; Academic Press: Boston, MA, USA, 2011; pp. 165–194. [Google Scholar]
- Cui, L.; Li, H.; Xi, Y.; Hu, Q.; Liu, H.; Fan, J.; Xiang, Y.; Zhang, X.; Shui, W.; Lai, Y. Vesicle trafficking and vesicle fusion: Mechanisms, biological functions, and their implications for potential disease therapy. Mol. Biomed. 2022, 3, 29. [Google Scholar] [CrossRef]
- Tellier, C.; Vallet-Strouve, C.; Akoka, S.; Poignant, S. Interactions between biliary lipid micelles and intestinal brush border membranes investigated by 1H and 31P nuclear magnetic resonance. Eur. Biophys. J. 1987, 15, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Gasperotti, M.; Passamonti, S.; Tramer, F.; Masuero, D.; Guella, G.; Mattivi, F.; Vrhovsek, U. Fate of microbial metabolites of dietary polyphenols in rats: Is the brain their target destination? ACS Chem. Neurosci. 2015, 6, 1341–1352. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Sun, P.; Lv, M.; Tong, G.; Jin, X.; Zhu, X. Mustard-inspired delivery shuttle for enhanced blood–brain barrier penetration and effective drug delivery in glioma therapy. Biomater. Sci. 2017, 5, 1041–1050. [Google Scholar] [CrossRef] [PubMed]
- Burton-Freeman, B.M.; Sesso, H.D. Whole Food versus Supplement: Comparing the Clinical Evidence of Tomato Intake and Lycopene Supplementation on Cardiovascular Risk Factors. Adv. Nutr. Int. Rev. J. 2014, 5, 457–485. [Google Scholar] [CrossRef] [PubMed]
- Palou, A.; Serra, F.; Pico, C. General aspects on the assessment of functional foods in the European Union. Eur. J. Clin. Nutr. 2003, 57, S12–S17. [Google Scholar] [CrossRef][Green Version]
- Rahman, M.M.; Rahaman, M.S. Role of Phenolic Compounds in Human Disease: Current Knowledge and Future Prospects. Molecules 2021, 27, 233. [Google Scholar] [CrossRef]
- Galleano, M.; Verstraeten, S.V.; Oteiza, P.I.; Fraga, C.G. Antioxidant actions of flavonoids: Thermodynamic and kinetic analysis. Arch. Biochem. Biophys. 2010, 501, 23–30. [Google Scholar] [CrossRef]
- Kostyuk, V.; Potapovich, A.; Strigunova, E.; Kostyuk, T.; Afanas’Ev, I. Experimental evidence that flavonoid metal complexes may act as mimics of superoxide dismutase. Arch. Biochem. Biophys. 2004, 428, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Yuting, C.; Rongliang, Z.; Zhongjian, J.; Yong, J. Flavonoids as superoxide scavengers and antioxidants. Free. Radic. Biol. Med. 1990, 9, 19–21. [Google Scholar] [CrossRef]
- Cos, P.; Ying, L.; Calomme, M.; Hu, J.P.; Cimanga, K.; Van Poel, B.; Pieters, L.; Vlietinck, A.A.J.; Berghe, D.V. Structure−Activity Relationship and Classification of Flavonoids as Inhibitors of Xanthine Oxidase and Superoxide Scavengers. J. Nat. Prod. 1998, 61, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.-Z.; Deng, G.; Zhang, Y.-C. Multiple free radical scavenging reactions of flavonoids. Dye. Pigment. 2022, 198, 109877. [Google Scholar] [CrossRef]
- Yin, Z.; Wu, Y.; Chen, Y.; Qie, X.; Zeng, M.; Wang, Z.; Qin, F.; Chen, J.; He, Z. Analysis of the interaction between cyanidin-3-O-glucoside and casein hydrolysates and its effect on the antioxidant ability of the complexes. Food Chem. 2020, 340, 127915. [Google Scholar] [CrossRef] [PubMed]
- Bors, W.; Heller, W.; Michel, C.; Saran, M. Flavonoids as antioxidants: Determination of radical-scavenging efficiencies. Methods Enzymol. 1990, 186, 343–355. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Li, L.; Chen, W.; Xu, S.; Feng, X.; Zhang, L. Natural products: The role and mechanism in low-density lipoprotein oxidation and atherosclerosis. Phytother. Res. 2021, 35, 2945–2967. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, S.; Ribeiro, C.; Paiva-Martins, F.; Catarino, C.; Santos-Silva, A. Protective effect of olive oil polyphenol phase II sulfate conjugates on erythrocyte oxidative-induced hemolysis. Food Funct. 2020, 11, 8670–8679. [Google Scholar] [CrossRef] [PubMed]
- Weinbrenner, T.; Fitó, M.; Albaladejo, M.F.; Sáez, G.; Rijken, P.; Tormos, C.; Coolen, S.; de la Torre, R.; Covas, M.I. Bioavailability of phenolic compounds from olive oil and oxidative/antioxidant status at postprandial state in healthy humans. Drugs Under Exp. Clin. Res. 2004, 30, 207–212. [Google Scholar]
- López-Yerena, A.; Vallverdú-Queralt, A.; Lamuela-Raventós, R.; Escribano-Ferrer, E. LC-ESI-LTQ-Orbitrap-MS for Profiling the Distribution of Oleacein and Its Metabolites in Rat Tissues. Antioxidants 2021, 10, 1083. [Google Scholar] [CrossRef]
- Paiva-Martins, F.; Fernandes, J.; Rocha, S.; Nascimento, H.; Vitorino, R.; Amado, F.; Borges, F.; Belo, L.; Santos-Silva, A. Effects of olive oil polyphenols on erythrocyte oxidative damage. Mol. Nutr. Food Res. 2009, 53, 609–616. [Google Scholar] [CrossRef]
- Paiva-Martins, F.; Silva, A.; Almeida, V.; Carvalheira, M.; Serra, C.; Rodrígues-Borges, J.E.; Fernandes, J.; Belo, L.; Santos-Silva, A. Protective Activity of Hydroxytyrosol Metabolites on Erythrocyte Oxidative-Induced Hemolysis. J. Agric. Food Chem. 2013, 61, 6636–6642. [Google Scholar] [CrossRef]
- Lotito, S.B.; Frei, B. The increase in human plasma antioxidant capacity after apple consumption is due to the metabolic effect of fructose on urate, not apple-derived antioxidant flavonoids. Free Radic. Biol. Med. 2004, 37, 251–258. [Google Scholar] [CrossRef]
- Ursini, F.; Sevanian, A. Wine Polyphenols and Optimal Nutrition. Ann. New York Acad. Sci. 2002, 957, 200–209. [Google Scholar] [CrossRef]
- Ruskovska, T.; Budić-Leto, I.; Corral-Jara, K.F.; Ajdžanović, V.; Arola-Arnal, A.; Bravo, F.I.; Deligiannidou, G.-E.; Havlik, J.; Janeva, M.; Kistanova, E.; et al. Systematic analysis of nutrigenomic effects of polyphenols related to cardiometabolic health in humans—Evidence from untargeted mRNA and miRNA studies. Ageing Res. Rev. 2022, 79, 101649. [Google Scholar] [CrossRef]
- Rubió, L.; Farràs, M.; de La Torre, R.; Macià, A.; Romero, M.-P.; Valls, R.M.; Solà, R.; Farré, M.; Fitó, M.; Motilva, M.-J. Metabolite profiling of olive oil and thyme phenols after a sustained intake of two phenol-enriched olive oils by humans: Identification of compliance markers. Food Res. Int. 2014, 65, 59–68. [Google Scholar] [CrossRef]
- Scalbert, A.; Brennan, L.; Manach, C.; Andres-Lacueva, C.; Dragsted, L.O.; Draper, J.; Rappaport, S.M.; van der Hooft, J.J.; Wishart, D.S. The food metabolome: A window over dietary exposure. Am. J. Clin. Nutr. 2014, 99, 1286–1308. [Google Scholar] [CrossRef] [PubMed]
- Kay, C.D.; Pereira-Caro, G.; Ludwig, I.A.; Clifford, M.N.; Crozier, A. Anthocyanins and Flavanones Are More Bioavailable than Previously Perceived: A Review of Recent Evidence. Annu. Rev. Food Sci. Technol. 2017, 8, 155–180. [Google Scholar] [CrossRef] [PubMed]
- Gee, J.M.; DuPont, M.S.; Day, A.J.; Plumb, G.W.; Williamson, G.; Johnson, I.T. Intestinal Transport of Quercetin Glycosides in Rats Involves Both Deglycosylation and Interaction with the Hexose Transport Pathway. J. Nutr. 2000, 130, 2765–2771. [Google Scholar] [CrossRef] [PubMed]
- Pinto, J.; Paiva-Martins, F.; Corona, G.; Debnam, E.S.; Jose Oruna-Concha, M.; Vauzour, D.; Gordon, M.H.; Spencer, J.P.E. Absorption and metabolism of olive oil secoiridoids in the small intestine. Br. J. Nutr. 2011, 105, 1607–1618. [Google Scholar] [CrossRef]
- Serreli, G.; Deiana, M. Biological Relevance of Extra Virgin Olive Oil Polyphenols Metabolites. Antioxidants 2018, 7, 170. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G.; Aeberli, I.; Miguet, L.; Zhang, Z.; Sanchez, M.-B.; Crespy, V.; Barron, D.; Needs, P.; Kroon, P.A.; Glavinas, H.; et al. Interaction of Positional Isomers of Quercetin Glucuronides with the Transporter ABCC2 (cMOAT, MRP2). Drug Metab. Dispos. 2007, 35, 1262–1268. [Google Scholar] [CrossRef]
- Williamson, G.; Kay, C.D.; Crozier, A. The Bioavailability, Transport, and Bioactivity of Dietary Flavonoids: A Review from a Historical Perspective. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1054–1112. [Google Scholar] [CrossRef]
- Patel, K.R.; Andreadi, C.; Britton, R.G.; Horner-Glister, E.; Karmokar, A.; Sale, S.; Brown, V.A.; Brenner, D.E.; Singh, R.; Steward, W.P.; et al. Sulfate Metabolites Provide an Intracellular Pool for Resveratrol Generation and Induce Autophagy with Senescence. Sci. Transl. Med. 2013, 5, 205ra133. [Google Scholar] [CrossRef] [PubMed]
- Oteiza, P.; Fraga, C.; Mills, D.; Taft, D. Flavonoids and the gastrointestinal tract: Local and systemic effects. Mol. Asp. Med. 2018, 61, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Caro, G.; Ludwig, I.A.; Polyviou, T.; Malkova, D.; García, A.; Moreno-Rojas, J.M.; Crozier, A. Identification of Plasma and Urinary Metabolites and Catabolites Derived from Orange Juice (Poly)phenols: Analysis by High-Performance Liquid Chromatography–High-Resolution Mass Spectrometry. J. Agric. Food Chem. 2016, 64, 5724–5735. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, C.; Marzorati, M.; Daghio, M.; Franzetti, A.; Innocenti, M.; Van de Wiele, T.; Mulinacci, N. Effects of Olive and Pomegranate By-Products on Human Microbiota: A Study Using the SHIME® In Vitro Simulator. Molecules 2019, 24, 3791. [Google Scholar] [CrossRef]
- Kim, M.T.; Sedykh, A.; Chakravarti, S.K.; Saiakhov, R.D.; Zhu, H. Critical Evaluation of Human Oral Bioavailability for Pharmaceutical Drugs by Using Various Cheminformatics Approaches. Pharm. Res. 2014, 31, 1002–1014. [Google Scholar] [CrossRef]
- Gadekar, V.; Borade, Y.; Kannaujia, S.; Rajpoot, K.; Anup, N.; Tambe, V.; Kalia, K.; Tekade, R.K. Nanomedicines accessible in the market for clinical interventions. J. Control. Release 2021, 330, 372–397. [Google Scholar] [CrossRef]
- Germain, M.; Caputo, F.; Metcalfe, S.; Tosi, G.; Spring, K.; Åslund, A.K.; Pottier, A.; Schiffelers, R.; Ceccaldi, A.; Schmid, R. Delivering the power of nanomedicine to patients today. J. Control. Release 2020, 326, 164–171. [Google Scholar] [CrossRef]
- Ratnam, D.V.; Ankola, D.; Bhardwaj, V.; Sahana, D.; Kumar, M.R. Role of antioxidants in prophylaxis and therapy: A pharmaceutical perspective. J. Control. Release 2006, 113, 189–207. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; del Pilar Rodriguez-Torres, M.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef]
- Weiser, J.R.; Saltzman, W.M. Controlled release for local delivery of drugs: Barriers and models. J. Control. Release 2014, 190, 664–673. [Google Scholar] [CrossRef] [PubMed]
- Pattni, B.S.; Torchilin, V.P. Targeted Drug Delivery Systems: Strategies and Challenges. In Targeted Drug Delivery: Concepts and Design; Devarajan, P.V., Jain, S., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 3–38. [Google Scholar]
- Rabanel, J.-M.; Aoun, V.; Elkin, I.; Mokhtar, M.; Hildgen, P. Drug-Loaded Nanocarriers: Passive Targeting and Crossing of Biological Barriers. Curr. Med. Chem. 2012, 19, 3070–3102. [Google Scholar] [CrossRef] [PubMed]
- Delshadi, R.; Bahrami, A.; McClements, D.J.; Moore, M.D.; Williams, L. Development of nanoparticle-delivery systems for antiviral agents: A review. J. Control. Release 2021, 331, 30–44. [Google Scholar] [CrossRef]
- Dahiya, R.; Dahiya, S. Chapter 13—Advanced drug delivery applications of self-assembled nanostructures and polymeric nanoparticles. In Handbook on Nanobiomaterials for Therapeutics and Diagnostic Applications; Anand, K., Ed.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 297–339. [Google Scholar]
- Malam, Y.; Loizidou, M.; Seifalian, A.M. Liposomes and nanoparticles: Nanosized vehicles for drug delivery in cancer. Trends Pharmacol. Sci. 2009, 30, 592–599. [Google Scholar] [CrossRef]
- Montoto, S.S.; Muraca, G.; Ruiz, M.E. Solid Lipid Nanoparticles for Drug Delivery: Pharmacological and Biopharmaceutical Aspects. Front. Mol. Biosci. 2020, 7, 587997. [Google Scholar] [CrossRef] [PubMed]
- Bayindir, Z.S.; Yuksel, N. Provesicles as novel drug delivery systems. Curr. Pharm. Biotechnol. 2015, 16, 344–364. [Google Scholar] [CrossRef] [PubMed]
- Sezgin, Z.; Yuksel, N.; Baykara, T. Preparation and characterization of polymeric micelles for solubilization of poorly soluble anticancer drugs. Eur. J. Pharm. Biopharm. 2006, 64, 261–268. [Google Scholar] [CrossRef]
- Barani, M.; Sangiovanni, E.; Angarano, M. Phytosomes as Innovative Delivery Systems for Phytochemicals: A Comprehensive Review of Literature. Int. J. Nanomed. 2021, 16, 6983–7022. [Google Scholar] [CrossRef]
- Danhier, F.; Feron, O.; Préat, V. To exploit the tumor microenvironment: Passive and active tumor targeting of nanocarriers for anti-cancer drug delivery. J. Control. Release 2010, 148, 135–146. [Google Scholar] [CrossRef]
- Basile, L.; Pignatello, R.; Passirani, C. Active targeting strategies for anticancer drug nanocarriers. Curr. Drug Deliv. 2012, 9, 255–268. [Google Scholar] [CrossRef]
- Yu, M.K.; Park, J.; Jon, S. Targeting Strategies for Multifunctional Nanoparticles in Cancer Imaging and Therapy. Theranostics 2012, 2, 3–44. [Google Scholar] [CrossRef]
- Attia, M.F.; Anton, N.; Wallyn, J.; Omran, Z.; Vandamme, T.F. An overview of active and passive targeting strategies to improve the nanocarriers efficiency to tumour sites. J. Pharm. Pharmacol. 2019, 71, 1185–1198. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.; Ramsey, J.D.; Kabanov, A.V. Polymeric micelles for the delivery of poorly soluble drugs: From nanoformulation to clinical approval. Adv. Drug Deliv. Rev. 2020, 156, 80–118. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Ma, C.; Yang, X.; Wang, J.; Long, G.; Zhou, J. Phytonanomaterials as therapeutic agents and drug delivery carriers. Adv. Drug Deliv. Rev. 2021, 176, 113868. [Google Scholar] [CrossRef]
- Zhang, S.; Peng, B.; Chen, Z.; Yu, J.; Deng, G.; Bao, Y.; Ma, C.; Du, F.; Sheu, W.C.; Kimberly, W.T.; et al. Brain-targeting, acid-responsive antioxidant nanoparticles for stroke treatment and drug delivery. Bioact. Mater. 2022, 16, 57–65. [Google Scholar] [CrossRef]
- Li, S.; Lv, H.; Chen, Y.; Song, H.; Zhang, Y.; Wang, S.; Luo, L.; Guan, X. N-trimethyl chitosan coated targeting nanoparticles improve the oral bioavailability and antioxidant activity of vitexin. Carbohydr. Polym. 2022, 286, 119273. [Google Scholar] [CrossRef]
- Poonia, N.; Lather, V.; Narang, J.K.; Beg, S.; Pandita, D. Resveratrol-loaded folate targeted lipoprotein-mimetic nanoparticles with improved cytotoxicity, antioxidant activity and pharmacokinetic profile. Mater. Sci. Eng. C 2020, 114, 111016. [Google Scholar] [CrossRef]
- Komeil, I.A.; Abdallah, O.Y.; El-Refaie, W.M. Surface modified genistein phytosome for breast cancer treatment: In-vitro appraisal, pharmacokinetics, and in-vivo antitumor efficacy. Eur. J. Pharm. Sci. 2022, 179, 106297. [Google Scholar] [CrossRef]
- Tian, J.-Y.; Chi, C.-L.; Bian, G.; Xing, D.; Guo, F.-J.; Wang, X.-Q. PSMA conjugated combinatorial liposomal formulation encapsulating genistein and plumbagin to induce apoptosis in prostate cancer cells. Colloids Surf. B Biointerfaces 2021, 203, 111723. [Google Scholar] [CrossRef] [PubMed]
- Binu, N.M.; Prema, D.; Prakash, J.; Balagangadharan, K.; Balashanmugam, P.; Selvamurugan, N.; Venkatasubbu, G.D. Folic acid decorated pH sensitive polydopamine coated honeycomb structured nickel oxide nanoparticles for targeted delivery of quercetin to triple negative breast cancer cells. Colloids Surf. A Physicochem. Eng. Asp. 2021, 630, 127609. [Google Scholar] [CrossRef]
- Zhang, J.; Nie, S.; Zu, Y.; Abbasi, M.; Cao, J.; Li, C.; Wu, D.; Labib, S.; Brackee, G.; Shen, C.-L.; et al. Anti-atherogenic effects of CD36-targeted epigallocatechin gallate-loaded nanoparticles. J. Control. Release 2019, 303, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Bhattacherjee, A.; Chakraborti, A.S. Argpyrimidine-tagged rutin-encapsulated biocompatible (ethylene glycol dimers) nanoparticles: Application for targeted drug delivery in experimental diabetes (Part 2). Int. J. Pharm. 2017, 528, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, Y.; Wada, K.; Nakatani, M.; Yamada, S.; Onoue, S. Formulation design for poorly water-soluble drugs based on biopharmaceutics classification system: Basic approaches and practical applications. Int. J. Pharm. 2011, 420, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wu, W. Ligand-mediated active targeting for enhanced oral absorption. Drug Discov. Today 2014, 19, 898–904. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Park, K. Polymeric micelles and alternative nanonized delivery vehicles for poorly soluble drugs. Int. J. Pharm. 2013, 453, 198–214. [Google Scholar] [CrossRef]
- Nishikawa, M.; Hasegawa, S.; Yamashita, F.; Takakura, Y.; Hashida, M. Electrical charge on protein regulates its absorption from the rat small intestine. Am. J. Physiol. Liver Physiol. 2002, 282, G711–G719. [Google Scholar] [CrossRef][Green Version]
- Reinholz, J.; Landfester, K.; Mailänder, V. The challenges of oral drug delivery via nanocarriers. Drug Deliv. 2018, 25, 1694–1705. [Google Scholar] [CrossRef]
- Pan-On, S.; Dilokthornsakul, P.; Tiyaboonchai, W. Trends in advanced oral drug delivery system for curcumin: A systematic review. J. Control. Release 2022, 348, 335–345. [Google Scholar] [CrossRef]
- Ezzat, H.M.; Elnaggar, Y.S.R.; Abdallah, O.Y. Improved oral bioavailability of the anticancer drug catechin using chitosomes: Design, in-vitro appraisal and in-vivo studies. Int. J. Pharm. 2019, 565, 488–498. [Google Scholar] [CrossRef]
- Saad, S.; Ahmad, I.; Kawish, S.M.; Khan, U.A.; Ahmad, F.J.; Ali, A.; Jain, G.K. Improved cardioprotective effects of hesperidin solid lipid nanoparticles prepared by supercritical antisolvent technology. Colloids Surf. B Biointerfaces 2020, 187, 110628. [Google Scholar] [CrossRef]
- Wang, Q.; Wei, C.; Weng, W.; Bao, R.; Adu-Frimpong, M.; Toreniyazov, E.; Ji, H.; Xu, X.-M.; Yu, J. Enhancement of oral bioavailability and hypoglycemic activity of liquiritin-loaded precursor liposome. Int. J. Pharm. 2021, 592, 120036. [Google Scholar] [CrossRef]
- Duong, T.T.; Yen, T.T.H.; Nguyen, L.T.; Nguyen, T.-D.; Nguyen, T.-Q.; Nghiem, T.-H.; Pham, H.T.; Raal, A.; Heinämäki, J.; Pham, T.-M. Berberine-loaded liposomes for oral delivery: Preparation, physicochemical characterization and in-vivo evaluation in an endogenous hyperlipidemic animal model. Int. J. Pharm. 2022, 616, 121525. [Google Scholar] [CrossRef]
- Thant, Y.; Wang, Q.; Wei, C.; Liu, J.; Zhang, K.; Bao, R.; Zhu, Q.; Weng, W.; Yu, Q.; Zhu, Y.; et al. TPGS conjugated pro-liposomal nano-drug delivery system potentiate the antioxidant and hepatoprotective activity of Myricetin. J. Drug Deliv. Sci. Technol. 2021, 66, 102808. [Google Scholar] [CrossRef]
- Behl, T.; Bungau, S.; Kumar, K.; Zengin, G.; Khan, F.; Kumar, A.; Kaur, R.; Venkatachalam, T.; Tit, D.M.; Vesa, C.M.; et al. Pleotropic Effects of Polyphenols in Cardiovascular System. Biomed. Pharmacother. 2020, 130, 110714. [Google Scholar] [CrossRef]
- Calabriso, N.; Gnoni, A.; Stanca, E.; Cavallo, A.; Damiano, F.; Siculella, L.; Carluccio, M.A. Hydroxytyrosol Ameliorates Endothelial Function under Inflammatory Conditions by Preventing Mitochondrial Dysfunction. Oxidat. Med. Cell. Longev. 2018, 2018, 9086947. [Google Scholar] [CrossRef]
- Song, J.; Huang, Y.; Zheng, W.; Yan, J.; Cheng, M.; Zhao, R.; Jia, W. Resveratrol reduces intracellular reactive oxygen species levels by inducing autophagy through the AMPK-mTOR pathway. Front. Med. 2018, 12, 697–706. [Google Scholar] [CrossRef] [PubMed]
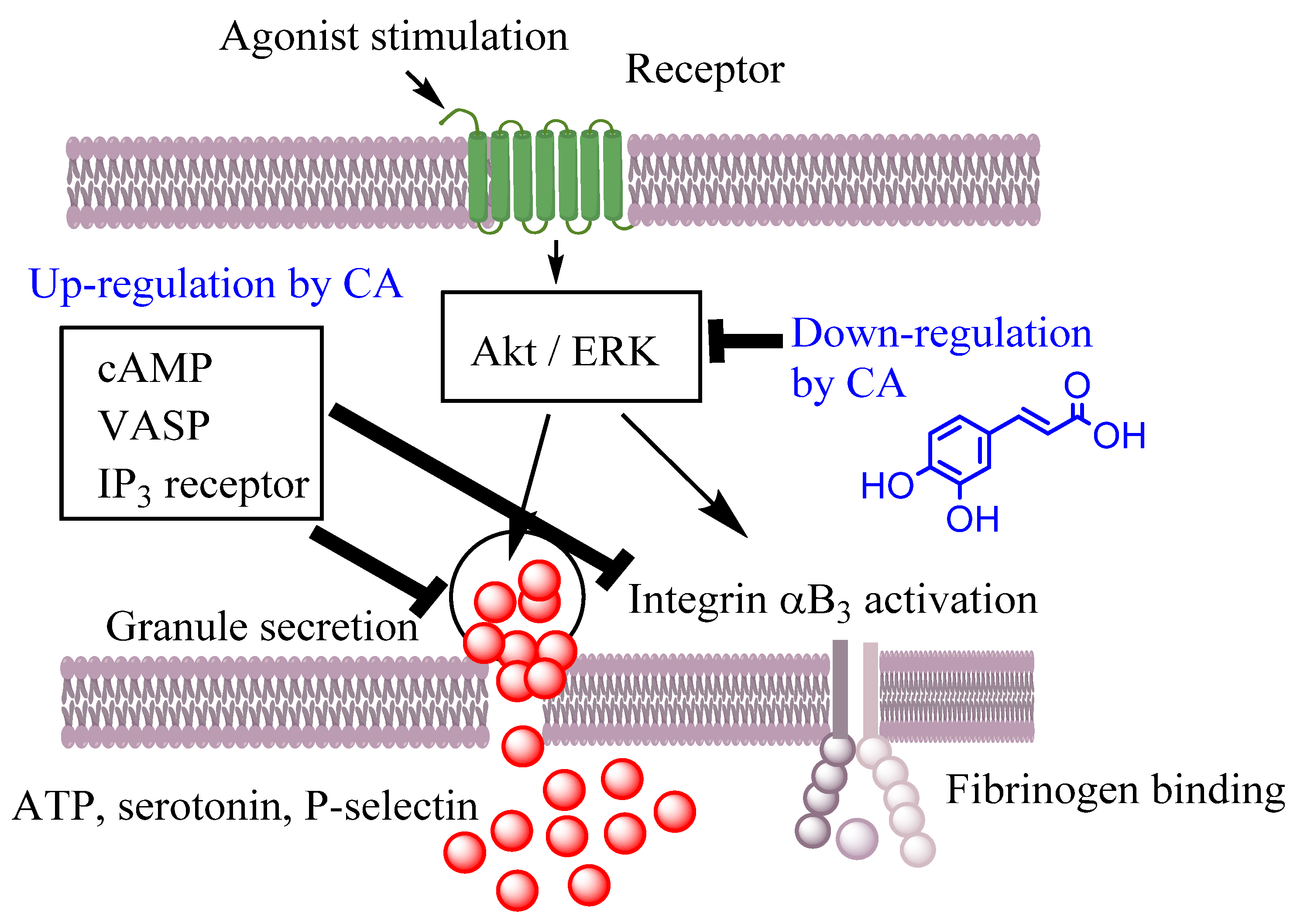
- Nam, G.S.; Park, H.J.; Nam, K.S. The antithrombotic effect of caffeic acid is associated with a cAMP-dependent pathway and clot retraction in human platelets. Thromb. Res. 2020, 195, 87–94. [Google Scholar] [CrossRef]
- Cai, Y.; Yu, S.S.; He, Y.; Bi, X.Y.; Gao, S.; Yan, T.D.; Liu, P.Q. EGCG inhibits pressure overload-induced cardiac hypertrophy via the PSMB5/Nmnat2/SIRT6-dependent signalling pathways. Acta Physiol. 2020, 231, e13602–e13619. [Google Scholar] [CrossRef] [PubMed]
- Safarpour, S.; Pirzadeh, M.; Ebrahimpour, A.; Shirafkan, F.; Madani, F.; Hosseini, M.; Moghadamnia, A.A.; Kazemi, S. Protective Effect of Kaempferol and Its Nanoparticles on 5-Fluorouracil-Induced Cardiotoxicity in Rats. BioMed. Res. Int. 2022, 2022, 2273000. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Liu, S.; Tang, H.; Song, S.; Lu, L.; Zhang, P.; Li, X. Effects of protocatechuic acid on ameliorating lipid profiles and cardio-protection against coronary artery disease in high fat and fructose diet fed in rats. J. Vet. Med. Sci. 2020, 82, 1387–1394. [Google Scholar] [CrossRef]
- Jin, L.; Piao, Z.H.; Sun, S.; Liu, B.; Kim, G.R.; Seok, Y.M.; Lin, M.Q.; Ryu, Y.; Choi, S.Y.; Kee, H.J.; et al. Gallic Acid Reduces Blood Pressure and Attenuates Oxidative Stress and Cardiac Hypertrophy in Spontaneously Hypertensive Rats. Sci. Rep. 2017, 7, 15607. [Google Scholar] [CrossRef]
- Kannan, M.M.; Quine, S.D. Ellagic acid inhibits cardiac arrhythmias, hypertrophy and hyperlipidaemia during myocardial infarction in rats. Metabolism 2013, 62, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Li, F.; Zhang, X.; Wang, W.; Shao, J.; Zheng, Y. Delphinidin-3-O-glucoside, an active compound of Hibiscus sabdariffa calyces, inhibits oxidative stress and inflammation in rabbits with atherosclerosis. Pharm. Biol. 2022, 60, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Quirós-Fernández, R.; López-Plaza, B.; Bermejo, L.M.; Palma-Milla, S.; Gómez-Candela, C. Supplementation with Hydroxytyrosol and Punicalagin Improves Early Atherosclerosis Markers Involved in the Asymptomatic Phase of Atherosclerosis in the Adult Population: A Randomized, Placebo-Controlled, Crossover Trial. Nutrients 2019, 11, 640. [Google Scholar] [CrossRef] [PubMed]
- Salazar, H.M.; Mendonça, R.D.D.; Laclaustra, M.; Moreno-Franco, B.; Åkesson, A.; Guallar-Castillón, P.; Donat-Vargas, C. The intake of flavonoids, stilbenes, and tyrosols, mainly consumed through red wine and virgin olive oil, is associated with lower carotid and femoral subclinical atherosclerosis and coronary calcium. Eur. J. Nutr. 2022, 61, 2697–2709. [Google Scholar] [CrossRef]
- Salehi, B.; Stojanović-Radić, Z.; Matejić, J.; Sharifi-Rad, M.; Anil Kumar, N.V.; Martins, N.; Sharifi-Rad, J. The therapeutic potential of curcumin: A review of clinical trials. Eur. J. Med. Chem. 2019, 163, 527–545. [Google Scholar] [CrossRef]
- DiSilvestro, R.A.; Joseph, E.; Zhao, S.; Bomser, J. Diverse effects of a low dose supplement of lipidated curcumin in healthy middle aged people. Nutr. J. 2012, 11, 79. [Google Scholar] [CrossRef] [PubMed]
- Di Meo, F.; Valentino, A.; Petillo, O.; Peluso, G.; Filosa, S.; Crispi, S. Bioactive Polyphenols and Neuromodulation: Molecular Mechanisms in Neurodegeneration. Int. J. Mol. Sci. 2020, 21, 2564. [Google Scholar] [CrossRef]
- Rahman, M.; Akter, R.; Bhattacharya, T.; Abdel-Daim, M.M.; Alkahtani, S.; Arafah, M.W.; Mittal, V. Resveratrol and Neuroprotection: Impact and Its Therapeutic Potential in Alzheimer’s Disease. Front. Pharmacol. 2020, 30, 2272. [Google Scholar] [CrossRef]
- Shen, D.; Sang, W.; Song, R.; Tian, X. Effect of Melatonin and Resveratrol against Memory Impairment and Hippocampal Damage in a Rat Model of Vascular Dementia. Neuroimmunomodulation 2016, 23, 318–331. [Google Scholar] [CrossRef]
- Rezai-Zadeh, K.; Arendash, G.W.; Hou, H.; Fernandez, F.; Jensen, M.; Runfeldt, M.; Shytle, R.D.; Tan, J. Green tea epigallocatechin-3-gallate (EGCG) reduces β-amyloid mediated cognitive impairment and modulates tau pathology in Alzheimer transgenic mice. Brain Res. 2008, 1214, 177–187. [Google Scholar] [CrossRef]
- Evin, G.; Barakat, A.; Masters, C.L. BACE: Therapeutic target and potential biomarker for Alzheimer’s disease. Int. J. Biochem. Cell Biol. 2010, 42, 1923–1926. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-López, L.; Márquez-Valadez, B.; Gómez-Sánchez, A.; Silva-Lucero, M.; Torres-Pérez, M.; Téllez-Ballesteros, R.; Ichwan, M.; Meraz-Ríos, M.; Kempermann, G.; Ramírez-Rodríguez, G. Green tea compound epigallo-catechin-3-gallate (EGCG) increases neuronal survival in adult hippocampal neurogenesis in vivo and in vitro. Neuroscience 2016, 322, 208–220. [Google Scholar] [CrossRef]
- Lu, Y.; Liu, Q.; Yu, Q. Quercetin enrich diet during the early-middle not middle-late stage of alzheimer’s disease ameliorates cognitive dysfunction. Am. J. Transl. Res. 2018, 10, 1237–1246. [Google Scholar] [PubMed]
- Kong, Y.; Li, K.; Fu, T.; Wan, C.; Zhang, D.; Song, H.; Zhang, Y.; Liu, N.; Gan, Z.; Yuan, L. Quercetin ameliorates Aβ toxicity in Drosophila AD model by modulating cell cycle-related protein expression. Oncotarget 2016, 7, 67716–67731. [Google Scholar] [CrossRef] [PubMed]
- Dauer, W.; Przedborski, S. Parkinson’s disease: Mechanisms and models. Neuron 2003, 39, 889–909. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-L.; Ju, B.; Zhang, Y.-Z.; Yin, H.-L.; Liu, Y.-J.; Wang, S.-S.; Zeng, Z.-L.; Yang, X.-P.; Wang, H.-T.; Li, J.-F. Protective Effect of Curcumin Against Oxidative Stress-Induced Injury in Rats with Parkinson’s Disease Through the Wnt/β-Catenin Signaling Pathway. Cell. Physiol. Biochem. 2017, 43, 2226–2241. [Google Scholar] [CrossRef] [PubMed]
- Visioli, F.; Rodríguez-Pérez, M.; Gómez-Torres, Ó; Pintado-Losa, C.; Burgos-Ramos, E. Hydroxytyrosol improves mitochondrial energetics of a cellular model of Alzheimer’s disease. Nutr. Neurosci. 2022, 25, 990–1000. [Google Scholar] [CrossRef] [PubMed]
- González-Correa, J.A.; Rodríguez-Pérez, M.D.; Márquez-Estrada, L.; López-Villodres, J.A.; Reyes, J.J.; Rodríguez-Gutiérrez, G.; Fernández-Bolaños, J.; De La Cruz, J.P. Neuroprotective Effect of Hydroxytyrosol in Experimental Diabetic Retinopathy: Relationship with Cardiovascular Biomarkers. J. Agric. Food Chem. 2018, 66, 637–644. [Google Scholar] [CrossRef]
- D’Andrea, G.; Ceccarelli, M.; Bernini, R.; Clemente, M.; Santi, L.; Caruso, C.; Micheli, L.; Tirone, F. Hydroxytyrosol stimulates neurogenesis in aged dentate gyrus by enhancing stem and progenitor cell proliferation and neuron survival. FASEB J. 2020, 34, 4512–4526. [Google Scholar] [CrossRef]
- Chiu, H.-F.; Venkatakrishnan, K.; Golovinskaia, O.; Wang, C.-K. Gastroprotective Effects of Polyphenols against Various Gastro-Intestinal Disorders: A Mini-Review with Special Focus on Clinical Evidence. Molecules 2021, 26, 2090. [Google Scholar] [CrossRef]
- Abdallah, D.M.; Ismael, N.R. Resveratrol abrogates adhesion molecules and protects against TNBS-induced ulcerative colitis in rats. Can. J. Physiol. Pharmacol. 2011, 89, 811–818. [Google Scholar] [CrossRef]
- Sharma, M.; Mohapatra, J.; Wagh, A.; Patel, H.M.; Pandey, D.; Kadam, S.; Argade, A.; Deshpande, S.S.; Shah, G.B.; Chatterjee, A.; et al. Involvement of TACE in colon inflammation: A novel mechanism of regulation via SIRT-1 activation. Cytokine 2014, 66, 30–39. [Google Scholar] [CrossRef]
- Ju, S.; Ge, Y.; Li, P.; Tian, X.; Wang, H.; Zheng, X.; Ju, S. Dietary quercetin ameliorates experimental colitis in mouse by remodeling the function of colonic macrophages via a heme oxygenase-1-dependent pathway. Cell Cycle 2018, 17, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Wan, F.; Zhong, R.; Wang, M.; Zhou, Y.; Chen, Y.; Yi, B.; Hou, F.; Liu, L.; Zhao, Y.; Chen, L.; et al. Caffeic Acid Supplement Alleviates Colonic Inflammation and Oxidative Stress Potentially Through Improved Gut Microbiota Community in Mice. Front. Microbiol. 2021, 12, 784211. [Google Scholar] [CrossRef] [PubMed]
- Larrosa, M.; Luceri, C.; Vivoli, E.; Pagliuca, C.; Lodovici, M.; Moneti, G.; Dolara, P. Polyphenol metabolites from colonic microbiota exert anti-inflammatory activity on different inflammation models. Mol. Nutr. Food Res. 2009, 53, 1044–1054. [Google Scholar] [CrossRef]
- Pal, C.; Bindu, S.; Dey, S.; Alam, A.; Goyal, M.; Iqbal, M.S.; Maity, P.; Adhikari, S.S.; Bandyopadhyay, U. Gallic acid prevents nonsteroidal anti-inflammatory drug-induced gastropathy in rat by blocking oxidative stress and apoptosis. Free Radic. Biol. Med. 2010, 49, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, A.; Young, K.; Moniruzzaman, M.; Beyene, A.; Do, K.; Kalaiselvi, S.; Min, T. Curcumin and Its Modified Formulations on Inflammatory Bowel Disease (IBD): The Story So Far and Future Outlook. Pharmaceutics 2021, 13, 484. [Google Scholar] [CrossRef]
- Briguglio, G.; Costa, C.; Pollicino, M.; Giambò, F.; Catania, S.; Fenga, C. Polyphenols in cancer prevention: New insights (Review). Int. J. Funct. Nutr. 2020, 1, 9. [Google Scholar] [CrossRef]
- Reyes-Farias, M.; Carrasco-Pozo, C. The Anti-Cancer Effect of Quercetin: Molecular Implications in Cancer Metabolism. Int. J. Mol. Sci. 2019, 20, 3177. [Google Scholar] [CrossRef]
- Balc-Okcanoğlu, T.; Yilma-Susluer, S.; Kayabasi, C.; Ozme-Yelken, B.; Biray-Avci, C.; Gunduz, C. The effect of caffeic acid phenethyl ester on cell cycle control gene expressions in breast cancer cells. Mol. Biol. Res. Commun. 2021, 10, 39–43. [Google Scholar] [CrossRef]
- Zeng, A.; Liang, X.; Zhu, S.; Liu, C.; Wang, S.; Zhang, Q.; Song, L. Chlorogenic acid induces apoptosis, inhibits metastasis and improves antitumor immunity in breast cancer via the NF-κB signaling pathway. Oncol. Rep. 2021, 45, 717–727. [Google Scholar] [CrossRef] [PubMed]
- Hormozi, M.; Salehi Marzijerani, A.; Baharvand, P. Effects of Hydroxytyrosol on Expression of Apoptotic Genes and Activity of Antioxidant Enzymes in LS180 Cells. Cancer Manag. Res. 2020, 12, 7913–7919. [Google Scholar] [CrossRef]
- Wei, R.; Hackman, R.M.; Wang, Y.; Mackenzie, G.G. Targeting Glycolysis with Epigallocatechin-3-Gallate Enhances the Efficacy of Chemotherapeutics in Pancreatic Cancer Cells and Xenografts. Cancers 2019, 11, 1496. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yi, P.; Tu, C.; Zhan, J.; Jiang, L.; Zhang, F. Curcumin Inhibits ERK/c-Jun Expressions and Phosphorylation against Endometrial Carcinoma. BioMed. Res. Int. 2019, 2019, 8912961. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-L.; Li, T.; Li, J.-H.; Miao, S.-Y.; Xiao, X.-Z. The Effects of Resveratrol on Inflammation and Oxidative Stress in a Rat Model of Chronic Obstructive Pulmonary Disease. Molecules 2017, 22, 1529. [Google Scholar] [CrossRef]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef]
- Santos, J.C.; Ribeiro, M.L.; Gambero, A. The Impact of Polyphenols-Based Diet on the Inflammatory Profile in COVID-19 Elderly and Obese Patients. Front. Physiol. 2021, 11, 612268–612280. [Google Scholar] [CrossRef]





| Solvent | ROO• | RO• | |
|---|---|---|---|
| H abstraction, LH | Nonpolar organic | <1–400 M−1s−1 | 104–107 M−1s−1 |
| H abstraction, LOOH | Nonpolar organic | 600 M−1s−1 | 2.5 × 108 M−1s−1 |
| Cyclization | Nonpolar organic | 101–103 s−1 | 104–105 s−1 |
| Addition | Nonpolar organic | — | 104–108 M−1s−1 |
| β-scission | Oleate Linoleate | 1–8 s−1 27–430 s−1 | 103–105 s−1 104–105 s−1 |
| R1 | R2 | R3 | R4 | R5 | 10−4 kinh (M−1 s−1) | |
|---|---|---|---|---|---|---|
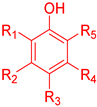 Phenols | H | H | CH3 | H | H | 0.917 |
| H | H | OCH3 | H | H | 4.78 | |
| C(CH3)3 | H | OCH3 | H | C(CH3)3 | 11 | |
| CH3 | H | OCH3 | H | CH3 | 94 | |
| CH3 | H | CH3 | H | CH3 | 8.5 | |
| CH3 | CH3 | CH3 | H | CH3 | 11 | |
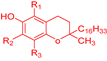 Tocopherols | CH3 | CH3 | CH3 | — | — | 320 |
| CH3 | H | CH3 | — | — | 130 | |
| H | H | CH3 | — | — | 44 | |
| H | CH3 | CH3 | — | — | 140 |
| Solvent | Dielectric Constant ∈ |
|---|---|
| Water | 78.5 |
| Ethanol | 24.3 |
| Propylene glycol | 32 |
| Olive oil | 3-0 |
| Sunflower | 3.1 |
| Corn | 3.1 |
| Modifications of the solvent phase | Cosolvents | Modification of the dielectric constant |
| Hydrotopes | Formation of complexes, modification solvent structure, and/or self-agregation to form “micelle-like” aggregates | |
| Micelles | Creation of one or more regions with different solvent properties | |
| Liposomes | ||
| Microemulsions | ||
| Emulsions | ||
| Complexation | Formation of host-guest complexes | |
| Combined methods | Simultaneous variation in one or more experimental parameters (pH, T, cosolvent, etc.) |
| Molecule | Membrane | T (°C) | Pm (cm/s) | Reference |
|---|---|---|---|---|
| O2 | CHO cells | 37 | 42 | [64] |
| NO• | RBC (human) | 20 | 18 | [65] |
| NO2• | EYPC | 25 | ~5 | [66] |
| H2O2 | PC12 cells HUVEC cells IMR-90 cells | - | 4 × 10−4 1.6 × 10−3 1.1 × 10−3 | [67] |
| HOO• | EYPC | 23 | 4.9 × 10−4 | [68] |
| O2− | EYPC | 23 | 7.6 × 10−8 | [68] |
| Antioxidant | Target | Carrier | Targeting Ligand | Result | Reference |
|---|---|---|---|---|---|
| Vitexin | Goblet cell | Soy peptide nanoparticles | Goblet cell targeting peptide CSKSSDYQC (CSK) coupled N-trimethyl chitosan (TMC) |
| [127] |
| Resveratrol | MCF-7 cells | Lipoprotein-mimetic nanoparticles | Folic Acid (FA) |
| [128] |
| Genistein | CD44 expressed breast tumor | Phytosomes | Hyaluronic acid (HA) |
| [129] |
| Plumbagin Genistein | Prostate cancer cells | Liposomes | PSMA specific antibodies |
| [130] |
| Quercetin | Triple negative breast cancer cells | pH sensitive nickel oxide nanoparticle | Folic Acid |
| [131] |
| Epigallocatechin gallate (EGCG) | CD36 receptor of macrophages | Nanoparticles | KOdiA-PC |
| [132] |
| Rutin | Cell surface receptors of AGEs (RAGE) | Nanoparticles | Argpyrimidine (Advanced Glycation End Product-AGE) |
| [133] |
| Antioxidant | Study Method | Formulation | Particle Size (nm) | Findings | Reference |
|---|---|---|---|---|---|
| Catechin |
| Chitosan coated liposomes (Chitosomes) | 137 ± 0.82 | Chitosan coated liposomes were stable against bile salts. Improved Catechin absorption was observed in uninverted rat intestine sac model. Pharmacokinetic studies on Wistar rats revealed an increase in AUC and Cmax values, thus increasing bioavailability. | [140] |
| Hesperidin |
| Solid lipid nanoparticles | 175.3 ± 3.6 | ~5 times higher apparent permeability coefficient was obtained compared to free hesperidin. Increased oral bioavailability due to improved solubility and permeation was observed. Due to effective suppression of oxidative stress and apoptosis, the cardiotoxicity caused by doxorubicin was reduced. | [141] |
| Liquiritin |
| Liposomes | 91.84 ± 1.85 | The solubility of liquiritin was improved via liposomes, and its oral bioavailability was 8.8 times higher than the free drug. In diabetic mice, improved hypoglycemic effect was observed due to antioxidant activity, and histopathological studies revealed the repairing ability of liposomal liquiritin to the organ damage. | [142] |
| Berberine |
| Proliposomes | 116.6 ± 5.8 * | Proliposomes were suggested as solid templates for liposomes. Reconstituted liposomes increased oral berberine bioavailability ~6 times in rats. The serum cholesterol levels significantly decreased in hyperlipidemic mice. | [143] |
| Myricetin |
| Proliposomes | 33.17 ± 0.32 * | Vitamin E TPGS-conjugated liposomes increased the oral myricetin bioavailability via facilitated mucoadhesion, permeation and controlled drug release. Further, pharmacodynamic studies revealed better antioxidant and hepatoprotective effects in CCl4 induced hepatotoxicity mice. | [144] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Losada-Barreiro, S.; Sezgin-Bayindir, Z.; Paiva-Martins, F.; Bravo-Díaz, C. Biochemistry of Antioxidants: Mechanisms and Pharmaceutical Applications. Biomedicines 2022, 10, 3051. https://doi.org/10.3390/biomedicines10123051
Losada-Barreiro S, Sezgin-Bayindir Z, Paiva-Martins F, Bravo-Díaz C. Biochemistry of Antioxidants: Mechanisms and Pharmaceutical Applications. Biomedicines. 2022; 10(12):3051. https://doi.org/10.3390/biomedicines10123051
Chicago/Turabian StyleLosada-Barreiro, Sonia, Zerrin Sezgin-Bayindir, Fátima Paiva-Martins, and Carlos Bravo-Díaz. 2022. "Biochemistry of Antioxidants: Mechanisms and Pharmaceutical Applications" Biomedicines 10, no. 12: 3051. https://doi.org/10.3390/biomedicines10123051
APA StyleLosada-Barreiro, S., Sezgin-Bayindir, Z., Paiva-Martins, F., & Bravo-Díaz, C. (2022). Biochemistry of Antioxidants: Mechanisms and Pharmaceutical Applications. Biomedicines, 10(12), 3051. https://doi.org/10.3390/biomedicines10123051






