Clinical Dilemmas in the Treatment of Elderly Patients Suffering from Hodgkin Lymphoma: A Review
Abstract
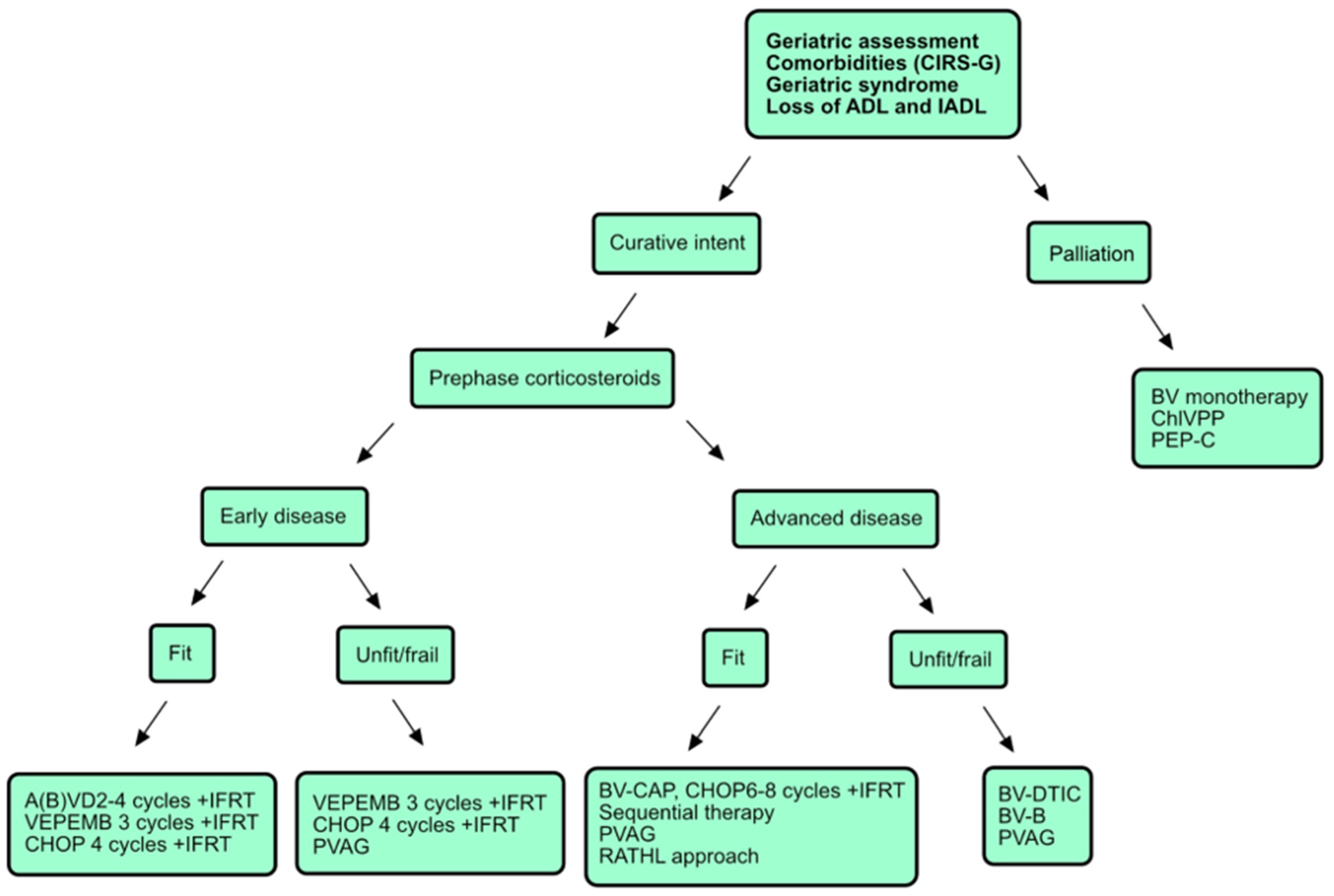
1. Introduction
2. Geriatric Approach to Aggressive Lymphomas
3. Does ABVD Regimen Treatment Have a Role in Elderly Patients with HL?
3.1. Early and Intermediate Disease
3.2. Advanced Disease
4. Should We Be Afraid of Bleomycin Use in Elderly Patients with HL?
5. ABVD and Leading Guidelines
6. Alternative Chemotherapy Regimens
7. The Use of Novel Agents in the Treatment of Elderly Patients Suffering from HL
7.1. Brentuximab Vedotin
7.2. BV in Frail Patients
7.3. BV in Fit Patients
7.4. Sequential Therapy
7.5. Brentuximab Vedotin: A New Role in First-Line Treatment?
8. Checkpoint Inhibitors: A Chemo-Free Approach?
9. Relapsed/Refractory Setting in Elderly Suffering from Hodgkin Lymphoma
9.1. Is There a Role for High-Dose Therapy and Autologous Stem Cell Transplant?
9.2. The Role of Novel Therapeutical Options: Are We There Yet?
9.2.1. Brentuximab Vedotin
9.2.2. Checkpoint Inhibitors
Nivolumab
Pembrolizumab
9.3. The Role of Novel Therapeutical Options: Are We There Yet? No…
10. Other Approaches
11. Conclusions: Is There Evidence-Based Medicine in the Treatment of Elderly Patients Suffering from Hodgkin Lymphoma?
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Swerdlow, S.H.; Campo, E.; Pileri, S.A.; Harris, N.L.; Stein, H.; Siebert, R.; Advani, R.; Ghielmini, M.; Salles, G.A.; Zelenetz, A.D.; et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood 2016, 127, 2375–2390. [Google Scholar] [CrossRef] [PubMed]
- Cancer Stat Facts: Hodgkin Lymphoma. Available online: https://seer.cancer.gov/statfacts/html/hodg.html (accessed on 29 March 2022).
- Engert, A.; Ballova, V.; Haverkamp, H.; Pfistner, B.; Josting, A.; Dühmke, E.; Müller-Hermelink, K.; Diehl, V.; German Hodgkin’s Study Group. Hodgkin’s lymphoma in elderly patients: A comprehensive retrospective analysis from the German Hodgkin’s Study Group. J. Clin. Oncol. 2005, 23, 5052–5060. [Google Scholar] [CrossRef] [PubMed]
- Mohile, S.G.; Dale, W.; Somerfield, M.R.; Schonberg, M.A.; Boyd, C.M.; Burhenn, P.S.; Canin, B.; Cohen, H.J.; Holmes, H.M.; Hopkins, J.O.; et al. Practical Assessment and Management of Vulnerabilities in Older Patients Receiving Chemotherapy: ASCO Guideline for Geriatric Oncology. J. Clin. Oncol. 2018, 36, 2326–2347. [Google Scholar] [CrossRef] [PubMed]
- Parmelee, P.A.; Thuras, P.D.; Katz, I.R.; Lawton, M.P. Validation of the Cumulative Illness Rating Scale in a geriatric residential population. J. Am. Geriatr. Soc. 1995, 43, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Eyre, T.A.; Martinez-Calle, N.; Hildyard, C.; Eyre, D.W.; Plaschkes, H.; Griffith, J.; Wolf, J.; Fields, P.; Gunawan, A.; Oliver, R.; et al. Impact of intended and relative dose intensity of R-CHOP in a large, consecutive cohort of elderly diffuse large B-cell lymphoma patients treated with curative intent: No difference in cumulative incidence of relapse comparing patients by age. J. Intern. Med. 2019, 285, 681–692. [Google Scholar] [CrossRef]
- Eyre, T.A.; Wilson, W.; Kirkwood, A.A.; Wolf, J.; Hildyard, C.; Plaschkes, H.; Griffith, J.; Fields, P.; Gunawan, A.; Oliver, R.; et al. Infection-related morbidity and mortality among older patients with DLBCL treated with full- or attenuated-dose R-CHOP. Blood Adv. 2021, 5, 2229–2236. [Google Scholar] [CrossRef]
- Sorror, M.L.; Maris, M.B.; Storb, R.; Baron, F.; Sandmaier, B.M.; Maloney, D.G.; Storer, B. Hematopoietic cell transplantation (HCT)-specific comorbidity index: A new tool for risk assessment before allogeneic HCT. Blood 2005, 106, 2912–2919. [Google Scholar] [CrossRef]
- Dahi, P.B.; Tamari, R.; Devlin, S.M.; Maloy, M.; Bhatt, V.; Scordo, M.; Goldberg, J.; Zelenetz, A.D.; Hamlin, P.A.; Matasar, M.J.; et al. Favorable outcomes in elderly patients undergoing high-dose therapy and autologous stem cell transplantation for non-Hodgkin lymphoma. Biol. Blood Marrow Transplant. 2014, 20, 2004–2009. [Google Scholar] [CrossRef]
- Geriatric Syndrome. Steadman’s Medical Dictionary. Available online: https://stedmansonline.com/ (accessed on 29 March 2022).
- Orellana-Noia, V.M.; Isaac, K.; Malecek, M.K.; Bartlett, N.L.; Voorhees, T.J.; Grover, N.S.; Hwang, S.R.; Bennani, N.N.; Hu, R.; Hill, B.T.; et al. Multicenter analysis of geriatric fitness and real-world outcomes in older patients with classical Hodgkin lymphoma. Blood Adv. 2021, 5, 3623–3632. [Google Scholar] [CrossRef]
- Martinez-Tapia, C.; Canoui-Poitrine, F.; Bastuji-Garin, S.; Soubeyran, P.; Mathoulin-Pelissier, S.; Tournigand, C.; Paillaud, E.; Laurent, M.; Audureau, E.; ELCAPA Study Group. Optimizing the G8 Screening Tool for Older Patients With Cancer: Diagnostic Performance and Validation of a Six-Item Version. Oncologist 2016, 21, 188–195. [Google Scholar] [CrossRef]
- Lee, S.; Fujita, K.; Morishita, T.; Oiwa, K.; Tsukasaki, H.; Negoro, E.; Hara, T.; Tsurumi, H.; Ueda, T.; Yamauchi, T. Association of the Geriatric 8 with treatment intensity and prognosis in older patients with diffuse large B-cell lymphoma. Br. J. Haematol. 2021, 194, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.; Downs, T.D.; Cash, H.R.; Grotz, R.C. Progress in development of the index of ADL. Gerontologist 1970, 10, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Blackwood, J.; Karczewski, H.; Huang, M.H.; Pfalzer, L. Katz activities of daily living disability in older cancer survivors by age, stage, and cancer type. J. Cancer Surviv. 2020, 14, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Garric, M.; Sourdet, S.; Cabarrou, B.; Steinmeyer, Z.; Gauthier, M.; Ysebaert, L.; Beyne-Rauzy, O.; Gerard, S.; Lozano, S.; Brechemier, D.; et al. Impact of a comprehensive geriatric assessment on decision-making in older patients with hematological malignancies. Eur. J. Haematol. 2021, 106, 616–626. [Google Scholar] [CrossRef] [PubMed]
- Schonberg, M.A.; Davis, R.B.; McCarthy, E.P.; Marcantonio, E.R. Index to predict 5-year mortality of community-dwelling adults aged 65 and older using data from the National Health Interview Survey. J. Gen. Intern. Med. 2009, 24, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, S.; Matsushima, T.; Minami, M.; Kadowaki, M.; Takase, K.; Iwasaki, H. Clinical impact of comprehensive geriatric assessment in patients aged 80 years and older with diffuse large B-cell lymphoma receiving rituximab-mini-CHOP: A single-institute retrospective study. Eur. Geriatr. Med. 2022, 13, 195–201. [Google Scholar] [CrossRef]
- Zhang, W.H.; Li, G.Y.; Ma, Y.J.; Li, Z.C.; Zhu, Y.; Chang, J.; Hao, S.G.; Tao, R. Reduced-dose EPOCH-R chemotherapy for elderly patients with advanced stage diffuse large B cell lymphoma. Ann. Hematol. 2018, 97, 1809–1816. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, C.L.; Feng, R.; Li, J.T.; Tian, Y.; Wang, T. Validation and Refinement of the Age, Comorbidities, and Albumin Index in Elderly Patients with Diffuse Large B-Cell Lymphoma: An Effective Tool for Comprehensive Geriatric Assessment. Oncologist 2018, 23, 722–729. [Google Scholar] [CrossRef]
- Hshieh, T.T.; Jung, W.F.; Grande, L.J.; Chen, J.; Stone, R.M.; Soiffer, R.J.; Driver, J.A.; Abel, G.A. Prevalence of Cognitive Impairment and Association With Survival Among Older Patients With Hematologic Cancers. JAMA Oncol. 2018, 4, 686–693. [Google Scholar] [CrossRef]
- Di, M.; Keeney, T.; Belanger, E.; Panagiotou, O.A.; Olszewski, A.J. Global Risk Indicator and Therapy for Older Patients With Diffuse Large B-Cell Lymphoma: A Population-Based Study. JCO Oncol. Pract. 2022, 18, 383–402. [Google Scholar] [CrossRef]
- Tuch, G.; Soo, W.K.; Luo, K.Y.; Frearson, K.; Oh, E.L.; Phillips, J.L.; Agar, M.; Lane, H. Cognitive Assessment Tools Recommended in Geriatric Oncology Guidelines: A Rapid Review. Curr. Oncol. 2021, 28, 3987–4003. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, J.I.; Yesavage, J.A.; Brooks, J.O.; Friedman, L.; Gratzinger, P.; Hill, R.D.; Zadeik, A.; Crook, T. Proposed factor structure of the Geriatric Depression Scale. Int. Psychogeriatr. 1991, 3, 23–28. [Google Scholar] [CrossRef]
- Surov, A.; Wienke, A. Sarcopenia predicts overall survival in patients with malignant hematological diseases: A meta-analysis. Clin. Nutr. 2021, 40, 1155–1160. [Google Scholar] [CrossRef] [PubMed]
- Hurria, A.; Cirrincione, C.T.; Muss, H.B.; Kornblith, A.B.; Barry, W.; Artz, A.S.; Schmieder, L.; Ansari, R.; Tew, W.P.; Weckstein, D.; et al. Implementing a geriatric assessment in cooperative group clinical cancer trials: CALGB 360401. J. Clin. Oncol. 2011, 29, 1290–1296. [Google Scholar] [CrossRef] [PubMed]
- Coiffier, B.; Thieblemont, C.; Van Den Neste, E.; Lepeu, G.; Plantier, I.; Castaigne, S.; Lefort, S.; Marit, G.; Macro, M.; Sebban, C.; et al. Long-term outcome of patients in the LNH-98.5 trial, the first randomized study comparing rituximab-CHOP to standard CHOP chemotherapy in DLBCL patients: A study by the Groupe d’Etudes des Lymphomes de l’Adulte. Blood 2010, 116, 2040–2045. [Google Scholar] [CrossRef] [PubMed]
- Tavares, A.; Moreira, I. Diffuse large B-cell lymphoma in very elderly patients: Towards best tailored treatment-A systematic review. Crit. Rev. Oncol. Hematol. 2021, 160, 103294. [Google Scholar] [CrossRef]
- Van Spronsen, D.J.; Janssen-Heijnen, M.L.; Breed, W.P.; Coebergh, J.W. Prevalence of co-morbidity and its relationship to treatment among unselected patients with Hodgkin’s disease and non-Hodgkin’s lymphoma, 1993–1996. Ann. Hematol. 1999, 78, 315–319. [Google Scholar] [CrossRef]
- Evens, A.M.; Helenowski, I.; Ramsdale, E.; Nabhan, C.; Karmali, R.; Hanson, B.; Parsons, B.; Smith, S.; Larsen, A.; McKoy, J.M.; et al. A retrospective multicenter analysis of elderly Hodgkin lymphoma: Outcomes and prognostic factors in the modern era. Blood 2012, 119, 692–695. [Google Scholar] [CrossRef]
- Kumar, A.J.; Nelson, J.; Rodday, A.M.; Evens, A.M.; Friedberg, J.W.; Wildes, T.M.; Parsons, S.K. Development and validation of a prediction model for 1-year mortality among older adults with Hodgkin Lymphoma who receive dose-intense chemotherapy. J. Geriatr. Oncol. 2021, 12, 1233–1239. [Google Scholar] [CrossRef]
- Charlson, M.; Szatrowski, T.P.; Peterson, J.; Gold, J. Validation of a combined comorbidity index. J. Clin. Epidemiol. 1994, 47, 1245–1251. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. National Library of Medicine (U.S.). Prospective Study on Elderly (≥65 Years) Patients Affected with Classical Hodgkin Lymphoma Undergoing Comprehensive Geriatric Assessment at Diagnosis. (11–14 June 2018) Identifier NCT03552003 Retrieved 4. April 2022. Available online: https://www.clinicaltrials.gov/ct2/show/NCT03552003 (accessed on 10 March 2022).
- Carter, J.; David, K.A.; Kritharis, A.; Evens, A.M. Current Treatment Options for Older Patients with Hodgkin Lymphoma. Curr. Treat. Options Oncol. 2020, 21, 42. [Google Scholar] [CrossRef] [PubMed]
- Evens, A.M.; Carter, J.; Loh, K.P.; David, K.A. Management of older Hodgkin lymphoma patients. Hematol. Am. Soc. Hematol. Educ. Program 2019, 2, 33–242. [Google Scholar] [CrossRef]
- Follows, G.A.; Barrington, S.F.; Bhuller, K.S.; Culligan, D.J.; Cutter, D.J.; Gallop-Evans, E.; Kassam, S.; Osborne, W.; Sadullah, S.; Townsend, W.; et al. Guideline for the First-Line Management of Classical Hodgkin Lymphoma-A British Society for Haematology Guideline. Br. J. Haematol. 2022, 197, 558–572. [Google Scholar] [CrossRef]
- Engert, A.; Plütschow, A.; Eich, H.T.; Lohri, A.; Dörken, B.; Borchmann, P.; Berger, B.; Greil, R.; Willborn, K.C.; Wilhelm, M.; et al. Reduced treatment intensity in patients with early-stage Hodgkin’s lymphoma. N. Engl. J. Med. 2010, 363, 640–652. [Google Scholar] [CrossRef] [PubMed]
- Eich, H.T.; Diehl, V.; Görgen, H.; Pabst, T.; Markova, J.; Debus, J.; Ho, A.; Dörken, B.; Rank, A.; Grosu, A.L.; et al. Intensified chemotherapy and dose-reduced involved-field radiotherapy in patients with early unfavorable Hodgkin’s lymphoma: Final analysis of the German Hodgkin Study Group HD11 trial. J. Clin. Oncol. 2010, 28, 4199–4206. [Google Scholar] [CrossRef]
- Böll, B.; Görgen, H.; Fuchs, M.; Pluetschow, A.; Eich, H.T.; Bargetzi, M.J.; Weidmann, E.; Junghanß, C.; Greil, R.; Scherpe, A.; et al. ABVD in older patients with early-stage Hodgkin lymphoma treated within the German Hodgkin Study Group HD10 and HD11 trials. J. Clin. Oncol. 2013, 31, 1522–1529. [Google Scholar] [CrossRef] [PubMed]
- Behringer, K.; Goergen, H.; Hitz, F.; Zijlstra, J.M.; Greil, R.; Markova, J.; Sasse, S.; Fuchs, M.; Topp, M.S.; Soekler, M.; et al. Omission of dacarbazine or bleomycin, or both, from the ABVD regimen in treatment of early-stage favourable Hodgkin’s lymphoma (GHSG HD13): An open-label, randomised, non-inferiority trial. Lancet 2015, 385, 1418–1427. [Google Scholar] [CrossRef]
- Böll, B.; Goergen, H.; Behringer, K.; Bröckelmann, P.J.; Hitz, F.; Kerkhoff, A.; Greil, R.; von Tresckow, B.; Eichenauer, D.A.; Bürkle, C.; et al. Bleomycin in older early-stage favorable Hodgkin lymphoma patients: Analysis of the German Hodgkin Study Group (GHSG) HD10 and HD13 trials. Blood 2016, 127, 2189–2192. [Google Scholar] [CrossRef]
- Gordon, L.I.; Hong, F.; Fisher, R.I.; Bartlett, N.L.; Connors, J.M.; Gascoyne, R.D.; Wagner, H.; Stiff, P.J.; Cheson, B.D.; Gospodarowicz, M.; et al. Randomized phase III trial of ABVD versus Stanford V with or without radiation therapy in locally extensive and advanced-stage Hodgkin lymphoma: An intergroup study coordinated by the Eastern Cooperative Oncology Group (E2496). J. Clin. Oncol. 2013, 31, 684–691. [Google Scholar] [CrossRef]
- Evens, A.M.; Hong, F.; Gordon, L.I.; Fisher, R.I.; Bartlett, N.L.; Connors, J.M.; Gascoyne, R.D.; Wagner, H.; Gospodarowicz, M.; Cheson, B.D.; et al. The efficacy and tolerability of adriamycin, bleomycin, vinblastine, dacarbazine and Stanford V in older Hodgkin lymphoma patients: A comprehensive analysis from the North American intergroup trial E2496. Br. J. Haematol. 2013, 161, 76–86. [Google Scholar] [CrossRef]
- Johnson, P.; Federico, M.; Kirkwood, A.; Fosså, A.; Berkahn, L.; Carella, A.; d’Amore, F.; Enblad, G.; Franceschetto, A.; Fulham, M.; et al. Adapted Treatment Guided by Interim PET-CT scan in Advanced Hodgkin’s Lymphoma. N. Engl. J. Med. 2016, 374, 2419–2429. [Google Scholar] [CrossRef] [PubMed]
- Bentur, O.S.; Dann, E.J.; Paran, E.; Lavie, D.; Nachmias, B.; Ron, Y.; Dally, N.; Gutwein, O.; Herishanu, Y.; Sarid, N.; et al. Interim PET-CT-guided therapy in elderly patients with Hodgkin lymphoma-a retrospective national multi-center study. Ann. Hematol. 2019, 98, 1665–1674. [Google Scholar] [CrossRef] [PubMed]
- Cheson, B.D.; Fisher, R.I.; Barrington, S.F.; Cavalli, F.; Schwartz, L.H.; Zucca, E.; Lister, T.A.; Alliance; Australasian Leukaemia and Lymphoma Group; Eastern Cooperative Oncology Group; et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: The Lugano classification. J. Clin. Oncol. 2014, 32, 3059–3068. [Google Scholar] [CrossRef] [PubMed]
- Pavlovsky, A.; Fernandez, I.; Kurgansky, N.; Prates, V.; Zoppegno, L.; Negri, P.; Milone, G.; Cerutti, I.; Zabaljauregui, S.; Mariano, R.; et al. PET-adapted therapy after three cycles of ABVD for all stages of Hodgkin lymphoma: Results of the GATLA LH-05 trial. Br. J. Haematol. 2019, 185, 865–873. [Google Scholar] [CrossRef] [PubMed]
- Ciliberti, E.I.; Fernandez, N.; Kurgansky, V.; Prates, L.; Zoppegno, P.; Negri, G.; Milone, I.; Cerutti, S.; Zabaljauregui, R.; Mariano, H.; et al. Safety and Efficacy Analysis of Elderly Patients Treated within the Gatla HL-05 Clinical Trial: Pet Adapted THerapy After 3 Cycles of ABVD for all Stages of Hodgkin Lymphoma. In Proceedings of the 14th International Conference on Malignant Lymphoma, Lugano, Switzerland, 14–17 June 2017; Wiley: Lugano, Switzerland, 2017. Abstract Number: 166. [Google Scholar]
- Leger, P.; Limper, A.H.; Maldonado, F. Toxicities from Conventional Chemotherapy. Clin. Chest Med. 2017, 38, 209–222. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, J.M.; Huddart, R.A.; Norman, A.R.; Nicholls, J.; Dearnaley DPHorwich, A. Predicting the risk of bleomycin lung toxicity in patients with germ-cell tumours. Ann. Oncol. 2003, 14, 91–96. [Google Scholar] [CrossRef]
- Martin, W.G.; Ristow, K.M.; Habermann, T.M.; Colgan, J.P.; Witzig, T.E.; Ansell, S.M. Bleomycin pulmonary toxicity has a negative impact on the outcome of patients with Hodgkin’s lymphoma. J. Clin. Oncol. 2005, 23, 7614–7620. [Google Scholar] [CrossRef]
- Stamatoullas, A.; Brice, P.; Bouabdallah, R.; Mareschal, S.; Camus, V.; Rahal, I.; Franchi, P.; Lanic, H.; Tilly, H. Outcome of patients older than 60 years with classical Hodgkin lymphoma treated with front line ABVD chemotherapy: Frequent pulmonary events suggest limiting the use of bleomycin in the elderly. Br. J. Haematol. 2015, 170, 179–184. [Google Scholar] [CrossRef]
- The GRADE Working Group. Available online: https://www.gradeworkinggroup.org/ (accessed on 7 April 2022).
- Hodgkin Lymphoma. Version 2.2022. NCCN Clinical Practice Guidelines in Oncology. Available online: https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1439 (accessed on 7 April 2022).
- Eichenauer, D.A.; Aleman, B.; André, M.; Federico, M.; Hutchings, M.; Illidge, T.; Engert, A.; Ladetto, M.; Hodgkin lymphoma. ESMO Guidelines Committee. Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2018, 29, iv19–iv29. [Google Scholar] [CrossRef]
- Ballova, V.; Rüffer, J.U.; Haverkamp, H.; Pfistner, B.; Müller-Hermelink, H.K.; Dühmke, E.; Worst, P.; Wilhelmy, M.; Naumann, R.; Hentrich, M.; et al. A prospectively randomized trial carried out by the German Hodgkin Study Group (GHSG) for elderly patients with advanced Hodgkin’s disease comparing BEACOPP baseline and COPP-ABVD (study HD9elderly). Ann. Oncol. 2005, 16, 124–131. [Google Scholar] [CrossRef]
- Halbsguth, T.V.; Nogová, L.; Mueller, H.; Sieniawski, M.; Eichenauer, D.A.; Schober, T.; Nisters-Backes, H.; Borchmann, P.; Diehl, V.; Engert, A.; et al. Phase 2 study of BACOPP (bleomycin, adriamycin, cyclophosphamide, vincristine, procarbazine, and prednisone) in older patients with Hodgkin lymphoma: A report from the German Hodgkin Study Group (GHSG). Blood 2010, 116, 2026–2032. [Google Scholar] [CrossRef] [PubMed]
- ChlVPP therapy for Hodgkin’s disease: Experience of 960 patients. The International ChlVPP Treatment Group. Ann. Oncol. 1995, 6, 167–172. [CrossRef]
- Coleman, M.; Ruan, G.; Elstrom, R.L.; Martin, P.; Leonard, J.P. Metronomic therapy for refractory/relapsed lymphoma: The PEP-C low-dose oral combination chemotherapy regimen. Hematology 2012, 17, S90–S92. [Google Scholar] [CrossRef]
- Kolstad, A.; Nome, O.; Delabie, J.; Lauritzsen, G.F.; Fossa, A.; Holte, H. Standard CHOP-21 as first line therapy for elderly patients with Hodgkin’s lymphoma. Leuk. Lymphoma 2007, 48, 570–576. [Google Scholar] [CrossRef]
- Böll, B.; Bredenfeld, H.; Görgen, H.; Halbsguth, T.; Eich, H.T.; Soekler, M.; Markova, J.; Keller, U.; Graeven, U.; Kremers, S.; et al. Phase 2 study of PVAG (prednisone, vinblastine, doxorubicin, gemcitabine) in elderly patients with early unfavorable or advanced stage Hodgkin lymphoma. Blood 2011, 118, 6292–6298. [Google Scholar] [CrossRef]
- Aussedat, G.; Idlhaj, M.; Durand, A.; Roussel, X.; Brice, P.; Salles, G.; Deau Fischer, B.; Chauchet, A.; Rossi, C.; Ghesquieres, H. PVAG Regimen (Prednisone, Vinblastine, Doxorubicin, Gemcitabine) Used in Real-Life Setting in First Line Therapy for Elderly Classical Hodgkin Lymphoma Patients: A Retrospective Study of Lysa Centers. Blood 2020, 136, 11. [Google Scholar] [CrossRef]
- Levis, A.; Anselmo, A.P.; Ambrosetti, A.; Adamo, F.; Bertini, M.; Cavalieri, E.; Gavarotti, P.; Genua, A.; Liberati, M.; Pavone, V.; et al. VEPEMB in elderly Hodgkin’s lymphoma patients. Results from an Intergruppo Italiano Linfomi (IIL) study. Ann. Oncol. 2004, 15, 123–128. [Google Scholar] [CrossRef]
- Epperla, N.; Herrera, A.F. How I incorporate novel agents into the treatment of classical Hodgkin lymphoma. Blood 2021, 138, 520–530. [Google Scholar] [CrossRef]
- Forero-Torres, A.; Holkova, B.; Goldschmidt, J.; Chen, R.; Olsen, G.; Boccia, R.V.; Bordoni, R.E.; Friedberg, J.W.; Sharman, J.P.; Palanca-Wessels, M.C.; et al. Phase 2 study of frontline brentuximab vedotin monotherapy in Hodgkin lymphoma patients aged 60 years and older. Blood 2015, 126, 2798–2804. [Google Scholar] [CrossRef] [PubMed]
- Gibb, A.; Pirrie, S.J.; Linton, K.; Warbey, V.; Paterson, K.; Davies, A.J.; Collins, G.P.; Menne, T.; McKay, P.; Fields, P.A.; et al. Phase II study of brentuximab vedotin using a response-adapted design in the first-line treatment of patients with classical Hodgkin lymphoma unsuitable for chemotherapy due to age, frailty or comorbidity (BREVITY). Br. J. Haematol. 2021, 193, 63–71. [Google Scholar] [CrossRef]
- Yasenchak, C.A.; Bordoni, R.; Patel-Donnelly, D.; Larson, T.; Goldschmidt, J.; Boccia, R.V.; Cline, V.J.M.; Sacchi, M.; Forero-Torres, A.; Sims, R.B.; et al. Frontline Brentuximab Vedotin as Monotherapy or in Combination for Older Hodgkin Lymphoma Patients. Blood 2020, 136, 18–19. [Google Scholar] [CrossRef]
- Friedberg, J.W.; Forero-Torres, A.; Bordoni, R.E.; Cline, V.; Patel Donnelly, D.; Flynn, P.J.; Olsen, G.; Chen, R.; Fong, A.; Wang, Y.; et al. Frontline brentuximab vedotin in combination with dacarbazine or bendamustine in patients aged ≥60 years with HL. Blood 2017, 130, 2829–2837. [Google Scholar] [CrossRef] [PubMed]
- Schiano de Colella, J.M.; Viviani, S.; Rapezzi, D.; Patti, C.; Clement Filliatre, L.; Rossi, A.; Bijou, F.; Cantonetti, M.; Borel, C.; Thamphya, B.; et al. Brentuximab vedotin and bendamustine as first-line treatment of Hodgkin lymphoma in the elderly (HALO Trial). J. Clin. Oncol. 2020, 38, 8029. [Google Scholar] [CrossRef]
- Connors, J.M.; Jurczak, W.; Straus, D.J.; Ansell, S.M.; Kim, W.S.; Gallamini, A.; Younes, A.; Alekseev, S.; Illés, Á.; Picardi, M.; et al. ECHELON-1 Study Group. Brentuximab Vedotin with Chemotherapy for Stage III or IV Hodgkin’s Lymphoma. N. Engl. J. Med. 2018, 378, 331–344. [Google Scholar] [CrossRef] [PubMed]
- Evens, A.M.; Connors, J.M.; Younes, A.; Ansell, S.M.; Kim, W.S.; Radford, J.; Feldman, T.A.; Tuscano, J.; Savage, K.J.; Oki, Y.; et al. Older Patients (pts) with Previously Untreated Classical Hodgkin Lymphoma (cHL): A Detailed Analysis from the Phase 3 ECHELON-1 Study. Blood 2018, 132, 1618. [Google Scholar] [CrossRef]
- Boell, B.; Fosså, A.; Goergen, H.; Kamper, P.; Leppa, S.; Molin, D.; Meissner, J.; Ellen Ritter, E.; Christensen, J.H.; Hutchings, M.; et al. B-CAP (brentuximab vedotin, cyclophosphamide, doxorubicin and predniso(lo)Ne) in Older Patients with Advanced-Stage Hodgkin Lymphoma: Results of a Phase II Intergroup Trial By the German Hodgkin Study Group (GHSG) and the Nordic Lymphoma Group (NLG). Blood 2018, 132, 926. [Google Scholar] [CrossRef]
- Evens, A.M.; Advani, R.H.; Helenowski, I.B.; Fanale, M.; Smith, S.M.; Jovanovic, B.D.; Bociek, G.R.; Klein, A.K.; Winter, J.N.; Gordon, L.I.; et al. A Multicenter Phase II Study of Sequential Brentuximab Vedotin and Doxorubicin, Vinblastine, and Dacarbazine Chemotherapy for Older Patients With Untreated Classical Hodgkin Lymphoma. J. Clin. Oncol. 2018, 36, 3015–3022. [Google Scholar] [CrossRef]
- Evens, A.M.; Advani, R.H.; Fanale, M.A.; Smith, S.M.; Helenowski, I.B.; Bociek, G.; Kirschmer Klein, A.; Winter, J.N.; Gordon, L.I.; Hamlin, P.A. Prognostication of older Hodgkin lymphoma (HL) patients (pts): Findings from a multicenter phase II study. J. Clin. Oncol. 2018, 36, 7540. [Google Scholar] [CrossRef]
- Yasenchak, C.A.; Bordoni, R.; Yazbeck, V.; Patel-Donnelly, D.; Anderson, T.; Larson, T.; Newhook, T.; Mei, M.; Ho, L.; Friedberg, J.W. Phase 2 Study of Frontline Brentuximab Vedotin Plus Nivolumab in Patients with Hodgkin Lymphoma Aged ≥60 Years. Blood 2019, 134, 237. [Google Scholar] [CrossRef]
- Cheson, B.D.; Bartlett, N.L.; LaPlant, B.; Lee, H.J.; Advani, R.J.; Christian, B.; Diefenbach, C.S.; Feldman, T.A.; Ansell, S.M. Brentuximab vedotin plus nivolumab as first-line therapy in older or chemotherapy-ineligible patients with Hodgkin lymphoma (ACCRU): A multicentre, single-arm, phase 2 trial. Lancet Haematol. 2020, 7, e808–e815. [Google Scholar] [CrossRef]
- Lazarovici, J.; Amorim, S.; Bouabdallah, K.; Guidez, S.; Molina, L.; Morschhauser, F.; Gac, A.C.; Gastinne, T.; Laribi, K.; Launay, V.; et al. Nivolumab First-Line Therapy for Elderly, Frail Hodgkin Lymphoma Patients: Niviniho, a Lysa Phase II Study. Blood 2021, 138, 232. [Google Scholar] [CrossRef]
- Puig, N.; Pintilie, M.; Seshadri, T.; al-Farsi, K.; Franke, N.; Keating, A.; Kuruvilla, J.; Crump, M. High-dose chemotherapy and auto-SCT in elderly patients with Hodgkin’s lymphoma. Bone Marrow Transplant. 2011, 46, 1339–1344. [Google Scholar] [CrossRef] [PubMed]
- Stamatoullas, A.; Brice, P.; Gueye, M.S.; Mareschal, S.; Chevallier, P.; Bouabdallah, R.; Nguyenquoc, S.; Francois, S.; Turlure, P.; Ceballos, P.; et al. Autologous stem cell transplantation for patients aged 60 years or older with refractory or relapsed classical Hodgkin’s lymphoma: A retrospective analysis from the French Society of Bone Marrow Transplantation and Cell Therapies (SFGM-TC). Bone Marrow Transplant. 2016, 51, 928–932. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Martínez, C.; Jorge, A.S.; Pereira, A.; Moreno, M.; Núñez, J.; Gayoso, J.; Gonzalez-Medina, J.; Revilla, N.; Sampol, A.; Domingo-Domenech, E.; et al. Comorbidities, not age, are predictive of survival after autologous hematopoietic cell transplantation for relapsed/refractory Hodgkin’s lymphoma in patients older than 50 years. Ann. Hematol. 2017, 96, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Moskowitz, C.H.; Nademanee, A.; Masszi, T.; Agura, E.; Holowiecki, J.; Abidi, M.H.; Chen, A.I.; Stiff, P.; Gianni, A.M.; Carella, A.; et al. Brentuximab vedotin as consolidation therapy after autologous stem-cell transplantation in patients with Hodgkin’s lymphoma at risk of relapse or progression (AETHERA): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2015, 385, 1853–1862. [Google Scholar] [CrossRef]
- Bartlett, N.L.; Chen, R.; Fanale, M.A.; Brice, P.; Gopal, A.; Smith, S.E.; Advani, R.; Matous, J.V.; Ramchandren, R.; Rosenblatt, J.D.; et al. Retreatment with brentuximab vedotin in patients with CD30-positive hematologic malignancies. J. Hematol. Oncol. 2014, 7, 24. [Google Scholar] [CrossRef]
- Ansell, S.M.; Lesokhin, A.M.; Borrello, I.; Halwani, A.; Scott, E.C.; Gutierrez, M.; Schuster, S.J.; Millenson, M.M.; Cattry, D.; Freeman, G.J.; et al. PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma. N. Engl. J. Med. 2015, 372, 311–319. [Google Scholar] [CrossRef]
- Armand, P.; Engert, A.; Younes, A.; Fanale, M.; Santoro, A.; Zinzani, P.L.; Timmerman, J.M.; Collins, G.P.; Ramchandren, R.; Cohen, J.B.; et al. Nivolumab for Relapsed/Refractory Classic Hodgkin Lymphoma After Failure of Autologous Hematopoietic Cell Transplantation: Extended Follow-Up of the Multicohort Single-Arm Phase II CheckMate 205 Trial. J. Clin. Oncol. 2018, 36, 1428–1439. [Google Scholar] [CrossRef]
- Zettler, M.E.; Nabhan, C.; Gajra, A.; Feinberg, B. Adverse Events in Elderly Hodgkin Lymphoma Patients Treated with Pembrolizumab or Nivolumab in the Real World. Blood 2019, 134, 3490. [Google Scholar] [CrossRef]
- Kuruvilla, J.; Ramchandren, R.; Santoro, A.; Paszkiewicz-Kozik, E.; Gasiorowski, R.; Johnson, N.A.; Fogliatto, L.M.; Goncalves, I.; de Oliveira, J.; Buccheri, V.; et al. Pembrolizumab versus brentuximab vedotin in relapsed or refractory classical Hodgkin lymphoma (KEYNOTE-204): An interim analysis of a multicentre, randomised, open-label, phase 3 study. Lancet Oncol. 2021, 22, 512–524. [Google Scholar] [CrossRef]
- Böll, B.; Goergen, H.; Arndt, N.; Meissner, J.; Krause, S.W.; Schnell, R.; von Tresckow, B.; Eichenauer, D.A.; Sasse, S.; Fuchs, M.; et al. Relapsed hodgkin lymphoma in older patients: A comprehensive analysis from the German hodgkin study group. J. Clin. Oncol. 2013, 31, 4431–4437. [Google Scholar] [CrossRef] [PubMed]
- Josting, A.; Franklin, J.; May, M.; Koch, P.; Beykirch, M.K.; Heinz, J.; Rudolph, C.; Diehl, V.; Engert, A. New prognostic score based on treatment outcome of patients with relapsed Hodgkin’s lymphoma registered in the database of the German Hodgkin’s lymphoma study group. J. Clin. Oncol. 2002, 20, 221–230. [Google Scholar] [PubMed]
- National Institute of Health. U.S. National Library of Health. Clinical Trials.gov. Available online: https://www.clinicaltrials.gov/ (accessed on 17 April 2022).
| Domain | Explanation | Tools Used in Aggressive Lymphomas and Proposed by ASCO |
|---|---|---|
| Non-cancer life expectancy | Comorbidities predicting the non-cancer-related mortality | CIRS-G [5,6,7] * HCT-CI [8,9] |
| Geriatric syndrome | The aggregate of “symptoms and signs associated with any morbid process, and constituting together the picture of the disease” [10] | GA [11] ** and G8 [12,13] *** |
| Function | Loss of activities of daily life (ADL) and instrumental activities of daily life (IADL) | Katz index for ADL [14,15,16] Schonberg index for IADL [17] ****; other indexes [16,18,19,20] |
| Falls | Number of falls in the last 6 months [4] | Single-item question |
| Cognition | Cognitive impairment is associated with worse outcomes [4] | Various indexes [21,22,23] ***** |
| Depression | Depression in cancer patients is related to multiple variables, ranging from treatment feasibility and mortality to functional decline [4] ****** | GDS [24] |
| Nutrition | Unintentional weight loss and BMI are associated with mortality [4] | SMM, CT and PET-CT [25] ******* |
| Toxicity | Risk factors for treatment-related toxicity | GA [11], G8 [12], GRI [22], nutrition [25] and CARG toxicity tool [26] |
| Study | n | Disease Stage | Regimen | CR | Outcome Rate * | p ** | AE Rate N (Rate) *** | CVD-Related Death N (Rate) | Respiratory AE N (Rate) *** | Infection AE N (Rate) *** | TRM Due to AEs |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Boll et al. [39] | 117 | early | ABVD 4x | 89% | 5-year PFS = 74.8% 5-year OS = 81.2% | p < 0.001 | 79 (68%) | 8 (7%) | 6 (5%) | 11(10%) | 5% |
| Boll et al. [41] **** | 287 | early | AVD 2x (HD13, N = 82) | 98% | 5-year PFS = 79% 5-year OS = 91% | p = NR | 31(40%) | NR | NR | 2(3%) | 1% |
| ABVD 2x (HD13, N = 67) | 99% | 5-year PFS = 78% 5-year OS = 86% | 26 (42%) | 3 (4%) | 1 (2%) | 4 (6%) | NR | ||||
| ABVD 2x (HD10, N = 70) | 96% | 5-year PFS = 79% 5-year OS = 84% | 24 (37%) | NR | 1(2%) | 5 (8%) | 3% | ||||
| ABVD 4x (HD10, N = 68) | 88% | 5-year PFS = 79% 5-year OS = 87% | 45(65%) | 5 (7%) | 7 (10%) | (8) 12% | 6% | ||||
| Evens et al. [43] ***** | 24 | advanced | ABVD x6 | 65% | 5-year FFS = 53% 5-year OS = 64% | p = 0.002 p < 0.0001 | 22 (92%) | NR | Grade 3 = 5 (20%) Grade 4 = 7 (27%) | Grade 3 = 10 (4%) Grade 4 = 10 (4%) | 9% |
| Study | n | Disease Stage | Regimen | CR | Outcomes Rate | AE N, (Rate) * | CVD AE * N (Rate) | Respiratory AE N (Rate) * | Infection AE N (Rate) * | TRM Due to AEs | Discontinuation Rate/Deviation |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ballova et al. [56] ** | 42 | advanced | BEACOPP baseline | 76% | 5-year FFTF= 46% 5-year OS = 50% *** | 34 (87%) ** | 6 (15%) | 3 (8%) | 9 (23%) | 21% | 50% |
| Halbsguth et al. [57] *** | 60 | early advanced | BACOPP | 85% | 3-year PFS = 62% 3-year OS = 66% | 52 (87%) | 5(8%) | 7(12%) | 14(23%) | 12% | 30% |
| Kolstad et al. [60] **** | 29 | early advanced | CHOP21+ irradiation | 93% | 3-year PFS = 76% Median OS not reached | 16 (51%) | 2(7%) | 1(3%) | 9(31%) | 7% | n.r. |
| Boll et al. [61] ***** | 59 | early advanced | PVAG | 78% | 3-year PFS = 58.4% 3-year OS = 66.1% | 43 (75.4%) | 4 (7%) | 4 (7%) | 13 (22.8%) | 1.7% | 36% |
| Levis et al. [63] ****** | 48 | early | VEBEMP | 98% | 5-year FFS = 79% 5-year OS = 94% | N/A | NR | NR | 2 (5%) | 0 | 3 (7%) |
| 57 | advanced | 58% | 5-year FFS= 34% 5-year OS = 32% | 8 (14%) | 2 (3%) | 15 (26%) |
| Study | N | Frailty Status | Disease Stage | Regimen | CR | Outcomes | AE Rate * | PN * | Infection AE Rate * | Discontinuation Rate ** |
|---|---|---|---|---|---|---|---|---|---|---|
| Forero-Torres et al. [65] | 26 | Frail | Early Advanced | BV monotherapy *** | 73% | Median PFS 10.5 mos Median OS not reached | NR | 26% | NR | 41% |
| Gibb et al. [66] | 31 | Frail | Advanced | BV monotherapy *** | 26% | Median PFS 6. mos Median OS 19 mos | 58% | 10% | NR | 29% |
| Yasenchak et al. [67] | 26 | Frail | Early Advanced | BV monotherapy *** | NR (ORR = 92%) | Median PFS 10 mos Median OS 77 mos | 50% | NR | NR | 42% |
| Yasenchak et al. [67] **** | 22 | Frail | Early Advanced | BV + DTIC | NR (ORR = 100%) | Median PFS 46 mos Median OS 64 mos | 37% | NR | NR | 42% |
| Schiano de Colella [69] ***** | 49 | Frail | Advanced | BV + B | 63% | 2-year PFS = 54% 2-year OS = 83% | NR | Not recorded | CMV reactivation 17 events ****** | 16% |
| Evans et al. [71] ******* | 98 | Fit | Advanced | ABVD | NR | 2-year PFS = 68% | 80% | NR | 17% | BV discontinuation 20% |
| 83 | BV-ABV | 2-year PFS =73% | 88% | NR | 37% | |||||
| Boell et al. [72] | 49 | Fit | Advanced | BV-CAP | 65% | 1-year PFS = 74% 1-year OS = 92% | NR | Not recorded | 27% | 2 events |
| Evens et al. [73] | 48 | Fit Frail ******* | Advanced | Sequential therapy | 90% | 2-year EFS = 80% 2-year OS = 93% | 42% | 4% | 16% | 35% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Milunović, V.; Hude, I.; Rinčić, G.; Galušić, D.; Grubešić, A.; Martinović, M.; Popović, N.; Divošević, S.; Brčić, K.; Međugorac, M.; et al. Clinical Dilemmas in the Treatment of Elderly Patients Suffering from Hodgkin Lymphoma: A Review. Biomedicines 2022, 10, 2917. https://doi.org/10.3390/biomedicines10112917
Milunović V, Hude I, Rinčić G, Galušić D, Grubešić A, Martinović M, Popović N, Divošević S, Brčić K, Međugorac M, et al. Clinical Dilemmas in the Treatment of Elderly Patients Suffering from Hodgkin Lymphoma: A Review. Biomedicines. 2022; 10(11):2917. https://doi.org/10.3390/biomedicines10112917
Chicago/Turabian StyleMilunović, Vibor, Ida Hude, Goran Rinčić, Davor Galušić, Aron Grubešić, Marko Martinović, Nika Popović, Sunčana Divošević, Klara Brčić, Marin Međugorac, and et al. 2022. "Clinical Dilemmas in the Treatment of Elderly Patients Suffering from Hodgkin Lymphoma: A Review" Biomedicines 10, no. 11: 2917. https://doi.org/10.3390/biomedicines10112917
APA StyleMilunović, V., Hude, I., Rinčić, G., Galušić, D., Grubešić, A., Martinović, M., Popović, N., Divošević, S., Brčić, K., Međugorac, M., Kužat, L., Strahija, D., Mrđenović, S., Mandac Smoljanović, I., Radić-Krišto, D., Gašparov, S., Aurer, I., & Ostojić Kolonić, S. (2022). Clinical Dilemmas in the Treatment of Elderly Patients Suffering from Hodgkin Lymphoma: A Review. Biomedicines, 10(11), 2917. https://doi.org/10.3390/biomedicines10112917









