Pathogenesis of Vascular Retinal Manifestations in COVID-19 Patients: A Review
Abstract
1. Introduction
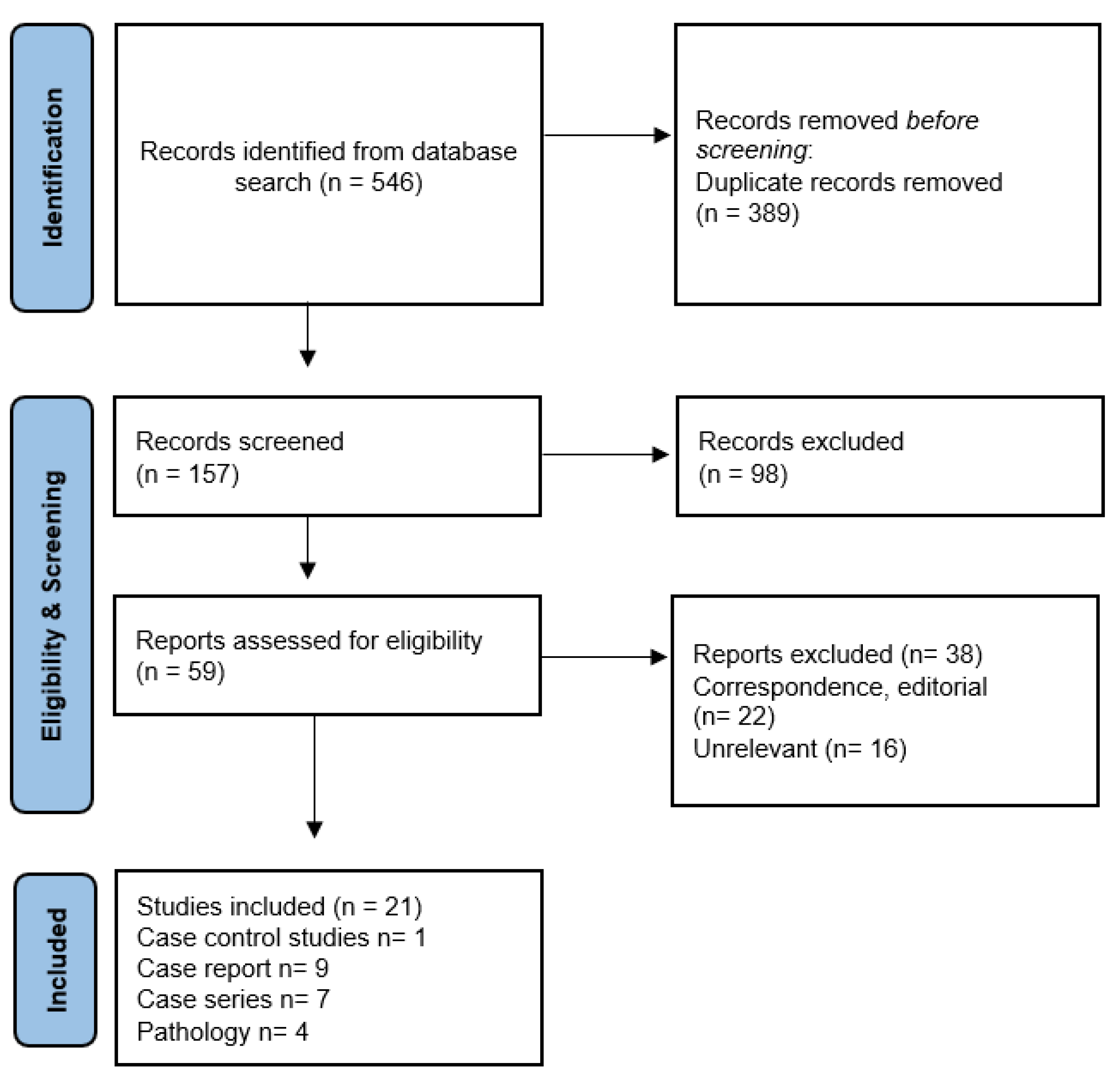
2. Methods
3. Retinal Findings in COVID-19 Patients
3.1. Retinal Microvasculopathy
| Author, Month, Year | Study Design | Location Country | Age, Gender | Total Eyes | Diagnosis | COVID Status | Biochemistry | Comorbidity |
|---|---|---|---|---|---|---|---|---|
| Marinho PM et al [14], May, 2020 | Case series | Brazil | Range 25–70 | 11 | Cotton wool spot and retinal microhemorrhages (n 4) | RT-PCR+ (n 9) Antibody+ (n 2) | NA | NA |
| Invernizzi A, et al [16], Oct, 2020 | Case series | Italy | 50 (range 23–82) | 54 | Cotton wool spot (7.4%) Retinal hemorrhages (9.25%) Dilated veins (11.1%) Tortuous veins (12.9%) | RT-PCR+ | PT (ratio) 1.19, PTT (ratio) 1.16 Fibrinogen (mg/dL) 550.2 CRP (mg/L) 26.2 Ferritin (mg/L) 662 LDH (U/L) 270.9 | Hypertension, diabetes, dyslipidemia, stroke |
| Aydemir E, et al [18], July, 2021 | Case control series | Turkey | Mean 38 | 46 cases | Dilated arteries and veins | RT-PCR+ | NA | NA |
| Pereira LA, et al [19], May 2020 | Case series | Brazil | Median 62.5 | 18 | Cotton wool spot (16.7%) Retinal flame-shaped hemorrhages (22.2%) | RT-PCR+ ICU (94.4%) | NA | Hypertension, diabetes |
| Author, Month, Year | Study Design | Location Country | Age, Gender | Total Eyes | Diagnosis | COVID Status | Biochemistry | Comorbidity |
|---|---|---|---|---|---|---|---|---|
| Sheth JU et al [20], July, 2020 | Case report | India | 52, M | 1 | HRVO | RT-PCR + No hospitalization | NA | no |
| Fonollosa A et al [21], March, 2022 | Case series | Spain | 39 (range 30–67) | 18 | CRVO (n 9) BRVO (n 4) CRVO + CRAO (n 3) CRAO (n2) | RT-PCR+ (n 18) Asymptomatic (n 3) Mild disease no hospitalization (n 8) ICU (n 4) | Elevated D-dimer and platelets | Hypertension, diabetes, dyslipidemia |
| Invernizzi A et al [22], June, 2020 | Case report | Italy | 54, F | 1 | Impending CRVO | RT-PCR+ | PT 13.8 s (INR 1.27) aPTT 36.6 s (RATIO 1.26) Fibrinogen 6.82 g/l D-dimer 426 lg/L | NA |
| Insausti-Garcia A et al [23], June, 2020 | Case report | Spain | 40, M | 1 | CRVO with papillophlebitis | IgM+ IgG+ SARS-CoV-2 (ELISA) | D-dimer 672 µg/L Fibrinogen 451 mg/dL | NA |
| Gaba WH et al [24], June, 2020 | Case report | UAE | 52, M | 1 | Bilateral CRVO + optic disc edema | RT-PCR+ Severe pneumonia DVT right leg (Doppler US) Severe dilation of right ventricle (echocardiography) | D-dimer >20 µcg/mL LDH 402 IU/L Ferritin 1518 µg/L | NA |
| Shroff D, et al [25], Mar, 2022 | Case series | India | Range 32–81 | 4 | CRVO, BRVO, CRAO, vitreous hemorrhage | PT-PCR+ Hospitalization ICU (n 3) | Elevated D-dimer and fibrinogen | no |
| Shiroma HF, et al [26], June, 2022 | Case series | Brazil | 48 (range 27–73) | 14 | CRVO (n 8) HRVO (n 1) BRVO (n 3) CRAO (n 2) | PT-PCR+ Hospitalization (n 3) | Elevated D-dimer and C-reactive protein | Hypertension, diabetes, dyslipidemia |
| Ashkenazy N et al [27], March, 2022 | Case series | USA | 32 (range 18–50) | 12 | CRVO (n 9) HRVO (n 3) | PT-PCR+ Hospitalization (n 3) | NA | no |
3.2. Retinal Venous Occlusion
3.3. Retinal Arterial Occlusion
| Author, Month, Year | Study Design | Location Country | Age, Gender | Total Patients | Diagnosis | COVID Status | Biochemistry | Comorbidity |
|---|---|---|---|---|---|---|---|---|
| Kulkarni MS et al [29], May, 2022 | Case report | India | 49, M 20, M | 2 | Choroidal artery occlusion Mixed CRAO + CRVO | RT-PCR+ Hospitalization RT-PCR+ Hospitalization | NA D-dimer: 321 ng/mL, C-reactive protein (CRP; 2.44 mg/dL), lactate dehydrogenase (LDH; 414 IU/L) | NA NA |
| Acharya S et al [30], June, 2020 | Case report | USA | 60, M | 1 | CRAO | RT-PCR+ ICU | D-dimer: 42.131 Fibrinogen: >700 C-reactive protein: 7.02 Ferritin: 324 Procalcitonin: 0.07 IL-6: 546.1 | NA |
| Gascon P et al [31], May, 2020 | Case report | France | 53, M | 1 | PAMM, AMN, deep retinal hemorrahages | RT-PCR+ Chest CT: bilateral ground glass opacities | C-reactive protein: 29 mg/L thrombocytosis | NA |
| Virgo P et al [32], June, 2020 | Case report | UK | 37 32 | 2 | PAMM, AMN | RT-PCR+ IgG+ | NA NA | NA NA |
| Montesel A et al [33], Dec, 2020 | Case report | Switzerland | 59, M | 1 | CRAO | RT-PCR+ ICU | D-Dimer: 2059 ng/mL (normal <500 ng/mL); Fibrinogen: 5.9 g/L (normal 2.0–4.0 g/L); C-reactive protein: 184 mg/L (normal <10 mg/L); IL-6: 42 pg/dl (normal <11 pg/dL) | Hypertension, hyperuricemia |
4. Detection of SARS-CoV-2 and its Receptors in Human Retina
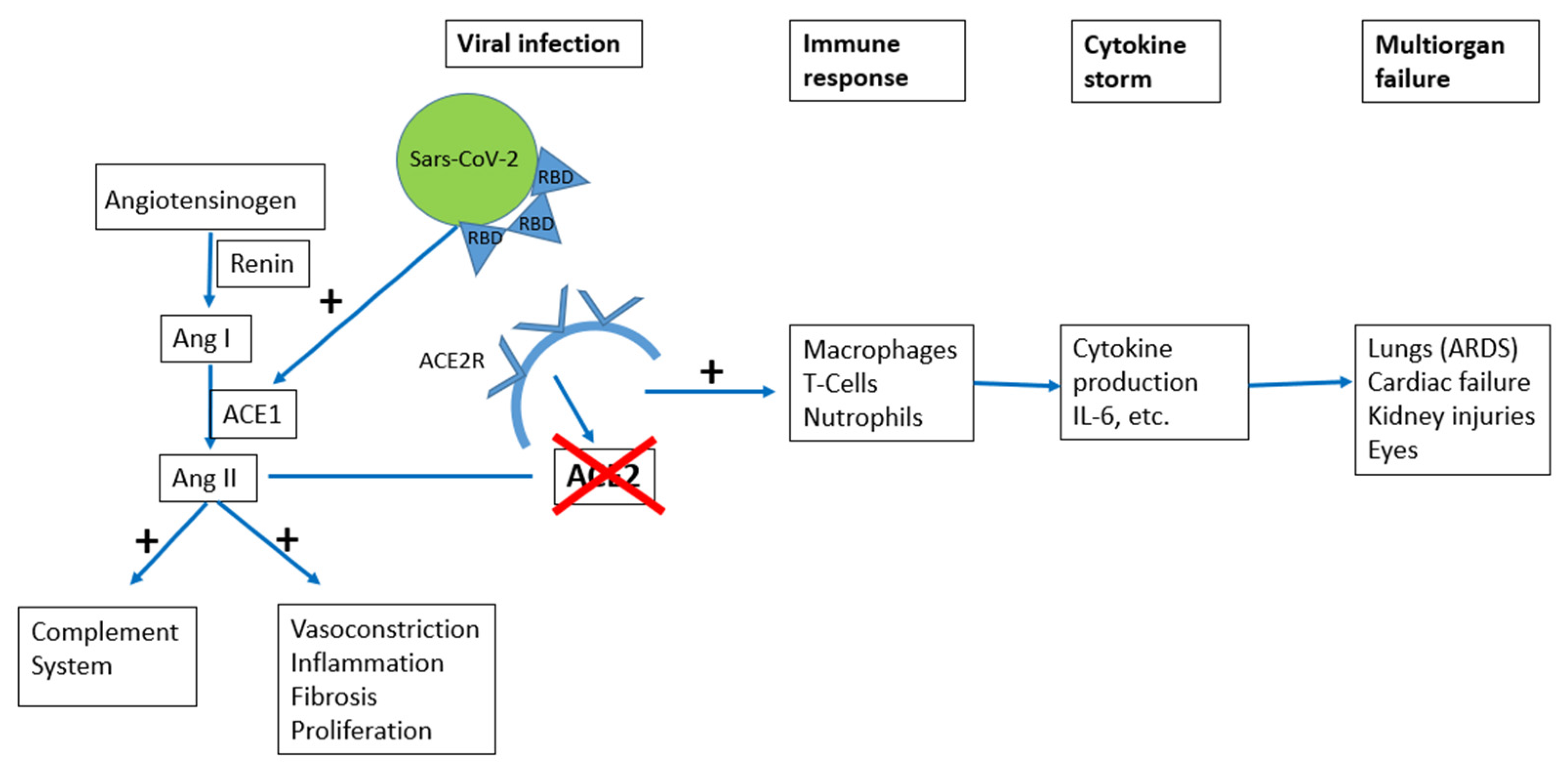
5. Pathophysiological Mechanisms
6. Discussion
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Trypsteen, W.; Van Cleemput, J.; van Snippenberg, W.; Gerlo, S.; Vandekerckhove, L. On the whereabouts of SARS-CoV-2 in the human body: A systematic review. PLoS Pathog. 2020, 16, e1009037. [Google Scholar] [CrossRef]
- Aggarwal, K.; Agarwal, A.; Jaiswal, N.; Dahiya, N.; Ahuja, A.; Mahajan, S.; Tong, L.; Duggal, M.; Singh, M.; Agrawal, R.; et al. Ocular surface manifestations of coronavirus disease 2019 (COVID-19): A systematic review and meta-analysis. PLoS ONE 2020, 15, e0241661. [Google Scholar] [CrossRef]
- Zhong, Y.; Wang, K.; Zhu, Y.; Lyu, D.; Yu, Y.; Li, S.; Yao, K. Ocular manifestations in COVID-19 patients: A systematic review and meta-analysis. Travel Med. Infect. Dis. 2021, 44, 102191. [Google Scholar] [CrossRef]
- Teo, K.Y.C.; Invernizzi, A.; Staurenghi, G.; Cheung, C.M.G. COVID-19-Related Retinal Micro-vasculopathy—A Review of Current Evidence. Am. J. Ophthalmol. 2022, 235, 98–110. [Google Scholar] [CrossRef]
- Erogul, O.; Gobeka, H.H.; Dogan, M.; Akdogan, M.; Balci, A.; Kasikci, M. Retinal microvascular morphology versus COVID-19: What to anticipate? Photodiagnosis Photodyn. Ther. 2022, 39, 102920. [Google Scholar] [CrossRef]
- Sim, R.; Cheung, G.; Ting, D.; Wong, E.; Wong, T.Y.; Yeo, I.; Wong, C.W. Retinal microvascular signs in COVID-19. Br. J. Ophthalmol. 2021, 106, 1308–1312. [Google Scholar] [CrossRef]
- Sen, S.; Kannan, N.B.; Kumar, J.; Rajan, R.P.; Kumar, K.; Baliga, G.; Reddy, H.; Upadhyay, A.; Ramasamy, K. Retinal manifestations in patients with SARS-CoV-2 infection and pathogenetic implications: A systematic review. Int. Ophthalmol. 2022, 42, 323–336. [Google Scholar] [CrossRef]
- Abrishami, M.; Emamverdian, Z.; Shoeibi, N.; Omidtabrizi, A.; Daneshvar, R.; Rezvani, R.S.; Saeedian, N.; Eslami, S.; Mazloumi, M.; Sadda, S.; et al. Optical coherence tomography angiography analysis of the retina in patients recovered from COVID-19: A case-control study. Can. J. Ophthalmol. 2021, 56, 24–30. [Google Scholar] [CrossRef]
- Riotto, E.; Mgevand, V.; Megevand, A.; Marti, C.; Pugin, J.; Stangos, A.; Marconi Archinto, L.; Sunaric Megevand, G. Retinal Manifestations in Patients with COVID-19: A Prospective Cohort Study. J. Clin. Med. 2022, 11, 1828. [Google Scholar] [CrossRef]
- Wong, T.Y.; Klein, R.; Sharrett, A.R.; Manolio, T.A.; Hubbard, L.D.; Marino, E.K.; Kuller, L.; Burke, G.; Tracy, R.P.; Polak, J.F.; et al. The prevalence and risk factors of retinal microvascular abnormalities in older persons: The Cardiovascular Health Study. Ophthalmology 2003, 110, 658–666. [Google Scholar] [CrossRef]
- Casagrande, M.; Fitzek, A.; Puschel, K.; Aleshcheva, G.; Schultheiss, H.-P.; Berneking, L.; Spitzer, M.S.; Schultheiss, M. Detection of SARS-CoV-2 in Human Retinal Biopsies of Deceased COVID-19 Patients. Ocul. Immunol. Inflam. 2020, 28, 721–725.vID. [Google Scholar] [CrossRef] [PubMed]
- Jidigam, V.K.; Singh, R.; Batoki, J.C.; Milliner, C.; Sawant, O.B.; Bonilha, V.L.; Rao, S. Histopathological assessments reveal retinal vascular changes, inflammation and gliosis in patients with lethal COVID-19. Graefe’s Arch. Clin. Exp. Ophthalmol. 2022, 260, 1275–1288. [Google Scholar] [CrossRef] [PubMed]
- Penkava, J.; Muenchhoff, M.; Badell, I.; Osterman, A.; Delbridge, C.; Niederbuchner, F.; Soliman, S.; Rudelius, M.; Graf, A.; Krebs, S.; et al. Detection of SARS-CoV-2-RNA in post-mortem samples of human eyes. Graefe’s Arch. Clin. Exp. Ophthalmol. 2022, 260, 1789–1797. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.E.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Marinho, P.M.; Marcos, A.A.A.; Romano, A.C.; Nascimento, H.; Belfort, R., Jr. Retinal Findings in patients with COVID-19. Lancet 2020, 395, 1610. [Google Scholar] [CrossRef]
- Duh, E.J. Seeking clarity on retinal findings in patients with COVID-19. Lancet 2020, 396, e39. [Google Scholar] [CrossRef]
- Invernizzi, A.; Torre, A.; Parrulli, S.; Zicarelli, F.; Schiuma, M.; Colombo, V.; Giacomelli, A.; Cigada, M.; Milazzo, L.; Ridolfo, A.; et al. Retinal findings in patients with COVID-19: Results from the SERPICO-19 study. EClinicalMedicine 2020, 27, 100550. [Google Scholar] [CrossRef]
- Invernizzi, A.; Schiuma, M.; Parrulli, S.; Torre, A.; Zicarelli, F.; Colombo, V.; Marini, S.; Villella, E.; Bertoni, A.; Antinori, S.; et al. Retinal vessels modifications in acute and post COVID-19. Sci. Rep. 2021, 11, 19373. [Google Scholar] [CrossRef]
- Aydemir, E.; Bayat, A.H.; Oren, B.; Atesoglu, H.I.; Goker, Y.S.; Ozcelik, K.C. Retinal vascular findings in patients with COVID-19. Ther. Adv. Ophthalmol. 2021, 13, 25158414211030419. [Google Scholar] [CrossRef]
- Pereira, L.A.; Soares, L.C.M.; Nascimento, P.A.; Cirillo, L.R.N.; Sakuma, H.T.; Veiga, G.L.D.; Fonseca, F.L.A.; Lima, V.L.; Abucham-Neto, J.Z. Retinal findings in hospitalized patients with severe COVID-19. Br. J. Ophthalmol. 2022, 106, 102–105. [Google Scholar] [CrossRef]
- Sheth, J.U.; Narayanan, R.; Goyal, J.; Goyal, V. Retinal vein occlusion in COVID-19: A novel entity. Indian J. Ophthalmol. 2020, 68, 2291–2293. [Google Scholar] [CrossRef] [PubMed]
- Fonollosa, A.; Hernandez-Rodriguez, J.; Cuadros, C.; Giralt, L.; Sacristan, C.; Artaraz, J.; Pelegrin, L.; Olate-Perez, A.; Romero, R.; Pastor-Idoate, S.; et al. Characterizing COVID-19-related retinal vascular occlusions: A case series and review of the literature. Retina 2022, 42, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Invernizzi, A.; Pellegrini, M.; Messenio, D.; Cereda, M.; Olivieri, P.; Brambilla, A.M.; Staurenghi, G. Impending Central Retinal Vein Occlusion in a Patient with Coronavirus Disease 2019 (COVID-19). Ocul. Immonol. Inflamm. 2020, 28, 1290–1292. [Google Scholar] [CrossRef] [PubMed]
- Insausti-Garcıa, A.; Reche-Sainz, J.A.; Ruiz-Arranz, C.; Lopez Vazquez, A.; Ferro-Osuna, M. Papillophlebitis in a COVID-19 patient: Inflammation and hypercoagulable state. Eur. J. Ophthalmol. 2020, 30, 1120672120947591. [Google Scholar] [CrossRef]
- Gaba, W.H.; Ahmed, D.; Al Nuaimi, R.K.; Dhanhani, A.A.; Eatamadi, H. Bilateral central retinal vein occlusion in a 40-year-old man with severe coronavirus disease 2019 (COVID-19) Pneumonia. Am. J. Case Rep. 2020, 21, e927691. [Google Scholar] [CrossRef]
- Shroff, D.; Kumar, S.; Naidu, A.; Gupta, C.; Shroff, C.M. Retinal vasoocclusive spectrum following COVID-19. Indian J. Ophthalmol. 2022, 70, 1412–1415. [Google Scholar]
- Shiroma, H.F.; Lima, L.H.; Shiroma, Y.B.; Kanadani, T.C.; Nobrega, M.J.; Andrade, G.; de Moraes Filho, M.N.; Penha, F.M. Retinal vascular occlusion in patients with the COVID-19 virus. Int. J. Retin. Vitr. 2022, 8, 45. [Google Scholar] [CrossRef]
- Ashkenazy, N.; Patel, N.A.; Sridhar, J.; Yannuzzi, N.A.; Belin, P.J.; Kaplan, R.; Kothari, N.; Benitez Bajandas, G.A.; Kohly, R.P.; Roizenblatt, R.; et al. Hemi- and Central Retinal Vein Occlusion Associated with COVID-19 Infection in Young Patients without Known Risk Factors. Ophthalmol. Retin. 2022, 6, 520–530. [Google Scholar] [CrossRef]
- Ucar, F.; Cetinkaya, S. Central retinal artery occlusion in a patient who contracted COVID-19 and review of similar cases. BMJ Case Rep. 2021, 14, e244181. [Google Scholar] [CrossRef]
- Kulkarni, M.S.; Rajesh, R.; Shanmugam, M.P. Ocular occlusions in two cases of COVID-19. Indian J. Ophthalmol. 2022, 70, 1825–1827. [Google Scholar] [CrossRef]
- Acharya, S.; Diamond, M.; Anwar, S.; Glaser, A.; Tyagi, P. Unique case of central retinal artery occlusion secondary to COVID-19 disease. IDCases 2020, 21, e00867. [Google Scholar] [CrossRef] [PubMed]
- Gascon, P.; Briantais, A.; Bertrand, E.; Ramtohul, P.; Comet, A.; Beylerian, M.; Sauvan, L.; Swiader, L.; Durand, J.M.; Denis, D. COVID-19-associated retinopathy: A case report. Ocul. Immunol. Inflamm. 2020, 28, 1293–1297. [Google Scholar] [CrossRef] [PubMed]
- Virgo, J.; Mohamed, M. Paracentral acute middle maculopathy and acute macular neuroretinopathy following SARS-CoV-2 infection. Eye 2020, 34, 2352–2353. [Google Scholar] [CrossRef]
- Montesel, A.; Bucolo, C.; Mouvet, V.; Moret, E.; Eandi, C.M. Case Report: Central Retinal Artery Occlusion in a COVID-19 Patient. Front. Pharmacol. 2020, 11, 588384. [Google Scholar] [CrossRef] [PubMed]
- Araujo-Silva, C.A.; Marcos, A.A.A.; Marinho, P.M.; Branco, A.M.C.; Roque, A.; Romano, A.C.; Matuoka, M.L.; Farah, M.; Burnier, M.; Moraes, N.F.; et al. Presumed SARS-CoV-2 Viral Particles in the Human Retina of Patients with COVID-19. JAMA Ophth 2021, 139, 1015–1021. [Google Scholar] [CrossRef]
- Saggioro de Figueiredo, C.; Raony, I.; Giestal-de-Araujo, E. SARS-CoV-2 Targeting the Retina: Host-virus Interaction and Possible Mechanisms of Viral Tropism. Ocul. Immunol. Inflam. 2020, 28, 1301–1304. [Google Scholar] [CrossRef]
- Zhang, H.; Penniger, J.M.; Li, Y.; Zhong, N.; Slutsky, A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: Molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020, 46, 586–590. [Google Scholar] [CrossRef]
- Matsuyama, S.; Nao, N.; Shirato, K.; Kawase, M.; Saito, S.; Takayama, I.; Nagata, N.; Sekizuka, T.; Katoh, H.; Kato, F.; et al. Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells. Proc. Natal. Acad. Sci. USA 2020, 117, 7001–7003. [Google Scholar] [CrossRef]
- Tortorici, M.A.; Walls, A.C.; Lang, Y.; Wang, C.; Li, Y.; Koerhuis, D.; Boons, G.-J.; Bosch, B.-J.; Rey, F.A.; de Groot, R.J.; et al. Structural basis for human coronavirus attachment to sialic acid receptors. Nat. Struct. Mol. Biol. 2019, 26, 481–489. [Google Scholar] [CrossRef]
- Zhou, L.; Xu, Z.; Guerra, J.; Rosenberg, A.Z.; Fenaroli, P.; Eberhart, C.G.; Duh, E.J. Expression of the SARS-CoV-2 Receptor ACE2 in Human Retina and Diabetes-Implications for Retinopathy. Investig. Ophthalmol. Vis. Sci. 2021, 62, 6. [Google Scholar] [CrossRef]
- Schnichels, S.; Rohrbach, J.M.; Bayyoud, T.; Thaler, S.; Ziemssen, F.; Hurst, J. Can SARS-CoV-2 infect the eye? An overview of the receptor status in ocular tissue. Ophtalmologe 2021, 118 (Suppl. 1), 81–84. [Google Scholar] [CrossRef] [PubMed]
- Sardu, C.; Gambardella, J.; Morelli, M.B.; Wang, X.; Marfella, R.; Santulli, G. Hypertension, Thrombosis, Kidney Failure, and Diabetes: Is COVID-19 an Endothelial Disease? A Comprehensive Evaluation of Clinical and Basic Evidence. J. Clin. Med. 2020, 9, 1417. [Google Scholar] [CrossRef] [PubMed]
- Vardecchia, P.; Cavallini, C.; Spanevello, A.; Angeli, F. COVID-19: ACE2 centric infective disease? Hypertension 2020, 76, 294–299. [Google Scholar] [CrossRef]
- Reinhold, A.; Tzankov, A.; Matter, M.S.; Mihic-Probst, D.; Scholl, H.P.N.; Meyer, P. Ocular Pathology and Occasionally Detectable Intraocular Severe Acute Respiratory Syndrome Coronavirus-2 RNA in Five Fatal Coronavirus Disease-19 Cases. Ophthalmic Res. 2021, 64, 785–792. [Google Scholar] [CrossRef]
- Connors, J.M.; Levy, J.H. COVID-19 and its implications for thrombosis and anticoagulation. Blood 2020, 135, 2033–2040. [Google Scholar] [CrossRef] [PubMed]
- Tang, N.; Li, D.; Wang, X.; Sun, Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 844–847. [Google Scholar] [CrossRef]
- Dumitrascu, O.; Volod, O.; Bose, S.; Wang, Y.; Biousse, V.; Lyden, P.D. Acute ophthalmic artery occlusion in COVID-19 patient on apixaban. J. Stroke Cerebrovasc. Dis. 2020, 29, 104982. [Google Scholar] [CrossRef]
- Giamarellos-Bourboulis, E.J.; Netea, M.G.; Rovina, N.; Akinosoglou, K.; Antoniadou, A.; Antonakos, N.; Damoraki, G.; Gkavogianni, T.; Adami, M.-E.; Katsaounou, P.; et al. Complex immune dysregulation in COVID-19 patients with severe respiratory failure. Cell Host Microbe 2020, 27, 992–1000. [Google Scholar] [CrossRef]
- Siddiqi, H.; Libby, P.; Ridker, P.M. COVID-19—A vascular disease. Trends Cardiovasc. Med. 2021, 31, 1–5. [Google Scholar] [CrossRef]
- Syed, F.; Relich, R.F.; Russell, P.M.; Zhang, S.; Zimmerman, M.K.; Yu, Q. Excessive Matrix Metalloproteinase-1 and Hyperactivation of Endothelial Cells Occurred in COVID-19 Patients and Were Associated with the Severity of COVID-19. J. Infect. Dis. 2021, 224, 60–69. [Google Scholar] [CrossRef]
- Seah, I.; Agrawal, R. Can the coronavirus disease 2019 (COVID-19) affect the eyes? a review of coronaviruses and ocular implications in humans and animals. Ocul. Immunol. Inflamm. 2020, 28, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Neri, P.; Pichi, F. SARS-CoV-2 and the Eye: The Pandora’s Box of Ocular Immunology. J. Ocul. Pharmacol. Ther. 2021, 37, 502–509. [Google Scholar] [CrossRef] [PubMed]
- De Roquetaillade, C.; Chousterman, B.G.; Tomasoni, D.; Zeitouni, M.; Houdart, E.; Guedon, A.; Reiner, P.; Bordier, R.; Gayat, E.; Montalescot, G.; et al. Unusual arterial thrombotic events in COVID-19 patients. Int. J. Cardiol. 2021, 323, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Raony, I.; Saggioro de Figueiredo, C. Retinal outcomes of COVID-19: Possible role of CD147 and cytokine storm in infected patients with diabetes mellitus. Diabetes Res. Clin. Pr. 2020, 165, 108280. [Google Scholar] [CrossRef]
- Weger, M.; Pichler, T.; Franke, G.H.; Haas, A.; Thaler, H.V.; Kraigher-Krainer, N.; Groselj-Strele, A.; Wedrich, A.; Rabensteiner, D.F. Assessment of vision-related quality of life in patients with central retinal artery occlusion. Retina 2014, 34, 539–545. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Alessandro, E.; Kawasaki, A.; Eandi, C.M. Pathogenesis of Vascular Retinal Manifestations in COVID-19 Patients: A Review. Biomedicines 2022, 10, 2710. https://doi.org/10.3390/biomedicines10112710
D’Alessandro E, Kawasaki A, Eandi CM. Pathogenesis of Vascular Retinal Manifestations in COVID-19 Patients: A Review. Biomedicines. 2022; 10(11):2710. https://doi.org/10.3390/biomedicines10112710
Chicago/Turabian StyleD’Alessandro, Elisa, Aki Kawasaki, and Chiara M. Eandi. 2022. "Pathogenesis of Vascular Retinal Manifestations in COVID-19 Patients: A Review" Biomedicines 10, no. 11: 2710. https://doi.org/10.3390/biomedicines10112710
APA StyleD’Alessandro, E., Kawasaki, A., & Eandi, C. M. (2022). Pathogenesis of Vascular Retinal Manifestations in COVID-19 Patients: A Review. Biomedicines, 10(11), 2710. https://doi.org/10.3390/biomedicines10112710





