Synthetic Mono-Carbonyl Curcumin Analogues Attenuate Oxidative Stress in Mouse Models
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Animals
2.2. In Vitro Antioxidant Activity
2.3. Acute Toxicity Study
2.4. Experimental Design and Animal Dosing
2.5. Behavioral Studies
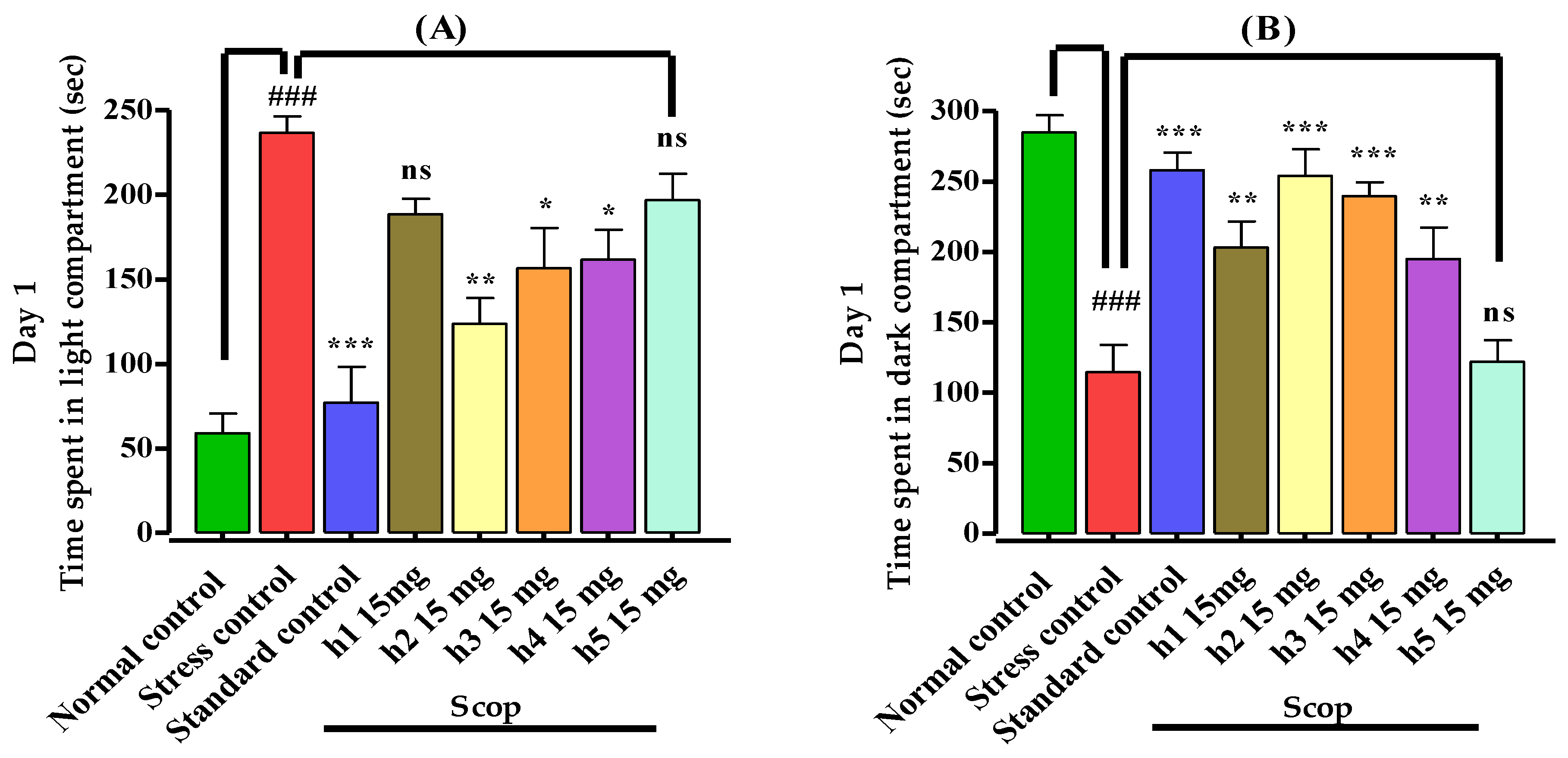
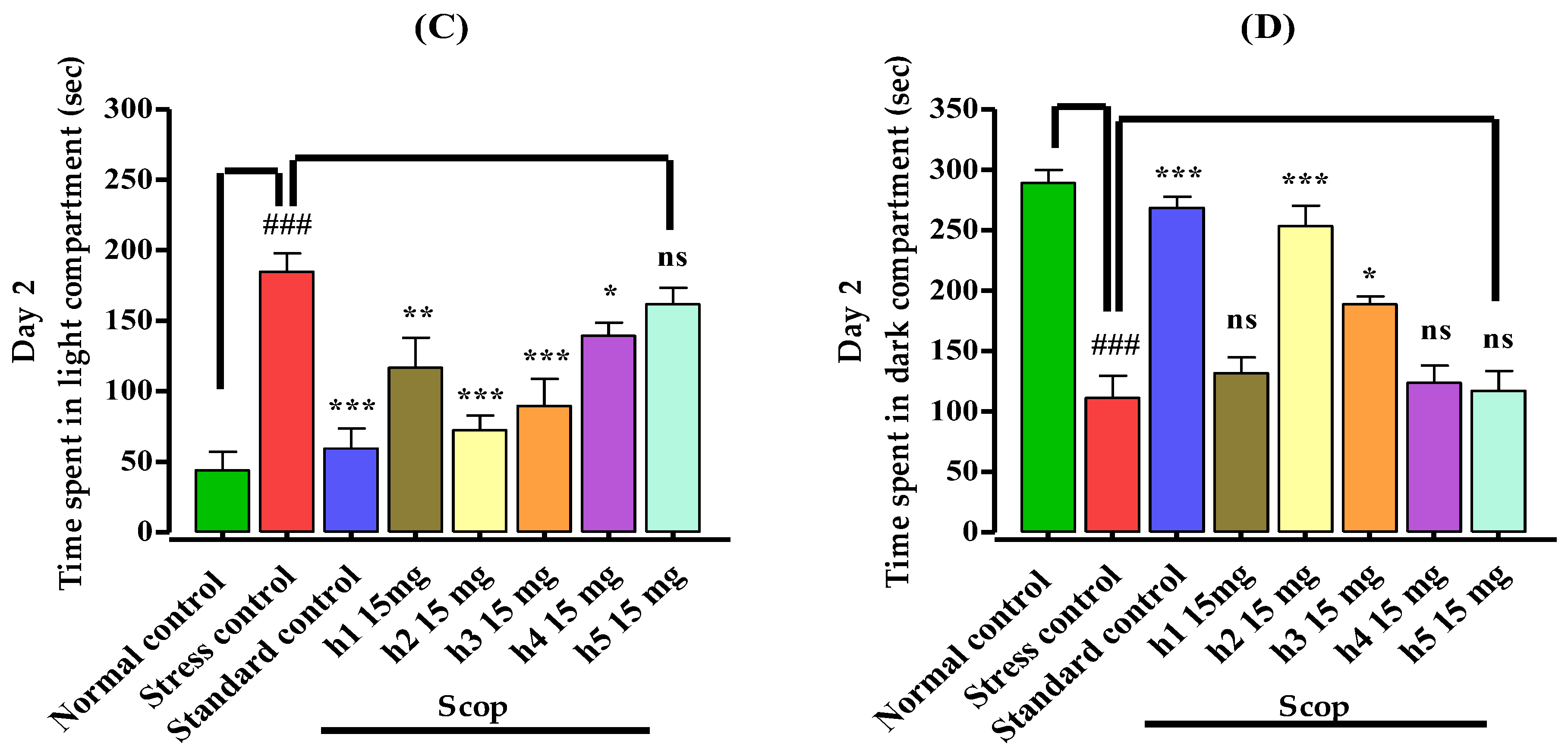
2.5.1. Light–Dark Box
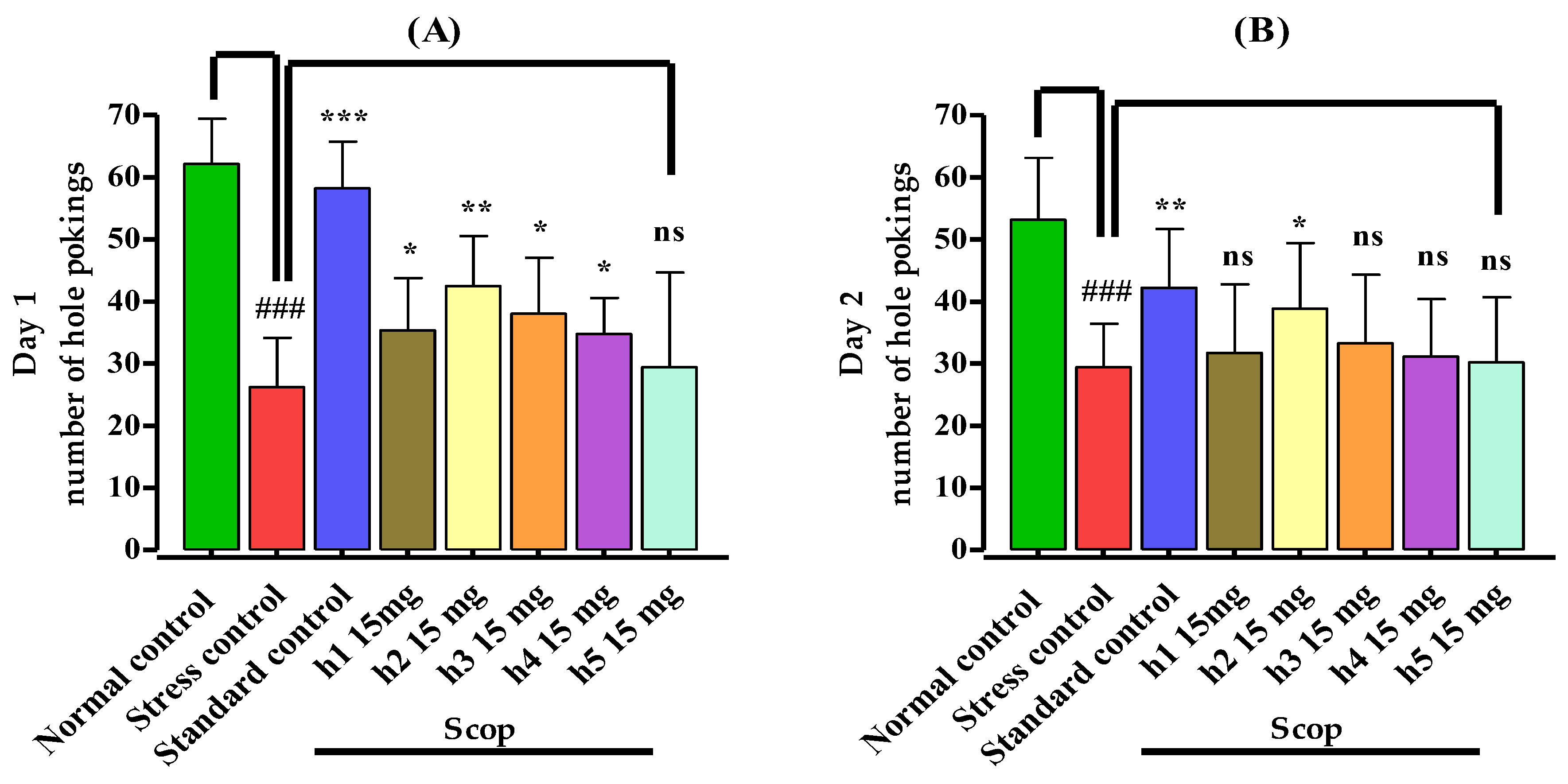
2.5.2. Hole Board Test
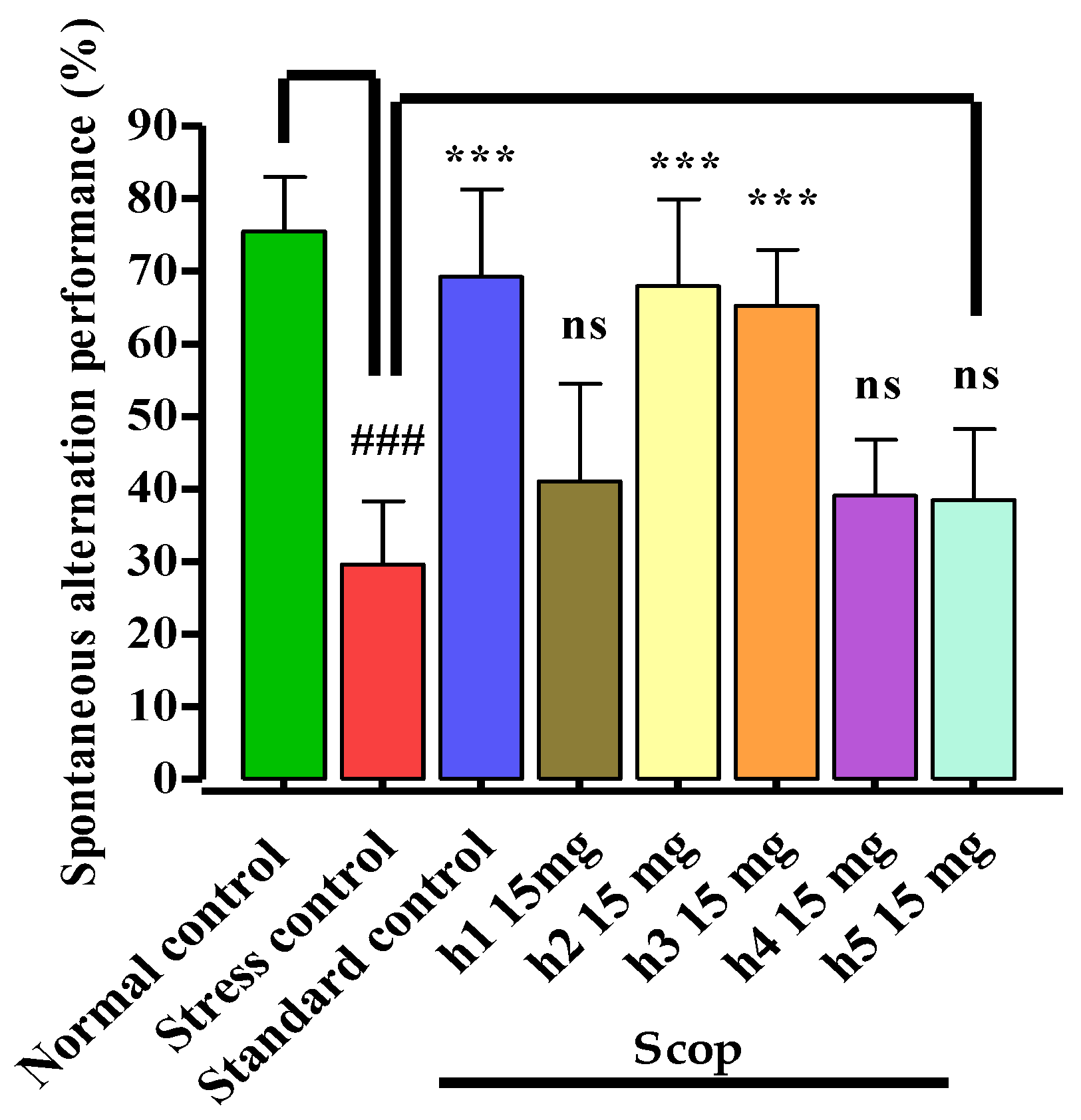
2.5.3. Y-Maze Test
2.6. Assessment of Biochemical Parameters and Biomarker Study
2.6.1. Measurement of Malondialdehyde (MDA) Level
2.6.2. Catalase (CAT) Activity
2.6.3. Superoxide Dismutase (SOD) Activity
2.6.4. Measurement of Glutathione (GSH) Activity
2.7. Statistical Analysis
3. Results
3.1. In Vitro Antioxidant Activity
3.2. Acute Toxicity
3.3. In Vivo Behavioral Studies
3.3.1. Light–Dark Box Apparatus
3.3.2. Hole Board Assay
3.3.3. Y-Maze Test
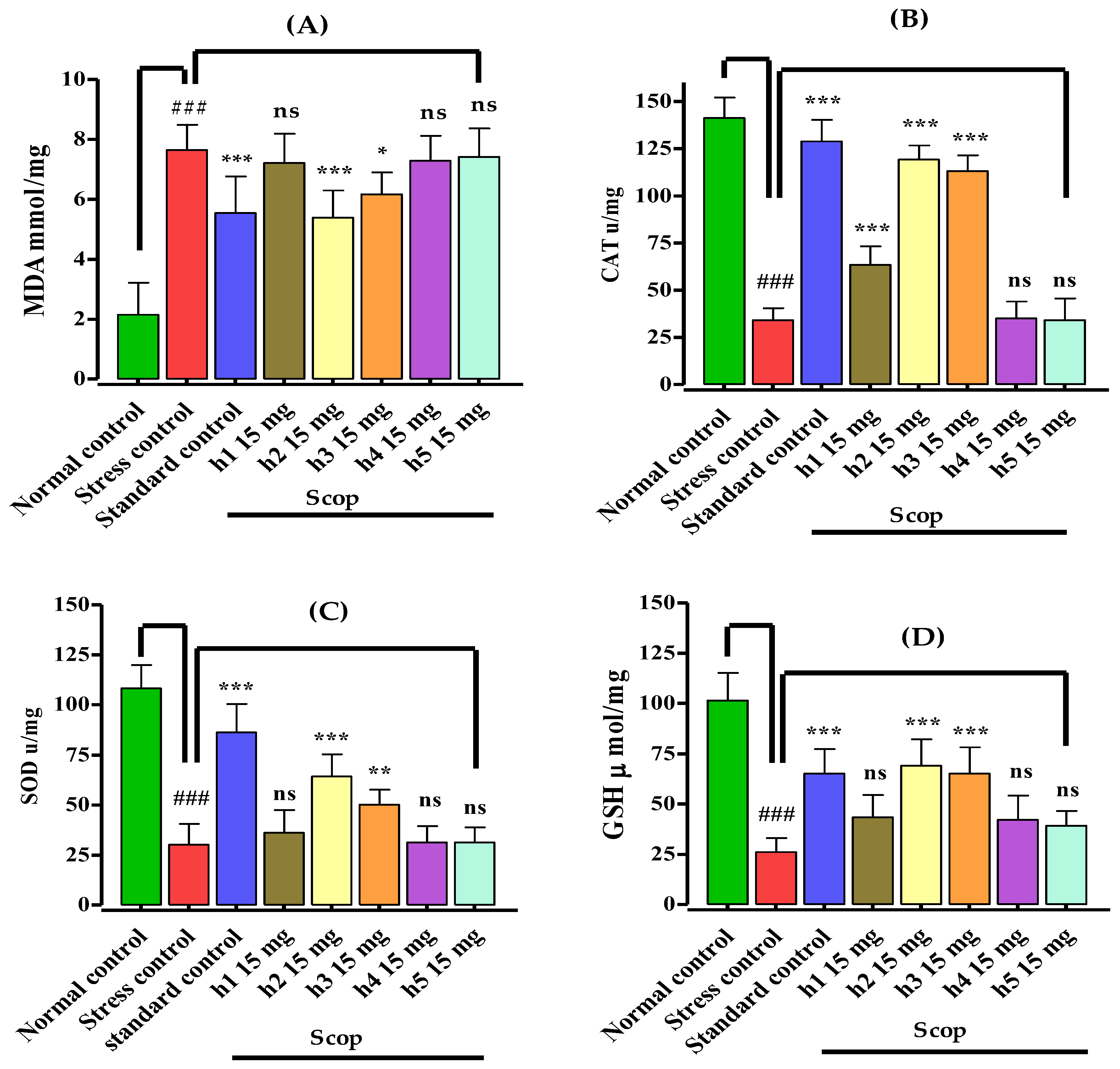
3.4. Assessment of Biochemical Parameters and Biomarker Study
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thomas, D.X.; Bajaj, S.; McRae-McKee, K.; Hadjichrysanthou, C.; Anderson, R.M.; Collinge, J. Association of TDP-43 proteinopathy, cerebral amyloid angiopathy, and Lewy bodies with cognitive impairment in individuals with or without Alzheimer’s disease neuropathology. Sci. Rep. 2020, 10, 14579. [Google Scholar] [CrossRef]
- Capatina, L.; Todirascu-Ciornea, E.; Napoli, E.M.; Ruberto, G.; Hritcu, L.; Dumitru, G. Thymus vulgaris essential oil protects zebrafish against cognitive dysfunction by regulating cholinergic and antioxidants systems. Antioxidants 2020, 9, 1083. [Google Scholar] [CrossRef]
- Stanciu, G.D.; Luca, A.; Rusu, R.N.; Bild, V.; Ioan, S.; Chiriac, B.; Solcan, C.; Bild, W.; Ababei, D.C. Alzheimer’s Disease Pharmacotherapy in Relation to Cholinergic System Involvement. Biomolecules 2020, 10, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shal, B.; Khan, A.; Khan, A.U.; Ullah, R.; Ali, G.; Islam, S.U.; Haq, I.U.; Ali, H.; Seo, E.K.; Khan, S. Alleviation of memory deficit by bergenin via the regulation of reelin and Nrf-2/NF-κB pathway in transgenic mouse model. Int. J. Mol. Sci. 2021, 22, 6603. [Google Scholar] [CrossRef] [PubMed]
- Hussain, H.; Ahmad, S.; Wadood, S.; Shah, A.; Ghias, M.; Ullah, A.; Rahman, S.U.; Kamal, Z.; Khan, F.A.; Khan, N.M.; et al. Neuroprotective Potential of Synthetic Mono-Carbonyl Curcumin Analogs Assessed by Molecular Docking Studies. Molecules 2021, 26, 7168. [Google Scholar] [CrossRef] [PubMed]
- Hussain, H.; Ahmad, S.; Wadood, S.; Shah, A.; Ullah, A.; Ali, N.; Almehmadi, M.; Ahmad, M.; Ali, A.; Khalil, K.; et al. Attenuation of Scopolamine-Induced Amnesia via Cholinergic Modulation in Mice by Synthetic Curcumin Analogs. Molecules 2022, 27, 2468. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, K.; Tomlinson, B.E.; Blessed, G.; Gibson, P.H.; Perry, R.H. Correlation of cholinergic abnormalities with senile plaques and mental test scores in senile dementia. Br. Med. J. 1978, 2, 1457–1459. [Google Scholar] [CrossRef] [Green Version]
- Sugimoto, H. The new approach in development of anti-Alzheimer’s disease drugs via the cholinergic hypothesis. Chem. Biol. Interact. 2008, 175, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Hammond, T.C.; Xing, X.; Wang, C.; Ma, D.; Nho, K.; Crane, P.K.; Elahi, F.; Ziegler, D.A.; Liang, G.; Cheng, Q.; et al. β-amyloid and tau drive early Alzheimer’s disease decline while glucose hypometabolism drives late decline. Commun. Biol. 2020, 3, 352. [Google Scholar] [CrossRef] [PubMed]
- Alisi, I.O.; Uzairu, A.; Abechi, S.E.; Idris, S.O. Quantitative structure activity relationship analysis of coumarins as free radical scavengers by genetic function algorithm. Phys. Chem. Res. 2018, 6, 208–222. [Google Scholar] [CrossRef]
- Shoaib, M.; Shah, S.W.A.; Ali, N.; Shah, I.; Naveed Umar, M.; Shafiullah; Ayaz, M.; Tahir, M.N.; Akhtar, S. In vitro enzyme inhibition potentials and antioxidant activity of synthetic flavone derivatives. J. Chem. 2015, 2015, 516878. [Google Scholar] [CrossRef] [Green Version]
- Valko, M.; Rhodes, C.J.; Moncol, J.; Izakovic, M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Biol. Interact. 2006, 160, 1–40. [Google Scholar] [CrossRef]
- Ighodaro, O.M.; Akinloye, O.A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alexandria J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef] [Green Version]
- Spiegel, M.; Kapusta, K.; Kołodziejczyk, W.; Saloni, J.; Zbikowska, B.; Hill, G.A.; Sroka, Z. Antioxidant Activity of Selected Phenolic Acids–Ferric Reducing Antioxidant Power Assay and QSAR Analysis of the Structural Features. Molecules 2020, 25, 3088. [Google Scholar] [CrossRef] [PubMed]
- Kancheva, V.D.; Dettori, M.A.; Fabbri, D.; Alov, P.; Angelova, S.E.; Slavova-Kazakova, A.K.; Carta, P.; Menshov, V.A.; Yablonskaya, O.I.; Trofimov, A.V.; et al. Natural chain-breaking antioxidants and their synthetic analogs as modulators of oxidative stress. Antioxidants 2021, 10, 624. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Chang, X.; Lang, M. Iron homeostasis disorder and alzheimer’s disease. Int. J. Mol. Sci. 2021, 22, 12442. [Google Scholar] [CrossRef]
- Persson, T.; Popescu, B.O.; Cedazo-Minguez, A. Oxidative stress in Alzheimer’s disease: Why did antioxidant therapy fail? Oxid. Med. Cell. Longev. 2014, 2014, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, Y.; Zhao, B. Oxidative Stress and the Pathogenesis of Alzheimer’s Disease. Oxid. Med. Cell. Longev. 2013, 2013, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, W.J.; Zhang, X.; Chen, W.W. Role of oxidative stress in Alzheimer’s disease (review). Biomed. Rep. 2016, 4, 519–522. [Google Scholar] [CrossRef] [Green Version]
- Vanzella, C.; Neves, J.D.; Vizuete, A.F.; Aristimunha, D.; Kolling, J.; Longoni, A.; Gonçalves, C.A.S.; Wyse, A.T.S.; Netto, C.A. Treadmill running prevents age-related memory deficit and alters neurotrophic factors and oxidative damage in the hippocampus of Wistar rats. Behav. Brain Res. 2017, 334, 78–85. [Google Scholar] [CrossRef]
- Nunomura, A.; Castellani, R.J.; Zhu, X.; Moreira, P.I.; Perry, G.; Smith, M.A. Involvement of oxidative stress in Alzheimer disease. J. Neuropathol. Exp. Neurol. 2006, 65, 631–641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pathway, B.T.; Baek, S.Y.; Li, F.Y.; Kim, D.H.; Kim, S.J.; Kim, M.R. Enteromorpha prolifera Extract Improves Memory in Scopolamine-Treated Mice via Downregulating Amyloid-β Expression and Upregulating. Antioxidants 2020, 9, 620. [Google Scholar]
- Slavova-Kazakova, A.; Angelova, S.; Fabbri, D.; Antonietta Dettori, M.; Kancheva, V.D.; Delogu, G. Antioxidant properties of novel curcumin analogues: A combined experimental and computational study. J. Food Biochem. 2021, 45, e13584. [Google Scholar] [CrossRef] [PubMed]
- Rabiei, Z.; Setorki, M. Effect of hydroalcoholic echium amoenum extract on scopolamine-induced learning and memory impairment in rats. Pharm. Biol. 2018, 56, 672–677. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fronza, M.G.; Baldinotti, R.; Fetter, J.; Sacramento, M.; Sabedra, F.S.; Seixas, F.K.; Collares, T.; Alves, D.; Pratico, D.; Savegnago, L. QTC-4-MeOBnE rescues scopolamine-induced memory deficits in mice by targeting oxidative stress, neuronal plasticity and apoptosis QTC-4-MeOBnE rescues scopolamine-induced memory deficits in mice by targeting oxidative stress, neuronal plasticity and apo. ACS Chem. Neurosci. 2020, 11, 1259–1269. [Google Scholar] [CrossRef] [PubMed]
- Mošovská, S.; Petáková, P. Antioxidant properties of curcuminoids isolated from Curcuma longa L. Acta Chim. Slovaca 2016, 9, 130–135. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.E.; Song, H.S.; Park, M.N.; Kim, S.H.; Shim, B.S.; Kim, B.; Lee, J.E. Ethanol Extract of Oldenlandia diffusa Herba Attenuates Scopolamine-Induced Cognitive Impairments in Mice via Activation of BDNF, P-CREB and Inhibition of Acetylcholinesterase. Int. J. Mol. Sci. 2018, 19, 996. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castro, A.; Martinez, A. Targeting Beta-Amyloid Pathogenesis Through Acetylcholinesterase Inhibitors. Curr. Pharm. Des. 2006, 12, 4377–4387. [Google Scholar] [CrossRef] [PubMed]
- Orhan, G.; Orhan, I.; Sener, B. Recent Developments in Natural and Synthetic Drug Research for Alzheimers Disease. Lett. Drug Des. Discov. 2006, 3, 268–274. [Google Scholar] [CrossRef]
- Sadiq, A.; Mahmood, F.; Ullah, F.; Ayaz, M.; Ahmad, S.; Haq, F.U.; Khan, G.; Jan, M.S. Synthesis, anticholinesterase and antioxidant potentials of ketoesters derivatives of succinimides: A possible role in the management of alzheimer’s. Chem. Cent. J. 2015, 9, 31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, W.; Wang, S.Y. Antioxidant Activity and Phenolic Compounds in Selected Herbs. J. Agric. Food Chem. 2001, 49, 5165–5170. [Google Scholar] [CrossRef] [PubMed]
- Von Gadow, A.; Joubert, E.; Hansmann, C.F. Comparison of the Antioxidant Activity of Aspalathin with That of Other Plant Phenols of Rooibos Tea (Aspalathus linearis), r-Tocopherol, BHT, and BHA. J. Agric. Food Chem. 1997, 45, 632–638. [Google Scholar] [CrossRef]
- Mbese, Z.; Khwaza, V.; Aderibigbe, B.A. Curcumin and Its Derivatives as Potential Therapeutic Agents in Prostate, Colon and Breast Cancers. Molecules 2019, 24, 4386. [Google Scholar] [CrossRef] [Green Version]
- Liang, G.; Yang, S.; Jiang, L.; Zhao, Y.; Shao, L.; Xiao, J.; Ye, F.; Li, Y.; Li, X. Synthesis and anti-bacterial properties of mono-carbonyl analogues of curcumin. Chem. Pharm. Bull. 2008, 56, 162–167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forms, P. Curcumin: Biological Activities and Modern. Antibiotics 2022, 11, 135. [Google Scholar]
- Ahmed, T.; Gilani, A.H. Inhibitory effect of curcuminoids on acetylcholinesterase activity and attenuation of scopolamine-induced amnesia may explain medicinal use of turmeric in Alzheimer’s disease. Pharmacol. Biochem. Behav. 2009, 91, 554–559. [Google Scholar] [CrossRef]
- Lee, W.; Loo, C.; Bebawy, M.; Luk, F.; Mason, R.S. Curcumin and its Derivatives: Their Application in Neuropharmacology and Neuroscience in the 21 st Century. Curr. Neuropharmacol. 2013, 11, 338–378. [Google Scholar] [CrossRef] [Green Version]
- Naqvi, F.; Haider, S.; Naqvi, F.; Saleem, S.; Perveen, T.; Batool, Z. A comparative study showing greater effects of curcumin compared to donepezil on memory function in rats. Pak. J. Pharm. Sci. 2019, 32, 53–60. [Google Scholar]
- Zhang, Y.; Li, L.; Zhang, J. Curcumin in antidepressant treatments: An overview of potential mechanisms, pre-clinical/clinical trials and ongoing challenges. Basic Clin. Pharmacol. Toxicol. 2020, 127, 243–253. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Barry, J.M.; Costall, B.; Kelly, M.E.; Naylor, R.J. Withdrawal syndrome following subchronic treatment with anxiolytic agents. Pharmacol. Biochem. Behav. 1987, 27, 239–245. [Google Scholar] [CrossRef]
- Kaufmann, F.N.; Gazal, M.; Bastos, C.R.; Kaster, M.P.; Ghisleni, G. Curcumin in depressive disorders: An overview of potential mechanisms, preclinical and clinical findings. Eur. J. Pharmacol. 2016, 784, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Durcan, M.J.; Lister, R.G. Time course of ethanol’s effects on locomotor activity, exploration and anxiety in mice. Psychopharmacology 1988, 96, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Mushtaq, A.; Anwar, R.; Ahmad, M. Lavandula stoechas (L) a very potent antioxidant attenuates dementia in scopolamine induced memory deficit mice. Front. Pharmacol. 2018, 9, 1375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghias, M.; Wadood, S.; Shah, A.; Al-joufi, F.A.; Shoaib, M.; Muhammad, S.; Shah, M.; Ahmed, M.N.; Zahoor, M. In Vivo Antistress Effects of Synthetic Flavonoids in Mice: Behavioral and Biochemical Approach. Molecules 2022, 27, 1402. [Google Scholar] [CrossRef] [PubMed]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Sinha, A.K. Colorimetric assay of catalase. Anal. Biochem. 1972, 47, 389–394. [Google Scholar] [CrossRef]
- Kakkar, P.; Das, B.; Viswanathan, P.N. A modified spectrophotometric assay of superoxide dismutase. Indian J. Biochem. Biophys. 1984, 21, 130–132. [Google Scholar]
- Moron, M.S.; Depierre, J.W.; Mannervik, B. Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. Biochim. Biophys. Acta 1979, 582, 67–78. [Google Scholar] [CrossRef]
- Rawat, N.; Ahmad, Y.; Kant, R.; Singh, T.G.; Arora, G.; Dhiman, S.; Singh, T.G. Anti-amnesic activity of ascophyllum nodosum polyphenols on trihexyphenidyl induced amnesia. Plant Arch. 2019, 19, 2141–2147. [Google Scholar]
- Ramos, S.; Filho, S.; Haroldo, J.; Barbosa, O.; Rondinoni, C.; Carlos, A.; Ernesto, C.; Salmon, G.; Kilza, N.; Ferriolli, E.; et al. Neuro-degeneration profile of Alzheimer’s patients: A brain morphometry study. NeuroImage Clin. J. 2017, 15, 15–24. [Google Scholar] [CrossRef]
- Rajesh, V.; Riju, T.; Venkatesh, S.; Babu, G. Memory enhancing activity of Lawsonia inermis Linn. leaves against scopolamine induced memory impairment in Swiss albino mice. Orient. Pharm. Exp. Med. 2017, 17, 127–142. [Google Scholar] [CrossRef]
- Zafar, R.; Ullah, H.; Zahoor, M.; Sadiq, A. Isolation of bioactive compounds from Bergenia ciliata (haw.) Sternb rhizome and their antioxidant and anticholinesterase activities. BMC Complement. Altern. Med. 2019, 19, 296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khushwant, S. Bhullar Curcumin and Its Carbocyclic Analogs: Structure-Activity in Relation to Antioxidant and Selected Biological Properties. Molecules 2013, 18, 5389–5404. [Google Scholar] [CrossRef] [Green Version]
- Anand, P.; Thomas, S.G.; Kunnumakkara, A.B.; Sundaram, C.; Harikumar, K.B.; Sung, B.; Tharakan, S.T.; Misra, K.; Priyadarsini, I.K.; Rajasekharan, K.N.; et al. Biological activities of curcumin and its analogues (Congeners) made by man and Mother Nature. Biochem. Pharmacol. 2008, 76, 1590–1611. [Google Scholar] [CrossRef] [PubMed]
- Bourin, M.; Hascoët, M. The mouse light/dark box test. Eur. J. Pharmacol. 2003, 463, 55–65. [Google Scholar] [CrossRef]
- Ru, M.; Liu, H. Association between Y-Maze Acquisition Learning and Major Histocompatibility Complex Class II Polymorphisms in Mice. BioMed Res. Int. 2018, 2018, 6381932. [Google Scholar] [CrossRef] [Green Version]
- Walf, A.A.; Frye, C.A. The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nat. Protoc. 2007, 2, 322–328. [Google Scholar] [CrossRef] [Green Version]
- Kumar, N.; Singh, N.; Jaggi, A.S. Anti-stress effects of cilnidipine and nimodipine in immobilization subjected mice. Physiol. Behav. 2012, 105, 1148–1155. [Google Scholar] [CrossRef]
- Rubab, S.; Naeem, K.; Rana, I.; Khan, N.; Afridi, M.; Ullah, I.; Shah, F.A.; Sarwar, S.; Din, F.; Choi, H.I.; et al. Enhanced neuroprotective and antidepressant activity of curcumin-loaded nanostructured lipid carriers in lipopolysaccharide-induced depression and anxiety rat model. Int. J. Pharm. 2021, 603, 120670. [Google Scholar] [CrossRef]
- Lee, G.Y.; Lee, C.; Park, G.H.; Jang, J.H. Amelioration of Scopolamine-Induced Learning and Memory Impairment by α-Pinene in C57BL/6 Mice. Evid.-Based Complement. Altern. Med. 2017, 2017, 4926815. [Google Scholar] [CrossRef] [Green Version]
- Budzynska, B.; Boguszewska-czubara, A.; Kruk-slomka, M.; Skalicka-wozniak, K.; Michalak, A.; Musik, I.; Biala, G. Effects of imperatorin on scopolamine-induced cognitive impairment and oxidative stress in mice. Psychopharmacology 2015, 232, 931–942. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.H.; Lee, H.K.; Kim, J.A.; Hong, S.I.; Kim, H.C.; Jo, T.H.; Park, Y.I.; Lee, C.K.; Kim, Y.B.; Lee, S.Y.; et al. Neuroprotective effects of chlorogenic acid on scopolamine-induced amnesia via anti-acetylcholinesterase and anti-oxidative activities in mice. Eur. J. Pharmacol. 2010, 649, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Saxena, G.; Singh, S.P.; Agrawal, R.; Nath, C. Effect of donepezil and tacrine on oxidative stress in intracerebral streptozotocin-induced model of dementia in mice. Eur. J. Pharmacol. 2008, 581, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Umukoro, S.; Adewole, F.A.; Eduviere, A.T.; Aderibigbe, A.O.; Onwuchekwa, C. Free radical scavenging effect of donepezil as the possible contribution to its memory enhancing activity in mice. Drug Res. 2014, 64, 236–239. [Google Scholar] [CrossRef]
- Nade, V.S.; Kawale, L.A.; Naik, R.A.; Yadav, A.V. Adaptogenic effect of Morus alba on chronic footshock-induced stress in rats. Indian J. Pharmacol. 2009, 41, 246–251. [Google Scholar] [CrossRef] [Green Version]
- Hatcher, H.; Planalp, R.; Cho, J.; Torti, F.M.; Torti, S.V. Curcumin: From ancient medicine to current clinical trials. Cell. Mol. Life Sci. 2008, 65, 1631–1652. [Google Scholar] [CrossRef] [Green Version]
- Du, C.N.; Min, A.Y.; Kim, H.J. Deer bone extract prevents against scopolamine-induced memory impairment in mice. J. Med. Food 2015, 18, 157–165. [Google Scholar] [CrossRef] [Green Version]
- First, M.; Gil-Ad, I.; Taler, M.; Tarasenko, I.; Novak, N.; Weizman, A. The effects of fluoxetine treatment in a chronic mild stress rat model on depression-related behavior, brain neurotrophins and ERK expression. J. Mol. Neurosci. 2011, 45, 246–255. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Zhu, Y.; Wang, M.; Jing, G.; Zhu, R.; Wang, S. Antidepressant effects of curcumin and HU-211 coencapsulated solid lipid nanoparticles against corticosterone-induced cellular and animal models of major depression. Int. J. Nanomed. 2016, 11, 4975–4990. [Google Scholar] [CrossRef] [Green Version]
- Teiten, M.H.; Dicato, M.; Diederich, M. Hybrid curcumin compounds: A new strategy for cancer treatment. Molecules 2014, 19, 20839–20863. [Google Scholar] [CrossRef] [PubMed]
- Tabanelli, R.; Brogi, S.; Calderone, V. Improving curcumin bioavailability: Current strategies and future perspectives. Pharmaceutics 2021, 13, 1715. [Google Scholar] [CrossRef] [PubMed]
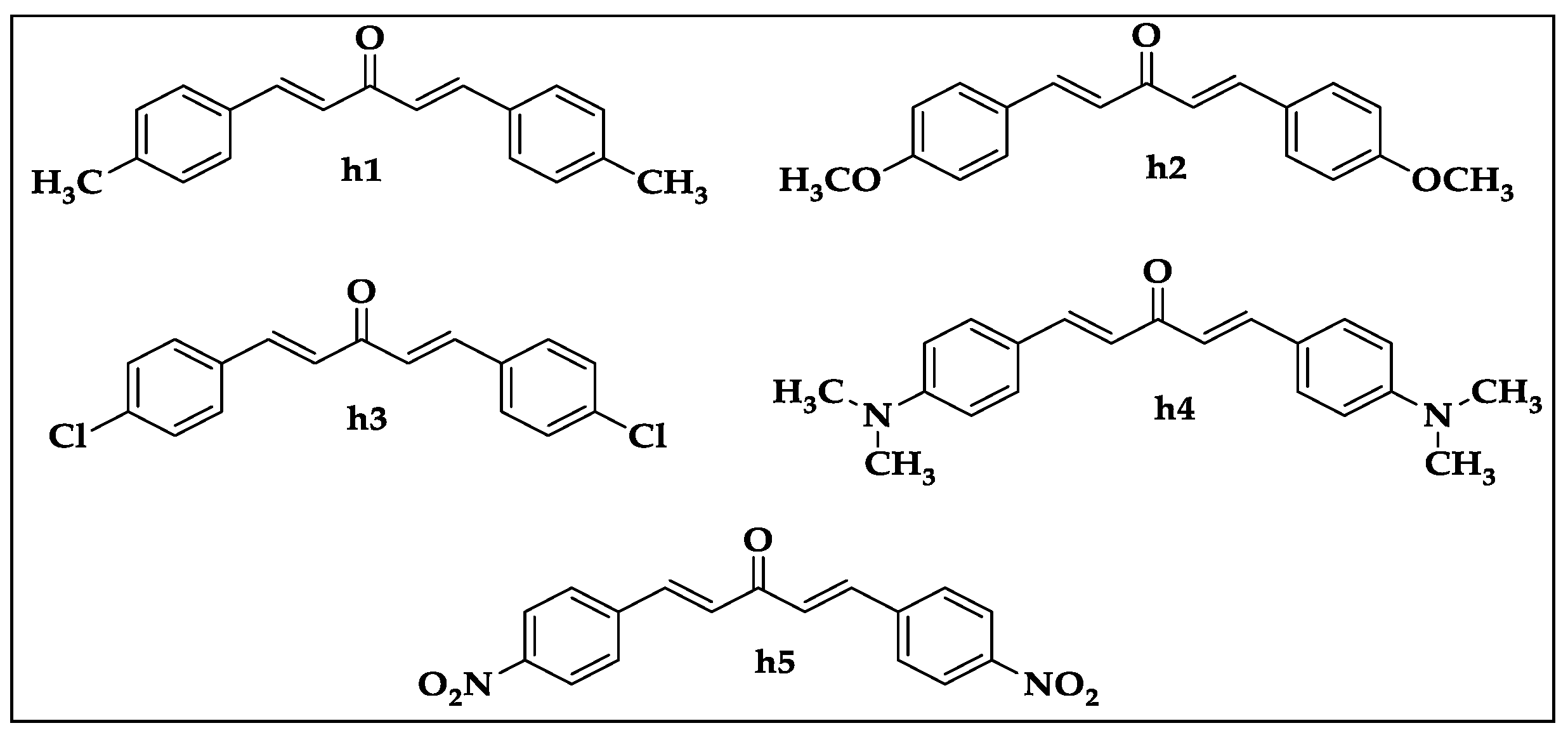
| Compound | DPPH (IC50 µg/mL) | ABTS (IC50 µg/mL) |
|---|---|---|
| h1 | 160.32 ± 1.37 | 184.19 ± 1.37 |
| h2 | 53.29 ± 2.13 | 70.21 ± 1.26 |
| h3 | 82.43 ± 2.17 | 143.65 ± 2.43 |
| h4 | 230.18 ± 2.15 | 356.38 ± 1.17 |
| h5 | 357.13 ± 1.16 | 435.31 ± 1.18 |
| Tocopherol | 9.16 ± 1.14 | 13.18 ± 1.16 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hussain, H.; Ahmad, S.; Shah, S.W.A.; Ullah, A.; Rahman, S.U.; Ahmad, M.; Almehmadi, M.; Abdulaziz, O.; Allahyani, M.; Alsaiari, A.A.; et al. Synthetic Mono-Carbonyl Curcumin Analogues Attenuate Oxidative Stress in Mouse Models. Biomedicines 2022, 10, 2597. https://doi.org/10.3390/biomedicines10102597
Hussain H, Ahmad S, Shah SWA, Ullah A, Rahman SU, Ahmad M, Almehmadi M, Abdulaziz O, Allahyani M, Alsaiari AA, et al. Synthetic Mono-Carbonyl Curcumin Analogues Attenuate Oxidative Stress in Mouse Models. Biomedicines. 2022; 10(10):2597. https://doi.org/10.3390/biomedicines10102597
Chicago/Turabian StyleHussain, Haya, Shujaat Ahmad, Syed Wadood Ali Shah, Abid Ullah, Shafiq Ur Rahman, Manzoor Ahmad, Mazen Almehmadi, Osama Abdulaziz, Mamdouh Allahyani, Ahad Amer Alsaiari, and et al. 2022. "Synthetic Mono-Carbonyl Curcumin Analogues Attenuate Oxidative Stress in Mouse Models" Biomedicines 10, no. 10: 2597. https://doi.org/10.3390/biomedicines10102597






