The Performance of FDA-Approved PET Imaging Agents in the Detection of Prostate Cancer
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Imaging Agents Targeting the Cell Division and Proliferation
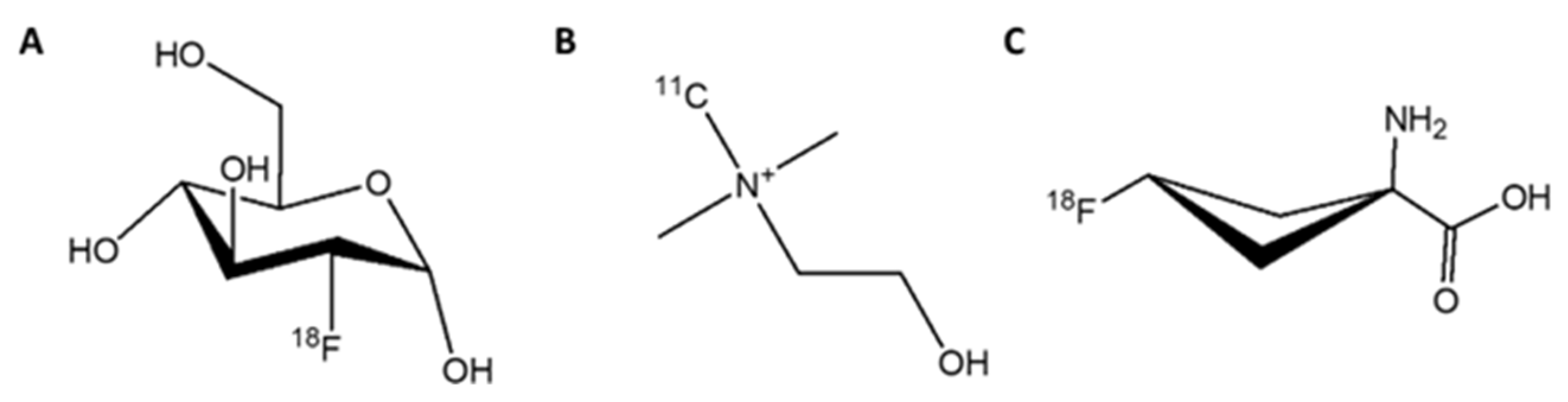
3.1.1. [18F]FDG
3.1.2. [11C]Choline
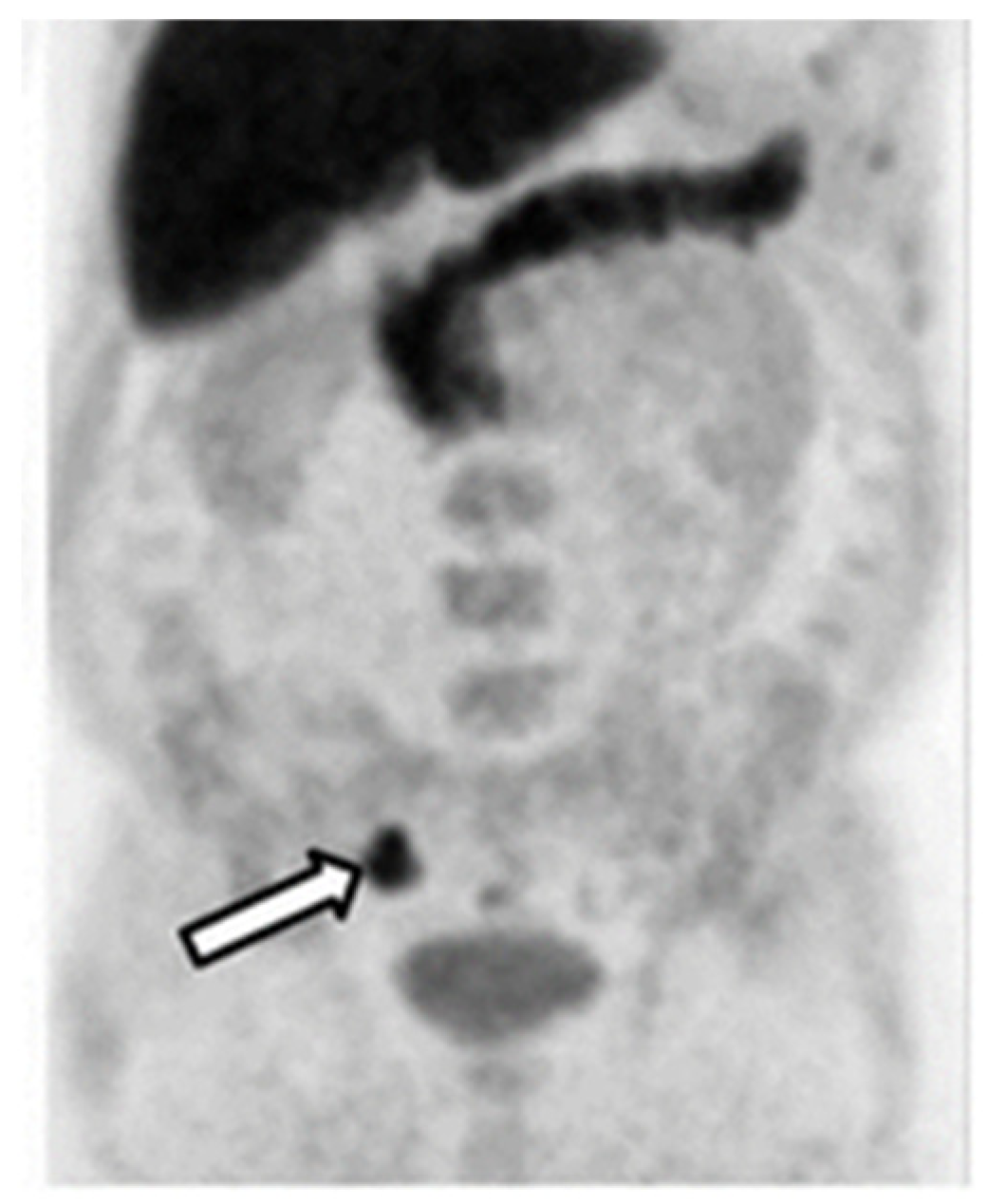
3.1.3. [18F]Fluciclovine (FACBC)
3.2. Imaging Agents Targeting Cancer-Specific Membrane Proteins or Receptors
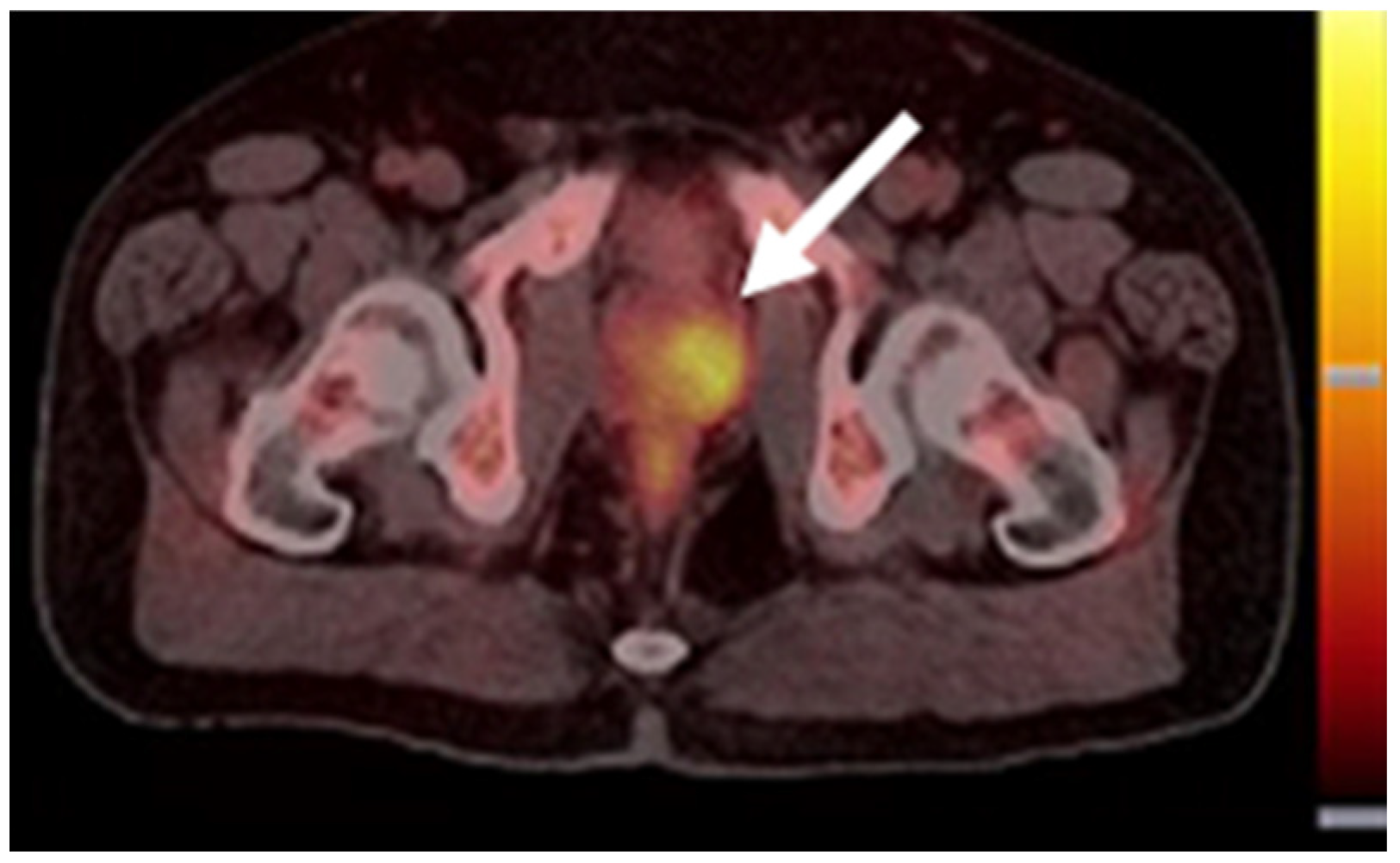
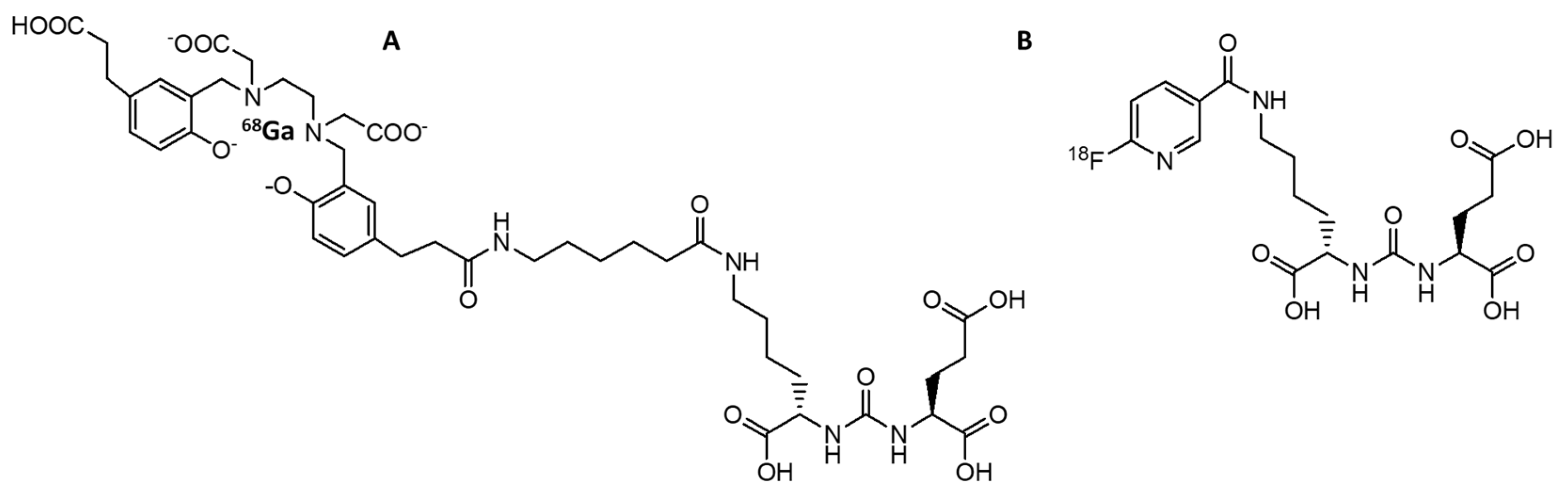
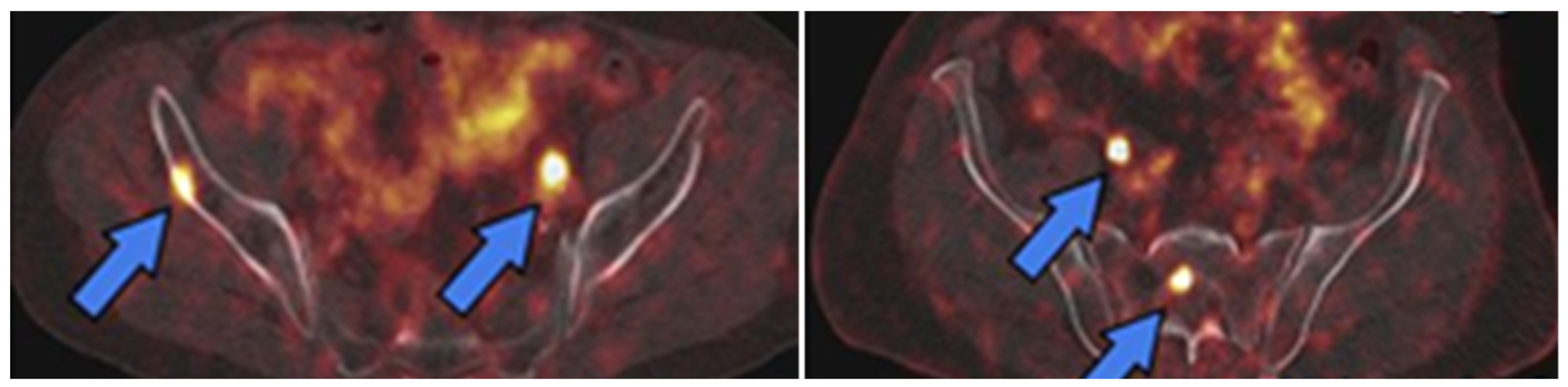
3.2.1. [68Ga]GaPSMA-11
3.2.2. [18F]DCFPyL
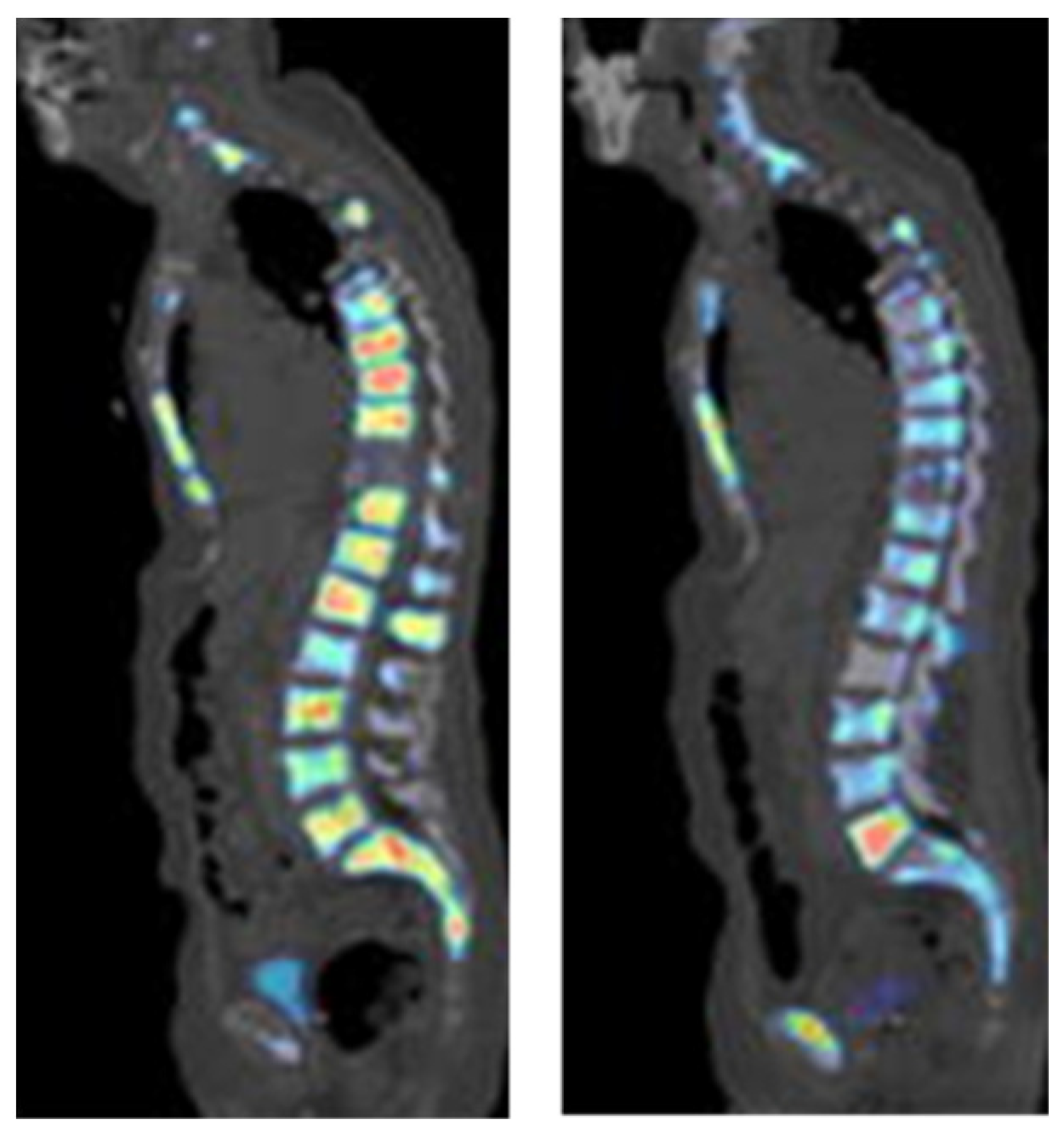
3.3. Imaging Agents Targeting Bone Lesions
[18F]-NaF
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Rawla, P. Epidemiology of Prostate Cancer. World J. Oncol. 2019, 10, 63–89. [Google Scholar] [CrossRef] [Green Version]
- Grozescu, T.; Popa, F. Prostate cancer between prognosis and adequate/proper therapy. J. Med. Life 2017, 10, 5–12. [Google Scholar]
- Key Statistics for Prostate Cancer. Prostate Cancer Facts. Available online: https://www.cancer.org/cancer/prostate-cancer/about/key-statistics.html (accessed on 10 November 2021).
- Matsushita, M.; Fujita, K.; Nonomura, N. Influence of Diet and Nutrition on Prostate Cancer. Int. J. Mol. Sci. 2020, 21, 1447. [Google Scholar] [CrossRef] [Green Version]
- Perry, E.; Talwar, A.; Taubman, K.; Ng, M.; Wong, L.-M.; Booth, R.; Sutherland, T.R. [18F]DCFPyL PET/CT in detection and localization of recurrent prostate cancer following prostatectomy including low PSA <0.5 ng/mL. Eur. J. Pediatr. 2021, 48, 2038–2046. [Google Scholar] [CrossRef]
- Mullerad, M.; Hricak, H.; Kuroiwa, K.; Pucar, D.; Chen, H.-N.; Kattan, M.; Scardino, P.T. Comparison of endorectal magnetic resonance imaging, guided prostate biopsy and digital rectal examination in the preoperative anatomical localization of prostate cancer. J. Urol. 2005, 174, 2158–2163. [Google Scholar] [CrossRef]
- Mottet, N.; Cornford, P. EAU Guidelines: Prostate Cancer. Uroweb. Available online: https://uroweb.org/guideline/prostate-cancer/#note_384 (accessed on 22 November 2021).
- Ferro, M.; de Cobelli, O.; Vartolomei, M.D.; Lucarelli, G.; Crocetto, F.; Barone, B.; Sciarra, A.; Del Giudice, F.; Muto, M.; Maggi, M.; et al. Prostate Cancer Radiogenomics—From Imaging to Molecular Characterization. Int. J. Mol. Sci. 2021, 22, 9971. [Google Scholar] [CrossRef]
- Cam, K.; Yucel, S.; Turkeri, L.; Akdas, A. Accuracy of transrectal ultrasound guided prostate biopsy: Histopathological correlation to matched prostatectomy specimens. Int. J. Urol. 2002, 9, 257–260. [Google Scholar] [CrossRef]
- Kumar, V.; Jagannathan, N.; Kumar, R.; Thulkar, S.; Gupta, S.D.; Hemal, A.; Gupta, N. Transrectal ultrasound-guided biopsy of prostate voxels identified as suspicious of malignancy on three-dimensional1H MR spectroscopic imaging in patients with abnormal digital rectal examination or raised prostate specific antigen level of 4–10 ng/mL. NMR Biomed. 2007, 20, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Yacoub, J.; Verma, S.; Moulton, J.S.; Eggener, S.; Oto, A. Imaging-guided Prostate Biopsy: Conventional and Emerging Techniques. RadioGraphics 2012, 32, 819–837. [Google Scholar] [CrossRef] [Green Version]
- Abuzallouf, S.; Dayes, I.; Lukka, H. Baseline staging of newly diagnosed prostate cancer: A summary of the literature. J. Urol. 2004, 171, 2122–2127. [Google Scholar] [CrossRef]
- Wibmer, A.G.; Burger, I.A.; Sala, E.; Hricak, H.; Weber, W.A.; Vargas, H.A. Molecular Imaging of Prostate Cancer. RadioGraphics 2016, 36, 142–159. [Google Scholar] [CrossRef]
- Jadvar, H. Prostate Cancer: PET with 18F-FDG, 18F- or 11C-Acetate, and 18F- or 11C-Choline. J. Nucl. Med. 2011, 52, 81–89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Osborne, J.R.; Green, D.A.; Spratt, D.E.; Lyashchenko, S.; Fareedy, S.B.; Robinson, B.D.; Beattie, B.J.; Jain, M.; Lewis, J.S.; Christos, P.; et al. A Prospective Pilot Study of 89 Zr-J591/Prostate Specific Membrane Antigen Positron Emission Tomography in Men with Localized Prostate Cancer Undergoing Radical Prostatectomy. J. Urol. 2014, 191, 1439–1445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fludeoxyglucose, F. 18 (Intravenous Route) Description and Brand Names—Mayo Clinic. Available online: https://www.mayoclinic.org/drugs-supplements/fludeoxyglucose-f-18-intravenous-route/description/drg-20074508 (accessed on 8 January 2022).
- Jadvar, H. PET of Glucose Metabolism and Cellular Proliferation in Prostate Cancer. J. Nucl. Med. 2016, 57, 25S–29S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jadvar, H.; Desai, B.; Ji, L.; Conti, P.S.; Dorff, T.B.; Groshen, S.G.; Pinski, J.K.; Quinn, D.I. Baseline 18F-FDG PET/CT Parameters as Imaging Biomarkers of Overall Survival in Castrate-Resistant Metastatic Prostate Cancer. J. Nucl. Med. 2013, 54, 1195–1201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jadvar, H.; Velez, E.M.; Desai, B.; Ji, L.; Colletti, P.M.; Quinn, D.I. Prediction of Time to Hormonal Treatment Failure in Metastatic Castration-Sensitive Prostate Cancer with 18F-FDG PET/CT. J. Nucl. Med. 2019, 60, 1524–1530. [Google Scholar] [CrossRef] [PubMed]
- Hillner, B.E.; Siegel, B.A.; Shields, A.F.; Liu, D.; Gareen, I.F.; Hunt, E.; Coleman, R.E. Relationship Between Cancer Type and Impact of PET and PET/CT on Intended Management: Findings of the National Oncologic PET Registry. J. Nucl. Med. 2008, 49, 1928–1935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, R.; Wang, Y.; Shi, Y.; Zhu, Y.; Xu, L.; Huang, G.; Liu, J. Diagnostic value of 18F-FDG PET/CT in patients with biochemical recurrent prostate cancer and negative 68Ga-PSMA PET/CT. Eur. J. Pediatr. 2021, 48, 2970–2977. [Google Scholar] [CrossRef] [PubMed]
- Hara, T.; Kosaka, N.; Kishi, H. PET imaging of prostate cancer using carbon-11-choline. J. Nucl. Med. 1998, 39, 990–995. [Google Scholar] [PubMed]
- Contractor, K.; Challapalli, A.; Barwick, T.; Winkler, M.; Hellawell, G.; Hazell, S.; Tomasi, G.; Al-Nahhas, A.; Mapelli, P.; Kenny, L.M.; et al. Use of [11C]Choline PET-CT as a Noninvasive Method for Detecting Pelvic Lymph Node Status from Prostate Cancer and Relationship with Choline Kinase Expression. Clin. Cancer Res. 2011, 17, 7673–7683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scattoni, V.; Picchio, M.; Suardi, N.; Messa, C.; Freschi, M.; Roscigno, M.; Da Pozzo, L.F.; Bocciardi, A.; Rigatti, P.; Fazio, F. Detection of Lymph-Node Metastases with Integrated [11C]Choline PET/CT in Patients with PSA Failure after Radical Retropubic Prostatectomy: Results Confirmed by Open Pelvic-Retroperitoneal Lymphadenectomy. Eur. Urol. 2007, 52, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Rinnab, L.; Mottaghy, F.M.; Simon, J.; Volkmer, B.G.; de Petriconi, R.; Hautmann, R.E.; Wittbrodt, M.; Egghart, G.; Moeller, P.; Blumstein, N.; et al. [11C]Choline PET/CT for Targeted Salvage Lymph Node Dissection in Patients with Biochemical Recurrence after Primary Curative Therapy for Prostate Cancer. Urol. Int. 2008, 81, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.R.; Lowe, V.J.; Rangel, L.J.; Hung, J.C.; Kwon, E.D.; Karnes, R.J. Operational Characteristics of 11 C-Choline Positron Emission Tomography/Computerized Tomography for Prostate Cancer with Biochemical Recurrence After Initial Treatment. J. Urol. 2013, 189, 1308–1313. [Google Scholar] [CrossRef]
- Reske, S.N. [11C]Choline uptake with PET/CT for the initial diagnosis of prostate cancer: Relation to PSA levels, tumour stage and anti-androgenic therapy. Eur. J. Pediatr. 2008, 35, 1740–1741. [Google Scholar] [CrossRef] [PubMed]
- Mayo Clinic Cancer Center’s Online Magazine—Mayo Clinic Gets Approval for New Prostate Cancer Imaging Agent. Mayo Clinic. Available online: https://www.mayo.edu/research/forefront/mayo-clinic-gets-approval-new-prostate-cancer-imaging-agent (accessed on 10 January 2022).
- Piert, M.; Park, H.; Khan, A.; Siddiqui, J.; Hussain, H.; Chenevert, T.; Wood, D.; Johnson, T.; Shah, R.B.; Meyer, C. Detection of Aggressive Primary Prostate Cancer with 11C-Choline PET/CT Using Multimodality Fusion Techniques. J. Nucl. Med. 2009, 50, 1585–1593. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Graziani, T.; Ceci, F.; Castellucci, P.; Polverari, G.; Lima, G.M.; Lodi, F.; Morganti, A.G.; Ardizzoni, A.; Schiavina, R.; Fanti, S. 11C-Choline PET/CT for restaging prostate cancer. Results from 4426 scans in a single-centre patient series. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1971–1979. [Google Scholar] [CrossRef]
- Giovacchini, G.; Guglielmo, P.; Mapelli, P.; Incerti, E.; Gajate, A.M.S.; Giovannini, E.; Riondato, M.; Briganti, A.; Gianolli, L.; Ciarmiello, A.; et al. 11C-choline PET/CT predicts survival in prostate cancer patients with PSA <1 NG/mL. Eur. J. Pediatr. 2019, 46, 921–929. [Google Scholar] [CrossRef]
- Giovacchini, G.; Picchio, M.; Garcia-Parra, R.; Briganti, A.; Abdollah, F.; Gianolli, L.; Schindler, C.; Montorsi, F.; Messa, C.; Fazio, F. 11C-Choline PET/CT Predicts Prostate Cancer–Specific Survival in Patients with Biochemical Failure during Androgen-Deprivation Therapy. J. Nucl. Med. 2014, 55, 233–241. [Google Scholar] [CrossRef] [Green Version]
- Shoup, T.M.; Olson, J.; Hoffman, J.M.; Votaw, J.; Eshima, D.; Eshima, L.; Camp, V.M.; Stabin, M.; Votaw, D.; Goodman, M.M. Synthesis and evaluation of [18F]1-amino-3-fluorocyclobutane-1-carboxylic acid to image brain tumors. J. Nucl. Med. 1999, 40, 331–338. [Google Scholar]
- Nanni, C.; Zanoni, L.; Pultrone, C.; Schiavina, R.; Brunocilla, E.; Lodi, F.; Malizia, C.; Ferrari, M.; Rigatti, P.; Fonti, C.; et al. 18F-FACBC (anti1-amino-3-18F-fluorocyclobutane-1-carboxylic acid) versus 11C-choline PET/CT in prostate cancer relapse: Results of a prospective trial. Eur. J. Pediatr. 2016, 43, 1601–1610. [Google Scholar] [CrossRef]
- FDA. Approves New Diagnostic Imaging Agent to Detect Recurrent Prostate Cancer. FDA, 24 March 2020. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-new-diagnostic-imaging-agent-detect-recurrent-prostate-cancer (accessed on 26 January 2022).
- Schuster, D.M.; Nanni, C.; Fanti, S. Evaluation of Prostate Cancer with Radiolabeled Amino Acid Analogs. J. Nucl. Med. 2016, 57, 61S–66S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamoto, R.; Harrison, C.; Song, H.; Guja, K.E.; Hatami, N.; Nguyen, J.; Moradi, F.; Franc, B.L.; Aparici, C.M.; Davidzon, G.; et al. The Clinical Utility of 18F-Fluciclovine PET/CT in Biochemically Recurrent Prostate Cancer: An Academic Center Experience Post FDA Approval. Mol. Imaging Biol. 2021, 23, 614–623. [Google Scholar] [CrossRef]
- Selnæs, K.M.; Krüger-Stokke, B.; Elschot, M.; Johansen, H.; Steen, P.A.; Langørgen, S.; Aksnessæther, B.Y.; Indrebø, G.; Sjøbakk, T.A.E.; Tessem, M.-B.; et al. Detection of Recurrent Prostate Cancer With 18F-Fluciclovine PET/MRI. Front. Oncol. 2020, 10, 582092. [Google Scholar] [CrossRef] [PubMed]
- Abiodun-Ojo, O.A.; Jani, A.B.; Akintayo, A.A.; Akin-Akintayo, O.O.; Odewole, O.A.; Tade, F.I.; Joshi, S.S.; Master, V.A.; Fielder, B.; Halkar, R.K.; et al. Salvage Radiotherapy Management Decisions in Postprostatectomy Patients with Recurrent Prostate Cancer Based on 18F-Fluciclovine PET/CT Guidance. J. Nucl. Med. 2021, 62, 1089–1096. [Google Scholar] [CrossRef] [PubMed]
- Andriole, G.L.; Kostakoglu, L.; Chau, A.; Duan, F.; Mahmood, U.; Mankoff, D.A.; Schuster, D.M.; Siegel, B.A.; Adler, L.P.; Belkoff, L.H.; et al. The Impact of Positron Emission Tomography with 18 F-Fluciclovine on the Treatment of Biochemical Recurrence of Prostate Cancer: Results from the LOCATE Trial. J. Urol. 2019, 201, 322–331. [Google Scholar] [CrossRef]
- Dreyfuss, A.D.; Ahn, G.S.; Barsky, A.R.; Gillman, J.A.; Vapiwala, N.; Pantel, A.R. 18F-Fluciclovine PET/CT in Therapeutic Decision Making for Prostate Cancer. Clin. Nucl. Med. 2020, 46, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Garza, D.; Kandathil, A.; Xi, Y.; Subramaniam, R.M. 18F-fluciclovine PET/CT detection of biochemical recurrent prostate cancer in patients with PSA levels <2.00 ng/mL. Nucl. Med. Commun. 2021, 8, 907–913. [Google Scholar] [CrossRef]
- Filippi, L.; Bagni, O.; Crisafulli, C.; Cerio, I.; Brunotti, G.; Chiaravalloti, A.; Schillaci, O.; Dore, F. Detection Rate and Clinical Impact of PET/CT with 18F-FACBC in Patients with Biochemical Recurrence of Prostate Cancer: A Retrospective Bicentric Study. Biomedicines 2022, 10, 177. [Google Scholar] [CrossRef]
- Marcus, C.; Abiodun-Ojo, O.A.; Jani, A.B.; Schuster, M.D. Clinical utility of 18F-Fluciclovine PET/CT in recurrent prostate cancer with very low (≤0.3 ng/mL) prostate-specific antigen levels. Am. J. Nucl. Med. Mol. Imaging 2021, 11, 406–414. [Google Scholar]
- Zanoni, L.; Bianchi, L.; Nanni, C.; Pultrone, C.; Giunchi, F.; Bossert, I.; Matti, A.; Schiavina, R.; Fiorentino, M.; Romagnoli, D.; et al. [18F]-Fluciclovine PET/CT for preoperative nodal staging in high-risk primary prostate cancer: Final results of a prospective trial. Eur. J. Pediatr. 2021, 49, 390–409. [Google Scholar] [CrossRef] [PubMed]
- Selnæs, K.M.; Krüger-Stokke, B.; Elschot, M.; Willoch, F.; Størkersen, Ø.; Sandsmark, E.; Moestue, S.A.; Tessem, M.-B.; Halvorsen, D.; Kjøbli, E.; et al. 18F-Fluciclovine PET/MRI for preoperative lymph node staging in high-risk prostate cancer patients. Eur. Radiol. 2018, 28, 3151–3159. [Google Scholar] [CrossRef] [PubMed]
- Seierstad, T.; Hole, K.H.; Tulipan, A.J.; Strømme, H.; Lilleby, W.; Revheim, M.-E.; Hernes, E. 18F-Fluciclovine PET for Assessment of Prostate Cancer with Histopathology as Reference Standard. PET Clin. 2021, 16, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Thoeny, H.C.; Froehlich, J.M.; Triantafyllou, M.; Huesler, J.; Bains, L.J.; Vermathen, P.; Fleischmann, A.; Studer, U.E. Metastases in Normal-sized Pelvic Lymph Nodes: Detection with Diffusion-weighted MR Imaging. Radiology 2014, 273, 125–135. [Google Scholar] [CrossRef]
- Evans, J.C.; Malhotra, M.; Cryan, J.F.; O’Driscoll, C.M. The therapeutic and diagnostic potential of the prostate specific membrane antigen/glutamate carboxypeptidase II (PSMA/GCPII) in cancer and neurological disease. J. Cereb. Blood Flow Metab. 2016, 173, 3041–3079. [Google Scholar] [CrossRef] [Green Version]
- Haffner, M.C.; Kronberger, I.E.; Ross, J.S.; Sheehan, C.E.; Zitt, M.; Mühlmann, G.; Öfner, D.; Zelger, B.; Ensinger, C.; Yang, X.J.; et al. Prostate-specific membrane antigen expression in the neovasculature of gastric and colorectal cancers. Hum. Pathol. 2009, 40, 1754–1761. [Google Scholar] [CrossRef]
- Ponsky, L.E.; Cherullo, E.E.; Starkey, R.; Nelson, D.; Neumann, D.; Zippe, C.D. Evaluation of preoperative ProstaScint™ scans in the prediction of nodal disease. Prostate Cancer Prostatic Dis. 2002, 5, 132–135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lawhn-Heath, C.; Salavati, A.; Behr, S.C.; Rowe, S.P.; Calais, J.; Fendler, W.P.; Eiber, M.; Emmett, L.; Hofman, M.S.; Hope, T.A. Prostate-specific Membrane Antigen PET in Prostate Cancer. Radiology 2021, 299, 248–260. [Google Scholar] [CrossRef]
- Liu, H.; Moy, P.; Kim, S.; Xia, Y.; Rajasekaran, A.; Navarro, V.; Knudsen, B.; Bander, N.H. Monoclonal antibodies to the extracellular domain of prostate-specific membrane antigen also react with tumor vascular endothelium. Cancer Res. 1997, 57, 3629–3634. [Google Scholar] [PubMed]
- Pandit-Taskar, N.; O’Donoghue, J.A.; Beylergil, V.; Lyashchenko, S.; Ruan, S.; Solomon, S.B.; Durack, J.C.; Carrasquillo, J.A.; Lefkowitz, R.A.; Gonen, M.; et al. 89Zr-huJ591 immuno-PET imaging in patients with advanced metastatic prostate cancer. Eur. J. Pediatr. 2014, 41, 2093–2105. [Google Scholar] [CrossRef] [Green Version]
- Chalasani, S.; Fareedy, S.; Lindgren-Belal, S.; Spratt, D.; Robinson, B.; Osborne, J.; Bander, N.; Scherr, D. MP51-14 A comparison of 111IN-J591 spect with 89ZR-J591 pet imaging for prostate cancer patients. J. Urol. 2014, 191, e602–e603. [Google Scholar] [CrossRef]
- Tagawa, S.T.; Vallabhajosula, S.; Christos, P.J.; Jhanwar, Y.S.; Batra, J.S.; Lam, L.; Osborne, J.; Beltran, H.; Molina, A.M.; Goldsmith, S.J.; et al. Phase 1/2 study of fractionated dose lutetium-177–labeled anti–prostate-specific membrane antigen monoclonal antibody J591 (177 Lu-J591) for metastatic castration-resistant prostate cancer. Cancer 2019, 125, 2561–2569. [Google Scholar] [CrossRef]
- Chen, Y.; Foss, C.A.; Byun, Y.; Nimmagadda, S.; Pullambhatla, M.; Fox, J.J.; Castanares, M.; Lupold, S.E.; Babich, J.W.; Mease, R.C.; et al. Radiohalogenated Prostate-Specific Membrane Antigen (PSMA)-Based Ureas as Imaging Agents for Prostate Cancer. J. Med. Chem. 2008, 51, 7933–7943. [Google Scholar] [CrossRef] [Green Version]
- Jeitner, T.M.; Babich, J.W.; Kelly, J.M. Advances in PSMA theranostics. Transl. Oncol. 2022, 22, 101450. [Google Scholar] [CrossRef] [PubMed]
- Afshar-Oromieh, A.; Malcher, A.; Eder, M.; Eisenhut, M.; Linhart, H.G.; Hadaschik, B.A.; Holland-Letz, T.; Giesel, F.L.; Kratochwil, C.; Haufe, S.; et al. PET imaging with a [68Ga]gallium-labelled PSMA ligand for the diagnosis of prostate cancer: Biodistribution in humans and first evaluation of tumour lesions. Eur. J. Pediatr. 2013, 40, 486–495. [Google Scholar] [CrossRef]
- Chen, Y.; Pullambhatla, M.; Foss, C.A.; Byun, Y.; Nimmagadda, S.; Senthamizhchelvan, S.; Sgouros, G.; Mease, R.C.; Pomper, M.G. 2-(3-{1-Carboxy-5-[(6-[18F]Fluoro-Pyridine-3-Carbonyl)-Amino]-Pentyl}-Ureido)-Pentanedioic Acid, [18F]DCFPyL, a PSMA-Based PET Imaging Agent for Prostate Cancer. Clin. Cancer Res. 2011, 17, 7645–7653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dietlein, F.; Kobe, C.; Neubauer, S.; Schmidt, M.; Stockter, S.; Fischer, T.; Schomäcker, K.; Heidenreich, A.; Zlatopolskiy, B.D.; Neumaier, B.; et al. PSA-Stratified Performance of 18F- and 68Ga-PSMA PET in Patients with Biochemical Recurrence of Prostate Cancer. J. Nucl. Med. 2017, 58, 947–952. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eder, M.; Schäfer, M.; Bauder-Wüst, U.; Hull, W.-E.; Wängler, C.; Mier, W.; Haberkorn, U.; Eisenhut, M. 68Ga-Complex Lipophilicity and the Targeting Property of a Urea-Based PSMA Inhibitor for PET Imaging. Bioconjug. Chem. 2012, 23, 688–697. [Google Scholar] [CrossRef] [PubMed]
- FDA. Approves First PSMA-Targeted PET Imaging Drug for Men with Prostate Cancer. 2 December 2020. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-first-psma-targeted-pet-imaging-drug-men-prostate-cancer (accessed on 29 November 2021).
- Park, S.Y.; Zacharias, C.; Harrison, C.; Fan, R.E.; Kunder, C.; Hatami, N.; Giesel, F.; Ghanouni, P.; Daniel, B.; Loening, A.M.; et al. Gallium 68 PSMA-11 PET/MR Imaging in Patients with Intermediate- or High-Risk Prostate Cancer. Radiology 2018, 288, 495–505. [Google Scholar] [CrossRef] [Green Version]
- Maurer, T.; Gschwend, J.E.; Rauscher, I.; Souvatzoglou, M.; Haller, B.; Weirich, G.; Wester, H.-J.; Heck, M.; Kübler, H.; Beer, A.J.; et al. Diagnostic Efficacy of 68 Gallium-PSMA Positron Emission Tomography Compared to Conventional Imaging for Lymph Node Staging of 130 Consecutive Patients with Intermediate to High Risk Prostate Cancer. J. Urol. 2016, 195, 1436–1443. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zang, S.; Zhang, C.; Fu, Y.; Lv, X.; Zhang, Q.; Deng, Y.; Zhang, C.; Luo, R.; Zhao, X.; et al. Comparison of 68Ga-PSMA-11 PET-CT with mpMRI for preoperative lymph node staging in patients with intermediate to high-risk prostate cancer. J. Transl. Med. 2017, 15, 230. [Google Scholar] [CrossRef] [Green Version]
- Uprimny, C.; Kroiss, A.S.; Decristoforo, C.; Fritz, J.; von Guggenberg, E.; Kendler, D.; Scarpa, L.; di Santo, G.; Roig, L.G.; Maffey-Steffan, J.; et al. 68Ga-PSMA-11 PET/CT in primary staging of prostate cancer: PSA and Gleason score predict the intensity of tracer accumulation in the primary tumour. Eur. J. Pediatr. 2017, 44, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Hope, T.A.; Armstrong, W.R.; Murthy, V.; Heath, C.L.; Behr, S.; Barbato, F.; Ceci, F.; Farolfi, A.; Schwarzenboeck, S.; Unterrainer, M.; et al. Accuracy of 68Ga-PSMA-11 for pelvic nodal metastasis detection prior to radical prostatectomy and pelvic lymph node dissection: A multicenter prospective phase III imaging study. J. Clin. Oncol. 2020, 38, 5502. [Google Scholar] [CrossRef]
- Hope, T.A.; Eiber, M.; Armstrong, W.R.; Juarez, R.; Murthy, V.; Lawhn-Heath, C.; Behr, S.C.; Zhang, L.; Barbato, F.; Ceci, F.; et al. Diagnostic Accuracy of 68Ga-PSMA-11 PET for Pelvic Nodal Metastasis Detection Prior to Radical Prostatectomy and Pelvic Lymph Node Dissection. JAMA Oncol. 2021, 7, 1635. [Google Scholar] [CrossRef] [PubMed]
- Van Leeuwen, P.J.; Donswijk, M.; Nandurkar, R.; Stricker, P.; Ho, B.; Heijmink, S.; Wit, E.M.; Tillier, C.; Van Muilenkom, E.; Nguyen, Q.; et al. Gallium-68-prostate-specific membrane antigen (68Ga-PSMA) positron emission tomography (PET)/computed tomography (CT) predicts complete biochemical response from radical prostatectomy and lymph node dissection in intermediate- and high-risk prostate cance. Br. J. Urol. 2019, 124, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, D.A.; Muehlematter, U.J.; Schüler, H.I.G.; Rupp, N.J.; Huellner, M.; Messerli, M.; Rüschoff, J.H.; ter Voert, E.E.G.W.; Hermanns, T.; Burger, I.A. 68Ga-PSMA-11 PET has the potential to improve patient selection for extended pelvic lymph node dissection in intermediate to high-risk prostate cancer. Eur. J. Pediatr. 2020, 47, 147–159. [Google Scholar] [CrossRef]
- Esen, T.; Falay, O.; Tarim, K.; Armutlu, A.; Koseoglu, E.; Kilic, M.; Seymen, H.; Sarikaya, A.F.; Kiremit, M.C.; Balbay, M.D.; et al. 68Ga-PSMA-11 Positron Emission Tomography/Computed Tomography for Primary Lymph Node Staging Before Radical Prostatectomy: Central Review of Imaging and Comparison with Histopathology of Extended Lymphadenectomy. Eur. Urol. Focus 2021, 7, 288–293. [Google Scholar] [CrossRef]
- Satapathy, S.; Singh, H.; Kumar, R.; Mittal, B.R. Diagnostic Accuracy of 68Ga-PSMA PET/CT for Initial Detection in Patients with Suspected Prostate Cancer: A Systematic Review and Meta-Analysis. Am. J. Roentgenol. 2021, 216, 599–607. [Google Scholar] [CrossRef]
- Sonni, I.; Eiber, M.; Fendler, W.P.; Alano, R.M.; Vangala, S.S.; Kishan, A.U.; Nickols, N.; Rettig, M.B.; Reiter, R.E.; Czernin, J.; et al. Impact of 68Ga-PSMA-11 PET/CT on Staging and Management of Prostate Cancer Patients in Various Clinical Settings: A Prospective Single-Center Study. J. Nucl. Med. 2020, 61, 1153–1160. [Google Scholar] [CrossRef]
- Ferraro, D.A.; Lehner, F.; Becker, A.S.; Kranzbühler, B.; Kudura, K.; Mebert, I.; Messerli, M.; Hermanns, T.; Eberli, D.; Burger, I.A. Improved oncological outcome after radical prostatectomy in patients staged with 68Ga-PSMA-11 PET: A single-center retrospective cohort comparison. Eur. J. Pediatr. 2021, 48, 1219–1228. [Google Scholar] [CrossRef]
- Afshar-Oromieh, A.; Holland-Letz, T.; Giesel, F.L.; Kratochwil, C.; Mier, W.; Haufe, S.; Debus, N.; Eder, M.; Eisenhut, M.; Schäfer, M.; et al. Diagnostic performance of 68Ga-PSMA-11 (HBED-CC) PET/CT in patients with recurrent prostate cancer: Evaluation in 1007 patients. Eur. J. Pediatr. 2017, 44, 1258–1268. [Google Scholar] [CrossRef] [Green Version]
- Afshar-Oromieh, A.; Sattler, L.P.; Mier, W.; Hadaschik, B.A.; Debus, J.; Holland-Letz, T.; Kopka, K.; Haberkorn, U. The Clinical Impact of Additional Late PET/CT Imaging with 68Ga-PSMA-11 (HBED-CC) in the Diagnosis of Prostate Cancer. J. Nucl. Med. 2017, 58, 750–755. [Google Scholar] [CrossRef] [Green Version]
- Deandreis, D.; Guarneri, A.; Ceci, F.; Lillaz, B.; Bartoncini, S.; Oderda, M.; Nicolotti, D.G.; Pilati, E.; Passera, R.; Zitella, A.; et al. 68Ga-PSMA-11 PET/CT in recurrent hormone-sensitive prostate cancer (HSPC): A prospective single-centre study in patients eligible for salvage therapy. Eur. J. Pediatr. 2020, 47, 2804–2815. [Google Scholar] [CrossRef] [PubMed]
- Pernthaler, B.; Kulnik, R.; Gstettner, C.; Šalamon, S.; Aigner, R.M.; Kvaternik, H. A Prospective Head-to-Head Comparison of 18F-Fluciclovine With 68Ga-PSMA-11 in Biochemical Recurrence of Prostate Cancer in PET/CT. Clin. Nucl. Med. 2019, 44, e566–e573. [Google Scholar] [CrossRef] [PubMed]
- Calais, J.; Fendler, W.P.; Herrmann, K.; Eiber, M.; Ceci, F. Comparison of 68Ga-PSMA-11 and 18F-Fluciclovine PET/CT in a Case Series of 10 Patients with Prostate Cancer Recurrence. J. Nucl. Med. 2018, 59, 789–794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sachpekidis, C.; Eder, M.; Kopka, K.; Mier, W.; Hadaschik, B.A.; Haberkorn, U.; Dimitrakopoulou-Strauss, A. 68Ga-PSMA-11 dynamic PET/CT imaging in biochemical relapse of prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1288–1299. [Google Scholar] [CrossRef] [PubMed]
- Kranzbühler, B.; Nagel, H.; Becker, A.S.; Müller, J.; Huellner, M.; Stolzmann, P.; Muehlematter, U.; Guckenberger, M.; Kaufmann, P.A.; Eberli, D.; et al. Clinical performance of 68Ga-PSMA-11 PET/MRI for the detection of recurrent prostate cancer following radical prostatectomy. Eur. J. Pediatr. 2017, 45, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Calais, J.; Czernin, J.; Cao, M.; Kishan, A.U.; Hegde, J.V.; Shaverdian, N.; Sandler, K.A.; Chu, F.-I.; King, C.R.; Steinberg, M.L.; et al. 68Ga-PSMA-11 PET/CT Mapping of Prostate Cancer Biochemical Recurrence After Radical Prostatectomy in 270 Patients with a PSA Level of Less Than 1.0 ng/mL: Impact on Salvage Radiotherapy Planning. J. Nucl. Med. 2018, 59, 230–237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farolfi, A.; Ceci, F.; Castellucci, P.; Graziani, T.; Siepe, G.; Lambertini, A.; Schiavina, R.; Lodi, F.; Morganti, A.G.; Fanti, S. 68Ga-PSMA-11 PET/CT in prostate cancer patients with biochemical recurrence after radical prostatectomy and PSA <0.5 ng/mL. Efficacy and impact on treatment strategy. Eur. J. Pediatr. 2018, 46, 11–19. [Google Scholar] [CrossRef]
- Hoffmann, M.A.; Buchholz, H.-G.; Wieler, H.J.; Müller-Hübenthal, J.; Trampert, L.; Richardsen, I.; Schreckenberger, M. Diagnostic Performance of 68Ga-PSMA-11 Positron-emission-tomography/Computed-tomography in a Large Cohort of Patients with Biochemical Recurrence of Prostate Carcinoma. Health Phys. 2020, 119, 141–147. [Google Scholar] [CrossRef]
- Hoffmann, M.A.; Buchholz, H.-G.; Wieler, H.J.; Miederer, M.; Rosar, F.; Fischer, N.; Müller-Hübenthal, J.; Trampert, L.; Pektor, S.; Schreckenberger, M. PSA and PSA Kinetics Thresholds for the Presence of 68Ga-PSMA-11 PET/CT-Detectable Lesions in Patients with Biochemical Recurrent Prostate Cancer. Cancers 2020, 12, 398. [Google Scholar] [CrossRef] [Green Version]
- Fassbind, S.; Ferraro, D.A.; Stelmes, J.-J.; Fankhauser, C.D.; Guckenberger, M.; Kaufmann, P.A.; Eberli, D.; Burger, I.A.; Kranzbühler, B. 68Ga-PSMA-11 PET imaging in patients with ongoing androgen deprivation therapy for advanced prostate cancer. Ann. Nucl. Med. 2021, 35, 1109–1116. [Google Scholar] [CrossRef] [PubMed]
- Dietlein, M.; Kobe, C.; Kuhnert, G.; Stockter, S.; Fischer, T.; Schomäcker, K.; Schmidt, M.; Dietlein, F.; Zlatopolskiy, B.; Krapf, P.; et al. Comparison of [18F]DCFPyL and [68Ga]Ga-PSMA-HBED-CC for PSMA-PET Imaging in Patients with Relapsed Prostate Cancer. Mol. Imaging Biol. 2015, 17, 575–584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szabo, Z.; Mena, E.; Rowe, S.P.; Plyku, D.; Nidal, R.; Eisenberger, M.A.; Antonarakis, E.S.; Fan, H.; Dannals, R.F.; Chen, Y.; et al. Initial Evaluation of [18F]DCFPyL for Prostate-Specific Membrane Antigen (PSMA)-Targeted PET Imaging of Prostate Cancer. Mol. Imaging Biol. 2015, 17, 565–574. [Google Scholar] [CrossRef]
- Durack, J.C.; Alva, A.S.; Preston, M.A.; Pouliot, F.; Saperstein, L.; Carroll, P.R.; Pienta, K.J.; Rowe, S.P.; Patnaik, A.; Probst, S.; et al. A prospective phase II/III study of PSMA-targeted 18F-DCFPyL-PET/CT in patients (pts) with prostate cancer (PCa) (OSPREY): A subanalysis of disease staging changes in PCa pts with recurrence or metastases on conventional imaging. J. Clin. Oncol. 2021, 39, 32. [Google Scholar] [CrossRef]
- Morris, M.J.; Rowe, S.P.; Gorin, M.A.; Saperstein, L.; Pouliot, F.; Josephson, D.Y.; Wong, J.Y.; Pantel, A.R.; Cho, S.Y.; Gage, K.L.; et al. Diagnostic Performance of 18F-DCFPyL-PET/CT in Men with Biochemically Recurrent Prostate Cancer: Results from the CONDOR Phase III, Multicenter Study. Clin. Cancer Res. 2021, 27, 3674–3682. [Google Scholar] [CrossRef] [PubMed]
- Fendler, W.P.; Calais, J.; Eiber, M.; Flavell, R.R.; Mishoe, A.; Feng, F.Y.; Nguyen, H.G.; Reiter, R.E.; Rettig, M.B.; Okamoto, S.; et al. Assessment of 68Ga-PSMA-11 PET Accuracy in Localizing Recurrent Prostate Cancer. JAMA Oncol. 2019, 5, 856–863. [Google Scholar] [CrossRef] [Green Version]
- Sun, J.; Lin, Y.; Wei, X.; Ouyang, J.; Huang, Y.; Ling, Z. Performance of 18F-DCFPyL PET/CT Imaging in Early Detection of Biochemically Recurrent Prostate Cancer: A Systematic Review and Meta-Analysis. Front. Oncol. 2021, 21, 649171. [Google Scholar] [CrossRef]
- “Osprey Trial,” PYLARIFY HCP. Available online: https://www.pylarify.com/high-risk-pca (accessed on 4 January 2022).
- Wondergem, M.; Van Der Zant, F.M.; Broos, W.A.M.; Roeleveld, T.A.; Donker, R.; Oever, D.T.; Geenen, R.W.F.; Knol, R.J.J. 18F-DCFPyL PET/CT for primary staging in 160 high-risk prostate cancer patients; metastasis detection rate, influence on clinical management and preliminary results of treatment efficacy. Eur. J. Pediatr. 2020, 48, 521–531. [Google Scholar] [CrossRef]
- Lu, M.; Lindenberg, L.; Mena, E.; Turkbey, B.; Seidel, J.; Ton, A.; McKinney, Y.; Eclarinal, P.; Merino, M.; Pinto, P. A Pilot Study of Dynamic 18F-DCFPyL PET/CT Imaging of Prostate Adenocarcinoma in High-Risk Primary Prostate Cancer Patients. Mol. Imaging Biol. 2021, 24, 444–452. [Google Scholar] [CrossRef]
- Rowe, S.P.; Li, X.; Trock, B.J.; Werner, R.A.; Frey, S.M.; DiGianvittorio, M.; Bleiler, J.K.; Reyes, D.K.; Abdallah, R.; Pienta, K.J.; et al. Prospective Comparison of PET Imaging with PSMA-Targeted 18F-DCFPyL Versus Na18F for Bone Lesion Detection in Patients with Metastatic Prostate Cancer. J. Nucl. Med. 2020, 61, 183–188. [Google Scholar] [CrossRef] [Green Version]
- Bertoldo, F.; Boccardo, F.; Bombardieri, E.; Evangelista, L.; Valdagni, R. Bone Metastases from Prostate Cancer: Biology, Diagnosis and Management; Springer: Cham, Switzerland, 2017. [Google Scholar]
- Yu, E.Y.; Duan, F.; Muzi, M.; Deng, X.; Chin, B.; Alumkal, J.J.; Taplin, M.-E.; Taub, J.M.; Herman, B.; Higano, C.S.; et al. Castration-Resistant Prostate Cancer Bone Metastasis Response Measured by 18F-Fluoride PET After Treatment with Dasatinib and Correlation with Progression-Free Survival: Results from American College of Radiology Imaging Network. J. Nucl. Med. 2015, 56, 354–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, J.C.; Kurdziel, K.A.; Lindenberg, L.; Gulley, J.L.; Madan, R.A.; Wood, L.V.; McKinney, Y.; Choyke, P.L.; Dahut, W.L.; Apolo, A.B. Preliminary results of a prospective study of 18F-NAF PET/CT in prostate cancer. J. Clin. Oncol. 2013, 31, 103. [Google Scholar] [CrossRef]
- Kim, J.W.; Lindenberg, M.L.; Dahut, W.L.; Gulley, J.L.; Madan, R.A.; Wood, L.V.; McKinney, Y.; Choyke, P.L.; Kurdziel, A.K.; Apolo, A.B. A pilot study on the clinical value of 18F-sodium fluoride PET/CT in advanced prostate cancer. J. Clin. Oncol. 2012, 30, 10589. [Google Scholar] [CrossRef]
- Langsteger, W.; Rezaee, A.; Pirich, C.; Beheshti, M. 18F-NaF-PET/CT and 99mTc-MDP Bone Scintigraphy in the Detection of Bone Metastases in Prostate Cancer. Semin. Nucl. Med. 2016, 46, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Sheikhbahaei, S.; Jones, K.M.; Werner, R.A.; Salas-Fragomeni, R.A.; Marcus, C.V.; Higuchi, T.; Rowe, S.P.; Solnes, L.B.; Javadi, M.S. 18F-NaF-PET/CT for the detection of bone metastasis in prostate cancer: A meta-analysis of diagnostic accuracy studies. Ann. Nucl. Med. 2019, 33, 351–361. [Google Scholar] [CrossRef]
- Uprimny, C.; Svirydenka, A.; Fritz, J.; Kroiss, A.S.; Nilica, B.; Decristoforo, C.; Haubner, R.; von Guggenberg, E.; Buxbaum, S.; Horninger, W.; et al. Comparison of [68Ga]Ga-PSMA-11 PET/CT with [18F]NaF PET/CT in the evaluation of bone metastases in metastatic prostate cancer patients prior to radionuclide therapy. Eur. J. Pediatr. 2018, 45, 1873–1883. [Google Scholar] [CrossRef]
- Madsen, C.; Østergren, P.; Haarmark, C. The Value of 68Ga-PSMA PET/CT Following Equivocal 18F-NaF PET/CT in Prostate Cancer Patients. Diagnostics 2020, 10, 352. [Google Scholar] [CrossRef]
- Huang, F.W.; Trinh, B.; Wang, S.; Hope, T.A. Combined 18F-naf and fluciclovine administration for PET imaging of prostate cancer. J. Clin. Oncol. 2020, 38, e17533. [Google Scholar] [CrossRef]
- Fourquet, A.; Rosenberg, A.; Mena, E.; Shih, J.J.; Turkbey, B.; Blain, M.; Bergvall, E.; Lin, I.F.; Adler, S.; Lim, I.; et al. A comparison of 18F-DCFPyL, 18F-NaF and 18F-FDG PET/CT in a prospective cohort of men with metastatic prostate cancer. J. Nucl. Med. 2022, 63, 735–741. [Google Scholar] [CrossRef]
- Sathianathen, N.J.; Butaney, M.; Konety, B.R. The utility of PET-based imaging for prostate cancer biochemical recurrence: A systematic review and meta-analysis. World J. Urol. 2019, 37, 1239–1249. [Google Scholar] [CrossRef]
- Wei, J.; Zhu, H.; Liao, X. Trigger pSA predicting recurrence from positive choline PET/CT with prostate cancer after initial treatment. Oncotarget 2018, 9, 14630–14641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bin, X.; Yong, S.; Kong, Q.-F.; Zhao, S.; Zhang, G.-Y.; Wu, J.-P.; Chen, S.-Q.; Zhu, W.-D.; Pan, K.-H.; Du, M.-L.; et al. Diagnostic Performance of PET/CT Using 18F-FACBC in Prostate Cancer: A Meta-Analysis. Front. Oncol. 2020, 9, 1438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hope, T.A.; Goodman, J.Z.; Allen, I.E.; Calais, J.; Fendler, W.P.; Carroll, P.R. Metaanalysis of 68Ga-PSMA-11 PET Accuracy for the Detection of Prostate Cancer Validated by Histopathology. J. Nucl. Med. 2019, 60, 786–793. [Google Scholar] [CrossRef] [PubMed]
- Pan, K.-H. Evaluation of 18F-DCFPyL PSMA PET/CT for Prostate Cancer: A Meta-Analysis. Front. Oncol. 2021, 10, 597422. [Google Scholar] [CrossRef] [PubMed]
- Glemser, P.A.; Rotkopf, L.T.; Ziener, C.H.; Beuthien-Baumann, B.; Weru, V.; Kopp-Schneider, A.; Schlemmer, H.P.; Dimitrakopoulou-Strauss, A.; Sachpekidis, C. Hybrid imaging with [68Ga]PSMA-11 PET-CT and PET-MRI in biochemically recurrent prostate cancer. Cancer Imaging 2022, 22, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Strauss, D.S.; Sachpekidis, C.; Kopka, K.; Pan, L.; Haberkorn, U.; Dimitrakopoulou-Strauss, A. Pharmacokinetic studies of [68 Ga]Ga-PSMA-11 in patients with biochemical recurrence of prostate cancer: Detection, differences in temporal distribution and kinetic modelling by tissue type. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 4472–4482. [Google Scholar] [CrossRef]
- Alberts, I.; Prenosil, G.; Sachpekidis, C.; Weitzel, T.; Shi, K.; Rominger, A.; Afshar-Oromieh, A. Digital versus analogue PET in [68Ga]Ga-PSMA-11 PET/CT for recurrent prostate cancer: A matched-pair comparison. Eur. J. Pediatr. 2020, 47, 614–623. [Google Scholar] [CrossRef]
- Filippi, L.; Bagni, O.; Schillaci, O. Digital PET/CT with 18F-FACBC in early castration-resistant prostate cancer: Our preliminary results. Expert Rev. Med. Devices 2022, 19, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Current, K.; Meyer, C.; Magyar, C.E.; Mona, C.E.; Almajano, J.; Slavik, R.; Stuparu, A.D.; Cheng, C.; Dawson, D.W.; Radu, C.G.; et al. Investigating PSMA-Targeted Radioligand Therapy Efficacy as a Function of Cellular PSMA Levels and Intratumoral PSMA Heterogeneity. Clin. Cancer Res. 2020, 26, 2946–2955. [Google Scholar] [CrossRef] [PubMed]
- Chevalme, Y.-M.; Boudali, L.; Gauthé, M.; Rousseau, C.; Skanjeti, A.; Merlin, C.; Robin, P.; Giraudet, A.-L.; Janier, M.; Talbot, J.-N. Survey by the French Medicine Agency (ANSM) of the imaging protocol, detection rate, and safety of 68Ga-PSMA-11 PET/CT in the biochemical recurrence of prostate cancer in case of negative or equivocal 18F-fluorocholine PET/CT: 1084 examinations. Eur. J. Pediatr. 2021, 48, 2935–2950. [Google Scholar] [CrossRef]
- Wang, B.; Liu, C.; Wei, Y.; Meng, J.; Zhang, Y.; Gan, H.; Xu, X.; Wan, F.; Pan, J.; Ma, X.; et al. A Prospective Trial of 68Ga-PSMA and 18F-FDG PET/CT in Nonmetastatic Prostate Cancer Patients with an Early PSA Progression During Castration. Clin. Cancer Res. 2020, 26, 4551–4558. [Google Scholar] [CrossRef]
- Beer, M.; Montani, M.; Gerhardt, J.; Wild, P.J.; Hany, T.F.; Hermanns, T.; Müntener, M.; Kristiansen, G. Profiling gastrin-releasing peptide receptor in prostate tissues: Clinical implicationns and molecular correlates. Prostate 2012, 72, 318–325. [Google Scholar] [CrossRef]
- Manyak, M.J. Indium-111 capromab pendetide in the management of recurrent prostate cancer. Expert Rev. Anticancer Ther. 2008, 8, 175–181. [Google Scholar] [CrossRef]
- Minamimoto, R.; Hancock, S.; Schneider, B.; Chin, F.T.; Jamali, M.; Loening, A.; Vasanawala, S.; Gambhir, S.S.; Iagaru, A. Pilot Comparison of 68Ga-RM2 PET and 68Ga-PSMA-11 PET in Patients with Biochemically Recurrent Prostate Cancer. J. Nucl. Med. 2016, 57, 557–562. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Li, J.; Hua, J.; Su, Y.; Beckford-Vera, D.R.; Zhao, W.; Jayaraman, M.; Huynh, T.L.; Zhao, N.; Wang, Y.-H.; et al. Molecular Imaging of Prostate Cancer Targeting CD46 Using ImmunoPET. Clin. Cancer Res. 2021, 27, 1305–1315. [Google Scholar] [CrossRef]
- Fuoco, V.; Argiroffi, G.; Mazzaglia, S.; Lorenzoni, A.; Guadalupi, V.; Franza, A.; Scalorbi, F.; Aliberti, G.; Chiesa, C.; Procopio, G.; et al. Update on radioligand therapy with 177Lu-PSMA for metastatic castration-resistant prostate cancer: Clinical aspects and survival effects. Tumori J. 2021, 108, 315–325. [Google Scholar] [CrossRef]
- Sathekge, M.M.; Bruchertseifer, F.; Vorster, M.; Morgenstern, A.; Lawal, I.O. Global experience with PSMA-based alpha therapy in prostate cancer. Eur. J. Pediatr. 2021, 49, 30–46. [Google Scholar] [CrossRef]
- Filippi, L.; Chiaravalloti, A.; Schillaci, O.; Bagni, O. The potential of PSMA-targeted alpha therapy in the management of prostate cancer. Expert Rev. Anticancer Ther. 2020, 20, 823–829. [Google Scholar] [CrossRef]
- Eder, M.; Schäfer, M.; Bauder-Wüst, U.; Haberkorn, U.; Eisenhut, M.; Kopka, K. Preclinical evaluation of a bispecific low-molecular heterodimer targeting both PSMA and GRPR for improved PET imaging and therapy of prostate cancer. Prostate 2014, 74, 659–668. [Google Scholar] [CrossRef]
- Mendoza-Figueroa, M.J. Preparation and preclinical evaluation of 68Ga-iPSMA-BN as a potential heterodimeric radiotracer for PET-imaging of prostate cancer. J. Radioanal. Nucl. Chem. 2018, 318, 2097–2105. [Google Scholar] [CrossRef]
- Lundmark, F.; Abouzayed, A.; Mitran, B.; Rinne, S.; Varasteh, Z.; Larhed, M.; Tolmachev, V.; Rosenström, U.; Orlova, A. Heterodimeric Radiotracer Targeting PSMA and GRPR for Imaging of Prostate Cancer—Optimization of the Affinity towards PSMA by Linker Modification in Murine Model. Pharmaceutics 2020, 12, 614. [Google Scholar] [CrossRef] [PubMed]
- Mitran, B.; Varasteh, Z.; Abouzayed, A.; Rinne, S.S.; Puuvuori, E.; De Rosa, M.; Larhed, M.; Tolmachev, V.; Orlova, A.; Rosenström, U. Bispecific GRPR-Antagonistic Anti-PSMA/GRPR Heterodimer for PET and SPECT Diagnostic Imaging of Prostate Cancer. Cancers 2019, 11, 1371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cardet, R.E.D.F.; Hofman, M.S.; Segard, T.; Yim, J.; Williams, S.; Francis, R.J.; Frydenberg, M.; Lawrentschuk, N.; Murphy, D.G.; Lourenco, R.D.A. Is Prostate-specific Membrane Antigen Positron Emission Tomography/Computed Tomography Imaging Cost-effective in Prostate Cancer: An Analysis Informed by the proPSMA Trial. Eur. Urol. 2021, 79, 413–418. [Google Scholar] [CrossRef] [PubMed]


| Target | Mechanism of Accumulation in Tumors | Compounds |
|---|---|---|
| Choline | Choline is used to synthesize phosphatidylcholine, a cell membrane component. Overexpression of choline kinase has been observed in human PCa | 11C- and 18F-choline |
| Acetate | Acetate is the substrate of lipid and cholesterol synthesis. In PCa cells, the increased cellular uptake of acetate was found | 11C-acetate, 18F-acetate |
| Glucose | Glucose is a ubiquitous fuel in the human body, particularly for hypermetabolic cells, and increased expressions of transmembrane glucose transporters and hexokinase have been observed in various cancer types | 18F-FDG |
| Amino acids | Amino acid transporter is upregulated in carcinomas because of increased amino acid use for energy requirements and protein synthesis | 18F-fluciclovine, 11C-methionine, 11C-serotonin, 18F-fluoro-methyl-arabinofuranosyl-uracil (FMAU) |
| PMSA | PSMA, a transmembrane cell protein, is heterogeneously expressed by normal prostate luminal epithelial cells and highly upregulated in PCa [15] | capromab pendetide (Prostascint), 89Zr-labeled J591111In-labeled J591, IAB2M,68Ga-PSMA-11, 18F-DCFPyL, 99mTc-MIP-1404 |
| GRPR | GRPR is overexpressed in many cancer types, including PCa, stimulating tumor cell proliferation when activated by agonistic molecules, while being non-detectable in normal prostate tissues | 68Ga-AMBA, 68Ga-BBN-RGD, 111In-RM1, 68Ga-RM2, 68Ga-SB3, NeoBOMB1, 68Ga-RM26 |
| uPA 1 | uPA binds with high affinity to uPAR, which is upregulated in most tumors, including PCa, and associated with advanced disease and poor prognosis [16] | 64Cu-DOTA-AE105 |
| PSCA 2 | PSCA is overexpressed in 83–100% of PCas; the elevated level of PSCA expression is directly associated with advanced stages and high GSC | 89Zr-A2cDb |
| STEAP1 3 | STEAP1 is overexpressed in many cancer cells, including PCa. STEAP1 shows a correlation between increased expression and tumor aggressiveness | 89Zr-2109A |
| CD46 | CD46 is overexpressed in primary tumor tissue and castration-resistant PCa | YS5 (89Zr-DFO-YS5) |
| AR | AR plays an important role in the growth of PCa because it is a transcription factor that influences many other genes, including the genes responsible for PSA | 18F-Fluorodihydrotestosterone (18F-FDHT) |
| Bone seeking agents | A bone-seeking agent is physiologically absorbed at the site of osteoblastic activity and accumulates in sclerotic metastases, due to its high affinity to the newly formed bone | 18F-NaF, 99mTc-MDP |
| Patients’ Condition | Imaging Modality | Detection Rate (%) | Reference |
|---|---|---|---|
| Number of patients: 213 GSC: ≥6 PSA (ng/mL): 4.24 (0.2–93.5) Mean age: 66.4 (46–90) | PET/CT | Overall: 57 31 (PSA: <0.5) 50 (PSA: 0.5–1.0) 66 (PSA: 1–2) | [40] |
| Number of patients: 84 GSC: ≥6 PSA (ng/mL): 0.7 (0.2–12.9) Mean age: 66.5 (53.0–84.7) | PET/MRI | Overall: 39.9 27.1 (PSA: <1) 55.6 (PSA: ≥1) | [38] |
| Number of patients: 328 GSC: unknown PSA (ng/mL): 1.6 (0.02–186.7) Mean age: 71(47–90) | PET/CT | Overall: 65 58 (PSA: 0–0.2) 50 (PSA: 0.2–0.5) 75 (PSA: 0.5–1.0) 73 (PSA: 1.0–2.0) 95 (PSA: 2.0–5.0) 97 (PSA: 5.0–10.0) 95 (PSA: ≥10.0) | [41] |
| Number of patients: 79 GSC: ≥6 PSA (ng/mL): 0.33 (0.02–31.00) Mean age: 61.6 ± 7.6 | PET/CT | Overall: 79.7 75.4 (PSA: <1) 90.9 (PSA: ≥1) | [39] |
| Number of patients: 165 GSC: unknown PSA (ng/mL): 3.1 (0.02–2975) Mean age: 71.1 (51–91) | PET/CT | Overall: 66.7 15.4 (PSA: <0.5) 50 (PSA: 0.5–0.99) 55.6 (PSA: 1–2) 68.2 (PSA: 2–5) 93.7 (PSA: ≥5) | [37] |
| Number of patients: 81 GSC: unknown PSA (ng/mL): median 0.99 Mean age:74 | PET/CT | Overall: 76.9 66.7 (PSA: 0.2–0.57) 71.4 (PSA: 0.58–0.99) 78.9 (PSA: 1–1.5) 90 (PSA: >1.5) | [43] |
| Patients’ Condition | Treatment | Imaging Modality | Detection Rate (%) (PSA:ng/mL) | Reference |
|---|---|---|---|---|
| Number of patients: 31 GSC: 7 (5–9) PSA (ng/mL): 2.0 (0.1–130.0) Mean age: 71 (51–78) | The combination or single use of RT, RP, HDR brachytherapy, and ADT and HIFU | dPET/CT and whole-body PET/CT | Overall: 71 46.7 (PSA: 0.1–2) 75.0 (PSA: >2) | [81] |
| Number of patients: 56 GSC: 6–9 (7/56 unknown) PSA (ng/mL): 0.99 (IQR: 3.1) Mean age: 69 (IQR: 11) | RP | PET/MRI | Overall: 78.6 44.4 (PSA: <0.2); 72.7 (PSA: 0.2–0.5) 80.0 (PSA: 0.5–2.0) 95.2 (PSA: >2.0) | [82] |
| Number of patients: 1007 GSC: 7 (5–10) PSA (ng/mL): 2.2 (0.01–1237) Mean age: 68 (39–90) | The combination or single use of RP, RT, ADT and HIFU | PET/CT | Overall: 79.5 46 (PSA: ≤0.2); 46 (PSA: 0.21–0.5) 73 (PSA: 0.51–1.0) 80 (PSA: 1.1–2.0) 86 (PSA: 2.1–3.0) 91 (PSA: 3.1–5.0) 94 (PSA: 5.1–7.0) 91 (PSA: 7.1–10.0) 96 (PSA: >10.0) | [76] |
| Number of patients: 270 GSC: ≥6 (6% unknown) PSA (ng/mL): 0.44 (0.03–1) Mean age: 68 (43–90) | RP without RT | PET/CT | Overall: 49 | [83] |
| Number of patients: 119 GSC: ≥6 PSA (ng/mL): 0.32 (0.2–0.5) Mean age: 66 (44–78) | RP without RT after recurrence | PET/CT | Overall: 34.4 | [84] |
| Number of patients: 223 PSA (ng/mL): 0.65 (0.2–8.9) Mean age: 70 (66–75) | RT, ADT-free six months before PET scan | PET/CT | Overall: 39.9 23.2 (PSA: 0.2–0.5) 49.6 (PSA: >0.5) | [78] |
| Number of patients: 660 GSC: >6, 45 patients GSC ≤ 6 PSA (ng/mL): median PSA 10.65 Mean age: 70 (49–88) | RP, RT, or brachytherapy | PET/CT | Overall: 76 41 (PSA: <0.2) 44.7 (PSA: 0.2–0.5) 61.7 (PSA: 0.5–1.0) 72.3 (PSA: 1.0–2.0) 82.5 (PSA: 2.0–5.0) 94 (PSA: ≥5) | [85] |
| Number of patients: 581 GSC: >6, 37 patients GSC ≤ 6 PSA (ng/mL): 2.98 (0.2–2000) Mean age: 71 (49–88) | RP, RT, and ADT | PET/CT | Overall: 77 Overall Post-RP: 75 40 (PSA: 0.2–0.5) 62 (PSA: 0.5–1) 70 (PSA: 1–2) 84 (PSA: 2–5) 94 (PSA: ≥5) Overall p-RT: 9188 (PSA: 2–5) 93 (PSA: ≥5) | [86] |
| Number of patients: 84 GSC: ≥8 PSA (ng/mL): 4.27 (0.8–18) Mean age: 71 (66–76) | Prior therapy: RT or RP Ongoing therapy: ADT | PET/CT or PET/MRI | Overall: 94 85.2 (PSA: 0–1) 97.3 (PSA: 1–20) 100 (PSA: >20) | [87] |
| Imaging Agent | Pooled Sensitivity, % | Pooled Specificity, % | Pooled Detection Rate, % | References |
|---|---|---|---|---|
| [11C]choline | 80–83 | 84–92 | 58–62.2 | [108,109] |
| [18F]FACBC | 79.7–88 | 61.9–73 | 58.6 | [108,110] |
| [68Ga]Ga-PSMA-11 | 74–97 | 66–99.8 | 63–82.8 | [73,108,111] |
| [18F]DCFPyL | 91 | 90 | 81–92% | [93,112] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, M.; Zelchan, R.; Orlova, A. The Performance of FDA-Approved PET Imaging Agents in the Detection of Prostate Cancer. Biomedicines 2022, 10, 2533. https://doi.org/10.3390/biomedicines10102533
Li M, Zelchan R, Orlova A. The Performance of FDA-Approved PET Imaging Agents in the Detection of Prostate Cancer. Biomedicines. 2022; 10(10):2533. https://doi.org/10.3390/biomedicines10102533
Chicago/Turabian StyleLi, Mei, Roman Zelchan, and Anna Orlova. 2022. "The Performance of FDA-Approved PET Imaging Agents in the Detection of Prostate Cancer" Biomedicines 10, no. 10: 2533. https://doi.org/10.3390/biomedicines10102533






