FYCO1 Increase and Effect of Arimoclomol–Treatment in Human VCP–Pathology
Abstract
:1. Introduction
2. Materials and Methods
2.1. Muscle MRI on VCP–Patients
2.2. Fibroblasts and Cell Culture
2.3. Proteomic Profiling on Human Skin Fibroblasts
2.4. Immunostaining on Quadriceps Muscle Biopsies
2.5. Immunostaining on Fibroblasts
2.6. Analyses of Cellular Fitness on Arimoclomol Treated Fibroblasts
3. Results
3.1. Clinical Findings
3.2. Proteomic Signature of Human VCP–Mutant Fibroblasts Revealed Increase of FYCO1
3.3. FYCO1 Is Increased in VCP–Patient Derived Muscle Biopsy Specimen
3.4. Increase of FYCO1 Concomitant with Protein Aggregate Markers and Decrease of Tenascin in VCP–Mutant Fibroblasts
3.5. Effect of Arimoclomol–Treatment on the Fitness of VCP–Patient Derived Fibroblasts
3.6. Effect of Arimoclomol on the Proteomic Signature of VCP–Mutant Fibroblasts
3.7. Validation of the Effect of Arimoclomol–Treatment on Protein Composition of Fibroblasts Derived from VCP–Patients
4. Discussion
5. Conclusions
- -
- FYCO1–increase also plays a role in the pathophysiology of VCP–related proteinopathy.
- -
- VCP–patient derived fibroblasts are a suitable in vitro model to study the underlying pathophysiology.
- -
- Arimoclomol–treatment enhances cell proliferation and viability and changes the proteomic signature of VCP–mutant fibroblasts toward the expression of:
- ○
- Pro–survival proteins such as tenascin and nestin
- ○
- Immune–relevant proteins (including interferon–modulated factors) such as GBP1, DDX58 and E3–ubiquitin–protein ligases with direct impact on proteolysis.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sun, X.; Qiu, H. Valosin–Containing Protein, a Calcium-Associated ATPase Protein, in Endoplasmic Reticulum and Mitochondrial Function and Its Implications for Diseases. Int. J. Mol. Sci. 2020, 21, 3842. [Google Scholar] [CrossRef]
- Yeo, B.K.; Hong, C.J.; Chung, K.M.; Woo, H.; Kim, K.; Jung, S.; Kim, E.-K.; Yu, S.-W. Valosin-containing protein is a key mediator between autophagic cell death and apoptosis in adult hippocampal neural stem cells following insulin withdrawal. Mol. Brain 2016, 9, 31. [Google Scholar] [CrossRef] [PubMed]
- Ikenaga, C.; Findlay, A.R.; Seiffert, M.; Peck, A.; Peck, N.; Johnson, N.E.; Statland, J.M.; Weihl, C.C. Phenotypic diversity in an international Cure VCP Disease registry. Orphanet J. Rare Dis. 2020, 15, 267. [Google Scholar] [CrossRef] [PubMed]
- Weihl, C.C.; Pestronk, A.; Kimonis, V.E. Valosin-containing protein disease: Inclusion body myopathy with Paget’s disease of the bone and fronto-temporal dementia. Neuromuscul. Disord. 2009, 19, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Kimonis, V.E.; Kovach, M.J.; Waggoner, B.; Leal, S.; Salam, A.; Rimer, L.; Davis, K.; Khardori, R.; Gelber, D. Clinical and molecular studies in a unique family with autosomal dominant limb-girdle muscular dystrophy and Paget disease of bone. Genet. Med. 2000, 2, 232–241. [Google Scholar] [CrossRef]
- Spina, S.; van Laar, A.D.; Murrell, J.R.; Hamilton, R.L.; Kofler, J.K.; Epperson, F.; Farlow, M.R.; Lopez, O.L.; Quinlan, J.; DeKosky, S.T.; et al. Phenotypic variability in three families with valosin-containing protein mutation. Eur. J. Neurol. 2013, 20, 251–258. [Google Scholar] [CrossRef]
- Kovach, M.J.; Waggoner, B.; Leal, S.M.; Gelber, D.; Khardori, R.; Levenstien, M.A.; Shanks, C.A.; Gregg, G.; Al-Lozi, M.T.; Miller, T.; et al. Clinical delineation and localization to chromosome 9p13.3–p12 of a unique dominant disorder in four families: Hereditary inclusion body myopathy, Paget disease of bone, and frontotemporal dementia. Mol. Genet. Metab. 2001, 74, 458–475. [Google Scholar] [CrossRef]
- González–Pérez, P.; Cirulli, E.T.; Drory, V.E.; Dabby, R.; Nisipeanu, P.; Carasso, R.L.; Sadeh, M.; Fox, A.; Festoff, B.W.; Sapp, P.C.; et al. Novel mutation in VCP gene causes atypical amyotrophic lateral sclerosis. Neurology 2012, 79, 2201–2208. [Google Scholar] [CrossRef]
- Bot, S.T.d.; Schelhaas, H.J.; Kamsteeg, E.-J.; van de Warrenburg, B.P.C. Hereditary spastic paraplegia caused by a mutation in the VCP gene. Brain 2012, 135, e223–e224. [Google Scholar] [CrossRef]
- Palmio, J.; Sandell, S.; Suominen, T.; Penttilä, S.; Raheem, O.; Hackman, P.; Huovinen, S.; Haapasalo, H.; Udd, B. Distinct distal myopathy phenotype caused by VCP gene mutation in a Finnish family. Neuromuscul. Disord. 2011, 21, 551–555. [Google Scholar] [CrossRef]
- Sacconi, S.; Camaño, P.; Greef, J.C.d.; Lemmers, R.J.L.F.; Salviati, L.; Boileau, P.; Lopez de Munain Arregui, A.; van der Maarel, S.M.; Desnuelle, C. Patients with a phenotype consistent with facioscapulohumeral muscular dystrophy display genetic and epigenetic heterogeneity. J. Med. Genet. 2012, 49, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, M.A.; Feely, S.M.; Speziani, F.; Strickland, A.V.; Danzi, M.; Bacon, C.; Lee, Y.; Chou, T.-F.; Blanton, S.H.; Weihl, C.C.; et al. A novel mutation in VCP causes Charcot-Marie-Tooth Type 2 disease. Brain 2014, 137, 2897–2902. [Google Scholar] [CrossRef]
- Mariani, L.-L.; Tesson, C.; Charles, P.; Cazeneuve, C.; Hahn, V.; Youssov, K.; Freeman, L.; Grabli, D.; Roze, E.; Noël, S.; et al. Expanding the Spectrum of Genes Involved in Huntington Disease Using a Combined Clinical and Genetic Approach. JAMA Neurol. 2016, 73, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.P. Multisystem proteinopathy: Intersecting genetics in muscle, bone, and brain degeneration. Neurology 2015, 85, 658–660. [Google Scholar] [CrossRef] [PubMed]
- Güttsches, A.-K.; Brady, S.; Krause, K.; Maerkens, A.; Uszkoreit, J.; Eisenacher, M.; Schreiner, A.; Galozzi, S.; Mertens-Rill, J.; Tegenthoff, M.; et al. Proteomics of rimmed vacuoles define new risk allele in inclusion body myositis. Ann. Neurol. 2017, 81, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Ma, Z.; Jiao, X.; Fariss, R.; Kantorow, W.L.; Kantorow, M.; Pras, E.; Frydman, M.; Pras, E.; Riazuddin, S.; et al. Mutations in FYCO1 cause autosomal-recessive congenital cataracts. Am. J. Hum. Genet. 2011, 88, 827–838. [Google Scholar] [CrossRef] [PubMed]
- Gang, Q.; Bettencourt, C.; Machado, P.M.; Brady, S.; Holton, J.L.; Pittman, A.M.; Hughes, D.; Healy, E.; Parton, M.; Hilton-Jones, D.; et al. Rare variants in SQSTM1 and VCP genes and risk of sporadic inclusion body myositis. Neurobiol. Aging 2016, 47, 218.e1–e218.e9. [Google Scholar] [CrossRef]
- Ahmed, M.; Machado, P.M.; Miller, A.; Spicer, C.; Herbelin, L.; He, J.; Noel, J.; Wang, Y.; McVey, A.L.; Pasnoor, M.; et al. Targeting protein homeostasis in sporadic inclusion body myositis. Sci. Transl. Med. 2016, 8, 331ra41. [Google Scholar] [CrossRef]
- Hentschel, A.; Czech, A.; Münchberg, U.; Freier, E.; Schara-Schmidt, U.; Sickmann, A.; Reimann, J.; Roos, A. Protein signature of human skin fibroblasts allows the study of the molecular etiology of rare neurological diseases. Orphanet J. Rare Dis. 2021, 16, 73. [Google Scholar] [CrossRef]
- Vaudel, M.; Barsnes, H.; Berven, F.S.; Sickmann, A.; Martens, L. SearchGUI: An open-source graphical user interface for simultaneous OMSSA and X!Tandem searches. Proteomics 2011, 11, 996–999. [Google Scholar] [CrossRef]
- Muth, T.; Vaudel, M.; Barsnes, H.; Martens, L.; Sickmann, A. XTandem Parser: An open–source library to parse and analyse X!Tandem MS/MS search results. Proteomics 2010, 10, 1522–1524. [Google Scholar] [CrossRef] [PubMed]
- Gungor, S.; Oktay, Y.; Hiz, S.; Aranguren-Ibáñez, Á.; Kalafatcilar, I.; Yaramis, A.; Karaca, E.; Yis, U.; Sonmezler, E.; Ekinci, B.; et al. Autosomal recessive variants in TUBGCP2 alter the γ-tubulin ring complex leading to neurodevelopmental disease. iScience 2021, 24, 101948. [Google Scholar] [CrossRef] [PubMed]
- Bayraktar, O.; Oral, O.; Kocaturk, N.M.; Akkoc, Y.; Eberhart, K.; Kosar, A.; Gozuacik, D. IBMPFD Disease-Causing Mutant VCP/p97 Proteins Are Targets of Autophagic-Lysosomal Degradation. PLoS ONE 2016, 11, e0164864. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.-L.; Zhang, H.-L.; Huang, Y.; Huang, J.-H.; Sun, P.; Zhou, N.-N.; Chen, Y.-H.; Mai, J.; Wang, Y.; Yu, Y.; et al. Autophagy deficiency promotes triple–negative breast cancer resistance to T cell-mediated cytotoxicity by blocking tenascin-C degradation. Nat. Commun. 2020, 11, 3806. [Google Scholar] [CrossRef]
- Gangfuß, A.; Czech, A.; Hentschel, A.; Münchberg, U.; Horvath, R.; Töpf, A.; O’Heir, E.; Lochmüller, H.; Stehling, F.; Kiewert, C.; et al. Homozygous WASHC4 variant in two sisters causes a syndromic phenotype defined by dysmorphisms, intellectual disability, profound developmental disorder, and skeletal muscle involvement. J. Pathol. 2022, 256, 93–107. [Google Scholar] [CrossRef]
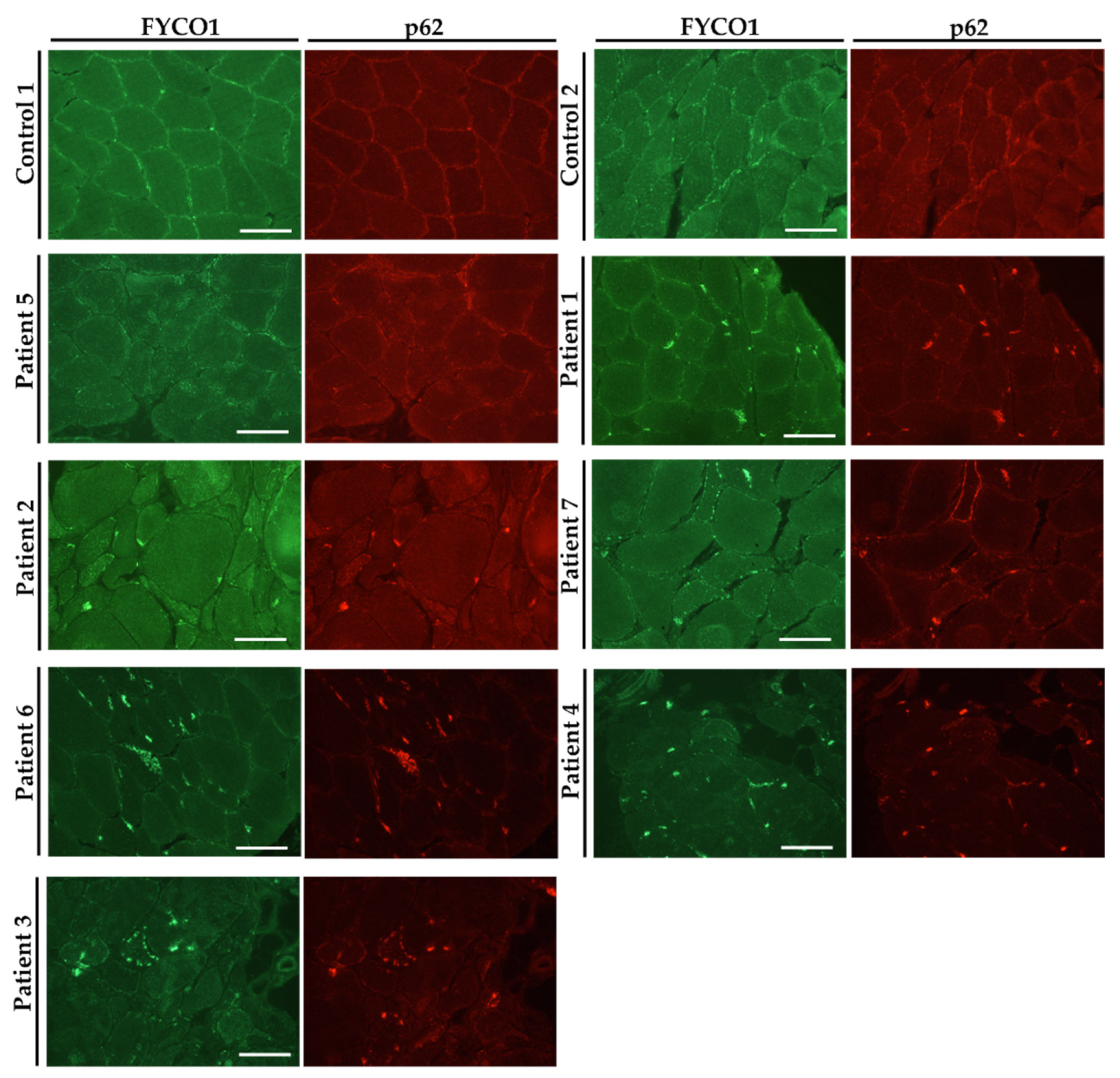
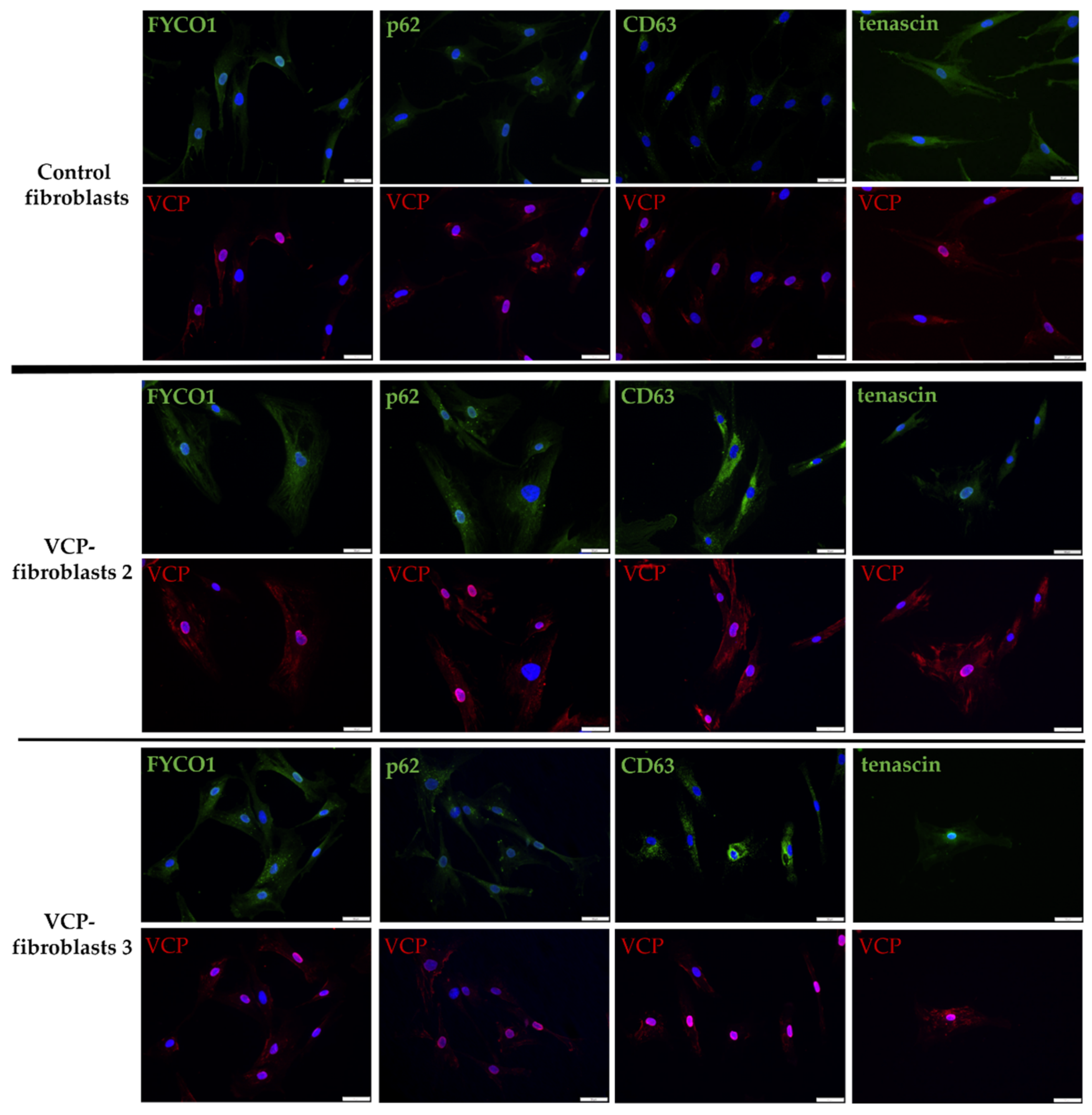
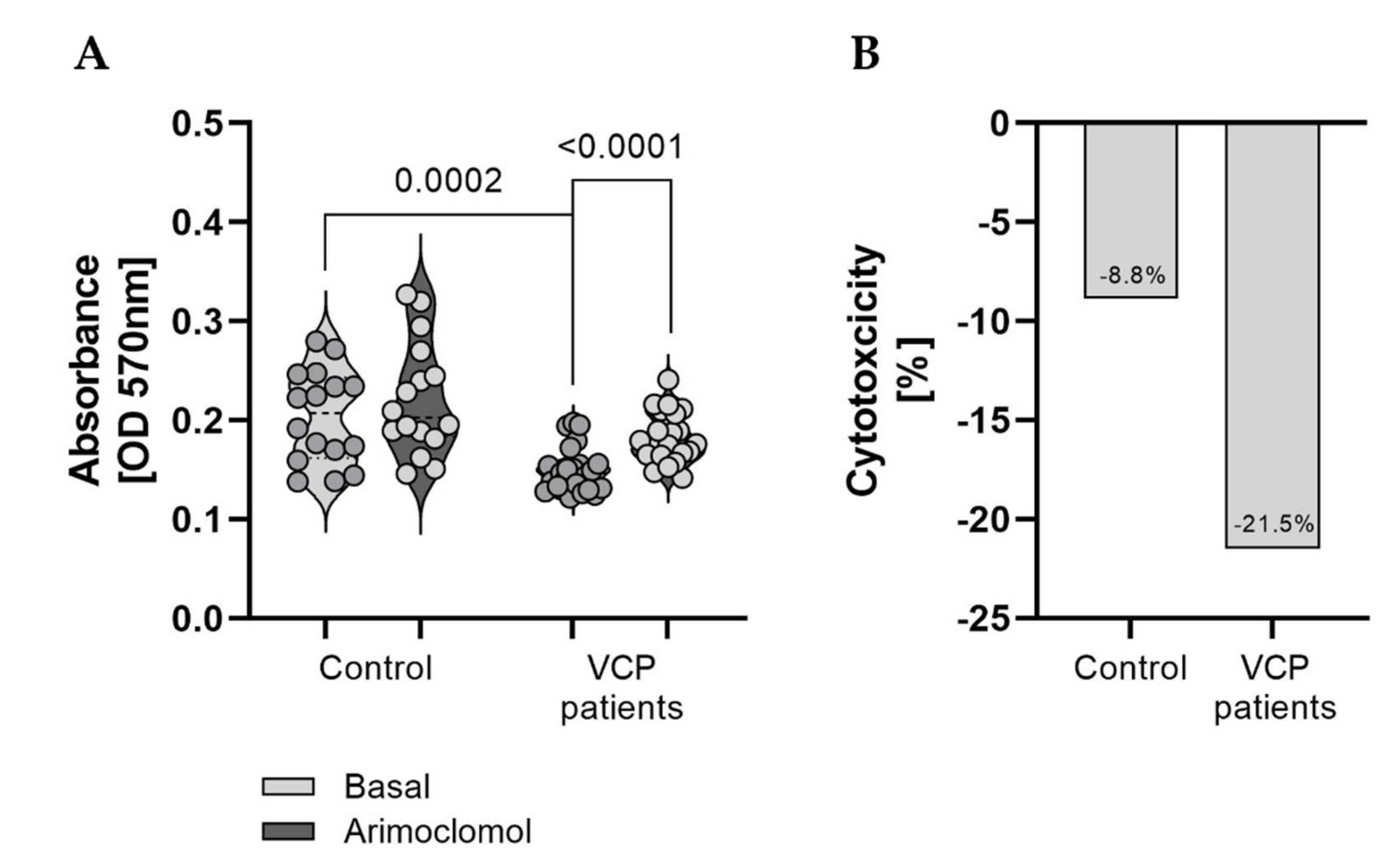
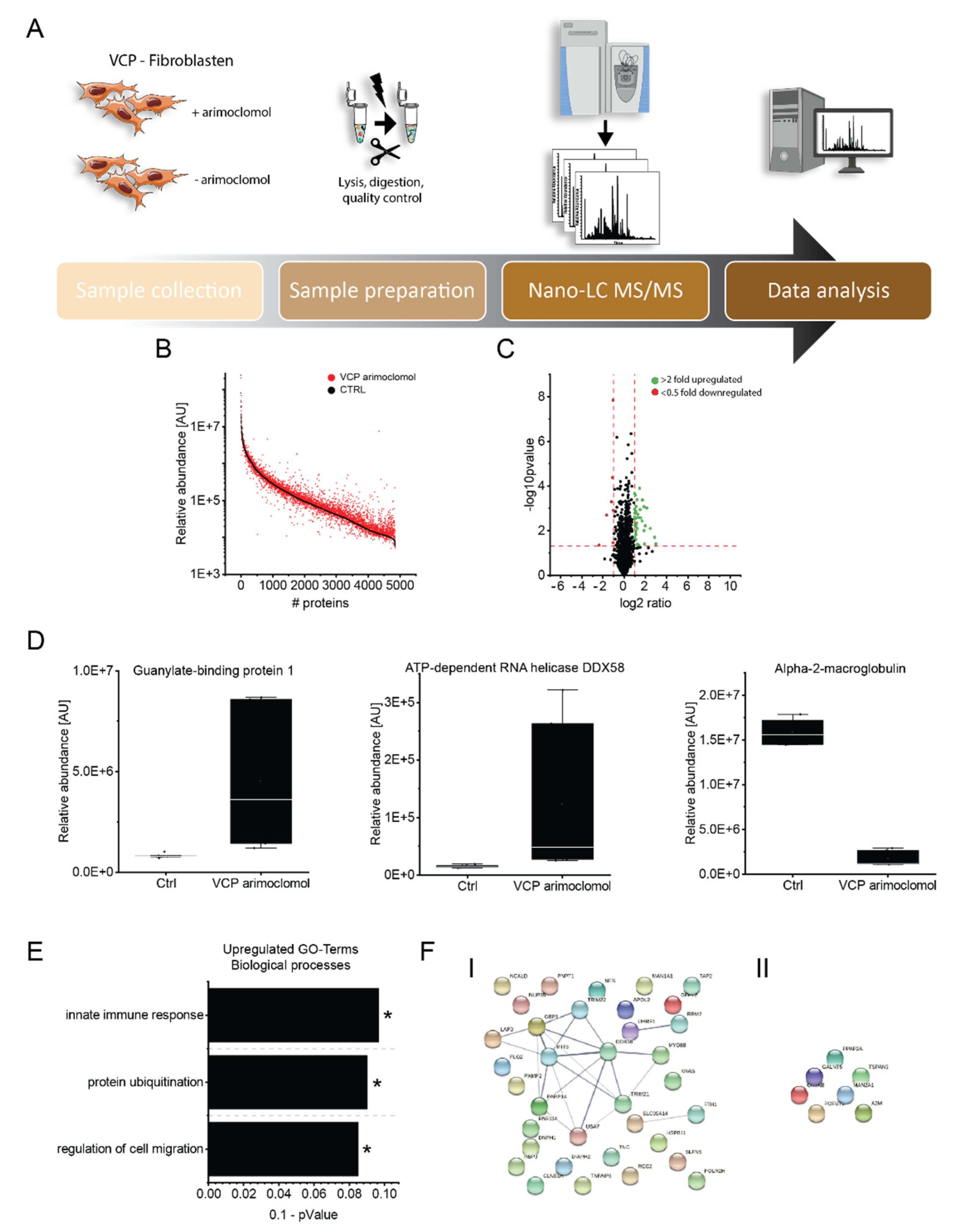

| Pat. No. | Sex | Age of Onset/at Biopsy | Neurological Findings at Time of Biopsy | Creatin–Kinase (U/L) | Muscle Biopsy Findings | Mono–Allelic Pathogenic Variants | Further Remarks |
|---|---|---|---|---|---|---|---|
| 1 | m | 44/46 | Scapula alata, atrophy of proximal UL, proximal paresis of LL | 200–300 | Muscle fiber variability, fiber splitting, focal lipofibromatosis, vacuoles, cell necrosis | c.464G>A, p.[Arg155His] | |
| 2 | m | 30/35 | Cramps and myalgia at muscle exercise, slight proximal paresis of LL | 700–1000 | c.464G>A, p.[Arg155His] | ||
| 3 | m | 50/58 | Cramps and myalgia Proximal and distal UL weakness Distal LL weakness | 432–483 | Rare small angular fibers; rimmed vacuoles; rare cytoplasmic bodies, fibrosis, necrosis (rare) Type 1 fiber predominance | c.648A>G p.[Ile216Met] | No family history |
| 4 | f | 43/45 | Scapula alata; distal UL and LL weakness | Normal | Fiber size variability; small atrophic fibers; some internalized nuclei; rimmed vacuoles; ragged red fibers (rare); Type 1 fiber predominance | c.474G>A p.[Met158Ile] | Scoliosis from childhood |
| 5 | m | 60/63 | Scapula alata; Proximal UL weakness; Distal UL and LL weakness (drop feet) | 317 | Slight fiber size variability; some internalized nuclei; scattered small atrophic fibers; focal increase of connective tissue | c.785C>G p.[Thr262Ser] | |
| 6 | m | 50/53 | Scapula alata Proximal UL, distal LL weakness | 557 | Moderate fiber size variability; some internalized nuclei; scattered small atrophic fibers; focal increase of connective tissue, Rimmed vacuoles | c.464G>A, p.[Arg155His] | Scoliosis, Paget’s bone disease |
| 7 | M | 69/71 | Scapula alata, proximal UL and LL, distal LL weakness | 230 | Slight fiber size variability; some internalized nuclei; scattered small atrophic fibers; focal increase of connective tissue | c.C277C>T, p.[Arg93Cys] | Scoliosis, axonal neuropathy of the legs |
| Accession | Protein Name | Fold Change | p–Value | Function |
|---|---|---|---|---|
| Upregulated upon Arimoclomol–Treatment | ||||
| O95786 | Probable ATP–dependent RNA helicase DDX58 | 8.10 | 0.039 | activates expression of IFN–alpha and IFN–beta interferons and recruits E3 ubiquitin–protein ligases |
| O14879 | Interferon–induced protein with tetratricopeptide repeats 3 | 7.40 | 0.020 | can negatively regulate the apoptotic effects |
| P32455 | Guanylate–binding protein 1 | 5.55 | 0.008 | interaction with ubiquitin–binding protein SQSTM1, which delivers monoubiquitylated proteins to autolysosomes |
| Q15043 | Zinc transporter ZIP14 | 4.47 | 0.001 | regulates insulin receptor signalling, glucose uptake, glycogen synthesis and gluconeogenesis |
| Q03519 | Antigen peptide transporter 2 | 3.97 | 0.006 | typically transports intracellular peptide antigens of 8 to 13 amino acids that arise from cytosolic proteolysis via IFNG–induced immunoproteasome |
| P61601 | Neurocalcin–delta | 3.81 | 0.041 | calcium–dependent regulation of rhodopsin phosphorylation |
| P31350 | Ribonucleoside–diphosphate reductase subunit M2 | 3.74 | 0.001 | catalyzes the biosynthesis of deoxyribonucleotides from the corresponding ribonucleotides and inhibits Wnt signaling |
| Q460N5 | Poly [ADP–ribose] polymerase 14 | 3.72 | 0.010 | regulates pro–inflammatory cytokine production in macrophages in response to IFNG stimulation |
| P02794 | Ferritin heavy chain | 3.32 | 0.000 | stores iron in a soluble, non–toxic, readily available form |
| O95379 | Tumor necrosis factor alpha–induced protein 8 | 3.23 | 0.004 | suppresses the TNF–mediated apoptosis by inhibiting caspase–8 activity |
| Q8IYM9 | E3 ubiquitin–protein ligase TRIM22 | 2.97 | 0.003 | interferon–induced antiviral protein with E3 ubiquitin–protein ligase activity |
| P24821 | Tenascin | 2.85 | 0.001 | implicated in guidance of migrating neurons as well as axons during development, synaptic plasticity as well as neuronal regeneration |
| Q9Y508 | E3 ubiquitin–protein ligase RNF114 | 2.81 | 0.000 | E3 ubiquitin–protein ligase that promotes the ubiquitination of various substrates |
| P48681 | Nestin | 2.56 | 0.045 | required for survival, renewal and mitogen–stimulated proliferation of neural progenitor cells |
| P28838 | Cytosol aminopeptidase | 2.47 | 0.002 | involved in the metabolism of glutathione and in the degradation of glutathione S–conjugates, |
| Q8TCS8 | Polyribonucleotide nucleotidyltransferase 1, mitochondrial | 2.45 | 0.003 | plays a role in mitochondrial morphogenesis and respiration; regulates the expression of the electron transport chain (ETC) components at the mRNA and protein levels |
| Q9NR77 | Peroxisomal membrane protein 2 | 2.42 | 0.008 | contributes to the unspecific permeability of the peroxisomal membrane |
| Q06330 | Recombining binding protein suppressor of hairless | 2.34 | 0.041 | transcriptional regulator that plays a central role in Notch signaling |
| Q9BQE5 | Apolipoprotein L2 | 2.33 | 0.000 | modulates movement of lipids in the cytoplasm or allows the binding of lipids to organelles |
| P01116 | GTPase KRas | 2.25 | 0.005 | plays an important role in the regulation of cell proliferation |
| P41226 | Ubiquitin–like modifier–activating enzyme 7 | 2.21 | 0.001 | activates ubiquitin by first adenylating with ATP its C–terminal glycine residue |
| Q9Y547 | Intraflagellar transport protein 25 homolog | 2.18 | 0.011 | component of the IFT complex B required for sonic hedgehog/SHH signaling |
| Q99836 | Myeloid differentiation primary response protein MyD88 | 2.16 | 0.006 | adapter protein involved in the Toll–like receptor and IL–1 receptor signaling pathway in the innate immune response |
| Q5D862 | Filaggrin–2 | 2.15 | 0.007 | essential for normal cell–cell adhesion in the cornified cell layers |
| Q9P258 | Protein RCC2 | 2.15 | 0.001 | multifunctional protein that may affect its functions by regulating the activity of small GTPases, such as RAC1 and RALA |
| O94808 | Glutamine––fructose–6–phosphate aminotransferase [isomerizing] 2 | 2.14 | 0.015 | controls the flux of glucose into the hexosamine pathway |
| Q96T88 | E3 ubiquitin–protein ligase UHRF1 | 2.14 | 0.025 | E3 ubiquitin–protein ligase activity by mediating the ubiquitination of target proteins |
| P19474 | E3 ubiquitin–protein ligase TRIM21 | 2.09 | 0.000 | E3 ubiquitin–protein ligase whose activity is dependent on E2 enzymes |
| P33908 | Mannosyl–oligosaccharide 1,2–alpha–mannosidase IA | 2.07 | 0.016 | involved in the maturation of Asn–linked oligosaccharides |
| Q08AF3 | Schlafen family member 5 | 2.07 | 0.002 | plays a role in hematopoietic cell differentiation |
| P52434 | DNA–directed RNA polymerases I, II, and III subunit RPABC3 | 2.03 | 0.003 | DNA–dependent RNA polymerase catalyzes the transcription of DNA into RNA |
| P54105 | Methylosome subunit pICln | 2.02 | 0.021 | chaperone that regulates the assembly of spliceosomal U1, U2, U4 and U5 small nuclear ribonucleoproteins |
| O43598 | 2’–deoxynucleoside 5’–phosphate N–hydrolase 1 | 2.02 | 0.001 | catalyzes the cleavage of the N–glycosidic bond of deoxyribonucleoside 5’–monophosphates to yield deoxyribose 5–phosphate |
| O60879 | Protein diaphanous homolog 2 | 2.02 | 0.006 | involved in the regulation of endosome dynamics |
| Q8NFH5 | Nucleoporin NUP35 | 2.01 | 0.000 | functions as a component of the nuclear pore complex |
| Downregulated upon arimoclomol–treatment | ||||
| Q7Z7M9 | Polypeptide N–acetylgalactosaminyltransferase 5 | 0.50 | 0.001 | catalyzes the initial reaction in O–linked oligosaccharide biosynthesis |
| Q16706 | Alpha–mannosidase 2 | 0.49 | 0.000 | catalyzes the first committed step in the biosynthesis of complex N–glycans |
| P02511 | Alpha–crystallin B chain | 0.48 | 0.036 | has chaperone–like activity, preventing aggregation of various proteins under a wide range of stress conditions |
| Q9Y2G5 | GDP–fucose protein O–fucosyltransferase 2 | 0.47 | 0.000 | catalyzes the reaction that attaches fucose through an O–glycosidic linkage to a conserved serine or threonine residue |
| O14494 | Phospholipid phosphatase 1 | 0.44 | 0.001 | regulates phospholipid–mediated signaling pathways |
| O60637 | Tetraspanin–3 | 0.32 | 0.002 | regulates the proliferation and migration of oligodendrocytes |
| P01023 | Alpha–2–macroglobulin | 0.19 | 0.045 | inhibits all four classes of proteinases by a unique ‘trapping’ mechanism |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guettsches, A.-K.; Meyer, N.; Zahedi, R.P.; Evangelista, T.; Muentefering, T.; Ruck, T.; Lacene, E.; Heute, C.; Gonczarowska-Jorge, H.; Schoser, B.; et al. FYCO1 Increase and Effect of Arimoclomol–Treatment in Human VCP–Pathology. Biomedicines 2022, 10, 2443. https://doi.org/10.3390/biomedicines10102443
Guettsches A-K, Meyer N, Zahedi RP, Evangelista T, Muentefering T, Ruck T, Lacene E, Heute C, Gonczarowska-Jorge H, Schoser B, et al. FYCO1 Increase and Effect of Arimoclomol–Treatment in Human VCP–Pathology. Biomedicines. 2022; 10(10):2443. https://doi.org/10.3390/biomedicines10102443
Chicago/Turabian StyleGuettsches, Anne-Katrin, Nancy Meyer, René P. Zahedi, Teresinha Evangelista, Thomas Muentefering, Tobias Ruck, Emmanuelle Lacene, Christoph Heute, Humberto Gonczarowska-Jorge, Benedikt Schoser, and et al. 2022. "FYCO1 Increase and Effect of Arimoclomol–Treatment in Human VCP–Pathology" Biomedicines 10, no. 10: 2443. https://doi.org/10.3390/biomedicines10102443
APA StyleGuettsches, A.-K., Meyer, N., Zahedi, R. P., Evangelista, T., Muentefering, T., Ruck, T., Lacene, E., Heute, C., Gonczarowska-Jorge, H., Schoser, B., Krause, S., Hentschel, A., Vorgerd, M., & Roos, A. (2022). FYCO1 Increase and Effect of Arimoclomol–Treatment in Human VCP–Pathology. Biomedicines, 10(10), 2443. https://doi.org/10.3390/biomedicines10102443






