Characteristics of SARS-CoV-2 Infection in an Actively Monitored Cohort of Patients with Lupus Nephritis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Data Collection
2.2. SARS-CoV-2 Infection Diagnosis and Assessment of Severity
2.3. Statistical Analysis
3. Results
3.1. Study Population
3.2. SARS-CoV-2 Infection Characteristics
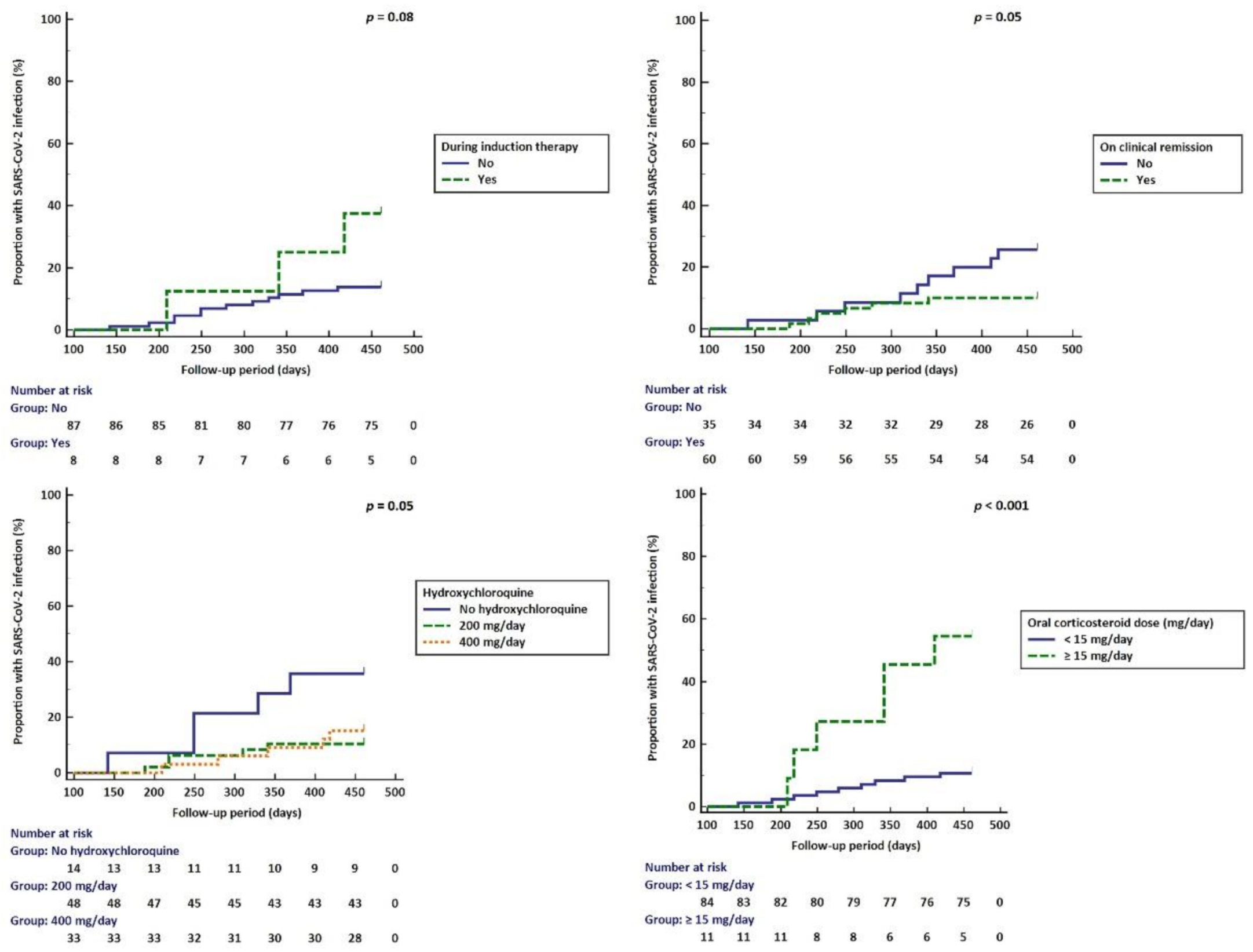
3.3. Risk Factors for SARS-CoV-2 Infection
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. WHO Coronavirus Disease (COVID-19) Dashboard. 2020. Available online: https://covid19.who.int/ (accessed on 1 August 2022).
- Gandhi, R.T.; Lynch, J.B.; del Rio, C. Mild or Moderate COVID-19. N. Engl. J. Med. 2020, 383, 1757–1766. [Google Scholar] [CrossRef] [PubMed]
- Obrișcă, B.; Jurubiță, R.; Andronesi, A.; Sorohan, B.; Achim, C.; Bobeica, R.; Gherghiceanu, M.; Mandache, E.; Ismail, G. Histological predictors of renal outcome in lupus nephritis: The importance of tubulointerstitial lesions and scoring of glomerular lesions. Lupus 2018, 27, 1455–1463. [Google Scholar] [CrossRef] [PubMed]
- Obrișcă, B.; Sorohan, B.; Tuță, L.; Ismail, G. Advances in lupus nephritis pathogenesis: From bench to bedside. Int. J. Mol. Sci. 2021, 22, 3766. [Google Scholar] [CrossRef] [PubMed]
- Saadoun, D.; Vieira, M.; Vautier, M.; Baraliakos, X.; Andreica, I.; da Silva, J.A.; Sousa, M.; Luis, M.; Khmelinskii, N.; Gracía, J.M.A.; et al. SARS-CoV-2 outbreak in immune-mediated inflammatory diseases: The Euro-COVIMID multicentre cross-sectional study. Lancet Rheumatol. 2021, 3, e481–e488. [Google Scholar] [CrossRef]
- Obrișcă, B.; Vornicu, A.; Jurubiță, R.; Achim, C.; Bobeică, R.; Andronesi, A.; Sorohan, B.; Herlea, V.; Procop, A.; Dina, C.; et al. Corticosteroids are the major contributors to the risk for serious infections in autoimmune disorders with severe renal involvement. Clin. Rheumatol. 2021, 40, 3285–3297. [Google Scholar] [CrossRef] [PubMed]
- Mageau, A.; Aldebert, G.; Van Gysel, D.; Papo, T.; Timsit, J.F.; Sacre, K. SARS-CoV-2 infection among inpatients with systemic lupus erythematosus in France: A nationwide epidemiological study. Ann. Rheum. Dis. 2021, 80, 1101–1102. [Google Scholar] [CrossRef]
- Ramirez, G.A.; Gerosa, M.; Beretta, L.; Bellocchi, C.; Argolini, L.M.; Moroni, L.; Della Torre, E.; Artusi, C.; Nicolosi, S.; Caporali, R.; et al. COVID-19 in systemic lupus erythematosus: Data from a survey on 417 patients. Semin. Arthritis. Rheum. 2020, 50, 1150–1157. [Google Scholar] [CrossRef]
- Espinosa, G.; Prieto-González, S.; Llevadot, M.; Marco-Hernández, J.; Martínez-Artuña, A.; Pérez-Isidro, A.; Rifé, E.; Cervera, R. The impact of SARS-CoV-2 coronavirus infection in patients with systemic lupus erythematosus from a single center in Catalonia. Clin. Rheumatol. 2021, 40, 2057–2063. [Google Scholar] [CrossRef]
- Akiyama, S.; Hamdeh, S.; Micic, D.; Sakuraba, A. Prevalence and clinical outcomes of COVID-19 in patients with autoimmune diseases: A systematic review and meta-analysis. Ann. Rheum. Dis. 2021, 80, 384–391. [Google Scholar] [CrossRef]
- Chen, C.; Yao, B.; Yan, M.; Su, K.; Wang, H.; Xu, C. The plight of patients with Lupus Nephritis during the outbreak of COVID-19 in Wuhan, China. J. Rheumatol. 2020, 47, 1452. [Google Scholar] [CrossRef]
- Batu, E.D.; Akca, U.K.; Kısaarslan, A.P.; Sağ, E.; Demir, F.; Demir, S.; Çiçek, S.Ö.; Poyrazoglu, H.; Sozeri, B.; Bilginer, Y.; et al. The performances of the ACR 1997, SLICC 2012, and EULAR/ACR 2019 classification criteria in pediatric systemic lupus erythematosus. J. Rheumatol. 2021, 48, 907–914. [Google Scholar] [CrossRef] [PubMed]
- Petri, M.; Orbai, A.M.; Alarcón, G.S.; Gordon, C.; Merrill, J.T.; Fortin, P.R.; Bruce, I.N.; Isenberg, D.; Wallace, D.J.; Nived, O.; et al. Derivation and validation of the systemic lupus international collaborating clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 2012, 64, 2677–2686. [Google Scholar] [CrossRef] [PubMed]
- Aringer, M.; Costenbader, K.; Daikh, D.; Brinks, R.; Mosca, M.; Ramsey-Goldman, R.; Smolen, J.S.; Wofsy, D.; Boumpas, D.T.; Kamen, D.L.; et al. 2019 European League Against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus. Ann. Rheum. Dis. 2019, 78, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Available online: https://www.covid19treatmentguidelines.nih.gov/ (accessed on 29 September 2021).
- Gianfrancesco, M.; Hyrich, K.L.; Al-Adely, S.; Carmona, L.; Danila, M.I.; Gossec, L.; Izadi, Z.; Jacobsohn, L.; Katz, P.; Lawson-Tovey, S.; et al. Characteristics associated with hospitalisation for COVID-19 in people with rheumatic disease: Data from the COVID-19 Global Rheumatology Alliance physician-reported registry. Ann. Rheum. Dis. 2020, 79, 859–866. [Google Scholar] [CrossRef]
- Fernandez-Ruiz, R.; Paredes, J.L.; Niewold, T.B. COVID-19 in patients with systemic lupus erythematosus: Lessons learned from the inflammatory disease. Transl. Res. 2021, 232, 13–36. [Google Scholar] [CrossRef] [PubMed]
- Sakthiswary, R.; Chuah, H.Y.; Chiang, K.S.; Liew, Y.S.; Aizat, N.A.M. COVID-19 in systemic lupus erythematosus: A pooled analysis and systematic review of case reports and series. Lupus 2021, 27, 9612033211045057. [Google Scholar] [CrossRef] [PubMed]
- Alzahrani, Z.A.; Alghamdi, K.A.; Almaqati, A.S. Clinical characteristics and outcome of COVID-19 in patients with rheumatic diseases. Rheumatol. Int. 2021, 41, 1097–1103. [Google Scholar] [CrossRef] [PubMed]
- Martelli Júnior, H.; Marques, N.P.; Marques, N.C.T.; Gomes De Lucena, E.; Martelli, D.R.B.; Oliveira, E.A. Correspondence on “Clinical course of coronavirus disease 2019 (COVID-19) in a series of 17 patients with systemic lupus erythematosus under long-term treatment with hydroxychloroquine". Ann. Rheum. Dis. 2021; Online ahead of print. [Google Scholar] [CrossRef]
- Boulware, D.R.; Pullen, M.F.; Bangdiwala, A.S.; Pastick, K.A.; Lofgren, S.M.; Okafor, E.C.; Skipper, C.P.; Nascene, A.A.; Nicol, M.R.; Abassi, M.; et al. A Randomized Trial of Hydroxychloroquine as Postexposure Prophylaxis for COVID-19. N. Engl. J. Med. 2020, 383, 517–525. [Google Scholar] [CrossRef]
- Abella, B.S.; Jolkovsky, E.L.; Biney, B.T.; Uspal, J.E.; Hyman, M.C.; Frank, I.; Hensley, S.E.; Gill, S.; Vogl, D.T.; Maillard, I.; et al. Efficacy and Safety of Hydroxychloroquine vs Placebo for Pre-exposure SARS-CoV-2 Prophylaxis among Health Care Workers: A Randomized Clinical Trial. JAMA Intern. Med. 2021, 181, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Geleris, J.; Sun, Y.; Platt, J.; Zucker, J.; Baldwin, M.; Hripcsak, G.; Labella, A.; Manson, D.K.; Kubin, C.; Barr, R.G.; et al. Observational Study of Hydroxychloroquine in Hospitalized Patients with COVID-19. N. Engl. J. Med. 2020, 382, 2411–2418. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.; Loutan, J.; Anders, H.-J. B-cell depletion or belimumab or voclosporin for lupus nephritis? Curr. Opin. Nephrol. Hypertens. 2021, 30, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Bomback, A.S.; Canetta, P.A.; Ahn, W.; Ahmad, S.B.; Radhakrishnan, J.; Appel, G.B. How COVID-19 has changed the management of glomerular diseases. Clin. J. Am. Soc. Nephrol. 2020, 15, 876–879. [Google Scholar] [CrossRef] [PubMed]
- Willicombe, M.; Thomas, D.; McAdoo, S. COVID-19 and Calcineurin Inhibitors: Should They Get Left Out in the Storm? J. Am. Soc. Nephrol. 2020, 31, 1145–1146. [Google Scholar] [CrossRef] [PubMed]



| Variable | Value |
|---|---|
| Number of patients | 95 |
| Number of patients with COVID-19 (n, %) | 15 (15.8%) |
| Symptoms | |
| Fatigue (%) | 73.3% |
| Fever (%) | 46.7% |
| Cough (%) | 20% |
| Odynophagia/sore throat (%) | 20% |
| Myalgia/arthralgia (%) | 40% |
| Dyspnea (%) | 13.3% |
| Loss of taste/smell (%) | 53.3% |
| Diarrhea (%) | 26.7% |
| Outcome | |
| Hospitalization (%) | 40% |
| Length of hospitalization (days) | 11.5 (IQR, 3.75–14) |
| Non-invasive ventilation (%) | 0% |
| Invasive mechanical ventilation (%) | 0% |
| Death due to COVID-19 (%) | 0% |
| Severity of SARS-CoV-2 infection | |
| Mild (%) | 73.3% |
| Moderate (%) | 26.7% |
| Severe (%) | 0% |
| LN evolution following SARS-CoV-2 infection | |
| Increase in SCr of at least 0.5 mg/dL (%) | 20% |
| Increase in proteinuria (%) | 13.3% |
| Increase in hematuria (%) | 6.6% |
| Variable | Entire Cohort | SARS-CoV-2 | No SARS-CoV-2 | p-Value |
|---|---|---|---|---|
| Number of patients | 95 | 15 | 80 | |
| Sex (% females) | 83.2% | 86.7% | 82.3% | 0.83 |
| Age (years) | 39 ± 14 | 38 ± 12 | 40 ± 14 | 0.77 |
| Arterial hypertension (%) | 45.3% | 53.3% | 43.8% | 0.49 |
| Diabetes (%) | 9.5% | 0% | 11.2% | 0.34 |
| Duration of SLE (years) | 7.3 (IQR: 2.3–13.6) | 6.7 (IQR: 2.6–10.7) | 7.3 (IQR: 2.3–15.3) | 0.25 |
| Extrarenal involvement | ||||
| Cardiac | 3.2% | 0% | 3.8% | 0.99 |
| Neurological | 11.6% | 13.3% | 11.2% | 0.68 |
| Hematological | 65% | 73.3% | 65% | 0.53 |
| Articular | 67.4% | 46.7% | 71.2% | 0.07 |
| Serositis | 22.1% | 20% | 22.5% | 0.99 |
| Cutaneous | 61.1% | 40% | 65% | 0.06 |
| Gastro-intestinal tract | 8.4% | 6.7% | 8.8% | 0.99 |
| Laboratory data | ||||
| Serum creatinine (mg/dL) | 1.6 ± 1.9 | 1.87 ± 2.26 | 1.63 ± 1.84 | 0.97 |
| eGFR (mL/min) | 69 ± 32 | 64 ± 33 | 70 ± 32 | 0.56 |
| Serum albumin (g/dL) | 4 ± 0.85 | 3.5 ± 1.2 | 4.1 ± 0.76 | 0.09 |
| Fibrinogen (mg/dL) | 418 ± 121 | 430 ± 169 | 415 ± 112 | 0.75 |
| Hematuria (cells/mmc) | 10 (IQR: 4.3–30.2) | 29 (IQR: 10–118) | 10 (IQR: 4–26.7) | 0.01 |
| 24-h proteinuria (g/day) | 0.3 (IQR: 0–1.2) | 0.8 (IQR: 0.4–2.5) | 0.2 (IQR: 0–1) | 0.13 |
| Leukocytes (cells/mmc) | 7411 ± 3174 | 8702 ± 2465 | 7169 ± 3246 | 0.04 |
| Neutrophils (cell/mmc) | 5007 ± 2447 | 6282 ± 2377 | 4768 ± 2399 | 0.03 |
| Lymphocytes (cells/mmc) | 1647 ± 954 | 1593 ± 789 | 1657 ± 986 | 0.78 |
| Hemoglobin (g/dL) | 12.1 ± 1.9 | 12.4 ± 1.9 | 12.1 ± 1.9 | 0.52 |
| Serum IgA (mg/dL) | 250 (IQR: 153–337) | 224 (IQR: 184–392) | 250 (IQR: 141–333) | 0.54 |
| Serum IgG (mg/dL) | 1141 (IQR: 811–1519) | 1260 (IQR: 573–1550) | 1130 (IQR: 824–1519) | 0.88 |
| Serum IgM (mg/dL) | 86 (IQR: 47–157) | 63 (IQR: 37–149) | 89 (IQR: 47–166) | 0.6 |
| C-reactive protein (mg/L) | 8.9 ± 16.8 | 14.3 ± 29.5 | 8 ± 13.7 | <0.001 |
| Serum C3 (mg/dL) | 98 ± 30 | 96 ± 31 | 98 ± 30 | 0.81 |
| Serum C4 (mg/dL) | 19 ± 9 | 18 ± 7 | 19 ± 9 | 0.63 |
| Immunosuppression | ||||
| On immunosuppression (%) | 83.2% | 80% | 83.8% | 0.71 |
| During induction (%) | 8.4% | 20% | 6.2% | 0.11 |
| During maintenance (%) | 74.7% | 60% | 77.5% | 0.19 |
| Hydroxychloroquine (%) | 85.3% | 66.7% | 88.8% | 0.04 |
| Hydroxychloroquine dose (%) | ||||
| • 200 mg | 50.5% | 33.3% | 53.8% | 0.07 |
| • 400 mg | 34.7% | 33.3% | 35% | |
| Calcineurin inhibitor (%) | 9.5% | 13.3% | 8.8% | 0.63 |
| Mycophenolate mofetil (%) | 34.7% | 46.7% | 32.5% | 0.29 |
| Mycophenolate mofetil dose (g/day) | 1 (IQR: 1–1.5) | 1 (IQR: 1–1.5) | 1 (IQR: 1–1.5) | 0.67 |
| Cumulative cyclophosphamide dose (g) | 3 (IQR: 0.125–7.5) | 2.87 (IQR: 1.1–7.1) | 3 (IQR: 0–8.75) | 0.75 |
| Rituximab (%) | 10.5% | 13.3% | 10% | 0.65 |
| Azathioprine (%) | 15.8% | 6.7% | 17.6% | 0.45 |
| Oral corticosteroids (%) | 60% | 66.7% | 58.8% | 0.56 |
| Corticosteroid dose (mg/day) | 8 (IQR: 5–10) | 16 (IQR: 7–21) | 6 (IQR: 4–10) | 0.007 |
| On clinical remission (%) | 63.2% | 40% | 67.5% | 0.04 |
| Variable | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| Odds Ratio (95%CI) | p Value | Odds Ratio (95%CI) | p Value | |
| eGFR (for 1 mL/min/1.73m2) | 0.99 (0.97–1.012) | 0.54 | - | - |
| Serum albumin (for 1 g/dL) | 0.55 (0.31–0.96) | 0.03 | 1.04 (0.35–3.03) | 0.94 |
| C-reactive protein (for 1 mg/L) | 1.01 (0.98–1.04) | 0.25 | 1.006 (0.97–1.04) | 0.75 |
| Hematuria (for 1 cell/mmc) | 1.00 (0.99–1.001) | 0.8 | - | - |
| On immunosuppression (yes vs. no) | 0.77 (0.19–3.14) | 0.72 | - | - |
| During induction therapy (yes vs. no) | 3.75 (0.79–17.7) | 0.09 | 3.91 (0.56–30.4) | 0.1 |
| During maintenance therapy (yes vs. no) | 0.43 (0.13–1.38) | 0.16 | - | - |
| On hydroxychloroquine (yes vs. no) | 0.25 (0.07–0.91) | 0.03 | 0.09 (0.01–0.59) | 0.01 |
| Hydroxychloroquine dose (vs. no HCQ) | - | - | - | - |
| • 200 mg | 0.2 (0.05–0.87) | 0.03 | - | - |
| • 400 mg | 0.32 (0.07–1.36) | 0.12 | - | - |
| On CNI (yes vs. no) | 1.6 (0.29–8.59) | 0.58 | - | - |
| On MMF (yes vs. no) | 1.81 (0.59–5.55) | 0.29 | - | - |
| On azathioprine (yes vs. no) | 0.33 (0.04–2.77) | 0.31 | - | - |
| On rituximab (yes vs. no) | 1.38 (0.26–7.26) | 0.7 | - | - |
| Current oral corticosteroid dose (≥15 mg/d vs. <15 mg/d) | 10 (2.53–39.49) | 0.005 | 7.69 (1.3–45.46) | 0.02 |
| Cumulative cyclophosphamide dose (≥3 g vs. <3 g) | 0.75 (0.22–2.6) | 0.65 | - | - |
| With clinical remission (yes vs. no) | 0.27 (0.08–0.85) | 0.02 | 0.28 (0.05–1.45) | 0.72 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Obrișcă, B.; Vornicu, A.; Jurubiță, R.; Mocanu, V.; Dimofte, G.; Andronesi, A.; Sorohan, B.; Achim, C.; Micu, G.; Bobeică, R.; et al. Characteristics of SARS-CoV-2 Infection in an Actively Monitored Cohort of Patients with Lupus Nephritis. Biomedicines 2022, 10, 2423. https://doi.org/10.3390/biomedicines10102423
Obrișcă B, Vornicu A, Jurubiță R, Mocanu V, Dimofte G, Andronesi A, Sorohan B, Achim C, Micu G, Bobeică R, et al. Characteristics of SARS-CoV-2 Infection in an Actively Monitored Cohort of Patients with Lupus Nephritis. Biomedicines. 2022; 10(10):2423. https://doi.org/10.3390/biomedicines10102423
Chicago/Turabian StyleObrișcă, Bogdan, Alexandra Vornicu, Roxana Jurubiță, Valentin Mocanu, George Dimofte, Andreea Andronesi, Bogdan Sorohan, Camelia Achim, Georgia Micu, Raluca Bobeică, and et al. 2022. "Characteristics of SARS-CoV-2 Infection in an Actively Monitored Cohort of Patients with Lupus Nephritis" Biomedicines 10, no. 10: 2423. https://doi.org/10.3390/biomedicines10102423
APA StyleObrișcă, B., Vornicu, A., Jurubiță, R., Mocanu, V., Dimofte, G., Andronesi, A., Sorohan, B., Achim, C., Micu, G., Bobeică, R., Dina, C., & Ismail, G. (2022). Characteristics of SARS-CoV-2 Infection in an Actively Monitored Cohort of Patients with Lupus Nephritis. Biomedicines, 10(10), 2423. https://doi.org/10.3390/biomedicines10102423





