Exosomal microRNA Differential Expression in Plasma of Young Adults with Chronic Mild Traumatic Brain Injury and Healthy Control
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Protocol
2.2. Plasma Collection
2.3. ExomiRNA Purification
2.4. ExomiRNA Profiling
2.5. Network Analysis
2.6. Statistical Analysis
3. Results
3.1. Demographics of the Study Population
3.2. Differential Expression of Plasma ExomiRNAs
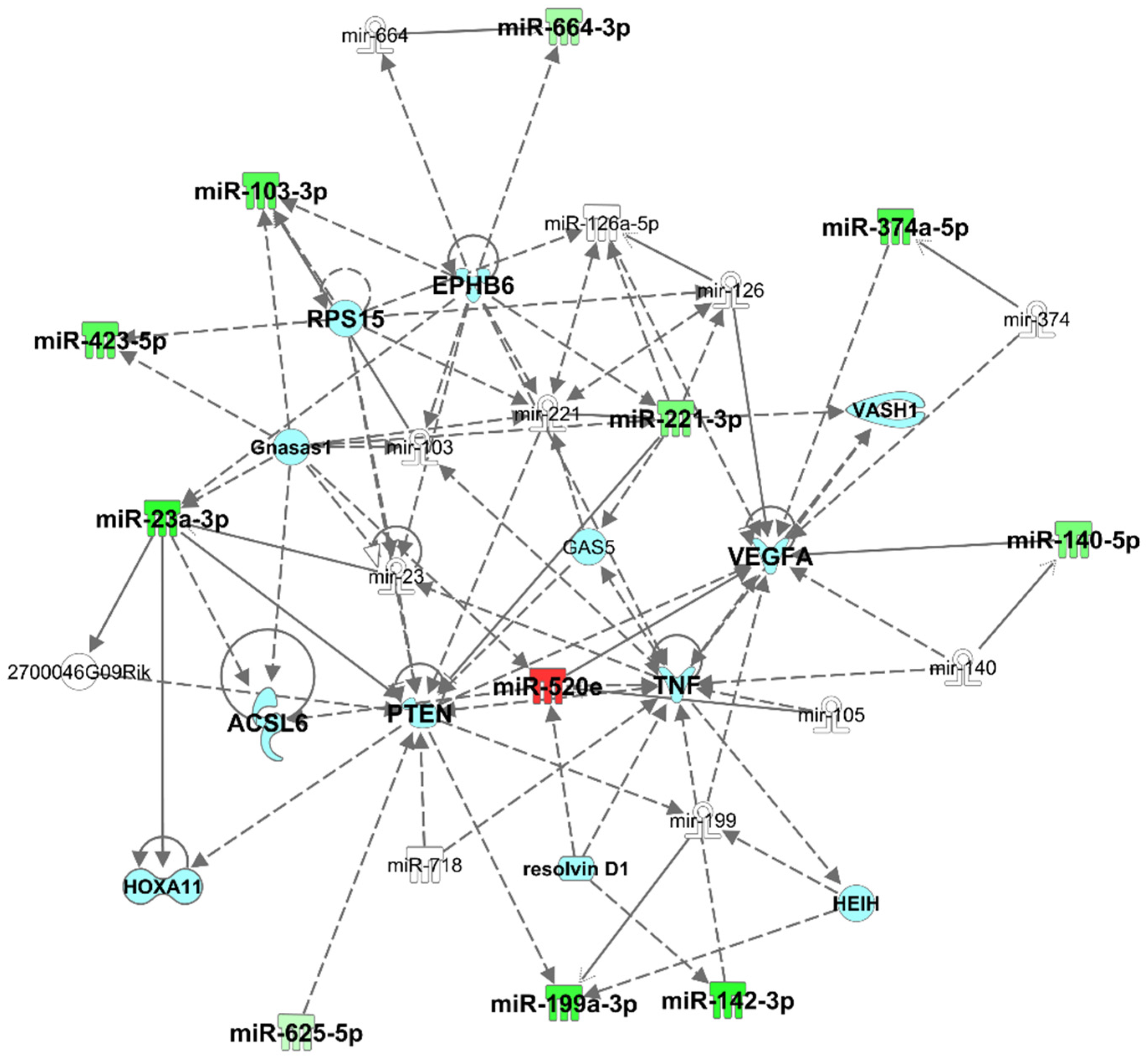
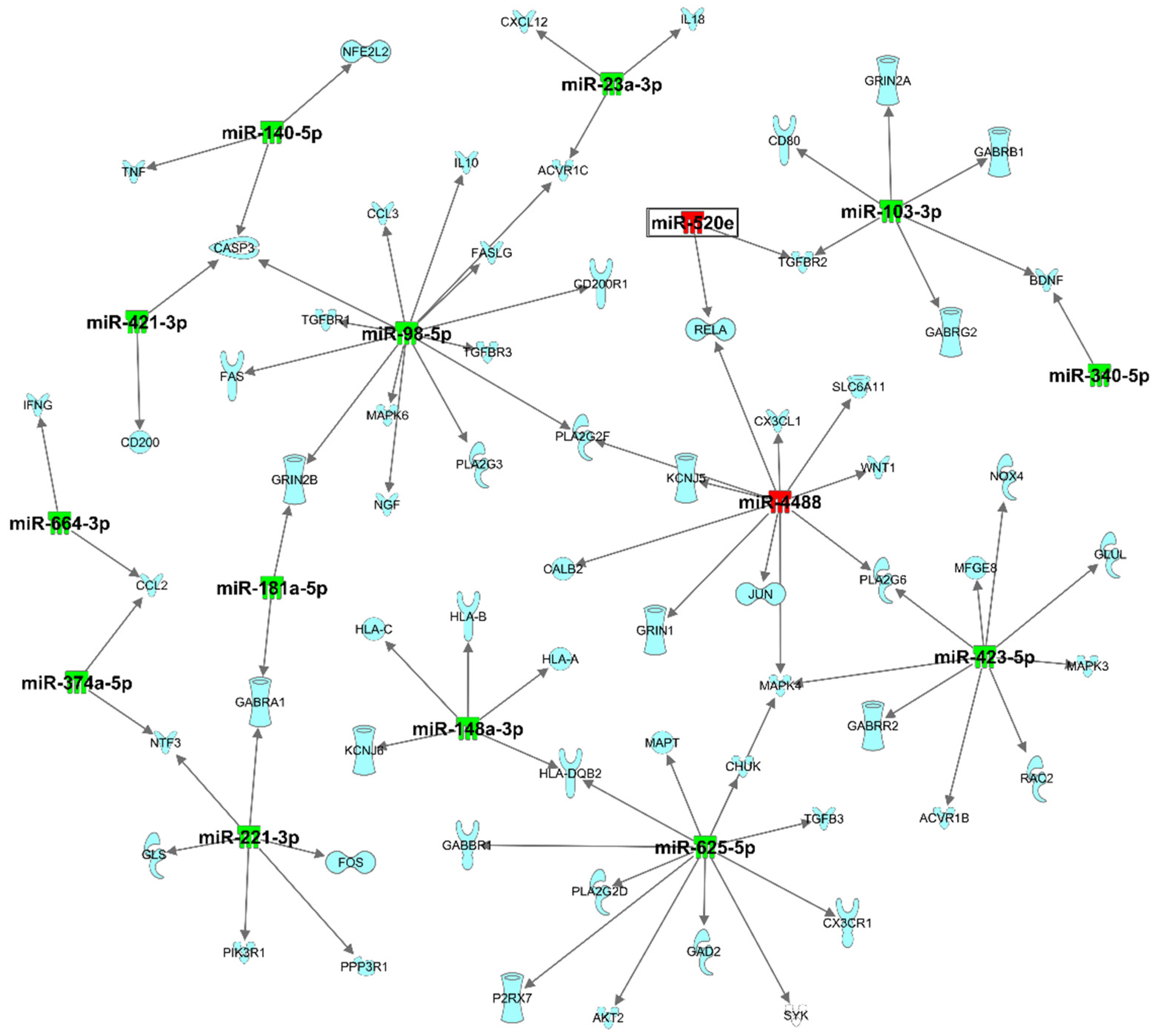
3.3. Pathway Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Langlois, J.A.; Rutland-Brown, W.; Wald, M.M. The epidemiology and impact of traumatic brain injury: A brief overview. J. Head Trauma Rehabil. 2006, 21, 375–378. [Google Scholar] [CrossRef] [Green Version]
- Dikmen, S.; Machamer, J.; Temkin, N. Mild traumatic brain injury: Longitudinal study of cognition, functional status, and post-traumatic symptoms. J. Neurotrauma 2017, 34, 1524–1530. [Google Scholar] [CrossRef]
- Frieden, T.R.; Houry, D.; Baldwin, G. Traumatic Brain Injury in the United States: Epidemiology and Rehabilitation. CDC NIH Rep. Congr. 2015, 1–74. Available online: https://www.cdc.gov/traumaticbraininjury/pdf/tbi_report_to_congress_epi_and_rehab-a.pdf (accessed on 7 August 2021).
- Lehman, E.J.; Hein, M.J.; Baron, S.L.; Gersic, C.M. Neurodegenerative causes of death among retired national football league players. Neurology 2012, 79, 1970–1974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Devoto, C.; Lai, C.; Qu, B.X.; Guedes, V.A.; Leete, J.; Wilde, E.; Walker, W.C.; Diaz-Arrastia, R.; Kenney, K.; Gill, J. Exosomal MicroRNAs in Military Personnel with Mild Traumatic Brain Injury: Preliminary Results from the Chronic Effects of Neurotrauma Consortium Biomarker Discovery Project. J. Neurotrauma 2020, 37, 2482–2492. [Google Scholar] [CrossRef] [PubMed]
- Seabury, S.A.; Gaudette, É.; Goldman, D.P.; Markowitz, A.J.; Brooks, J.; McCrea, M.A.; Okonkwo, D.O.; Manley, G.T.; Adeoye, O.; Badjatia, N.; et al. Assessment of Follow-up Care After Emergency Department Presentation for Mild Traumatic Brain Injury and Concussion: Results From the TRACK-TBI Study. JAMA Netw. Open 2018, 1, e180210. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.H.; Johnson, V.E.; Stewart, W. Chronic neuropathologies of single and repetitive TBI: Substrates of dementia? Nat. Rev. Neurol. 2013, 9, 211–221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Centers for Disease Control and Prevention. Surveillance Report of Traumatic Brain Injury-Related Emergency Department Visits, Hospitalizations, and Deaths—United States, 2014. 2019; Available online: https://www.cdc.gov/traumaticbraininjury/pdf/TBI-Surveillance-Report-FINAL_508.pdf (accessed on 7 August 2021).
- McGregor, K.; Pentland, B. Head injury rehabilitation in the U.K.: An economic perspective. Soc. Sci. Med. 1997, 45, 295–303. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Report to Congress on Mild Traumatic Brain Injury in the United States: Steps to Prevent a Serious Public Health Problem; CDC: Atlanta, GA, USA, 2003. [Google Scholar]
- Friedman, R.C.; Farh, K.K.H.; Burge, C.B.; Bartel, D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009, 19, 92–105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, Y.; Croce, C.M. The role of MicroRNAs in human cancer. Signal Transduct. Target. Ther. 2016, 1, 15004. [Google Scholar] [CrossRef] [Green Version]
- Mitchell, P.S.; Parkin, R.K.; Kroh, E.M.; Fritz, B.R.; Wyman, S.K.; Pogosova-Agadjanyan, E.L.; Peterson, A.; Noteboom, J.; O’Briant, K.C.; Allen, A.; et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. USA 2008, 105, 10513–10518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watson, C.N.; Belli, A.; Di Pietro, V. Small non-coding RNAs: New class of biomarkers and potential therapeutic targets in neurodegenerative disease. Front. Genet. 2019, 10, 364. [Google Scholar] [CrossRef]
- Xi, Y.; Nakajima, G.; Gavin, E.; Morris, C.G.; Kudo, K.; Hayashi, K.; Ju, J. Systematic analysis of microRNA expression of RNA extracted from fresh frozen and formalin-fixed paraffin-embedded samples. RNA 2007, 13, 1668–1674. [Google Scholar] [CrossRef] [Green Version]
- Atif, H.; Hicks, S.D. A Review of MicroRNA Biomarkers in Traumatic Brain Injury. J. Exp. Neurosci. 2019, 13, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kay, T.; Harrington, D.; Adams, R. Definition of mild traumatic brain injury: American congress of rehabilitation medicine. J. Head Trauma Rehabil. 1993, 8, 74–85. [Google Scholar] [CrossRef]
- Caplan, L.J.; Ivins, B.; Poole, J.H.; Vanderploeg, R.D.; Jaffee, M.S.; Schwab, K. The structure of postconcussive symptoms in 3 us military samples. J. Head Trauma Rehabil. 2010, 25, 447–458. [Google Scholar] [CrossRef]
- King, P.R.; Donnelly, K.T.; Donnelly, J.P.; Dunnam, M.; Warner, G.; Kittleson, C.J.; Bradshaw, C.B.; Alt, M.; Meier, S.T. Psychometric study of the Neurobehavioral Symptom Inventory. J. Rehabil. Res. Dev. 2012, 49, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Medvedev, O.N.; Theadom, A.; Barker-Collo, S.; Feigin, V. Distinguishing between enduring and dynamic concussion symptoms: Applying Generalisability theory to the Rivermead Post Concussion Symptoms Questionnaire (RPQ). PeerJ 2018, 6, e5676. [Google Scholar] [CrossRef] [PubMed]
- Geiss, G.K.; Bumgarner, R.E.; Birditt, B.; Dahl, T.; Dowidar, N.; Dunaway, D.L.; Fell, H.P.; Ferree, S.; George, R.D.; Grogan, T.; et al. Direct multiplexed measurement of gene expression with color-coded probe pairs. Nat. Biotechnol. 2008, 26, 317–325. [Google Scholar] [CrossRef]
- Wienholds, E.; Plasterk, R.H.A. MicroRNA function in animal development. FEBS Lett. 2005, 579, 5911–5922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sulhan, S.; Lyon, K.A.; Shapiro, L.A.; Huang, J.H. Neuroinflammation and blood–brain barrier disruption following traumatic brain injury: Pathophysiology and potential therapeutic targets. J. Neurosci. Res. 2020, 98, 19–28. [Google Scholar] [CrossRef] [Green Version]
- Zacchigna, S.; Lambrechts, D.; Carmeliet, P. Neurovascular signalling defects in neurodegeneration. Nat. Rev. Neurosci. 2008, 9, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Argandona, E.G.; Bengoetxea, H.; Ortuzar, N.; Bulnes, S.; Rico-Barrio, I.; Vicente Lafuente, J. Vascular Endothelial Growth Factor: Adaptive Changes in the Neuroglialvascular Unit. Curr. Neurovascular Res. 2012, 9, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Nasr, I.W.; Chun, Y.; Kannan, S. Neuroimmune responses in the developing brain following traumatic brain injury. Exp. Neurol. 2019, 320, 112957. [Google Scholar] [CrossRef]
- Edwards, K.A.; Pattinson, C.L.; Guedes, V.A.; Peyer, J.; Moore, C.; Davis, T.; Devoto, C.; Turtzo, L.C.; Latour, L.; Gill, J.M. Inflammatory Cytokines Associate With Neuroimaging After Acute Mild Traumatic Brain Injury. Front. Neurol. 2020, 11, 348. [Google Scholar] [CrossRef]
- Johnson, V.E.; Stewart, J.E.; Begbie, F.D.; Trojanowski, J.Q.; Smith, D.H.; Stewart, W. Inflammation and white matter degeneration persist for years after a single traumatic brain injury. Brain 2013, 136, 28–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loane, D.J.; Kumar, A.; Stoica, B.A.; Cabatbat, R.; Faden, A.I. Progressive neurodegeneration after experimental brain trauma: Association with chronic microglial activation. J. Neuropathol. Exp. Neurol. 2014, 73, 14–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scott, G.; Zetterberg, H.; Jolly, A.; Cole, J.H.; De Simoni, S.; Jenkins, P.O.; Feeney, C.; Owen, D.R.; Lingford-Hughes, A.; Howes, O.; et al. Minocycline reduces chronic microglial activation after brain trauma but increases neurodegeneration. Brain 2018, 141, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Krichevsky, A.; Grad, Y.; Hayes, G.D.; Kosik, K.S.; Church, G.M.; Ruvkun, G. Identification of many microRNAs that copurify with polyribosomes in mammalian neurons. Proc. Natl. Acad. Sci. USA 2004, 101, 360–365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaban, V.; Clarke, G.J.B.; Skandsen, T.; Islam, R.; Einarsen, C.E.; Vik, A.; Damås, J.K.; Mollnes, T.E.; Håberg, A.K.; Pischke, S.E. Systemic Inflammation Persists the First Year after Mild Traumatic Brain Injury: Results from the Prospective Trondheim Mild Traumatic Brain Injury Study. J. Neurotrauma 2020, 37, 2120–2130. [Google Scholar] [CrossRef] [Green Version]
- Benigni, M.; Ricci, C.; Jones, A.R.; Giannini, F.; Al-Chalabi, A.; Battistini, S. Identification of miRNAs as Potential Biomarkers in Cerebrospinal Fluid from Amyotrophic Lateral Sclerosis Patients. Neuromol. Med. 2016, 18, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Joilin, G.; Leigh, P.N.; Newbury, S.F.; Hafezparast, M. An Overview of MicroRNAs as Biomarkers of ALS. Front. Neurol. 2019, 10, 186. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.X.; Visavadiya, N.P.; Pandya, J.D.; Nelson, P.T.; Sullivan, P.G.; Springer, J.E. Mitochondria-associated microRNAs in rat hippocampus following traumatic brain injury. Exp. Neurol. 2015, 265, 84–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Landgraf, P.; Rusu, M.; Sheridan, R.; Sewer, A.; Iovino, N.; Aravin, A.; Pfeffer, S.; Rice, A.; Kamphorst, A.O.; Landthaler, M.; et al. A Mammalian microRNA Expression Atlas Based on Small RNA Library Sequencing. Cell 2007, 129, 1401–1414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sonkoly, E.; Ståhle, M.; Pivarcsi, A. MicroRNAs and immunity: Novel players in the regulation of normal immune function and inflammation. Semin. Cancer Biol. 2008, 18, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Deneen, B.; Ho, R.; Lukaszewicz, A.; Hochstim, C.J.; Gronostajski, R.M.; Anderson, D.J. The Transcription Factor NFIA Controls the Onset of Gliogenesis in the Developing Spinal Cord. Neuron 2006, 52, 953–968. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, A.Y.S.; Woo, J.; Sardar, D.; Lozzi, B.; Bosquez Huerta, N.A.; Lin, C.C.J.; Felice, D.; Jain, A.; Paulucci-Holthauzen, A.; Deneen, B. Region-Specific Transcriptional Control of Astrocyte Function Oversees Local Circuit Activities. Neuron 2020, 106, 992–1008. [Google Scholar] [CrossRef] [PubMed]
- Guerriero, R.M.; Giza, C.C.; Rotenberg, A. Glutamate and GABA Imbalance Following Traumatic Brain Injury. Curr. Neurol. Neurosci. Rep. 2015, 15, 27. [Google Scholar] [CrossRef]
- Drexel, M.; Puhakka, N.; Kirchmair, E.; Hörtnagl, H.; Pitkänen, A.; Sperk, G. Expression of GABA receptor subunits in the hippocampus and thalamus after experimental traumatic brain injury. Neuropharmacology 2015, 88, 122–133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, H.; Low, C.M.; Moody, O.A.; Jenkins, A.; Traynelis, S.F. Ionotropic GABA and glutamate receptor mutations and human neurologic diseases. Mol. Pharmacol. 2015, 88, 203–217. [Google Scholar] [CrossRef]
- Sheinerman, K.S.; Toledo, J.B.; Tsivinsky, V.G.; Irwin, D.; Grossman, M.; Weintraub, D.; Hurtig, H.I.; Chen-Plotkin, A.; Wolk, D.A.; McCluskey, L.F.; et al. Circulating brain-enriched microRNAs as novel biomarkers for detection and differentiation of neurodegenerative diseases. Alzheimer’s Res. Ther. 2017, 9, 89. [Google Scholar] [CrossRef] [PubMed]
- Rokad, D.; Ghaisas, S.; Harischandra, D.S.; Jin, H.; Anantharam, V.; Kanthasamy, A.; Kanthasamy, A.G. Role of neurotoxicants and traumatic brain injury in α-synuclein protein misfolding and aggregation. Brain Res. Bull. 2017, 133, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Goedert, M. Alpha-synuclein and neurodegenerative diseases. Nat. Rev. Neurosci. 2001, 2, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Acosta, S.A.; Tajiri, N.; de la Pena, I.; Bastawrous, M.; Sanberg, P.R.; Kaneko, Y.; Borlongan, C.V. Alpha-Synuclein as a pathological link between chronic traumatic brain injury and parkinson’s disease. J. Cell. Physiol. 2015, 230, 1024–1032. [Google Scholar] [CrossRef] [PubMed]
- Ninkina, N.; Peters, O.; Millership, S.; Salem, H.; van der Putten, H.; Buchman, V.L. γ-Synucleinopathy: Neurodegeneration associated with overexpression of the mouse protein. Hum. Mol. Genet. 2009, 18, 1779–1794. [Google Scholar] [CrossRef] [Green Version]
- Surgucheva, I.; He, S.; Rich, M.C.; Sharma, R.; Ninkina, N.N.; Stahel, P.F.; Surguchov, A. Role of synucleins in traumatic brain injury—An experimental in vitro and in vivo study in mice. Mol. Cell. Neurosci. 2014, 63, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Winder, A.D.; Maniar, K.P.; Wei, J.J.; Liu, D.; Scholtens, D.M.; Lurain, J.R.; Schink, J.C.; Buttin, B.M.; Filiaci, V.L.; Lankes, H.A.; et al. Synuclein-γ in uterine serous carcinoma impacts survival: An NRG Oncology/Gynecologic Oncology Group study. Cancer 2017, 123, 1144–1155. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.X.; Rajeev, B.W.; Stromberg, A.J.; Ren, N.; Tang, G.; Huang, Q.; Rigoutsos, I.; Nelson, P.T. The expression of microRNA miR-107 decreases early in Alzheimer’s disease and may accelerate disease progression through regulation of β-site amyloid precursor protein-cleaving enzyme 1. J. Neurosci. 2008, 28, 1213–1223. [Google Scholar] [CrossRef]
- Zhao, J.; Fu, Y.; Yasvoina, M.; Shao, P.; Hitt, B.; O’Connor, T.; Logan, S.; Maus, E.; Citron, M.; Berry, R.; et al. β-site amyloid precursor protein cleaving enzyme 1 levels become elevated in neurons around amyloid plaques: Implications for Alzheimer’s disease pathogenesis. J. Neurosci. 2007, 27, 3639–3649. [Google Scholar] [CrossRef]
- Guedes, V.A.; Kenney, K.; Shahim, P.; Qu, B.X.; Lai, C.; Devoto, C.; Walker, W.C.; Nolen, T.; Diaz-Arrastia, R.; Gill, J.M. Exosomal neurofilament light: A prognostic biomarker for remote symptoms after mild traumatic brain injury? Neurology 2020, 94, e2412–e2423. [Google Scholar] [CrossRef]
| Characteristic | Overall (n = 40) | Chronic mTBI (n = 29) | Control (n = 11) | χ2 or t | p |
|---|---|---|---|---|---|
| Demographic | |||||
| Age, Mean (SD) | 24.80 (5.22) | 25.59 (5.36) | 22.73 (4.41) | 1.576 | 0.123 |
| Gender, n (%) | |||||
| Males | 19 (47.5) | 15 (51.7) | 4 (36.4) | 0.755 | 0.488 |
| Females | 21 (52.5) | 14 (48.3) | 7 (63.6) | ||
| Weight (kg), Mean (SD) | 69.83 (14.64) | 70.18 (13.29) | 68.91 (18.45) | 0.243 | 0.810 |
| Height (cm), Mean (SD) | 167.52 (10.94) | 168.26 (10.48) | 165.56 (12.38) | 0.691 | 0.494 |
| BMI, Mean (SD) | 24.74 (3.77) | 24.65 (3.40) | 24.98 (4.81) | −0.247 | 0.807 |
| Ethnicity/Race, n (%) | |||||
| Hispanic | 6 (15.0) | 4 (13.8) | 2 (18.2) | 1.038 | 0.904 |
| White | 23 (57.5) | 17 (58.6) | 6 (54.5) | ||
| Black | 1 (2.5) | 1 (3.4) | 0 (0.0) | ||
| Asian | 8 (20.0) | 6 (20.7) | 2 (18.2) | ||
| Other | 2 (5.0) | 1 (3.4) | 1 (9.1) | ||
| Handedness, n (%) | |||||
| Right | 37 (92.5) | 27 (93.1) | 10 (90.9) | 0.055 | 1.000 |
| Left | 3 (7.5) | 2 (6.9) | 1 (9.1) | ||
| Education, n (%) | |||||
| In college | 31 (77.5) | 22 (75.9) | 9 (81.8) | 0.162 | 1.000 |
| In graduate school | 9 (22.5) | 7 (24.1) | 2 (18.2) | ||
| Marital Status, n (%) | |||||
| Single | 36 (90.0) | 26 (89.7) | 10 (90.9) | 0.412 | 0.814 |
| Married | 4 (10.0) | 3 (10.3) | 1 (9.1) | ||
| Employment Status, n (%) | |||||
| Yes | 30 (75.0) | 23 (79.3) | 7 (63.6) | 1.045 | 0.418 |
| No | 10 (25.0) | 6 (20.7) | 4 (36.4) | ||
| Clinical | |||||
| RPQ Total, Mean (SD) | 12.58 (12.42) | 16.76 (12.14) | 1.55 (2.07) | 6.505 | <0.001 |
| NSI Total, Mean (SD) | 15.43 (14.06) | 19.86 (13.91) | 3.73 (4.65) | 5.489 | <0.001 |
| Somatic/Sensory, Mean (SD) | 5.58 (5.81) | 7.34 (5.83) | 0.91 (1.81) | 5.304 | <0.001 |
| Cognitive, Mean (SD) | 2.75 (2.88) | 3.55 (2.89) | 0.64 (1.50) | 4.156 | <0.001 |
| Affective, Mean (SD) | 7.10 (6.42) | 8.97 (6.56) | 2.18 (1.94) | 5.023 | <0.001 |
| Injury Characteristics | |||||
| Number of Injuries, Mean (SD) | 2.55 (1.33) | N/A | |||
| Single Injury | 9 (31.0) | N/A | |||
| Multiple Injuries | 20 (69.0) | N/A | |||
| Time since the last Injury (years), Mean (SD) | 4.48 (5.00) | N/A | |||
| Mechanism of Injury, n (%) | |||||
| Sports-related | 14 (48.3) | N/A | |||
| Head hit | 6 (20.7) | N/A | |||
| High-level Falls | 5 (17.2) | N/A | |||
| Military-related | 3 (10.3) | N/A | |||
| Car accident | 1 (3.4) | N/A |
| Probe Name | Target Sequence | Log2FC | Adjusted p-Value |
|---|---|---|---|
| Upregulated | |||
| hsa-miR-520e | AAAGUGCUUCCUUUUUGAGGG | 0.98 | 0.03 |
| hsa-miR-499b-3p | AACAUCACUGCAAGUCUUAACA | 0.83 | 0.01 |
| hsa-miR-520b | AAAGUGCUUCCUUUUAGAGGG | 0.43 | 0.04 |
| hsa-miR-4488 | AGGGGGCGGGCUCCGGCG | 0.42 | 0.03 |
| Downregulated | |||
| hsa-miR-625-5p | AGGGGGAAAGUUCUAUAGUCC | −0.96 | 0.08 |
| hsa-miR-421 | AUCAACAGACAUUAAUUGGGCGC | −1.39 | 0.08 |
| hsa-miR-664a-3p | UAUUCAUUUAUCCCCAGCCUACA | −1.43 | 0.08 |
| hsa-miR-28-3p | CACUAGAUUGUGAGCUCCUGGA | −1.49 | 0.04 |
| hsa-miR-125a-5p | UCCCUGAGACCCUUUAACCUGUGA | −2.10 | 0.04 |
| hsa-miR-222-3p | AGCUACAUCUGGCUACUGGGU | −2.14 | 0.09 |
| hsa-miR-140-5p | CAGUGGUUUUACCCUAUGGUAG | −2.17 | 0.07 |
| hsa-miR-98-5p | UGAGGUAGUAAGUUGUAUUGUU | −2.32 | 0.09 |
| hsa-miR-148a-3p | UCAGUGCACUACAGAACUUUGU | −2.63 | 0.06 |
| hsa-miR-423-5p | UGAGGGGCAGAGAGCGAGACUUU | −2.65 | 0.09 |
| hsa-miR-107 | AGCAGCAUUGUACAGGGCUAUCA | −2.75 | 0.07 |
| hsa-miR-181a-5p | AACAUUCAACGCUGUCGGUGAGU | −2.81 | 0.09 |
| hsa-miR-374a-5p | UUAUAAUACAACCUGAUAAGUG | −2.86 | 0.09 |
| hsa-miR-340-5p | UUAUAAAGCAAUGAGACUGAUU | −2.87 | 0.07 |
| hsa-miR-29b-3p | UAGCACCAUUUGAAAUCAGUGUU | −2.95 | 0.05 |
| hsa-miR-191-5p | CAACGGAAUCCCAAAAGCAGCUG | −3.03 | 0.08 |
| hsa-miR-199a-3p | ACAGUAGUCUGCACAUUGGUUA | −3.13 | 0.05 |
| hsa-miR-126-3p | UCGUACCGUGAGUAAUAAUGCG | −3.13 | 0.09 |
| hsa-miR-23a-3p | AUCACAUUGCCAGGGAUUUCC | −3.36 | 0.04 |
| hsa-miR-142-3p | UGUAGUGUUUCCUACUUUAUGGA | −3.39 | 0.07 |
| hsa-miR-223-3p | UGUCAGUUUGUCAAAUACCCCA | −3.62 | 0.04 |
| Diseases and Disorders | p-Value Range | Number of Molecules |
|---|---|---|
| Neurological disease | 4.58 × 10−2–4.85 × 10−14 | 14 |
| Organismal injury and abnormality | 4.95 × 10−2–4.85 × 10−14 | 23 |
| Psychological disease | 4.58 × 10−2–4.85 × 10−14 | 13 |
| Cancer | 4.95 × 10−2–7.96 × 10−13 | 21 |
| Reproductive system disease | 4.85 × 10−2–1.49 × 10−12 | 20 |
| Molecular and Cellular Functions | p-Value Range | Number of Molecules |
| Cell cycle | 4.02 × 10−2–2.46 × 10−6 | 4 |
| Cellular movement | 4.88 × 10−2–2.46 × 10−6 | 12 |
| Cellular development | 4.47 × 10−2–5.25 × 10−6 | 12 |
| Cellular growth and proliferation | 4.47 × 10−2–5.25 × 10−6 | 12 |
| Cell death and survival | 3.99 × 10−2–8.71 × 10−5 | 11 |
| Physiological System Development and Function | p-Value Range | Number of Molecules |
| Organismal development | 3.74 × 10−2–6.45 × 10−9 | 11 |
| Organismal functions | 8.69 × 10−4–7.19 × 10−4 | 2 |
| Tissue morphology | 8.90 × 10−5–1.72 × 10−3 | 3 |
| Hematological system development and functions | 4.47 × 10−2–1.98 × 10−3 | 6 |
| Immune cell trafficking | 2.74 × 10−2–1.98 × 10−3 | 2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vorn, R.; Suarez, M.; White, J.C.; Martin, C.A.; Kim, H.-S.; Lai, C.; Yun, S.-J.; Gill, J.M.; Lee, H. Exosomal microRNA Differential Expression in Plasma of Young Adults with Chronic Mild Traumatic Brain Injury and Healthy Control. Biomedicines 2022, 10, 36. https://doi.org/10.3390/biomedicines10010036
Vorn R, Suarez M, White JC, Martin CA, Kim H-S, Lai C, Yun S-J, Gill JM, Lee H. Exosomal microRNA Differential Expression in Plasma of Young Adults with Chronic Mild Traumatic Brain Injury and Healthy Control. Biomedicines. 2022; 10(1):36. https://doi.org/10.3390/biomedicines10010036
Chicago/Turabian StyleVorn, Rany, Maiko Suarez, Jacob C. White, Carina A. Martin, Hyung-Suk Kim, Chen Lai, Si-Jung Yun, Jessica M. Gill, and Hyunhwa Lee. 2022. "Exosomal microRNA Differential Expression in Plasma of Young Adults with Chronic Mild Traumatic Brain Injury and Healthy Control" Biomedicines 10, no. 1: 36. https://doi.org/10.3390/biomedicines10010036
APA StyleVorn, R., Suarez, M., White, J. C., Martin, C. A., Kim, H.-S., Lai, C., Yun, S.-J., Gill, J. M., & Lee, H. (2022). Exosomal microRNA Differential Expression in Plasma of Young Adults with Chronic Mild Traumatic Brain Injury and Healthy Control. Biomedicines, 10(1), 36. https://doi.org/10.3390/biomedicines10010036







