Tick-Borne Surveillance Patterns in Perceived Non-Endemic Geographic Areas: Human Tick Encounters and Disease Outcomes
Abstract
1. Introduction
2. Materials and Methods
2.1. Texans and Ticks (TTS) Survey
TTS Respondent Tick Bite Encounters
2.2. Multi-Layer Thematic Mapping and Statistical Analysis
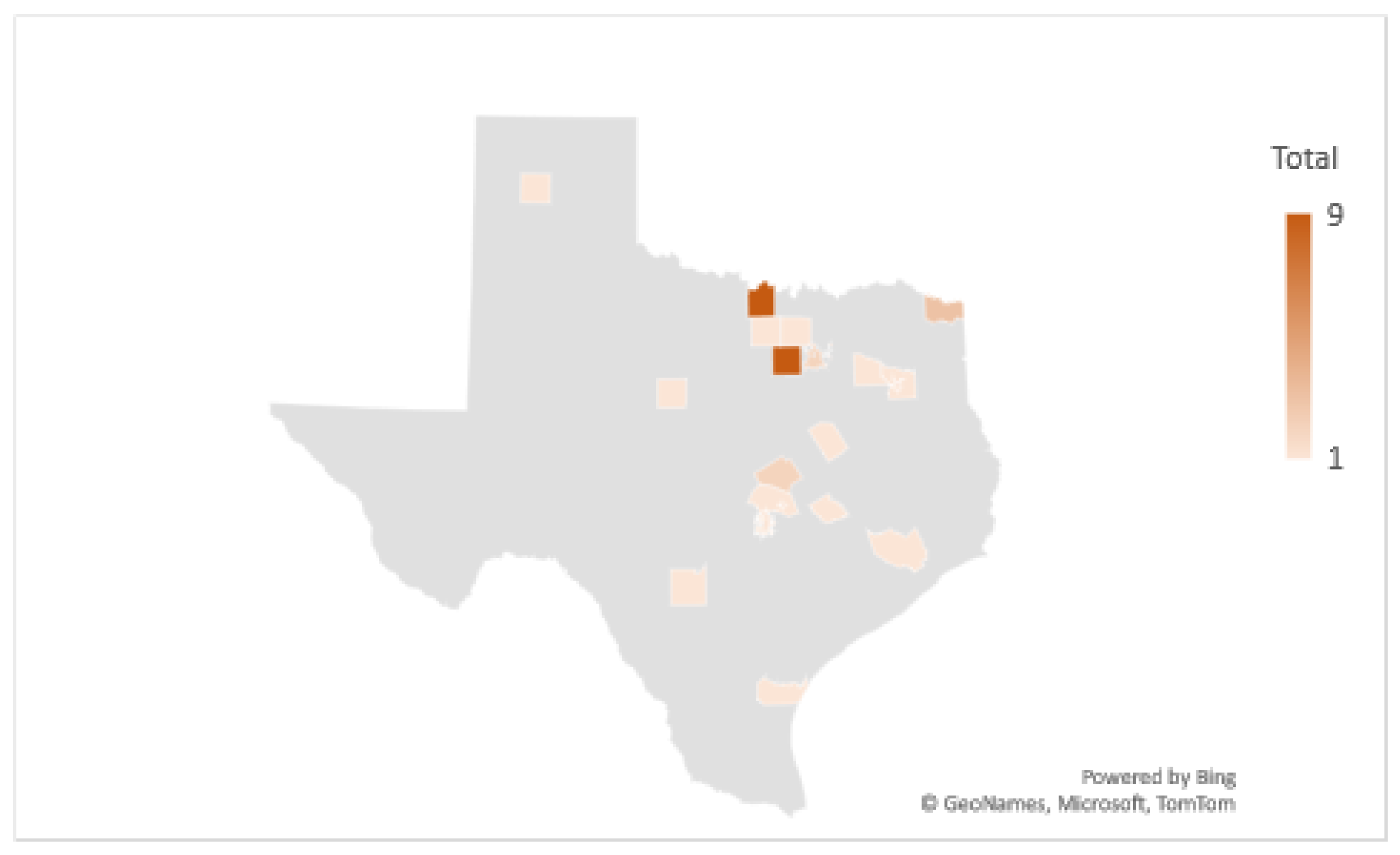
2.2.1. Comparison Mapping of TTS Respondents to Human Lyme Disease Cases
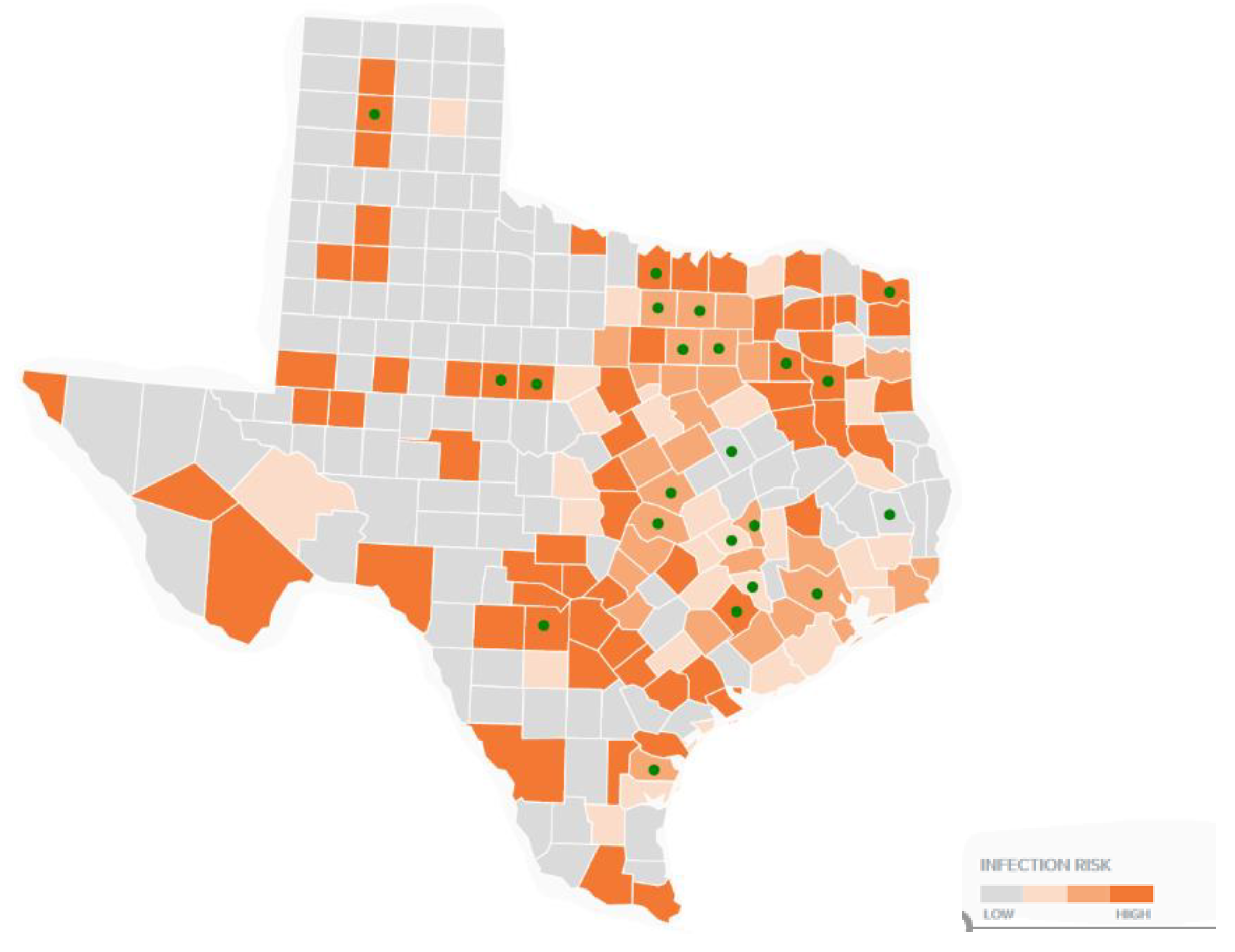
2.2.2. Comparison Mapping of TTS Respondents to Canine Tick-Borne Diseases
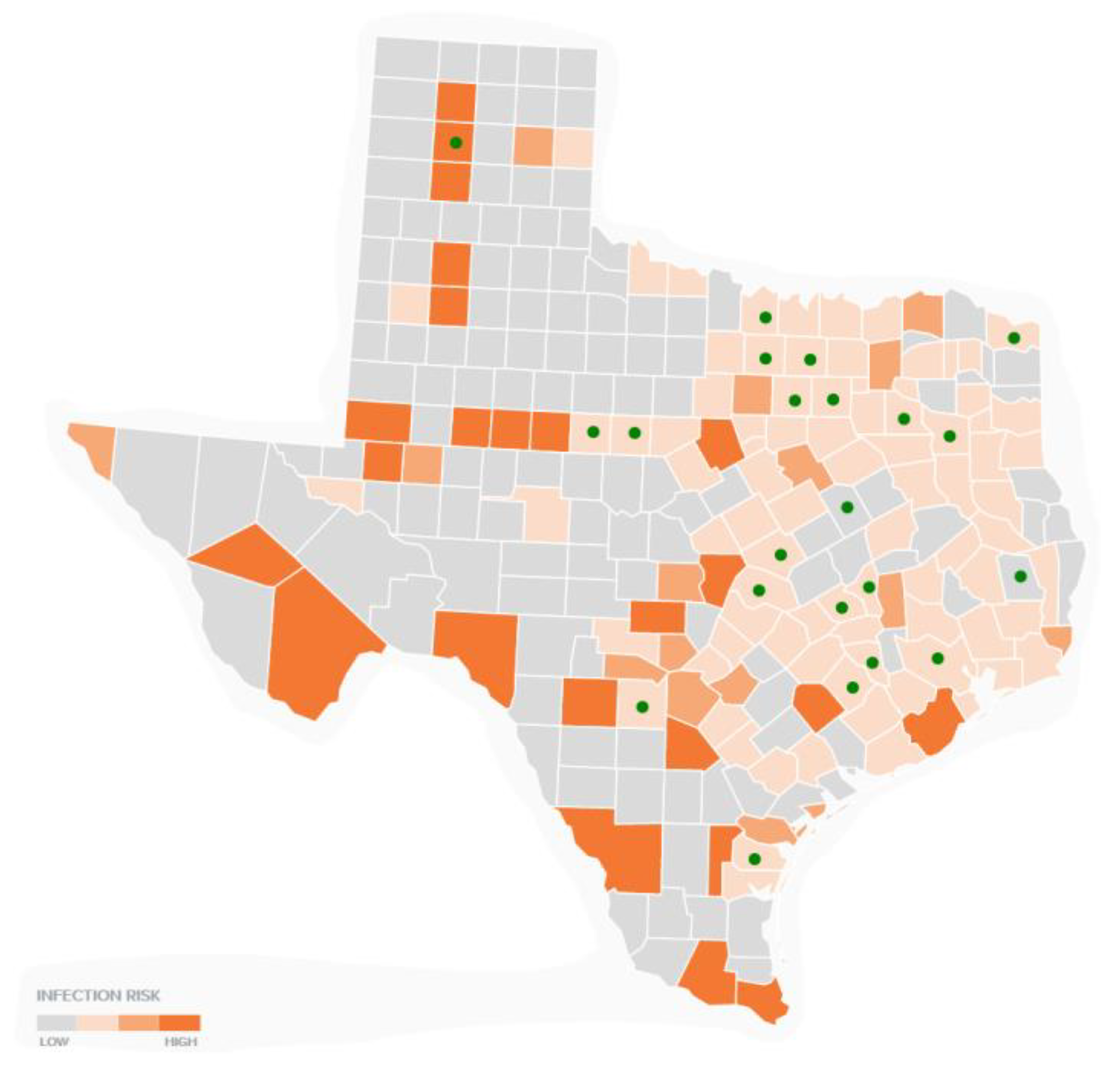
2.2.3. Comparison Mapping of TTS Respondents by Neighboring County and Eco-Regions
3. Results
3.1. TTS Respondent Demographics, Symptoms, and Diagnostic Information
3.2. Multi-Layer Thematic Mapping of TTS Respondents to Human Lyme Disease Cases
3.3. Multi-Layer Thematic Mapping of TTS Respondents to Canine Tick-Borne Disease Cases
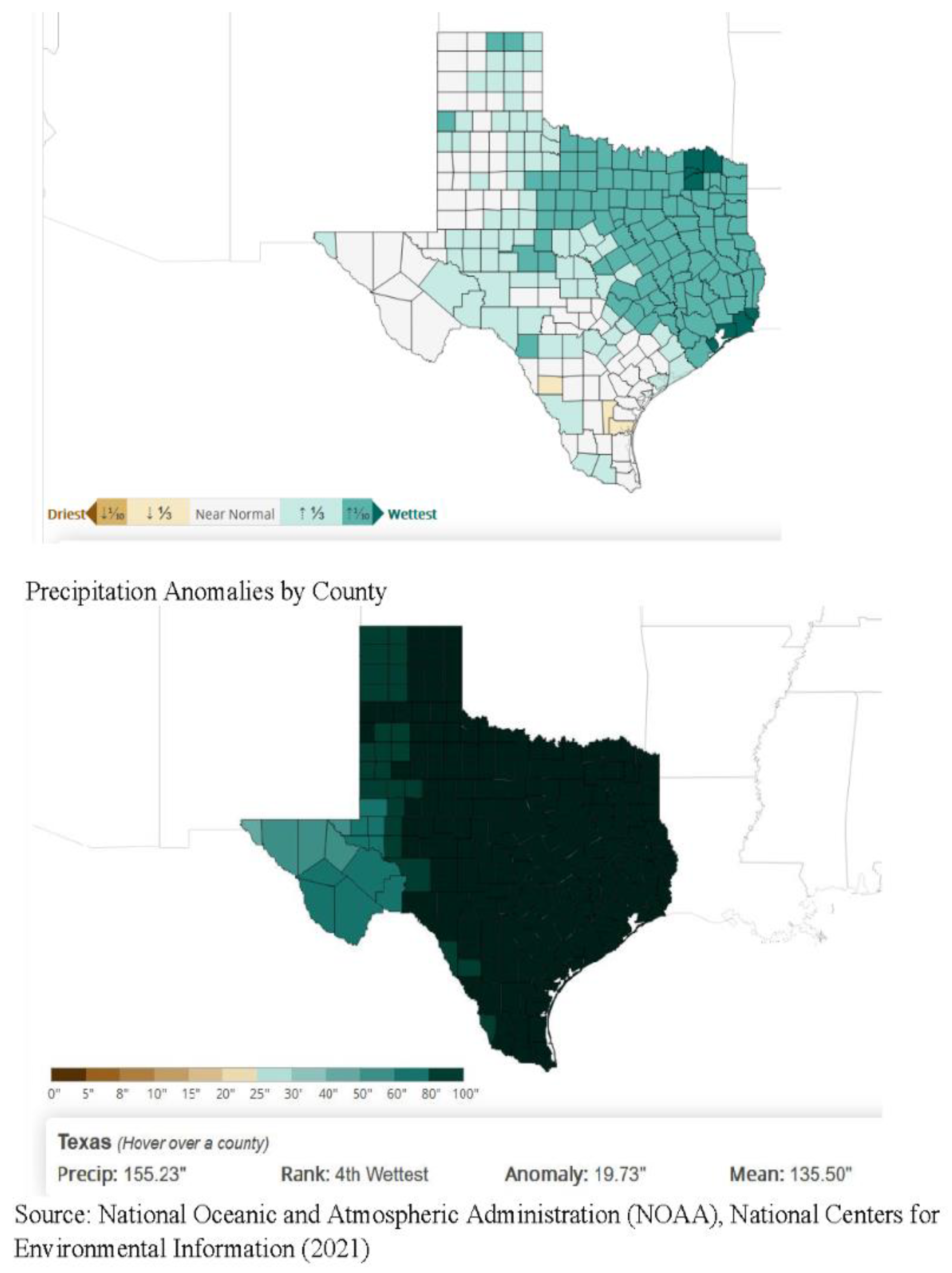
3.4. Multi-Layer Thematic Mapping of TTS Respondents to Eco-Regions and Precipitation Suitable for Tick Populations
4. Discussion
4.1. Significance of Findings
4.2. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hazen, B. Vector-Borne Diseases in the United States. SciLine. Available online: www.sciline.org/evidence-blog/vector-borne-diseases (accessed on 18 December 2019).
- U.S. Department of Health and Human Services. Tick-Borne Disease Working Group. HHS.Gov. Available online: www.hhs.gov/ash/advisory-committees/tick-bornedisease/index.html (accessed on 18 December 2019).
- Hinckley, A.F.; Connally, N.P.; Meek, J.I.; Johnson, B.J.; Kemperman, M.M.; Feldman, K.A.; Feldman, K.A.; White, J.L.; Mead, P.S. Lyme Disease Testing by Large Commercial Laboratories in the United States. Clin. Infect. Dis. 2014, 59, 676–681. [Google Scholar] [CrossRef]
- Nelson, C.A.; Saha, S.; Kugeler, K.J.; Delorey, M.J.; Shankar, M.B.; Hinckley, A.F.; Mead, P.S. Incidence of Clinician-Diagnosed Lyme Disease, United States, 2005–2010. Emerg. Infect. Dis. 2015, 21, 1625–1631. [Google Scholar] [CrossRef]
- DeLong, A.; Hsu, M.; Kotsoris, H. Estimation of cumulative Number of Post-Treatment Lyme Disease Cases in the US, 2016 and 2020. BMC Public Health 2019, 19. [Google Scholar] [CrossRef]
- Institute of Medicine. Critical Needs and Gaps in Understanding Prevention, Amelioration, and Resolution of Lyme and Other Tick-Borne Diseases; National Academies Press (US): Washington, DC, USA, 2011. [Google Scholar] [CrossRef]
- Berghoff, W. Chronic Lyme Disease and Co-Infections: Differential Diagnosis. Open Neurol. J. 2012, 6, 158–178. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Borrelia Miyamotoi Disease Tick-Borne Diseases Ticks CDC. Available online: www.cdc.gov/ticks/tick-bornediseases/borrelia-miyamotoi.html (accessed on 18 December 2019).
- Stricker, R.B.; Fesler, M.C. Chronic Lyme Disease: A Working Case Definition. Am. J. Infect. Dis. 2018, 14, 1–44. [Google Scholar] [CrossRef]
- Hook, S.A.; Nawrocki, C.C.; Meek, J.I.; Feldman, K.A.; White, J.L.; Connally, N.P.; Hinckley, A.F. Human-Tick Encounters as a Measure of Tickborne Disease Risk in Lyme Disease Endemic Areas. Zoonoses Public Health 2021, 2021. [Google Scholar] [CrossRef]
- Szonyi, B.; Srinath, I.; Esteve-Gassent, M.; Lupiani, B.; Ivanek, R. Exploratory Spatial Analysis of Lyme Disease in Texas—What Can We Learn from the Reported Cases? BMC Public Health 2015, 15, 924. [Google Scholar] [CrossRef]
- Dandashi, J.A.; Nizamutdinov, D.; Dayawansa, S.; Fonkem, E.; Huang, J.H. Texas Occurrence of Lyme Disease and Its Neurological Manifestations. J. Neuroinfect. Dis. 2016, 7, 217. [Google Scholar] [CrossRef]
- Harvey, W.T.; Salvato, P. ‘Lyme Disease’: Ancient Engine of an Unrecognized Borreliosis Pandemic? Med. Hypotheses 2003, 60, 742–759. [Google Scholar] [CrossRef]
- Feria-Arroyo, T.P.; Castro-Arellano, I.; Gordillo-Perez, G.; Cavazos, A.L.; Vargas-Sandoval, M.; Grover, A.; Torres, J.; Medina, R.F.; de León, A.A.P.; Esteve-Gassent, M.D. Implications of Climate Change on the Distribution of the Tick Vector Ixodes scapularis and Risk for Lyme Disease in the Texas-Mexico Transboundary Region. Parasites Vectors 2014, 25, 199. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, S.P. The Elusive Understanding of Lyme Disease in Non-Endemic Geographic Areas: An Exploratory Survey of Patients with Chronic Symptoms in Texas. J. Patient Exp. 2020, 7, 1621–1626. [Google Scholar] [CrossRef] [PubMed]
- Rebman, A.W.; Bechtold, K.T.; Yang, T.; Mihm, E.A.; Soloski, M.J.; Novak, C.B.; Aucott, J.N. The Clinical, Symptom, and Quality-of-Life Characterization of a Well-Defined Group of Patients with Posttreatment Lyme Disease Syndrome. Front. Med. 2017, 4, 224. [Google Scholar] [CrossRef]
- Centers for Disease Control. County-Level Lyme Disease Data from 2000–2018. Available online: https://www.cdc.gov/lyme/datasurveillance/index.html (accessed on 18 June 2020).
- Companion Animal Parasite Council (CAPC). Available online: capcvet.org/maps/#/ (accessed on 18 November 2020).
- Steere, A.C.; Dhar, A.; Hernandez, J.; Fischer, P.A.; Sikand, V.K.; Schoen, R.T.; Nowakowski, J.; McHugh, G.; Persing, D.H. Systemic Symptoms without Erythema Migrans as the Presenting Picture of Early Lyme Disease. Am. J. Med. 2003, 114, 58–62. [Google Scholar] [CrossRef]
- Omodior, O.; Luetke, M.; Kianersi, S.; Colón, A. Predictors of Tick Exposure Risk-Reduction Behavior in Indiana. J. Community Health 2020, 45, 862–870. [Google Scholar] [CrossRef]
- Aucott, J.N.; Seifter, A. Misdiagnosis of Early Lyme Disease as the Summer Flu. Orthop. Rev. 2011, 3, e14. [Google Scholar] [CrossRef]
- Belongia, E.A.; Reed, K.D.; Mitchell, P.D.; Mueller-Rizner, N.; Vandermause, M.; Finkel, M.F.; Kazmierczak, J.J. Tick-Borne Infections as a Cause of Nonspecific Febrile Illness in Wisconsin. Clin. Infect. Dis. 2001, 32, 1434–1439. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, S.A.; Singer, C. Gastrointestinal and Hepatic Manifestations of Tick-Borne Diseases in the United States. Clin. Infect. Dis. 2002, 34, 1206–1212. [Google Scholar] [CrossRef]
- Mitchell, E.A.; Williamson, P.C.; Billingsley, P.M.; Seals, J.P.; Ferguson, E.E.; Allen, M.S. Frequency and Distribution of Rickettsiae, Borreliae, and Ehrlichiae Detected in Human-Parasitizing Ticks, Texas, USA. Emerg. Infect. Dis. 2016, 22, 312–315. [Google Scholar] [CrossRef]
- Bowman, D.; Little, S.E.; Lorentzen, L.; Shields, J.; Sullivan, M.P.; Carlin, E.P. Prevalence and Geographic Distribution of Dirofilaria Immitis, Borrelia Burgdorferi, Ehrlichia Canis, and Anaplasma Phagocytophilum in Dogs in the United States: Results of a National Clinic-Based Serologic Survey. Vet. Parasitol. 2009, 160, 138–148. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Epidemiology and Statistics. Available online: www.cdc.gov/ehrlichiosis/stats/index.html (accessed on 15 June 2020).
- Little, S.E.; Heise, S.R.; Blagburn, B.L.; Callister, S.M.; Mead, P.S. Lyme Borreliosis in Dogs and Humans in the USA. Trends Parasitol. 2010, 26, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Texas Parks & Wildlife Foundation. n.d. Texas Ecoregions Texas Parks & Wildlife Department. Available online: tpwd.texas.gov/education/hunter-education/online-course/wildlife-conservation/texas-ecoregions (accessed on 18 December 2019).
- Eisen, R.J.; Eisen, L.; Beard, C.B. County-Scale Distribution of Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae) in the Continental United States. J. Med. Entomol. 2016, 53, 349–386. [Google Scholar] [CrossRef]
- Eisen, R.J.; Kugeler, K.J.; Eisen, L.; Beard, C.B.; Paddock, C.D. Tick-Borne Zoonoses in the United States: Persistent and Emerging Threats to Human Health. ILAR J. 2017, 58, 319–335. [Google Scholar] [CrossRef]
- Centers for Disease Control. One Health Model. Available online: https://www.cdc.gov/onehealth/basics/index.html (accessed on 16 May 2021).
- Omodior, O.; Kianersi, S.; Luetke, M. Spatial clusters and Non-Spatial Predictors of Tick-Borne Disease Diagnosis in Indiana. J. Community Health 2019, 44, 1111–1119. [Google Scholar] [CrossRef]
- Hill, D.; Holmes, T. Provider Knowledge, Attitudes, and Practices Regarding Lyme Disease in Arkansas. J. Community Health 2015, 40, 339–346. [Google Scholar] [CrossRef]
- Johnson, L.; Shapiro, M.; Mankoff, J. Removing the Mask of Average Treatment Effects in Chronic Lyme Disease Research Using Big Data and Subgroup Analysis. Healthcare 2018, 6, 124. [Google Scholar] [CrossRef]
- Mader, E.M.; Ganser, C.; Geiger, A.; Harrington, L.C.; Foley, J.; Smith, R.L.; Mateus-Pinilla, N.; Teel, P.D.; Eisen, R.J. A Survey of Tick Surveillance and Control Practices in the United States. J. Med. Entomol. 2020. [Google Scholar] [CrossRef]
- Andreasen, A.M.; Dehlendorff, P.B.; Knudtzen, F.C.; Bødker, R.; Kjær, L.J.; Skarphedinsson, S. Spatial and Temporal Patterns of Lyme Neuroborreliosis on Funen, Denmark from 1995–2014. Science 2020, 10, 7796. [Google Scholar] [CrossRef]




| Disease | Pathogen(s) | Life Stages Infected |
|---|---|---|
| Anaplasmosis | Anaplasma phagocytophilum | Nymphs, Adults |
| Babesiosis | Babesia microti | Nymphs, Adults |
| Borrelia miyamotoi disease | Borrelia miyamotoi | Larvae, Nymphs, Adults |
| Ehrlichiosis | Ehrlichia muris eauclairensis | Nymphs, Adults |
| Lyme disease | Borrelia burgdorferi sensu stricto, Borrelia mayonii | Nymphs, Adults |
| Powassan virus disease | Powassan virus (lineage II/deer tick lineage) | Larvae, Nymphs, Adults |
| Age (N = 95) | ||
|---|---|---|
| 18–30 | 19 | 20% |
| 31–45 | 25 | 26% |
| 46–60 | 35 | 37% |
| A family member, 17 or younger | 7 | 7% |
| older than 60 years of age | 9 | 9% |
| p < 0.001 | ||
| Occupation (N = 95) | ||
| Not working | 32 | 34% |
| Student | 20 | 21% |
| Work Outdoors | 6 | 6% |
| Work indoors | 31 | 33% |
| Work indoors, but spend a lot of time outside | 18 | 19% |
| Diagnostic Type (N = 95) | ||
| Clinical diagnosis by a doctor | 46 | 48% |
| Western Blot with 5 or more bands, i.e., “CDC Positive” | 22 | 23% |
| Western Blot with some bands positive, Not “CDC Positive” | 33 | 35% |
| IGenX or other speciality lab | 39 | 41% |
| Tick Bite Encounter Location | ||
| Recalled a tick bite | 43 | 45% |
| p = 0.4119 | ||
| Symptoms Experienced over the Last Five Years (N = 92) | ||
| Extreme or unusual fatigue | 84 | 91% |
| Brain fog or concentration problems | 84 | 91% |
| Headaches | 82 | 89% |
| Neck or back pain | 82 | 89% |
| Depression or anxiety | 80 | 87% |
| Flu-like illness | 79 | 86% |
| Other unusual or ongoing symptoms | 78 | 85% |
| Joint Pain or Swelling/Migrating Joint Pain | 77 | 84% |
| Gastrointestinal/stomach problems | 76 | 83% |
| Night sweats | 71 | 77% |
| Fever | 62 | 67% |
| Rash | 57 | 62% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maxwell, S.P.; McNeely, C.L.; Thomas, K.; Brooks, C. Tick-Borne Surveillance Patterns in Perceived Non-Endemic Geographic Areas: Human Tick Encounters and Disease Outcomes. Healthcare 2021, 9, 771. https://doi.org/10.3390/healthcare9060771
Maxwell SP, McNeely CL, Thomas K, Brooks C. Tick-Borne Surveillance Patterns in Perceived Non-Endemic Geographic Areas: Human Tick Encounters and Disease Outcomes. Healthcare. 2021; 9(6):771. https://doi.org/10.3390/healthcare9060771
Chicago/Turabian StyleMaxwell, Sarah P., Connie L. McNeely, Kevin Thomas, and Chris Brooks. 2021. "Tick-Borne Surveillance Patterns in Perceived Non-Endemic Geographic Areas: Human Tick Encounters and Disease Outcomes" Healthcare 9, no. 6: 771. https://doi.org/10.3390/healthcare9060771
APA StyleMaxwell, S. P., McNeely, C. L., Thomas, K., & Brooks, C. (2021). Tick-Borne Surveillance Patterns in Perceived Non-Endemic Geographic Areas: Human Tick Encounters and Disease Outcomes. Healthcare, 9(6), 771. https://doi.org/10.3390/healthcare9060771






