An Observational Study to Evaluate the Association between Intestinal Permeability, Leaky Gut Related Markers, and Metabolic Health in Healthy Adults
Abstract
1. Introduction
2. Materials and Methods
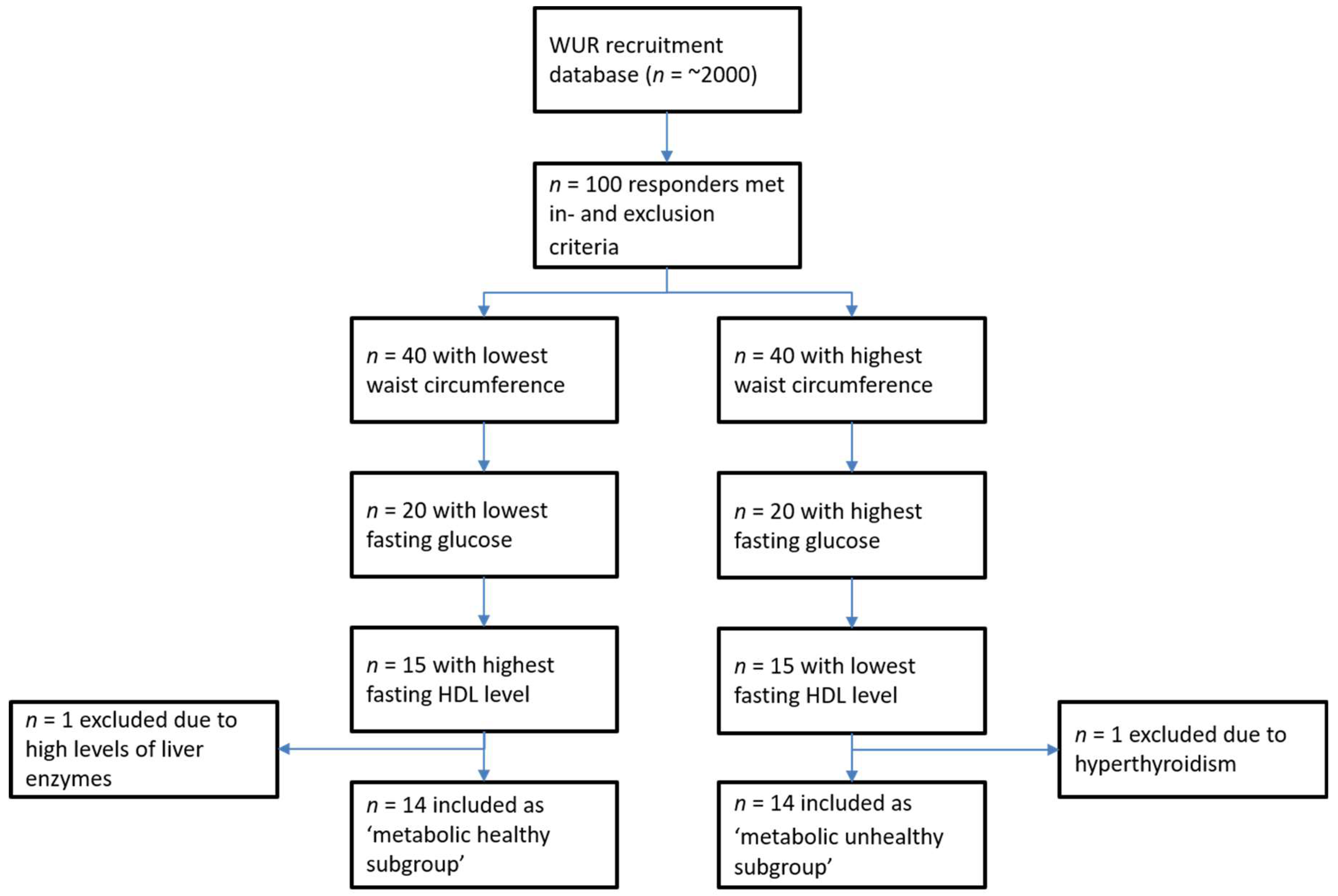
2.1. Subjects
2.2. Ethical Considerations
2.3. Study Design
2.4. MS Gut Permeability Test
2.5. Analysis of Metabolic Markers and LGM
2.6. Statistics
3. Results
3.1. Selection and Baseline Characteristics of Subjects
3.2. Unchallenged MS Permeability Test
3.3. Challenged MS Permeability Test
3.4. Correlation between Metabolic Health and Gut Permeability
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Saltiel, A.R.; Olefsky, J.M. Inflammatory mechanisms linking obesity and metabolic disease. J. Clin. Investig. 2017, 127, 1–4. [Google Scholar] [CrossRef]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Kern, P.A.; Di Gregorio, G.B.; Lu, T.; Rassouli, N.; Ranganathan, G. Adiponectin expression from human adipose tissue: Relation to obesity, insulin resistance, and tumor necrosis factor-alpha expression. Diabetes 2003, 52, 1779–1785. [Google Scholar] [CrossRef] [PubMed]
- Esser, N.; Legrand-Poels, S.; Piette, J.; Scheen, A.J.; Paquot, N. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Res. Clin. Pract. 2014, 105, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Feehley, T.J.; Nagler, C.R. The role of commensal bacteria in the regulation of sensitization to food allergens. FEBS Lett. 2014, 588, 4258–4266. [Google Scholar] [CrossRef]
- Camilleri, M. Leaky gut: Mechanisms, measurement and clinical implications in humans. Gut 2019, 68, 1516–1526. [Google Scholar] [CrossRef]
- Zhou, Q.; Zhang, B.; Verne, G.N. Intestinal membrane permeability and hypersensitivity in the irritable bowel syndrome. Pain 2009, 146, 41–46. [Google Scholar] [CrossRef]
- Michielan, A.; D’Inca, R. Intestinal Permeability in Inflammatory Bowel Disease: Pathogenesis, Clinical Evaluation, and Therapy of Leaky Gut. Mediat. Inflamm. 2015, 2015, 628157. [Google Scholar] [CrossRef]
- Visser, J.; Rozing, J.; Sapone, A.; Lammers, K.; Fasano, A. Tight junctions, intestinal permeability, and autoimmunity: Celiac disease and type 1 diabetes paradigms. Ann. N. Y. Acad. Sci. 2009, 1165, 195–205. [Google Scholar] [CrossRef]
- Gummesson, A.; Carlsson, L.M.; Storlien, L.H.; Backhed, F.; Lundin, P.; Lofgren, L.; Stenlof, K.; Lam, Y.Y.; Fagerberg, B.; Carlsson, B. Intestinal permeability is associated with visceral adiposity in healthy women. Obesity 2011, 19, 2280–2282. [Google Scholar] [CrossRef]
- Hoshiko, H.; Feskens, E.J.M.; Oosterink, E.; Ariens, R.M.C.; Mes, J.J.; de Wit, N.J.W. Identification of leaky gut-related markers as indicators of metabolic health in Dutch adults: The Nutrition Questionnaires plus (NQplus) study. PLoS ONE 2021, 16, e0252936. [Google Scholar] [CrossRef] [PubMed]
- Jayashree, B.; Bibin, Y.S.; Prabhu, D.; Shanthirani, C.S.; Gokulakrishnan, K.; Lakshmi, B.S.; Mohan, V.; Balasubramanyam, M. Increased circulatory levels of lipopolysaccharide (LPS) and zonulin signify novel biomarkers of proinflammation in patients with type 2 diabetes. Mol. Cell. Biochem. 2014, 388, 203–210. [Google Scholar] [CrossRef]
- Awoyemi, A.; Trøseid, M.; Arnesen, H.; Solheim, S.; Seljeflot, I. Markers of metabolic endotoxemia as related to metabolic syndrome in an elderly male population at high cardiovascular risk: A cross-sectional study. Diabetol. Metab. Syndr. 2018, 10, 59. [Google Scholar] [CrossRef] [PubMed]
- Scarpellini, E.; Lupo, M.; Iegri, C.; Gasbarrini, A.; De Santis, A.; Tack, J. Intestinal permeability in non-alcoholic fatty liver disease: The gut-liver axis. Rev. Recent Clin. Trials 2014, 9, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, T.F.; Souza, N.C.; Chiarello, P.G.; Franceschini, S.C.; Bressan, J.; Ferreira, C.L.; Peluzio Mdo, C. Intestinal permeability parameters in obese patients are correlated with metabolic syndrome risk factors. Clin. Nutr. 2012, 31, 735–740. [Google Scholar] [CrossRef]
- Teixeira, T.F.; Collado, M.C.; Ferreira, C.L.; Bressan, J.; Peluzio Mdo, C. Potential mechanisms for the emerging link between obesity and increased intestinal permeability. Nutr. Res. 2012, 32, 637–647. [Google Scholar] [CrossRef]
- Farhadi, A.; Gundlapalli, S.; Shaikh, M.; Frantzides, C.; Harrell, L.; Kwasny, M.M.; Keshavarzian, A. Susceptibility to gut leakiness: A possible mechanism for endotoxaemia in non-alcoholic steatohepatitis. Liver Int. Off. J. Int. Assoc. Study Liver 2008, 28, 1026–1033. [Google Scholar] [CrossRef]
- Van Wijck, K.; Verlinden, T.J.; van Eijk, H.M.; Dekker, J.; Buurman, W.A.; Dejong, C.H.; Lenaerts, K. Novel multi-sugar assay for site-specific gastrointestinal permeability analysis: A randomized controlled crossover trial. Clin. Nutr. 2013, 32, 245–251. [Google Scholar] [CrossRef]
- Lambert, G.P.; Schmidt, A.; Schwarzkopf, K.; Lanspa, S. Effect of aspirin dose on gastrointestinal permeability. Int. J. Sports Med. 2012, 33, 421–425. [Google Scholar] [CrossRef]
- Oshima, T.; Miwa, H.; Joh, T. Aspirin induces gastric epithelial barrier dysfunction by activating p38 MAPK via claudin-7. Am. J. Physiol. Cell. Physiol. 2008, 295, C800–C806. [Google Scholar] [CrossRef]
- International Diabetes Federation: The IDF Consensus Worldwide Definition of the Metabolic Syndrome. 2006. Available online: https://www.idf.org/component/attachments/attachments.html?id=705&task=download (accessed on 18 November 2021).
- Cariello, R.; Federico, A.; Sapone, A.; Tuccillo, C.; Scialdone, V.R.; Tiso, A.; Miranda, A.; Portincasa, P.; Carbonara, V.; Palasciano, G.; et al. Intestinal permeability in patients with chronic liver diseases: Its relationship with the aetiology and the entity of liver damage. Dig. Liver Dis. 2010, 42, 200–204. [Google Scholar] [CrossRef] [PubMed]
- Secondulfo, M.; Iafusco, D.; Carratù, R.; deMagistris, L.; Sapone, A.; Generoso, M.; Mezzogiomo, A.; Sasso, F.C.; Cartenì, M.; De Rosa, R.; et al. Ultrastructural mucosal alterations and increased intestinal permeability in non-celiac, type I diabetic patients. Dig. Liver Dis. 2004, 36, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Di Palo, D.M.; Garruti, G.; Di Ciaula, A.; Molina-Molina, E.; Shanmugam, H.; De Angelis, M.; Portincasa, P. Increased Colonic Permeability and Lifestyles as Contributing Factors to Obesity and Liver Steatosis. Nutrients 2020, 12, 564. [Google Scholar] [CrossRef] [PubMed]
- De Courten, B.; Moreno-Navarrete, J.M.; Lyons, J.; Soldatos, G.; de Courten, M.; Dougherty, S.; Forbes, J.; Fernández-Real, J.M. Contrasting association of circulating sCD14 with insulin sensitivity in non-obese and morbidly obese subjects. Mol. Nutr Food Res. 2016, 60, 103–109. [Google Scholar] [CrossRef]
- Masuda, D.; Sakai, N.; Sugimoto, T.; Kitazume-Taneike, R.; Yamashita, T.; Kawase, R.; Nakaoka, H.; Inagaki, M.; Nakatani, K.; Yuasa-Kawase, M.; et al. Fasting serum apolipoprotein B-48 can be a marker of postprandial hyperlipidemia. J. Atheroscler. Thromb. 2011, 18, 1062–1070. [Google Scholar] [CrossRef]
- Michalski, M.-C.; Vors, C.; Lecomte, M.; Laugerette, F. Dietary lipid emulsions and endotoxemia. OCL 2016, 23, D306. [Google Scholar] [CrossRef][Green Version]
- Tan, Y.; Kagan, J.C. A cross-disciplinary perspective on the innate immune responses to bacterial lipopolysaccharide. Mol. Cell 2014, 54, 212–223. [Google Scholar] [CrossRef]
- Salguero, M.V.; Al-Obaide, M.A.I.; Singh, R.; Siepmann, T.; Vasylyeva, T.L. Dysbiosis of Gram-negative gut microbiota and the associated serum lipopolysaccharide exacerbates inflammation in type 2 diabetic patients with chronic kidney disease. Exp. Ther. Med. 2019, 18, 3461–3469. [Google Scholar] [CrossRef]
- Giloteaux, L.; Goodrich, J.K.; Walters, W.A.; Levine, S.M.; Ley, R.E.; Hanson, M.R. Reduced diversity and altered composition of the gut microbiome in individuals with myalgic encephalomyelitis/chronic fatigue syndrome. Microbiome 2016, 4, 30. [Google Scholar] [CrossRef]
- Kasai, C.; Sugimoto, K.; Moritani, I.; Tanaka, J.; Oya, Y.; Inoue, H.; Tameda, M.; Shiraki, K.; Ito, M.; Takei, Y.; et al. Comparison of the gut microbiota composition between obese and non-obese individuals in a Japanese population, as analyzed by terminal restriction fragment length polymorphism and next-generation sequencing. BMC Gastroenterol. 2015, 15, 100. [Google Scholar] [CrossRef]
- Botelho, P.B.; Ferreira, M.V.R.; Araújo, A.M.; Mendes, M.M.; Nakano, E.Y. Effect of multispecies probiotic on gut microbiota composition in individuals with intestinal constipation: A double-blind, placebo-controlled randomized trial. Nutrition 2020, 78, 110890. [Google Scholar] [CrossRef] [PubMed]
- Szulińska, M.; Łoniewski, I.; van Hemert, S.; Sobieska, M.; Bogdański, P. Dose-Dependent Effects of Multispecies Probiotic Supplementation on the Lipopolysaccharide (LPS) Level and Cardiometabolic Profile in Obese Postmenopausal Women: A 12-Week Randomized Clinical Trial. Nutrients 2018, 10, 773. [Google Scholar] [CrossRef] [PubMed]
- Ghoshal, S.; Witta, J.; Zhong, J.; de Villiers, W.; Eckhardt, E. Chylomicrons promote intestinal absorption of lipopolysaccharides. J. Lipid Res. 2009, 50, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Stehle, J.R., Jr.; Leng, X.; Kitzman, D.W.; Nicklas, B.J.; Kritchevsky, S.B.; High, K.P. Lipopolysaccharide-binding protein, a surrogate marker of microbial translocation, is associated with physical function in healthy older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 1212–1218. [Google Scholar] [CrossRef]
- Wilson, N.L.; Vance, D.E.; Moneyham, L.D.; Raper, J.L.; Mugavero, M.J.; Heath, S.L.; Kempf, M.C. Connecting the dots: Could microbial translocation explain commonly reported symptoms in HIV disease? J. Assoc. Nurses AIDS Care 2014, 25, 483–495. [Google Scholar] [CrossRef][Green Version]
- Canipe, A.; Chidumayo, T.; Blevins, M.; Bestawros, M.; Bala, J.; Kelly, P.; Filteau, S.; Shepherd, B.E.; Heimburger, D.C.; Koethe, J.R. A 12 week longitudinal study of microbial translocation and systemic inflammation in undernourished HIV-infected Zambians initiating antiretroviral therapy. BMC Infect. Dis. 2014, 14, 521. [Google Scholar] [CrossRef]
- Gonzalez-Quintela, A.; Alonso, M.; Campos, J.; Vizcaino, L.; Loidi, L.; Gude, F. Determinants of serum concentrations of lipopolysaccharide-binding protein (LBP) in the adult population: The role of obesity. PLoS ONE 2013, 8, e54600. [Google Scholar] [CrossRef]
- Laugerette, F.; Alligier, M.; Bastard, J.P.; Drai, J.; Chanséaume, E.; Lambert-Porcheron, S.; Laville, M.; Morio, B.; Vidal, H.; Michalski, M.C. Overfeeding increases postprandial endotoxemia in men: Inflammatory outcome may depend on LPS transporters LBP and sCD14. Mol. Nutr. Food Res. 2014, 58, 1513–1518. [Google Scholar] [CrossRef]
- Fasano, A. All disease begins in the (leaky) gut: Role of zonulin-mediated gut permeability in the pathogenesis of some chronic inflammatory diseases. F1000Research 2020, 9, 32051759. [Google Scholar] [CrossRef]


| Metabolic Parameter | Metabolically Healthy | Metabolically Unhealthy | p-Value * |
|---|---|---|---|
| Weight (kg) | 65.7 (3.9) | 88.7 (6.3) | <0.01 |
| BMI (kg/m2) | 21.7 (0.8) | 27.9 (1.7) | <0.01 |
| Waist circumference (cm) | 81.2 (2.5) | 101.1 (3.8) | <0.01 |
| Glucose (mmol/L) | 4.9 (0.1) | 5.3 (0.2) | <0.01 |
| Total cholesterol (mmol/L) | 5.8 (0.4) | 5.7 (0.6) | 0.86 |
| HDL (mmol/L) | 1.8 (0.2) | 1.3 (0.1) | <0.01 |
| LDL (mmol/L) | 3.4 (0.3) | 3.6 (0.5) | 0.44 |
| Triglycerides (mmol/L) | 0.8 (0.2) | 1.5 (0.5) | 0.02 |
| ALT (U/L) | 20.7 (3.1) | 22.3 (3.0) | 0.49 |
| GGT (U/L) | 13.7 (1.8) | 24.7 (8.0) | 0.02 |
| CRP (mg/mL) | 4.5 (1.9) | 8.1 (3.3) | 0.07 |
| Location Permeability | Sugar Markers | Metabolically Healthy | Metabolically Unhealthy | p-Value * |
|---|---|---|---|---|
| Gastroduodenum | Sucrose/rhamnose (×103) (F1) | 80.3 (49.8) | 55.7 (12.9) | 0.36 |
| Proximal small intestine | Lactulose/rhamnose (× 103) (F1) | 25.0 (12.0) | 18.7 (2.5) | 0.33 |
| Distal small intestine | Lactulose/rhamnose (×103) (F2) | 46.4 (19.8) | 45.7 (7.6) | 0.95 |
| Colon | Sucralose/erythritol (×103) (F3) | 17.8 (3.9) | 13.8 (3.8) | 0.16 |
| Whole intestine | Sucralose/erythritol (×103) (F1–3) | 15.4 (3.2) | 13.4 (2.2) | 0.32 |
| Leaky Gut Markers (LGM) | Metabolically Healthy | Metabolically Unhealthy | p-Value * |
|---|---|---|---|
| Zonulin (ng/mL) | 33.5 (2.9) | 37.3 (4.1) | 0.15 |
| LBP (µg/mL) | 12.5 (1.3) | 14.2 (2.3) | 0.22 |
| sCD14 (µg/mL) | 1.4 (0.1) | 1.5 (0.1) | 0.11 |
| Location Permeability | Sugar Markers | Metabolically Healthy | Metabolically Unhealthy | p-Value * |
|---|---|---|---|---|
| Gastroduodenum | Sucrose/rhamnose (×103) (F1) | 52.3 (17.3) | 65.7 (14.5) | 0.26 |
| Proximal small intestine | Lactulose/rhamnose (×103) (F1) | 64.6 (14.3) | 75.0 (13.0) | 0.30 |
| Distal small intestine | Lactulose/rhamnose (×103) (F2) | 84.8 (24.0) | 81.0 (9.5) | 0.77 |
| Colon | Sucralose/erythritol (×103) (F3) | 15.9 (2.6) | 17.2 (3.0) | 0.52 |
| Whole intestine | Sucralose/erythritol (×103) (F1–3) | 13.4 (1.4) | 16.3 (3.0) | 0.09 |
| Metabolic Parameter | Metabolically Healthy | Metabolically Unhealthy | p-Value * |
|---|---|---|---|
| Glucose (mmol/L) | 4.8 (0.1) | 5.2 (0.2) | <0.01 |
| Total cholesterol (mmol/L) | 5.7 (0.5) | 5.3 (0.6) | 0.86 |
| HDL (mmol/L) | 1.8 (0.2) | 1.2 (0.1) | <0.01 |
| LDL (mmol/L) | 3.5 (0.4) | 3.5 (0.5) | 0.84 |
| Triglycerides (mmol/L) | 0.8 (0.2) | 1.5 (0.4) | 0.01 |
| ALT (U/L) | 21.3 (2.3) | 22.6 (4.0) | 0.58 |
| GGT (U/L) | 13.1 (1.5) | 22.4 (7.0) | 0.02 |
| CRP (mg/mL) | 3.7 (1.9) | 7.6 (3.3) | 0.06 |
| Leaky Gut Markers (LGM) | Metabolically Healthy | Metabolically Unhealthy | p-Value * |
|---|---|---|---|
| Zonulin (ng/mL) | 31.2 (4.0) | 36.3 (4.0) | 0.09 |
| LBP (µg/mL) | 12.7 (1.8) | 14.5 (2.2) | 0.24 |
| sCD14 (µg/mL) | 1.5 (0.1) | 1.6 (0.1) | 0.17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoshiko, H.; Zeinstra, G.G.; Lenaerts, K.; Oosterink, E.; Ariens, R.M.C.; Mes, J.J.; de Wit, N.J.W. An Observational Study to Evaluate the Association between Intestinal Permeability, Leaky Gut Related Markers, and Metabolic Health in Healthy Adults. Healthcare 2021, 9, 1583. https://doi.org/10.3390/healthcare9111583
Hoshiko H, Zeinstra GG, Lenaerts K, Oosterink E, Ariens RMC, Mes JJ, de Wit NJW. An Observational Study to Evaluate the Association between Intestinal Permeability, Leaky Gut Related Markers, and Metabolic Health in Healthy Adults. Healthcare. 2021; 9(11):1583. https://doi.org/10.3390/healthcare9111583
Chicago/Turabian StyleHoshiko, Hiroyuki, Gertrude G. Zeinstra, Kaatje Lenaerts, Els Oosterink, Renata M. C. Ariens, Jurriaan J. Mes, and Nicole J. W. de Wit. 2021. "An Observational Study to Evaluate the Association between Intestinal Permeability, Leaky Gut Related Markers, and Metabolic Health in Healthy Adults" Healthcare 9, no. 11: 1583. https://doi.org/10.3390/healthcare9111583
APA StyleHoshiko, H., Zeinstra, G. G., Lenaerts, K., Oosterink, E., Ariens, R. M. C., Mes, J. J., & de Wit, N. J. W. (2021). An Observational Study to Evaluate the Association between Intestinal Permeability, Leaky Gut Related Markers, and Metabolic Health in Healthy Adults. Healthcare, 9(11), 1583. https://doi.org/10.3390/healthcare9111583






