The Influence of Nordic Walking on Spinal Posture, Physical Function, and Back Pain in Community-Dwelling Older Adults: A Pilot Study
Abstract
:1. Introduction
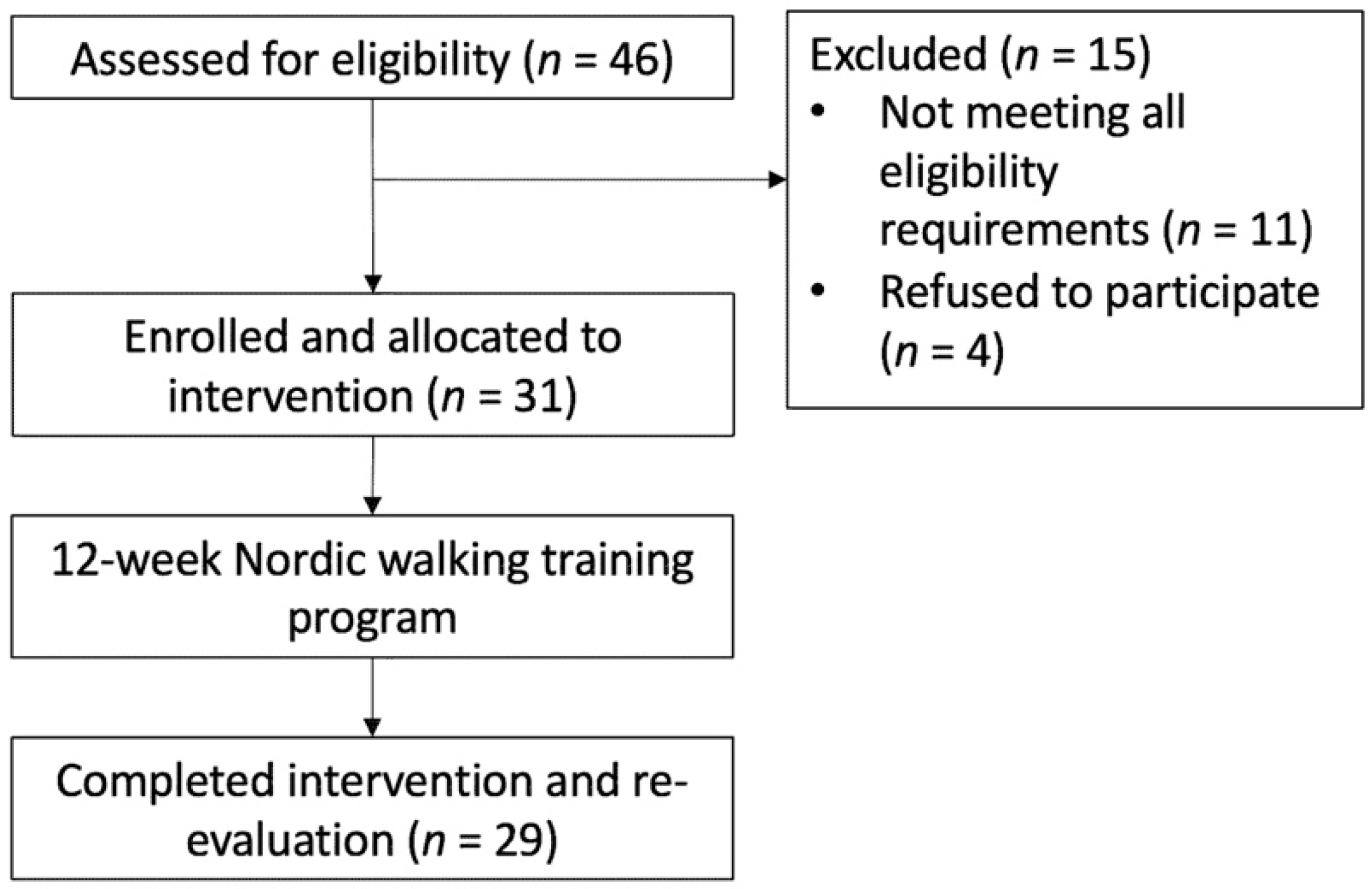
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Intervention
2.4. Outcome Measures
2.4.1. Thoracic Kyphosis Angle
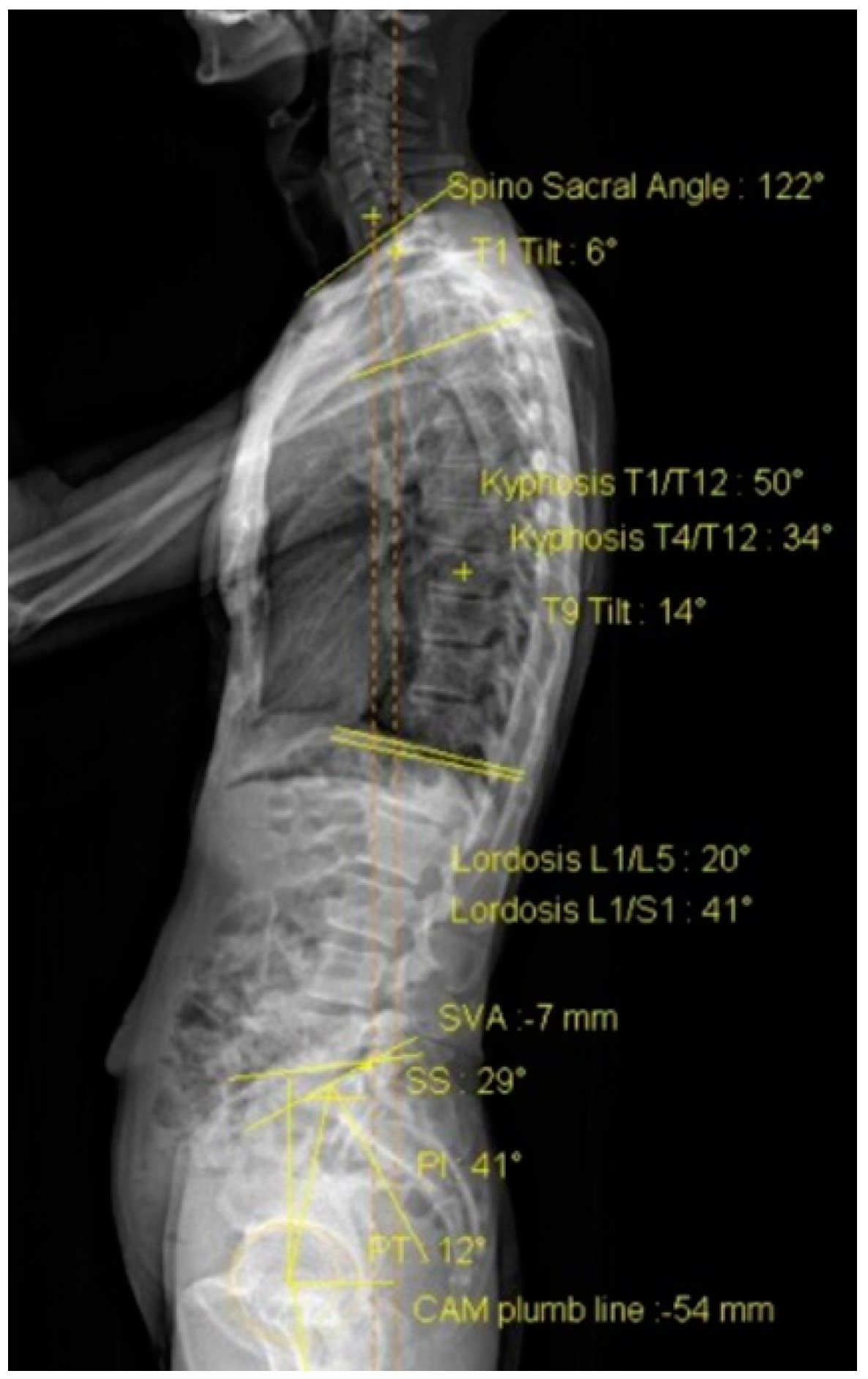
2.4.2. Sagittal Alignment
2.4.3. Physical Function
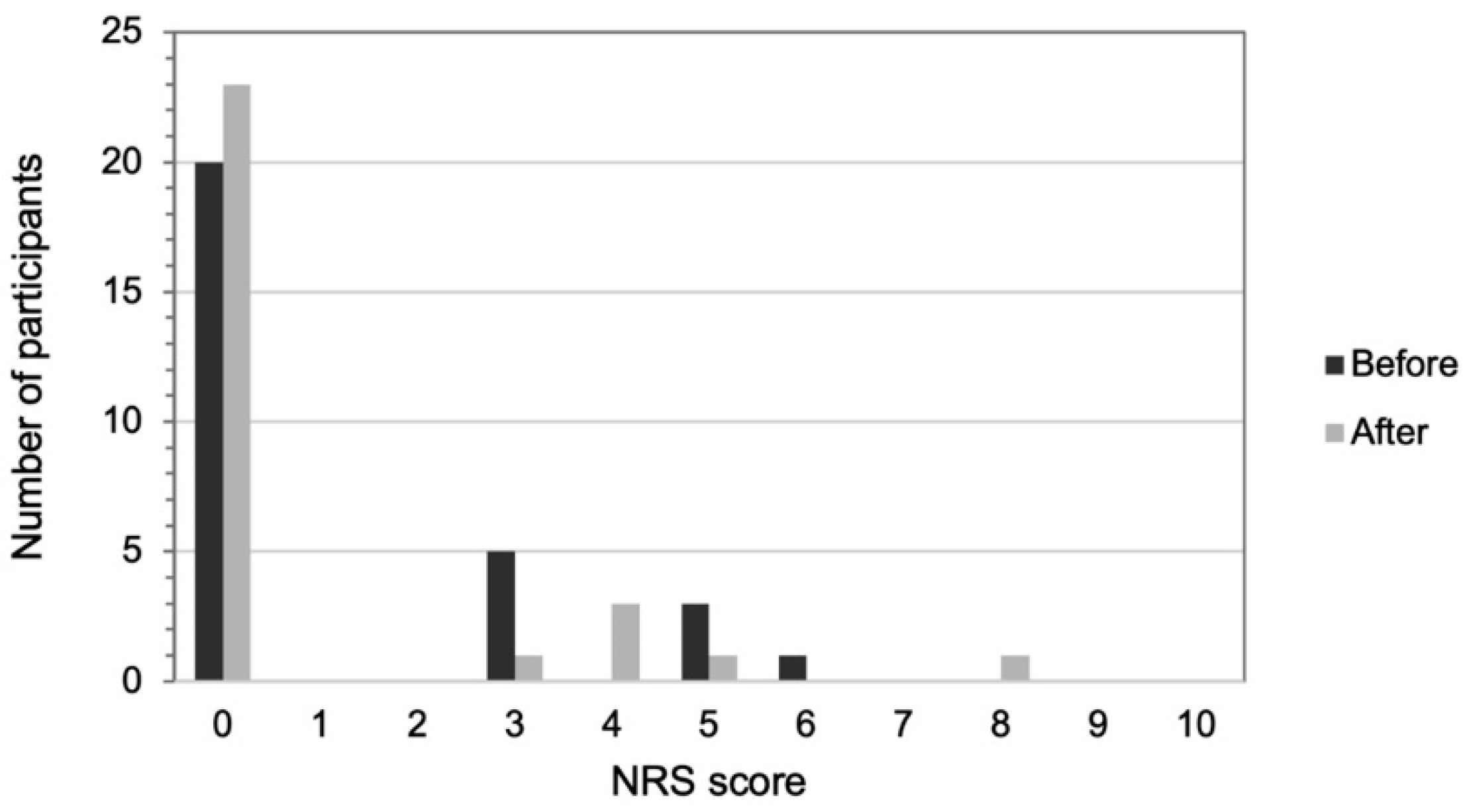
2.4.4. Back Pain
2.4.5. Strength and Endurance of Back Extensor Muscles
2.5. Statistical Analysis
3. Results
4. Discussion
4.1. Spinal Posture
4.2. Physical Function
4.3. Back Pain, Back Extensor Strength, and Back Extensor Endurance
4.4. Limitation and Strength
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roghani, T.; Zavieh, M.K.; Manshadi, F.D.; King, N.; Katzman, W. Age-related hyperkyphosis: Update of its potential causes and clinical impacts-narrative review. Aging Clin. Exp. Res. 2017, 29, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Katzman, W.B.; Wanek, L.; Shepherd, J.A.; Sellmeyer, D.E. Age-related hyperkyphosis: Its causes, consequences, and management. J. Orthop. Sports Phys. Ther. 2010, 40, 352–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koelé, M.C.; Lems, W.F.; Willems, H.C. The clinical relevance of hyperkyphosis: A narrative review. Front. Endocrinol. 2020, 11, 5. [Google Scholar] [CrossRef] [Green Version]
- Yokoyama, K.; Kawanishi, M.; Yamada, M.; Tanaka, H.; Ito, Y.; Kawabata, S.; Kuroiwa, T. Age-related variations in global spinal alignment and sagittal balance in asymptomatic Japanese adults. Neurol. Res. 2017, 39, 414–418. [Google Scholar] [CrossRef]
- NK, O.G.; Koseoglu, F.; Albayrak, N. Reduced aerobic capacity in patients with severe osteoporosis: A cross sectional study. Eur. J. Phys. Rehabil. Med. 2008, 44, 141–147. [Google Scholar]
- Kado, D.M.; Huang, M.-H.; Nguyen, C.B.; Barrett-Connor, E.; Greendale, G.A. Hyperkyphotic posture and risk of injurious falls in older persons: The Rancho Bernardo Study. J. Gerontol. A Biol. Sci. Med. Sci. 2007, 62, 652–657. [Google Scholar] [CrossRef] [Green Version]
- Kado, D.M.; Huang, M.-H.; Karlamangla, A.S.; Barrett-Connor, E.; Greendale, G.A. Hyperkyphotic posture predicts mortality in older community-dwelling men and women: A prospective study. J. Am. Geriatr. Soc. 2004, 52, 1662–1667. [Google Scholar] [CrossRef]
- Milne, J.S.; Williamson, J. A longitudinal study of kyphosis in older people. Age Ageing 1983, 12, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Mika, A.; Unnithan, V.B.; Mika, P. Differences in thoracic kyphosis and in back muscle strength in women with bone loss due to osteoporosis. Spine 2005, 30, 241–246. [Google Scholar] [CrossRef] [Green Version]
- Granito, R.N.; Aveiro, M.C.; Renno, A.C.; Oishi, J.; Driusso, P. Comparison of thoracic kyphosis degree, trunk muscle strength and joint position sense among healthy and osteoporotic elderly women: A cross-sectional preliminary study. Arch. Gerontol. Geriatr. 2012, 54, e199–e202. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, M.G.; Berti, L.; Presti, C.; Frizziero, A.; Giannini, S. Effects of an adapted physical activity program in a group of elderly subjects with flexed posture: Clinical and instrumental assessment. J. Neuroeng. Rehabil. 2008, 5, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bennell, K.L.; Matthews, B.; Greig, A.; Briggs, A.; Kelly, A.; Sherburn, M.; Larsen, J.; Wark, J. Effects of an exercise and manual therapy program on physical impairments, function and quality-of-life in people with osteoporotic vertebral fracture: A randomised, single-blind controlled pilot trial. BMC Musculoskelet. Disord. 2010, 11, 36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Katzman, W.B.; Vittinghoff, E.; Lin, F.; Schafer, A.; Long, R.K.; Wong, S.; Gladin, A.; Fan, B.; Allaire, B.; Kado, D.M.; et al. Targeted spine strengthening exercise and posture training program to reduce hyperkyphosis in older adults: Results from the study of hyperkyphosis, exercise, and function (SHEAF) randomized controlled trial. Osteoporos. Int. 2017, 28, 2831–2841. [Google Scholar] [CrossRef] [Green Version]
- Senthil, P.; Sudhakar, S.; Radhakrishnan, R.; Jeyakumar, S. Efficacy of corrective exercise strategy in subjects with hyperkyphosis. J. Back Musculoskelet. Rehabil. 2017, 30, 1285–1289. [Google Scholar] [CrossRef] [PubMed]
- Bansal, S.; Katzman, W.B.; Giangregorio, L.M. Exercise for improving age-related hyperkyphotic posture: A systematic review. Arch. Phys. Med. Rehabil. 2014, 95, 129–140. [Google Scholar] [CrossRef] [Green Version]
- Dalton, C.; Nantel, J. Nordic walking improves postural alignment and leads to a more normal gait pattern following weeks of training: A pilot study. J. Aging Phys. Act. 2016, 24, 575–582. [Google Scholar] [CrossRef]
- Hanuszkiewicz, J.; Woźniewski, M.; Malicka, I. The influence of Nordic walking on isokinetic trunk muscle endurance and sagittal spinal curvatures in women after breast cancer treatment: Age-specific indicators. Int J. Environ. Res. Public Health 2021, 18, 2409. [Google Scholar] [CrossRef] [PubMed]
- Gougeon, M.A.; Zhou, L.; Nantel, J. Nordic Walking improves trunk stability and gait spatial-temporal characteristics in people with Parkinson disease. NeuroRehabilitation 2017, 41, 205–210. [Google Scholar] [CrossRef]
- Bullo, V.; Gobbo, S.; Vendramin, B.; Duregon, F.; Cugusi, L.; Di Blasio, A.; Bocalini, D.S.; Zaccaria, M.; Bergamin, M.; Ermolao, A. Nordic walking can be incorporated in the exercise prescription to increase aerobic capacity, strength, and quality of life for elderly: A systematic review and meta-analysis. Rejuvenation Res. 2018, 21, 141–161. [Google Scholar] [CrossRef]
- Tschentscher, M.; Niederseer, D.; Niebauer, J. Health benefits of Nordic walking: A systematic review. Am. J. Prev. Med. 2013, 44, 76–84. [Google Scholar] [CrossRef]
- Li, H.; Jia, J.; Yang, Z. Mini-mental state examination in elderly Chinese: A population-based normative study. J. Alzheimer’s Dis. 2016, 53, 487–496. [Google Scholar] [CrossRef]
- Katzman, W.B.; Vittinghoff, E.; Kado, D.M.; Schafer, A.L.; Wong, S.S.; Gladin, A.; Lane, N.E. Study of hyperkyphosis, exercise and function (SHEAF) protocol of a randomized controlled trial of multimodal spine-strengthening exercise in older adults with hyperkyphosis. Phys. Ther. 2016, 96, 371–381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wiyanad, A.; Chokphukiao, P.; Suwannarat, P.; Thaweewannakij, T.; Wattanapan, P.; Gaogasigam, C.; Amatachaya, P.; Amatachaya, S. Is the occiput-wall distance valid and reliable to determine the presence of thoracic hyperkyphosis? Musculoskelet. Sci. Pract. 2018, 38, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Rehm, J.; Germann, T.; Akbar, M.; Pepke, W.; Kauczor, H.-U.; Weber, M.-A.; Spira, D. 3D-modeling of the spine using EOS imaging system: Inter-reader reproducibility and reliability. PLoS ONE 2017, 12, e0171258. [Google Scholar] [CrossRef]
- Kim, S.B.; Heo, Y.M.; Hwang, C.M.; Kim, T.G.; Hong, J.Y.; Won, Y.G.; Ham, C.U.; Min, Y.K.; Yi, J.W. Reliability of the eos imaging system for assessment of the spinal and pelvic alignment in the sagittal plane. Clin. Orthop. Surg. 2018, 10, 500–507. [Google Scholar] [CrossRef]
- Rikli, R.E.; Jones, C.J. Development and validation of criterion-referenced clinically relevant fitness standards for maintaining physical independence in later years. Gerontologist 2013, 53, 255–267. [Google Scholar] [CrossRef]
- Childs, J.D.; Piva, S.R.; Fritz, J.M. Responsiveness of the numeric pain rating scale in patients with low back pain. Spine 2005, 30, 1331–1334. [Google Scholar] [CrossRef]
- Shipp, K.; Purser, J.; Gold, D.; Pieper, C.; Sloane, R.; Schenkman, M.; Lyles, K. Timed loaded standing: A measure of combined trunk and arm endurance suitable for people with vertebral osteoporosis. Osteoporos. Int. 2000, 11, 914–922. [Google Scholar] [CrossRef]
- Lakens, D. Calculating and reporting effect sizes to facilitate cumulative science: A practical primer for t-tests and ANOVAs. Front. Psychol. 2013, 4, 863. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zoffoli, L.; Lucertini, F.; Federici, A.; Ditroilo, M. Trunk muscles activation during pole walking vs. walking performed at different speeds and grades. Gait Posture 2016, 46, 57–62. [Google Scholar] [CrossRef]
- Uehara, M.; Takahashi, J.; Ikegami, S.; Tokida, R.; Nishimura, H.; Sakai, N.; Kato, H. Sagittal spinal alignment deviation in the general elderly population: A Japanese cohort survey randomly sampled from a basic resident registry. Spine J. 2019, 19, 349–356. [Google Scholar] [CrossRef]
- Oe, S.; Yamato, Y.; Hasegawa, T.; Yoshida, G.; Kobayashi, S.; Yasuda, T.; Banno, T.; Arima, H.; Mihara, Y.; Ushirozako, H.; et al. Deterioration of sagittal spinal alignment with age originates from the pelvis not the lumbar spine: A 4-year longitudinal cohort study. Eur. Spine J. 2020, 29, 2329–2339. [Google Scholar] [CrossRef]
- Nemoto, Y.; Sakurai, R.; Ogawa, S.; Maruo, K.; Fujiwara, Y. Effects of an unsupervised Nordic walking intervention on cognitive and physical function among older women engaging in volunteer activity. J. Exerc. Sci. Fit. J. 2021, 19, 209–215. [Google Scholar] [CrossRef]
- Lee, H.S.; Park, J.H. Effects of Nordic walking on physical functions and depression in frail people aged 70 years and above. J. Phys. Ther. Sci. 2015, 27, 2453–2456. [Google Scholar] [CrossRef] [Green Version]
- Song, M.S.; Yoo, Y.K.; Choi, C.H.; Kim, N.C. Effects of nordic walking on body composition, muscle strength, and lipid profile in elderly women. Asian Nurs. Res. 2013, 7, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Takeshima, N.; Islam, M.M.; Rogers, M.E.; Rogers, N.L.; Sengoku, N.; Koizumi, D.; Kitabayashi, Y.; Imai, A.; Naruse, A. Effects of nordic walking compared to conventional walking and band-based resistance exercise on fitness in older adults. J. Sports Sci. Med. 2013, 12, 422–430. [Google Scholar] [PubMed]
- Kraemer, W.J.; Ratamess, N.A.; French, D.N. Resistance training for health and performance. Curr. Sports Med. Rep. 2002, 1, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Shahtahmassebi, B.; Hebert, J.J.; Hecimovich, M.; Fairchild, T.J. Trunk exercise training improves muscle size, strength, and function in older adults: A randomized controlled trial. Scand. J. Med. Sci. Sports 2019, 29, 980–991. [Google Scholar] [CrossRef] [PubMed]

| Variable | All Participants |
|---|---|
| Gender | 8 men, 23 women |
| Age (year) | 70 (8) |
| Height (cm) | 157.2 (8.0) |
| Weight (kg) | 57.2 ± 12.0 |
| Body mass index (kg/m2) | 22.6 (4.6) |
| Mini-Mental State Examination (score) | 29 (3) |
| High blood pressure | 8/34.5% |
| Diabetes | 5/17.2% |
| Osteoporosis | 7/24.1% |
| Knee arthritis | 5 (17.2%) |
| Heart diseases | 5 (17.2%) |
| Parkinson disease | 1 (3.4%) |
| Stroke | 1 (3.4%) |
| Education | |
| Primary school and illiterate | 4/13.7% |
| High school | 10/34.5% |
| College and above | 15/51.7% |
| Marital Status | |
| Married | 23/79.3% |
| Single, widowed, and divorced | 6/20.7% |
| Physical Activity Level 1 | |
| High | 11/37.9% |
| Moderate | 17/58.6% |
| Low | 1/3.4% |
| Outcome Measures | Pre-Test | Post-Test | Δ | p | d |
|---|---|---|---|---|---|
| Thoracic kyphosis angle | |||||
| OWD (usual, cm) | 6.79 ± 3.64 | 6.44 ± 2.53 | −0.35 | 0.419 | 0.15 |
| OWD (best, cm) | 4.86 ± 4.81 | 4.81 ± 2.15 | −0.04 | 0.917 | 0.02 |
| Sagittal alignment | |||||
| T1/T12 kyphosis (°) | 44.31 ± 9.80 | 44.42 ± 10.03 | −0.12 | 0.891 | 0.03 |
| L1/L5 lordosis (°) | 26.04 ± 14.99 | 25.54 ± 14.79 | 0.50 | 0.383 | 0.17 |
| Pelvic incidence (°) | 48.50 ± 12.05 | 48.04 ± 12.07 | 0.46 | 0.474 | 0.14 |
| Sacral slope (°) | 31.27 ± 9.41 | 31.31 ± 9.37 | −0.04 | 0.952 | 0.12 |
| Pelvic tilt (°) | 17.12 ± 8.09 | 16.81 ± 8.34 | 0.31 | 0.415 | 0.16 |
| Sagittal vertical axis (mm) | 36.04 ± 27.06 | 40.38 ± 26.10 | −4.35 | 0.204 | 0.26 |
| Physical function | |||||
| 30-s biceps curl (time) | 17.07 ± 3.67 | 18.28 ± 3.74 | 1.21 | 0.034 * | 0.41 |
| 30-s chair stand (time) | 17.07 ± 4.54 | 18.00 ± 4.33 | 0.93 | 0.026 * | 0.44 |
| Back scratch (cm) | 1.37 ± 9.72 | 1.97 ± 8.23 | 0.60 | 0.400 | 0.16 |
| Chair sit and reach (cm) | 9.84 ± 17.34 | 7.98 ± 17.28 | −1.86 | 0.051 | 0.38 |
| 2-min step (time) | 95.62 ± 21.27 | 98.93 ± 19.82 | 3.31 | 0.308 | 0.19 |
| 2.44-m up-and-go (s) | 5.83 ± 1.38 | 5.86 ± 1.16 | 0.03 | 0.798 | 0.05 |
| Single leg stance (s) | 19.74 ± 11.48 | 25.85 ± 7.76 | 6.10 | 0.001 * | 0.70 |
| Back extensor strength (N) | 39.27 ± 7.83 | 39.52 ± 7.44 | 0.25 | 0.855 | 0.03 |
| Back extensor endurance (s) | 148.55 ± 91.37 | 170.33 ± 115.10 | 21.79 | 0.053 | 0.38 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.-H.; Fang, I.-Y.; Kuo, Y.-L. The Influence of Nordic Walking on Spinal Posture, Physical Function, and Back Pain in Community-Dwelling Older Adults: A Pilot Study. Healthcare 2021, 9, 1303. https://doi.org/10.3390/healthcare9101303
Huang Y-H, Fang I-Y, Kuo Y-L. The Influence of Nordic Walking on Spinal Posture, Physical Function, and Back Pain in Community-Dwelling Older Adults: A Pilot Study. Healthcare. 2021; 9(10):1303. https://doi.org/10.3390/healthcare9101303
Chicago/Turabian StyleHuang, Yi-Hung, I-Yao Fang, and Yi-Liang Kuo. 2021. "The Influence of Nordic Walking on Spinal Posture, Physical Function, and Back Pain in Community-Dwelling Older Adults: A Pilot Study" Healthcare 9, no. 10: 1303. https://doi.org/10.3390/healthcare9101303
APA StyleHuang, Y.-H., Fang, I.-Y., & Kuo, Y.-L. (2021). The Influence of Nordic Walking on Spinal Posture, Physical Function, and Back Pain in Community-Dwelling Older Adults: A Pilot Study. Healthcare, 9(10), 1303. https://doi.org/10.3390/healthcare9101303






