Development and Themes of Diagnostic and Treatment Procedures for Secondary Leg Lymphedema in Patients with Gynecologic Cancers
Abstract
1. Introduction
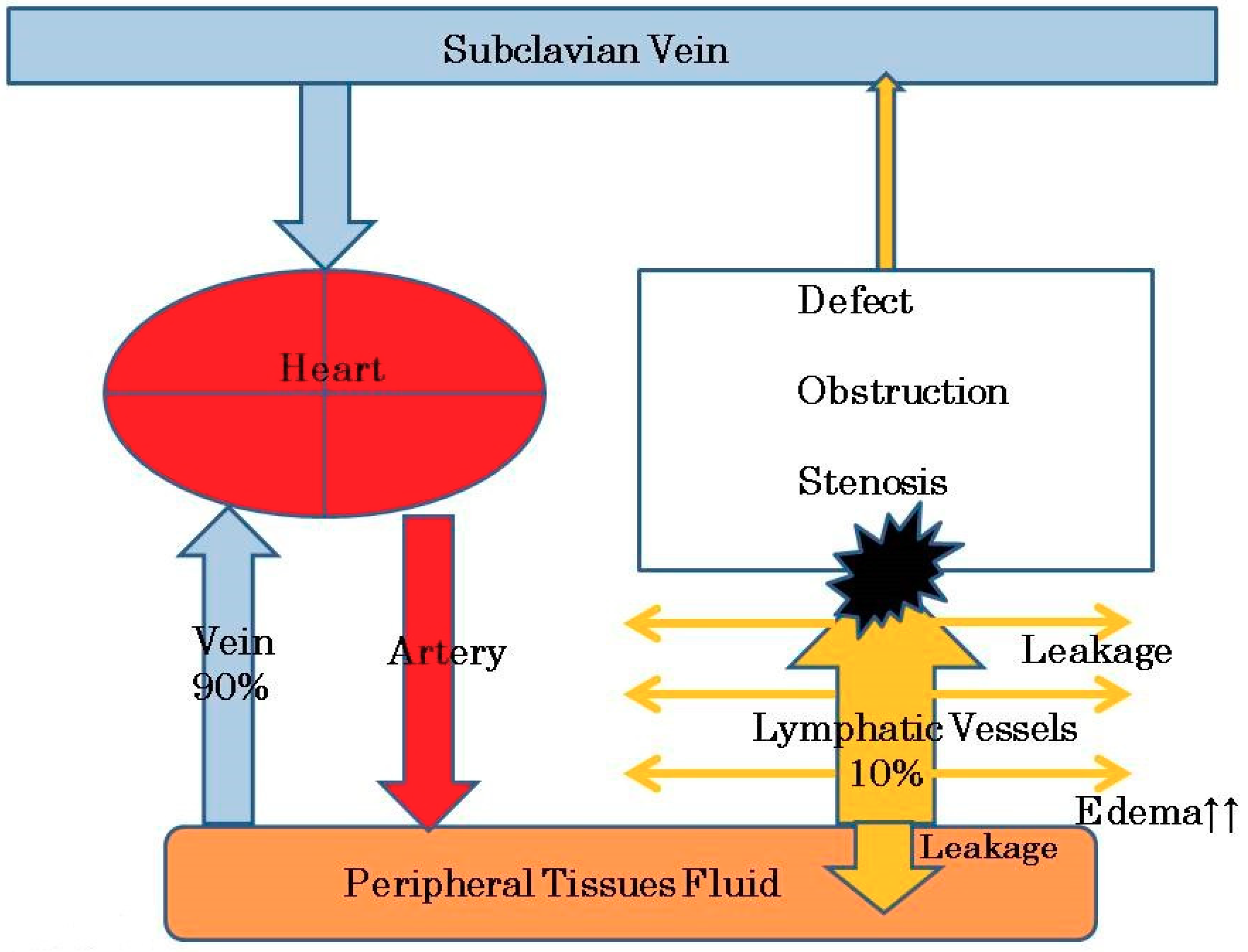
2. Etiology and Pathophysiology of Leg Lymphedema after Pelvic Lymphadenectomy
3. Symptoms
4. The Diagnosis
4.1. Physical Examination Findings
4.2. Ultrasonography
4.3. Magnetic Resonance Imaging (MRI)
4.4. MRI-Lymphography/Computed Tomography-Lymphography (MRI-LG/CT-LG)
4.5. Lymphosintiography
4.6. Indocyanine Green Lymphography (ICG-LG)
5. Treatments
5.1. MLD
5.2. Bandaging
5.3. PCT
5.4. Lymphaticovenous Anastomosis (LVA) and Vascularized Lymph Node Transfer (VLNT)
5.5. Liposuction and Surgical Excision
5.6. Prevention of Secondary Lymphedema
6. Discussion
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Achouri, A.; Huchon, C.; Bats, A.S.; Bensaid, C.; Nos, C.; Lecuru, F. Complications of lymphadenectomy for gynecologic cancer. Eur. J. Surg. Oncol. 2013, 39, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Biglia, N.; Zanfagnin, V.; Daniele, A.; Robba, E.; Bounous, V.E. Lower body lymphedema in patients with gynecologic cancer. Anticancer Res. 2017, 37, 4005–4015. [Google Scholar] [PubMed]
- Ki, E.T.; Park, J.S.; Lee, K.H.; Hur, S.Y. Incidence and risk factors of lower extremity lymphedema after gynecologic surgery in ovarian cancer. Int. J. Gynecol. Cancer 2016, 26, 1327–1332. [Google Scholar] [CrossRef] [PubMed]
- Togami, D.; Kawamura, T.; Fukuda, M.; Yanazume, S.; Kamio, M.; Kobayashi, H. Risk factors for lymphatic complications following lymphadenectomy in patients with cervical cancer. Jpn. J. Clin. 2018, 48, 1036–1040. [Google Scholar] [CrossRef] [PubMed]
- Hayes, S.C.; Janda, M.; Ward, L.C.; Reul-Hirche, H.; Steele, M.L.; Carter, J.; Quinn, M.; Cornish, B.; Obermair, A. Lymphedema following gynecological cancer: Resuts from a prospective, longitudinal cohort study on prevalence, incidence and risk factors. Gynecol. Oncol. 2017, 146, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Bae, D.S.; Nam, J.H.; Park, C.T.; Cho, C.H.; Lee, J.M.; Lee, M.K.; Kim, S.H.; Park, S.M.; Yun, Y.H. Quality of life and sexual problems in disease-free survivors of cervical cancer compared with the general population. Cancer 2007, 110, 2716–2725. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.F.; Li, S.H.; Huang, H.Y. The efficacy of complex decongestive physiotherapy (CDP) and predictive factors of response to CDP in lower limb lymphedema (LLL) after pelvic cancer treatment. Gynecol. Oncol. 2012, 125, 712–715. [Google Scholar] [CrossRef] [PubMed]
- Szuba, A.; Shin, W.S.; Strauss, H.W.; Rockson, S. The third circulation: Radionuclide lymphoscintigraphy in the evaluation of lymphedema. J. Nucl. Med. 2003, 44, 43–57. [Google Scholar] [PubMed]
- Keo, H.H.; Gretener, S.B.; Staub, D. Clinical and diagnostic aspects of lymphedema. Vasa 2017, 46, 255–261. [Google Scholar] [CrossRef][Green Version]
- Rockson, S.G. Lymphedema. Am. J. Med. 2001, 110, 288–295. [Google Scholar] [CrossRef]
- Todo, Y.; Yamamoto, R.; Minobe, S.; Suzuki, Y.; Takeshi, U.; Nakatani, M.; Aoyagi, Y.; Ohba, Y.; Okamoto, K.; Kato, H. Risk factors for postoperative lower-extremity lymphedema in endometrial cancer survivors who had treatment including lymphadenectomy. Gynecol. Oncol. 2010, 119, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Hopp, E.E.; Osborne, J.L.; Schneider, D.K.; Bojar, C.J.; Uyar, D.S. A prospective pilot study on the incidence of post-operative lymphedema in women with endometrial cancer. Gynecol. Oncol. Rep. 2015, 15, 25–28. [Google Scholar] [CrossRef] [PubMed]
- Szuba, A.; Rockson, S.G. Lymphedema: Anatomy, physiology and pathogenesis. Vasc. Med. 1997, 2, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Hauck, G. capillary permeability and micro-lymph drainage. Vasa 1994, 23, 451–458. [Google Scholar]
- Ellis, S. Structure and function of the lymphatic system: An overview. Br. J. Community 2006, 11, S4–S6. [Google Scholar] [CrossRef]
- Warren, A.G.; Brorson, H.; Borud, L.J.; Slavin, S.A. Lymphedema: A comprehensive review. Ann. Plast. Surg. 2007, 59, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Comford, M.E.; Oldendorf, W.H. Terminal endothelial cells of lymph capillaries as active transport structures involved in the formation of lymph in rat skin. Lymphology 1993, 26, 67–78. [Google Scholar]
- Olszewski, W.L. Contractility patterns of human leg lymphatics in various stages of obstructive lymphedema. Ann. N. Y. Acad. Sci. 2008, 1131, 110–118. [Google Scholar] [CrossRef]
- Olszewski, W.L.; Engeset, A. Intrinsiccontractility of prenodal lymph vessels and lymph flow in human leg. Am. J. Physiol. 1980, 239, H775–H783. [Google Scholar]
- Dobbins, D.E.; Dabney, J.M. Endothelin-mediated constriction of prenodal lymphatic vessels in the canine forelimb. Regul. Pept. 1991, 35, 81–91. [Google Scholar] [CrossRef]
- Sjoberg, T.; Steen, S. Contractile properties from human lower leg. Lymphology 1991, 24, 16–21. [Google Scholar] [PubMed]
- Tiwari, P.; Coriddi, M.; Lamp, S. Lymphedema strategies for investigation and treatment: A review. Plast. Surg. Nurs. 2012, 32, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Mihara, M.; Hara, H.; Hayashi, Y.; Narushima, M.; Yamamoto, T.; Todokoro, T.; Iida, T.; Sawamoto, N.; Araki, J.; Kikuchi, K.; et al. Pathological steps of cancer-related lymphedema: Histological changes in the collecting lymphatic vessels after lymphadenectomy. PLoS ONE 2012, 7, e41126. [Google Scholar] [CrossRef] [PubMed]
- Franzeck, U.K.; Spiegel, I.; Fischer, M.; Bortzler, C.; Stahel, H.U.; Bollinger, A. Combined physical therapy for lymphedema evaluated by fluorescence microlymphography and lymph capillary pressure measurements. J. Vasc. Res. 1997, 34, 306–311. [Google Scholar] [CrossRef] [PubMed]
- Zaugg-Vesti, B.; Dorffler-Melly, J.; Spiegel, M.; Wen, S.; Franzeck, U.K.; Bollinger, A. Lymphatic capillary pressure in patients with primary lymphedema. Microvasc. Res. 1993, 46, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Olszewski, W.L. Lymphology and the lymphatic system. In Lymph STASIS: Pathophysiology, Diagnosis and Treatment; Olszewski, W.L., Ed.; CRC Press: Boca Raton, FL, USA, 1991; pp. 4–12. [Google Scholar]
- Witte, C.L.; Witte, M.H. Pathophysiology of lymphatic insufficiency and principles of treatment. In Lymph Stasis: Pathophysiology, Diagnosis and Treatment; Olszewski, W.L., Ed.; CRC Press: Boca Raton, FL, USA, 1991; pp. 327–344. [Google Scholar]
- Olszewski, W.L. Pathophysiological and clinical observations of obstructive lymphedema of the limbs. In Lymphedema Stuuugart; Clodius, L., Ed.; Georg Thieme Publishers: Stuttgart, Germany, 1977; pp. 79–102. [Google Scholar]
- Bates, D.O.; Levick, J.R.; Mortimer, P.S. Subcutaneous interstitial fluid pressure and arm volume in lymphedema. Int. J. Microcirc. Clin. Exp. 1992, 11, 359–373. [Google Scholar] [PubMed]
- Adair, T.H.; Montani, J.P. Dynamics of lymph formation and its modification. In Lymph stasis: Pathophysiology, Diagnosis and Treatment; Olszewski, W.L., Ed.; CRC Press: Boca Raton, FL, USA, 1991; pp. 43–53. [Google Scholar]
- Ostgaard, G.; Reed, K. Hyaluronan turnover in the rat small intestine. Acta Physiol. Scand. 1993, 149, 237–244. [Google Scholar] [CrossRef]
- Granzows, J.W.; Soderberg, J.M.; Kaji, A.H.; Dauphine, C. Review of current surgical treatments for lymphedema. Ann. Surg. Oncol. 2014, 21, 1195–1201. [Google Scholar] [CrossRef] [PubMed]
- Casley-Smith, J.R.; Gaffney, R.M. Excess plasma proteins as a cause of chronic inflammation and lymphedema: Quantitative electron microscopy. J. Pathol. 1981, 133, 243–272. [Google Scholar] [CrossRef]
- Gaffiney, R.M.; Casley-Smith, J.R. Excess plasma proteins as a cause of chronic inflammation and lymphedema: Biochemical estimations. J. pathol. 1981, 133, 229–242. [Google Scholar] [CrossRef]
- Yuan, Y.; Arcucci, V.; Levy, S.M.; Achen, M.G. Modulation of immunity by lymphatic dysfunction in lymphedema. Front. Immunol. 2019, 10, 76. [Google Scholar] [CrossRef] [PubMed]
- Garcia Nores, G.D.; Ly, C.L.; Cuzzone, D.A.; Kataru, R.P.; Hespe, G.E.; Torrisi, J.S.; Huang, J.J.; Gardenier, J.C.; Savetsky, I.L.; Nitti, M.D.; et al. CD4(+) T cells are activated in regional lymph nodes and migrate to skin to initiate lymphedema. Nat. Commun. 2018, 9, 1970. [Google Scholar] [CrossRef] [PubMed]
- International Society of Lymphology (ISL). The diagnosis and treatment of peripheral lymphedema: 2016 consensus document of the international society of lymphology. Lymphology 2016, 49, 170–184. [Google Scholar]
- Dixon, J.B.; Weiler, M.J. Bridging the divide between pathogenesis and detection in lymphedema. Semin. Cell Dev. Biol. 2015, 38, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Swartz, M.A.; Kaipainen, A.; Netti, P.A.; Brekken, C.; Boucher, Y.; Grodzinsky, A.J.; Jain, R.K. Mechanics of interstitial-lymphatic fluid transport: Theoretical foundation and experimental validation. J. Biomech. 1999, 32, 1297–1307. [Google Scholar] [CrossRef]
- Rutkowski, J.M.; Markhus, C.E.; Gyenge, C.C.; Alitalo, K.; Wiig, H.; Swartz, M.A. Dermal collagen and lipid deposition correlate with tissue swelling anf hydraulic conductivity in murine primary lymphedema. Am. J. Pathol. 2010, 176, 1122–1129. [Google Scholar] [CrossRef] [PubMed]
- Avraham, T.; Clavin, N.W.; Daluvoy, S.V.; Femandez, J.; Soares, M.A.; Cordeiro, A.P.; Mehrara, B.J. Fibrosis is a key inhibitor of lymphatic regeneration. Plast. Reconstr. Surg. 2009, 124, 438–450. [Google Scholar] [CrossRef] [PubMed]
- Zampell, J.C.; Yan, A.; Elhadad, S.; Avraham, T.; Weitman, E.; Mehrara, B.J. CD4(+) cells regulate fibrosis and lymphangiogenesis in response to lymphatic fluid stasis. PLoS ONE 2012, 7, e49940. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.; Cheng, G.; Conner, D.A.; Huang, Y.; Kucheriapati, R.S.; Munn, L.L.; Ruddle, N.H.; Jain, R.K.; Fukumura, D.; Padera, T.P. Impaired lymphatic contraction associated with immunosuppression. Proc. Natl. Acad. Sci. USA 2011, 108, 18784–18789. [Google Scholar] [CrossRef]
- Zawieja, S.D.; Wang, W.; Wu, X.; Nepiyushchikh, Z.V.; Zawieja, D.C.; Muthuchamy, M. Impairments in the intrinsic contractility of mesenteric collecting lymphatics in a rat model of metabolic syndrome. Am. J. Physiol Heart Circ. Physiol. 2012, 302, H643–H653. [Google Scholar] [CrossRef]
- Hotta, E.; Asai, J.; Okuzawa, Y.; Hanada, K.; Nomiyama, T.; Takenaka, H.; Katoh, N. Verrucous lesions arising in lymphedema and diabetic neuropathy: Elephantiasis nostras verrucosa or verrucous skin lesions on the feet of patients with diabetic neuropathy? J. Dermatol. 2016, 43, 329–331. [Google Scholar] [CrossRef] [PubMed]
- Park, S.I.; Yang, E.J.; Kim, D.K.; Jeong, H.J.; Kim, G.C.; Sim, Y.J. Prevalence and epidemiological factors involved in cellulitis in Korean patients with lymphedema. Ann. Rehabil. Med. 2016, 40, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Teerachaisakul, M.; Ekataksin, W.; Durongwatana, S.; Taneepanichskul, S. Diet, C-reactive protein levels and cellulitis in patients with lymphedema: A cross-sectional study. J. Med. Med. Sci. 2011, 2, 129–1301. [Google Scholar]
- Cox, N.H. Management of lower leg cellulitis. Clin. Med. (Lond.) 2002, 2, 23–27. [Google Scholar] [CrossRef]
- Gethin, G.; Byrne, D.; Tierney, S.; Strapp, H.; Cowman, S. Prevalence of lymphedema and quality of life among patients attending a hospital-based wound management and vascular clinic. Int. Wound J. 2012, 9, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Trayes, K.P.; Studdiford, J.S. Edema: Diagnosis and management. Am. Fam. Physician 2013, 88, 102–110. [Google Scholar]
- Yanagisawa, N.; Koshiyama, M.; Watanabe, Y.; Sato, S.; Sakamoto, S. A quantitative method to measure skin thickness in leg edema in pregnant women using B-scan portable ultrasonography: A comparison between obese and non-obese women. Med. Sci. Monit. 2019, 25, 1–9. [Google Scholar] [CrossRef]
- Suehiro, K.; Morikage, N.; Murakami, M.; Yamashita, O.; Samura, M.; Hamano, K. Significance of ultrasound examination of skin and subcutaneous tissue in secondary lower extremity lymphedema. Ann. Vasc. Dis. 2013, 6, 180–188. [Google Scholar] [CrossRef]
- Suehiro, K.; Morikage, N.; Murakami, M.; Yamashita, O.; Ueda, K.; Samura, M.; Nakamura, K.; Hamano, K. Subcutaneous tissue ultrasonography in legs with dependent edema and secondary lymphedema. Ann. Vasc. Dis. 2014, 7, 21–27. [Google Scholar] [CrossRef]
- Hoffner, M.; Peterson, P.; Månsson, S.; Brorson, H. Lymphedema leads to fat deposition in muscle and decreased muscle/water volume after liposuction: A magnetic resonance imaging study. Lymphat. Res. Biol. 2018, 16, 174–181. [Google Scholar] [CrossRef]
- Lohrmann, C.; Foeldi, E.; Langer, M. Indirect magnetic resonance lymphangiography in patients with lymphedema preliminary results in humans. Eur. J. Radiol. 2006, 59, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Shinaoka, A.; Kimata, Y. Three-dimensional imaging of lymphatic system in lymphedema legs using interstitial computed tomography-lymphography. Acta Med. Okayama 2017, 71, 171–177. [Google Scholar] [PubMed]
- Mazzei, F.G.; Gentili, F.; Guerrini, S.; Squitieri, N.C.; Guerrieri, D.; Gennaro, P.; Scialpi, M.; Volterrani, L.; Mazzei, M.A. MR lymphangiography: A practical guide to perform it and a brief review of the literature from a technical point of view. Biomed. Res. Int. 2017, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.F.; Lu, Q.; Jiang, Z.H.; Wang, C.G.; Zhou, J.G. Anatomic and functional evaluation of the lymphatics and lymph nodes in diagnosis of lymphatic circulation disorders with contrast magnetic resonance lymphangiography. J. Vasc. Surg. 2009, 49, 980–987. [Google Scholar] [CrossRef] [PubMed]
- White, R.D.; Weir-McCall, J.R.; Budak, M.J.; Waugh, S.A.; Munnoch, D.A.; Sudarshan, T.A. Contrast-enhanced magnetic resonance lymphography in the assessment of lower limb lymphedema. Clin. Radiol. 2014, 69, e435–e444. [Google Scholar] [CrossRef] [PubMed]
- Suga, K.; Ogasawara, N.; Okada, M.; Matsunaga, N. Interstitial CT lymphography-guided localization of breast sentinel lymph node: Preliminary results. Surgery 2003, 133, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Sherman, A.; Ter-Pogossian, M. Lymph node concentration of radioactive colloidal gold following interstistial injection. Cancer 1953, 6, 1238–1240. [Google Scholar] [CrossRef]
- Yuan, Z.; Chen, L.; Luo, Q.; Zhu, J.; Lu, H.; Zhu, R. The role of radionuclide lymphoscintigraphy in extremity lymphedema. Ann. Nucl. Med. 2006, 20, 341–344. [Google Scholar] [CrossRef]
- Ter, S.E.; Alavi, A.; Kim, C.K.; Merli, G. Lymphoscintigraphy: A reliable test for the diagnosis of lymphedema. Clin. Nucl. Med. 1993, 18, 646–654. [Google Scholar] [CrossRef]
- Weissleder, H.; Weissleder, R. Lymphedema: Evaluation of qualitative and quantitative lymphoscintigraphy in 238 patients. Radiology 1988, 167, 729–735. [Google Scholar] [CrossRef]
- Moshiri, M.; Katz, D.S.; Boris, M.; Yung, E. Using lymphoscintigraphy to evaluate suspected lymphedema of the extremities. Am. J. Roentgenol. 2002, 178, 405–412. [Google Scholar] [CrossRef]
- Williams, W.H.; Witte, C.L.; Witte, M.H.; McNeill, G.C. Radionuclide lymphangioscintigraphy in the evaluation of peripheral lymphedema. Clin. Nucl. Med. 2000, 25, 451–464. [Google Scholar] [CrossRef] [PubMed]
- Mihara, M.; Hara, H.; Araki, J.; Kikuchi, K.; Narushima, M.; Yamamoto, T.; Iida, T.; Yoshimatsu, H.; Murai, N.; Mitsui, K.; et al. Indocyanine green (ICG) lymphography is superior to lymphoscintigraphy for diagnostic imaging of early lymphedema of the upper limbs. PLoS ONE 2012, 7, e38182. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Matsuda, N.; Doi, K.; Oshima, A.; Yoshimatsu, H.; Todokoro, T.; Ogata, F.; Mihara, M.; Narushima, M.; Iida, T.; et al. The earliest finding of indocyanine green lymphography in asymptomatic limbs of lower extremity lymphedema patients secondary to cancer treatment: The modified dermal backflow stage and concept of subclinical lymphedema. Plast. Reconstr. Surg. 2011, 128, 314e–321e. [Google Scholar] [CrossRef] [PubMed]
- Unno, N.; Nishiyama, M.; Suzuki, M.; Yamamoto, N.; Inuzuka, K.; Sagara, D.; Tanaka, H.; Konno, H. Quantitative lymph imaging for assessment of lymph function using indocyanine green fluorescence lymphography. Eur. J. Endovasc. Surg. 2008, 36, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Yamamoto, N.; Yoshimatsu, H.; Narushima, M.; Koshima, I. Factors associated with lower extremity dysmorphia caused by lower extremity lymphedema. Eur. J. Vasc. Endovasc. Surg. 2017, 54, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Suami, H.; Chang, D.; Skoracki, R.; Yamada, K.; Kimata, Y. Using indocyanine green fluorescent lymphography to demonstrate lymphatic architecture. J. Lymphedema 2012, 7, 25–29. [Google Scholar]
- Best Practice for the Management of Lymphoedema International Consensus. Available online: https://www.lympho.org/wp-content/uploads/2016/03/Best_practice.pdf (accessed on 17 July 2019).
- Watanabe, Y.; Koshiyama, M.; Yanagisawa, N. Treatment of leg and foot edema in women. Women’s Health Open J. 2017, 68–73. [Google Scholar] [CrossRef]
- Casley-Amith Judith, R.; Casley-Smith, J.R. Modern Treatment for Lymphedema, 5th ed.; The Walkerville SA 5081 Australia Lymphedema Association of Australia. Inc.: Adelaide, Australia, 1997; 228p. [Google Scholar]
- Yamamoto, R.; Yamamoto, T. Effectiveness of the treatment-phase of two-phase complex decongestive physiotherapy for the treatment of extremity lymphedema. Int. J. Clin. Oncol. 2007, 12, 463–468. [Google Scholar] [CrossRef]
- Williams, A. Manual lymphatic drainage: Exploring the history and evidence base. Br. J. Community Nurs. 2010, 15, S18–S24. [Google Scholar] [CrossRef]
- Hara, H.; Hamanaka, N.; Yoshida, M.; Ikehara, N.; Tachibana, S.; Nakakawaji, K.; Mihara, M. Variability in compression pressure of multi-layer bandaging applies by lymphedema therapists. Support Care Cancer 2019, 27, 959–963. [Google Scholar] [CrossRef] [PubMed]
- Ergin, G.; Şahinoğlu, E.; Karadibak, D.; Yavuzşen, T. Effect of bandage compliance on upper extremity volume in patients with breast cancer-related lymphedema. Lymphat. Res. Biol. 2018, 16, 553–558. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, K.; Mawaki, A.; Oshima, C.; Takeno, Y.; Kurono, F.; Taniho, Y.; Murotani, K.; Kikumori, K.; Fujimoto, E. Nighttime bandaging to reduce lymphedema swelling: A clinical pre-post study. Sage Open 2017, 3, 1–8. [Google Scholar]
- Scheer, R. Compression garments for managing lymphoedema. J. Lymphoedema 2017, 12, 39–45. [Google Scholar]
- International Society of Lymphology. The diagnosis and treatment of peripheral lymphedema: 2013 consensus document of the international society of lymphology. Lymphology 2013, 46, 1–11. [Google Scholar]
- Lawenda, B.D.; Mondry, T.E.; Johnstone, P.A. Lymphedema: A primer on the identification and management of a chronic condition in oncologic treatment. CA Cancer J. Clin. 2009, 59, 8–24. [Google Scholar] [CrossRef] [PubMed]
- Muluk, S.C.; Hirsch, A.T.; Taffe, E.C. Pneumatic compression device treatment of lower extremity lymphedema elicits improved limb volume and patient-reported outcomes. Eur. J. Vasc. Endovasc. Surg. 2013, 46, 480–487. [Google Scholar] [CrossRef]
- Zaleska, M.; Olszewski, W.L.; Cakala, M.; Cwikla, J.; Budlewski, T. Intermittent pneumatic compression enhances formation of edema tissue fluid channels in lymphedema of lower limbs. Lymphat. Res. Biol. 2015, 13, 146–153. [Google Scholar] [CrossRef]
- Aldrich, M.B.; Gross, D.; Morrow, J.R.; Fife, C.E.; Rasmussen, J.C. Effect of pneumatic compression therapy on lymp movement in lymphedema-affected extremities, as assessed by near-infrared fluorescence lymphatic imaging. J. Innov. Opt. Health Sci. 2017, 10, 1–12. [Google Scholar] [CrossRef]
- Campisi, C. Use of autologous interposition vein graft in management of lymphedema: Preliminary experimental and clinical observation. Lymphology 1991, 24, 71–76. [Google Scholar]
- Koshima, I.; Inagawa, K.; Urushibara, K.; Moriguchi, T. Supermicrosurgical lymphaticovenular anastomosis for the treatment of lymphedema in the upper extremities. J. Reconstr. Microsurg. 2000, 16, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Campisi, C.; Boccardo, F.; Zilli, A.; Macciò, A.; Napoli, F. Long-term results after lymphatic venous anastomoses for the treatment of obstructive lymphedema. Microsurgery 2001, 21, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Koshima, I.; Nanba, Y.; Tsutsui, T.; Takahashi, Y.; Itoh, S. Long-term results follow-up after lymphaticovenular anastomosis for lymphedema in the leg. J. Reconstr. Microsurg. 2003, 19, 209–215. [Google Scholar] [PubMed]
- Nielubowicz, J.; Olszewski, W.L. Experimental lymphovenous anastomosis. Br. J. Surg. 1968, 55, 449–451. [Google Scholar] [CrossRef] [PubMed]
- Olszewski, W.L. Lymphovenous microsurgical shunts in treatment of lymphedema of lower limbs: A 45-year experience of one surgeon/one center. Eur. J. Vasc. Endovasc. Surg. 2013, 45, 282–290. [Google Scholar] [CrossRef]
- Olszewski, W.L.; Zaleska, M. A novel method of edema fluid drainage in obstructive lymphedema of limbs by implantation of hydrophobic silicone tubes. J. Vasc. Surg. Venous Lymphat. Dis. 2015, 3, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Mihara, M.; Hara, H.; Zhou, H.P.; Tange, S.; Kikuchi, K. Lymphaticovenous anastomosis releases the lower extremity lymphedema-associated pain. Plast. Reconstr. Surg. Glob. Open 2017, 5, e1–e9. [Google Scholar] [CrossRef]
- Hara, H.; Mihara, M.; Ohtsu, H.; Narushima, M.; Iisa, T.; Koshima, I. Indication of lymphaticovenous anastomosis for lower limb primary lymphedema. Plast. Reconstr. Surg. 2015, 136, 883–893. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Sakuma, H.; Yamazaki, S. Evaluation of patency rates of different lymphaticovenous anastomosis techniques and risk factors for obstruction in secondary upper extremity lymphedema. J. Vasc. Surg. Venous Lymphat. Dis. 2019, 7, 113–117. [Google Scholar] [CrossRef]
- Maegawa, J.; Yabuki, Y.; Tomoeda, H.; Hosono, M.; Yasumura, K. Outcomes of lymphaticovenous side-to-end anastomosis in pheripheral lymphedema. J. Vasc. Surg. 2012, 55, 753–760. [Google Scholar] [CrossRef]
- Basta, M.N.; Gao, L.L.; Wu, L.C. Operative treatment of peripheral lymphedema: A systematic meta-analysis of the efficacy and safety of lymphovenous microsurgery and tissue transplantation. Plast. Reconstr. Surg. 2014, 133, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.I.; Lee, B.B.; Huh, S.; Lee, S.J.; Hwang, J.H.; Kim, Y.I. Excision of subcutaneous tissue and deep muscle fascia for advanced lymphedema. Lymphology 1998, 31, 190–194. [Google Scholar] [PubMed]
- Granzow, J.W. Lymphedema surgery: The current state of the art. Clin. Exp. Metastasis 2018, 35, 553–558. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.J.; Cheng, M.H. Lymphedema surgery: Patient selection and an overview of surgical technique. J. Surg. Oncol. 2016, 113, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.H.; Huang, J.J.; Wu, C.W.; Yang, C.Y.; Lin, C.Y.; Henry, S.L.; Kolios, L. The mechanism of vascularized lymph node transfer for lymphedema: Natural lymphaticovenous drainage. Plast. Reconstr. Surg. 2014, 133, 192e–198e. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, C.N.; Ozturk, C.; Glasgow, M.; Platek, M.; Ashary, Z.; Kuhn, J.; Aronoff, N.; Lohman, R.; Djohan, R.; Gurunluoglu, R. Free vascularized lymph node transfer for treatment of lymphedema: A systematic evidence based review. J. Plast. Reconstr. Aesthet. Surg. 2016, 69, 1234–1247. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.K.; Perrault, D.P.; Bouz, A.; Patel, K.M.; Hong, Y.K.; Wong, A.K. Surgical treatment modalities for lymphedema. J. Aesthet. Reconstr. Surg. 2016, 2. [Google Scholar] [CrossRef]
- Silva, A.K.; Chang, D.W. Vascularized lymph node transfer and lymphovenous bypass: Novel treatment strategies for symptomatic lymphedema. J. Surg. Oncol. 2016, 113, 932–939. [Google Scholar] [CrossRef]
- Saaristo, A.M.; Niemi, T.S.; Viitanen, T.P.; Tervala, T.V.; Hartiala, P. Microvascular breast reconstruction and lymph node transfer for postmastectomy lymphedema patients. Ann. Surg. 2012, 255, 468–473. [Google Scholar] [CrossRef]
- Gallagher, K.; Marulanda, K.; Gray, S. Surgical intervention for lymphedema. Surg. Oncol. Clin. N. Am. 2018, 27, 195–215. [Google Scholar] [CrossRef]
- Granzow, J.W.; Soderberg, J.M.; Kaji, A.H.; Dauphine, C. An effective system of surgical treatment of lymphedema. Ann. Surg. Oncol. 2014, 21, 1189–1194. [Google Scholar] [CrossRef] [PubMed]
- Scaverien, M.; Munnoch, D. Chapter-02 Liposuction for chronic lymphedema of the upper limb. In New Frontiers in Plastic and Cosmetic Surgery; Giuseppe, A.D., Shiffman, M.A., Eds.; Jaypee Brothers Medical Publishers Ltd.: New Delhi, India, 2015; pp. 13–22. [Google Scholar]
- Brorson, H.; Svensson, B.; Ohlin, K. Suction-Assisted Lipectomy. In Lymphedema; Greene, A.K., Slavin, S.A., Brorson, H., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 313–324. [Google Scholar]
- Lamprou, D.A.; Voesten, H.G.J.; Damstra, R.J.; Wikkeling, O.R.M. Circumferential suction-assisted lipectomy in the treatment of primary and secondary end-stage lymphoedema of the leg. Br. J. Surg. 2017, 104, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Frick, A.; Baumeister, R.G.H.; Hoffmann, J.N. Liposuction technique and lymphatics in liposuction. In Liposuction; Shiffman, M.A., Giuseppe, A.D., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 179–183. [Google Scholar]
- Granzow, J.W.; Soderberg, J.M.; Dauphine, C. A novel two-stage surgical approach to treat chronic lymphedema. Breast J. 2014, 20, 420–422. [Google Scholar] [CrossRef] [PubMed]
- Charles, R. Elephantiasis Scroti; Churchill: London, UK, 1912. [Google Scholar]
- Van der Walt, J.C.; Perks, T.J.; Zeeman, B.J.; Bruce-Chwatt, A.J.; Graewe, F.R. Modified Charles procedure using negative pressure dressings for primary lymphedema. Ann. Plast. Surg. 2009, 62, 669–675. [Google Scholar] [CrossRef] [PubMed]
- Sapountzis, S.; Ciudad, P.; Lim, S.Y.; Chilgar, R.M.; Kiranantawat, K.; Nicoli, F.; Constantinides, J.; Wei, M.Y.; Sönmez, T.T.; Singhal, D.; et al. Modified Charles procedure and lymph node flap transfer for advanced lower extremity lymphedema. Microsurgery 2014, 34, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Hareyama, H.; Ito, K.; Hada, K.; Uchida, A.; Hayakashi, Y.; Hirayama, E.; Oikawa, M.; Okuyama, K. Reduction/prevention of lower extremity lymphedema after pelvic and para-aortic lymphadenectomy for patients with gynecologic malignancies. Ann. Surg. Oncol. 2012, 19, 268–273. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Control Programme. Prevention of Clinical Lymphoedema after Cancer Treatment: Early Detection and Risk Reduction. A guide for Health Professionals. Available online: https://www.hse.ie/eng/services/list/5/cancer/patient/leaflets/prevention-of-clinical-lymphoedema-after-cancer-treatment.pdf (accessed on 11 July 2019).
- Cheifetz, O.; Haley, L. Management of secondary lymphedema related to breast cancer. Can. Fam. Physician 2010, 56, 1277–1284. [Google Scholar] [PubMed]
- Finegold, D.N.; Schacht, V.; Kimak, M.A.; Lawrence, E.C.; Foeldi, E.; Karlsson, J.M.; Baty, C.J.; Ferrell, R.E. HGF and MET mutations in primary and secondary lymphedema. Lymphat. Res. Biol. 2008, 6, 65–68. [Google Scholar] [CrossRef]
- American Cancer Society. Treatment & Support. For People at Risk of Lymphedema. Available online: https://www.cancer.org/treatment/treatments-and-side-effects/physical-side-effects/lymphedema/for-people-at-risk-of-lymphedema.html (accessed on 17 July 2019).
- Petrek, J.A.; Pressman, P.I.; Smith, R.A. Lymphoedema: Current issues in research and management. CA Cancer J. Clin. 2000, 50, 292–307. [Google Scholar] [CrossRef]
- National Breast and Ovarian Cancer Center (NBOCC). Review of Research Evidence on Secondary Lymphoedema: Incidence, Prevention, Risk Factors and Treatment. March 2008. Available online: https://canceraustralia.gov.au/sites/default/files/publications/slerw-review-research-secondary-lymphoedema_504af03e2a20c.pdf (accessed on 19 August 2019).
- Planinšek Ručigaj, T.; Tlaker Žunter, V. Lymphedema after breast and gynecological cancer—A frequent, chronic, disabling condition in cancer survivors. Acta Dermatovenerol. Croat. 2015, 23, 101–107. [Google Scholar]
| ISL Stage | Clinical Manifestation |
|---|---|
| 0 (or 1a) | Swelling is not yet evident despite impaired lymphtransport, subtle alterations in tissue fluid/composition, and changes in subjective symptoms. |
| 1 | Pitting may occur. An early accumulation of fluid relatively high in protein content occurs, which subsides with limb elevation. |
| 2 | Limb elevation alone rarely reduces the tissue swelling and pitting is manifest. |
| Later in Stage 2 | The limb may not pit as excess subcutaneous fat and fibrosis develop. |
| 3 | Lymphostatic elephantiasis occurs, where pitting can be absent and trophic skin changes such as acanthosis can be found. |
| Diagnostic Procedure | Features |
|---|---|
| Physical Examination | It may not be able to make a quantitative measurement of lymphedema. |
| Ultrasonography | Ultrasonography shows increased skin thickness, increased subcutaneous tissue thickness, and increased subcutaneous echogenicity. [52] |
| MRI | Water-fat MRI fails to differentiate water signals between fluid and muscle, which makes it difficult to evaluate subfacial tissue changes. [54] |
| MRI-LG | There are some limitations associated with MRI-LG, such as the long duration (about an hour) of the MR examination. [57] |
| CT-LG | CT-LG images can be obtained within a short time (about 5 min.). However, CT-LG has a disadvantage in its associated radiation exposure. [56] |
| Lymphoscintigraphy | Lymphoscintigraphy shows obstruction with visualization of discrete lymphatic trunks and slow transport. [65,66] |
| ICG-LG | ICG-LG detects the dermal lymph backflow sign in asymptomatic limbs and the splash pattern at the earliest stage. [68] |
| LDB Stage | ICG-LG Findings |
|---|---|
| satge 0 | Linear pattern only |
| stage 1 | Linear pattern + Splash pattern a |
| stage 2 | Linear pattern + Stardust pattern (1 region) b |
| stage 3 | Linear pattern + Stardust pattern (2 region) b |
| stage 4 | Linear pattern + Stardust pattern (3 region) b |
| stage 5 | Stardust pattern (associated with Diffuse pattern) |
| ISL Stage | Treatment |
|---|---|
| 0 (or 1a) | None |
| CDT [7,72,74] | |
| 1 | CDT [7,72,74] |
| LVA [92,94,100] | |
| 2 | LVA [92,94,100] |
| VLNT [100,103] | |
| Later in Stage 2 | VLNT [100,103] |
| Liposuction [97,107] | |
| 3 | VLNT [100,103,111,112] |
| Liposuction [97,107], Surgical Excision [113,114] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Watanabe, Y.; Koshiyama, M.; Seki, K.; Nakagawa, M.; Ikuta, E.; Oowaki, M.; Sakamoto, S.-i. Development and Themes of Diagnostic and Treatment Procedures for Secondary Leg Lymphedema in Patients with Gynecologic Cancers. Healthcare 2019, 7, 101. https://doi.org/10.3390/healthcare7030101
Watanabe Y, Koshiyama M, Seki K, Nakagawa M, Ikuta E, Oowaki M, Sakamoto S-i. Development and Themes of Diagnostic and Treatment Procedures for Secondary Leg Lymphedema in Patients with Gynecologic Cancers. Healthcare. 2019; 7(3):101. https://doi.org/10.3390/healthcare7030101
Chicago/Turabian StyleWatanabe, Yumiko, Masafumi Koshiyama, Keiko Seki, Miwa Nakagawa, Eri Ikuta, Makiko Oowaki, and Shin-ichi Sakamoto. 2019. "Development and Themes of Diagnostic and Treatment Procedures for Secondary Leg Lymphedema in Patients with Gynecologic Cancers" Healthcare 7, no. 3: 101. https://doi.org/10.3390/healthcare7030101
APA StyleWatanabe, Y., Koshiyama, M., Seki, K., Nakagawa, M., Ikuta, E., Oowaki, M., & Sakamoto, S.-i. (2019). Development and Themes of Diagnostic and Treatment Procedures for Secondary Leg Lymphedema in Patients with Gynecologic Cancers. Healthcare, 7(3), 101. https://doi.org/10.3390/healthcare7030101






